The laminins are a secreted family of heterotrimeric molecules essential for basement membrane formation, structure, and function. It is now well established that the α3 subunit of laminins-332, -321, and -311 plays an important role in mediating epidermal-dermal integrity and is essential for the skin to withstand mechanical stresses. These laminins also regulate cell migration and mechanosignal transduction. This article provides an overview of the gene, transcripts, and protein structures of laminin α3. Also discussed are the proposed functions for the α3 subunit–containing laminins.
The laminins are a secreted family of heterotrimeric molecules essential for basement membrane (BM) formation, structure, and function. Through the study of blistering skin diseases, it is now well established that the α3 subunit of laminins-332, -321, and -311 plays an important role in mediating epidermal-dermal integrity and is essential for the skin to withstand mechanical stresses. These laminins, however, also regulate cell migration and mechanosignal transduction. The precise mechanisms involved in cell migration and signaling are not yet fully clarified. This article provides an overview of the gene, transcripts, and protein structures of laminin α3, and briefly discusses the proposed functions for the α3 subunit–containing laminins.
LAMA3 gene structure and expression regulation
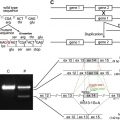
The human LAMA3 gene encodes 76 exons from 318 kb of genomic DNA at chromosomal location 18q11.2 ( Fig. 1 A ). Isolation of cDNA clones has revealed the presence of two major transcripts: LAMA3A and LAMA3B ( Fig. 1 B). Both of these transcripts share a common 3′ end that includes exons 40 through 76. Through alternate promoter usage, however, their 5′ ends are markedly different. LAMA3A , encoding laminin α3A, is expressed from a promoter within intron 38 and its protein product is encoded by exons 39 to 76 (5175 bp open reading frame, encoding 1724 amino acids, calculated molecular weight 190 kDa (see Fig. 1 B)). LAMA3B is much longer, consisting of exons 1 to 38 and the common 3′ exons 40 to 76; exon 39 is skipped (10002 bp open reading frame, encoding 3333 amino acids, calculated molecular weight 366 kDa (see Fig. 1 B)). In addition, at the message level about 20% of keratinocyte LAMA3B has exon 10 skipped. This shorter isoform has been termed “laminin α3B2,” whereas the full length transcript encodes laminin α3B1 (see Fig. 1 B).
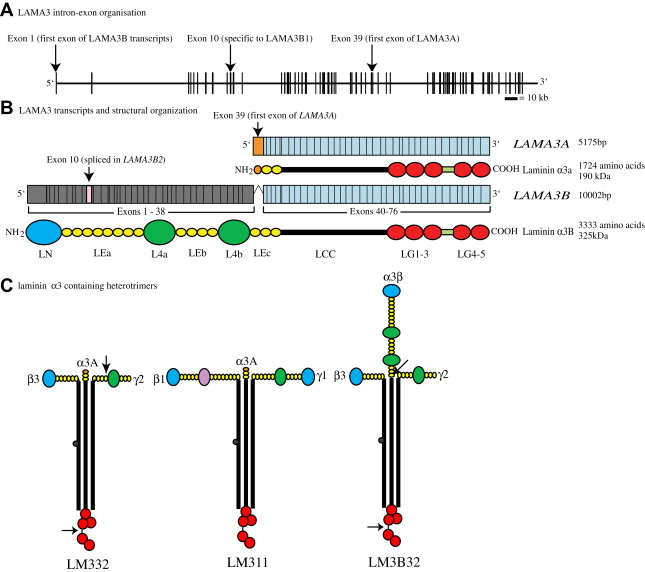
Testing of the promoter regions for both LAMA3 transcripts reveals them to be responsive to typical epithelial-mesenchymal transcription factors: epidermal growth factor, keratinocyte growth factor, insulin-like growth factor-1, thymosin beta 4, interferon-γ, transforming growth factor-α and -β1, and tumor necrosis factor-α. Both promoters also contain acute-phase reactant sequences and interleukin-6 binding sequences, both of which are found in many proteins upregulated at sites of trauma.
Through RNase protection assays on total RNA from adult human tissues, Doliana and colleagues have investigated the expression pattern of LAMA3A and LAMA3B . Spleen, stomach, kidney, skeletal muscle, pancreas, and adrenal gland express similar levels of both transcripts, whereas the salivary gland expresses only LAMA3A . Placenta expresses the highest LAMA3A message, whereas the uterus expresses the highest LAMA3B message. In situ reverse transcriptase polymerase chain reaction of human embryonic tissues reveals positive staining for LAMA3 (not LAMA3/B specific) message in developing tubules and developing comma-shaped bodies of the kidney, in epithelial cells of the developing lung, in the basal layer of developing skin at gestational week 6.5, and in all layers of the epidermis from gestational week 8 onward.
Laminin α3A/B subunit domain architecture and assembly isoforms
The laminin family of proteins shares a common architecture with regions of conserved protein folding. Laminins are secreted as heterotrimeric cross-shaped molecules consisting of one α, one β, and one γ subunit that assemble intracellularly through a coiled-coil domain termed the “LCC” (formerly known as “domains I and II”). In laminin α subunits, this LCC is followed by five globular domains (termed “LG1–5”). The link between LG3 and 4 is slightly extended in the laminin α3 subunit relative to other laminin α subunits, and is the site of an extracellular processing event (see later). In the laminin α3A subunit, the LCC is preceded by a short stretch of rodlike, laminin-type epithelial growth factor-like domains (LE, formerly domain V). In contrast, the amino terminus of laminin α3B subunit is much longer, consisting of an approximately 250 amino acid laminin N-terminal domain (LN domain, previously domain VI), which has been shown to be important for higher-order network formation through copolymerization and self-polymerization. The LN domain is followed by three stretches of rodlike LE domains (of eight, four, and three repeats, respectively), which are interspersed by two approximately 250 amino acid globular domains (termed “L4a” and “L4b,” previously domain IV (see Fig. 1 B)).
The functionality of individual laminin subunits depends not only on their own domain composition but also on that of the laminin subunits with which they associate. In terms of laminin α3A subunit, the most abundant and most studied isoform is laminin 332, comprising laminin α3A, β3, and γ2 (LM332, formerly known as “laminin 5/kalinin/epiligrin/ladsin” ( Fig. 1 C)). In addition, laminin α3A associates with laminin β1 and γ1 forming LM311 (laminin 6, k-laminin (see Fig. 1 C)) and, from co-immunoprecipitation data, with β2 and γ1 to form LM321 (laminin 7 (see Fig. 1 C)). The expression profile LM332, as expected, roughly matches that of its constituent mRNAs. Immunofluorescence staining for LM332 in adult tissues gives positive results in the BM of glomeruli and tubuli in kidney, the BM of alveoli, bronchioli and bronchi in lung, in the dermal-epidermal junction of skin, corneal BM, and in the enteric BM zone of the small intestine under the intestinal epithelium.
Laminin heterotrimer formation proceeds by a βγ dimer stage and seems to be dependent on sequences toward the C-terminus of the LCC. In theory, the laminin α3B subunit should be capable of associating with the same repertoire of β and γ laminin subunits as α3A. Although immunohistologic analyses have suggested the presence of LM3B11 in the BM of blood vessels, only LM3B32 has been studied in any detail to date (see Fig. 1 C). Interestingly, although βγ dimers require α laminin subunit incorporation to drive secretion, there is evidence that laminin α subunits can be secreted independently of trimerization; the functional significance of this observation is yet to be established.
Human LM332 is secreted as a 460-kDa species that is subsequently processed to a predominant 440-kDa form in keratinocytes maintained in low calcium medium (0.035 mM) and to a predominant 400-kDa form in keratinocytes maintained at higher concentrations of calcium (1 mM). These size shifts are caused by processing of the C-terminus of the α3A subunit, which removes LG domains 4 and 5 and converts it from approximately 190 kDa to 165 kDa, and processing of the γ2 subunit toward its N-terminus, converting it from a 155-kDa form to 105 kDa. LM332 containing the 165-kDa α3A and 105-kDa γ2 processed subunits is sometimes termed “mature” (matLM332). Processing of the laminin α3B subunit converts it from approximately 325 kDa to an approximately 280-kDa mature form. An additional minor product of 145 kDa, which is recognized by laminin α3 antibodies, has also been identified in extracts from human amnion, with the secondary processing occurring at the N-terminus, just before the LCC. Interestingly, in vitro studies have demonstrated that laminin α3A in LM311 is processed at a much lower rate than when it is incorporated into LM332, which may be relevant with regards to some of the functional differences between these heterotrimers (discussed later).
Laminin α3A/B subunit domain architecture and assembly isoforms
The laminin family of proteins shares a common architecture with regions of conserved protein folding. Laminins are secreted as heterotrimeric cross-shaped molecules consisting of one α, one β, and one γ subunit that assemble intracellularly through a coiled-coil domain termed the “LCC” (formerly known as “domains I and II”). In laminin α subunits, this LCC is followed by five globular domains (termed “LG1–5”). The link between LG3 and 4 is slightly extended in the laminin α3 subunit relative to other laminin α subunits, and is the site of an extracellular processing event (see later). In the laminin α3A subunit, the LCC is preceded by a short stretch of rodlike, laminin-type epithelial growth factor-like domains (LE, formerly domain V). In contrast, the amino terminus of laminin α3B subunit is much longer, consisting of an approximately 250 amino acid laminin N-terminal domain (LN domain, previously domain VI), which has been shown to be important for higher-order network formation through copolymerization and self-polymerization. The LN domain is followed by three stretches of rodlike LE domains (of eight, four, and three repeats, respectively), which are interspersed by two approximately 250 amino acid globular domains (termed “L4a” and “L4b,” previously domain IV (see Fig. 1 B)).
The functionality of individual laminin subunits depends not only on their own domain composition but also on that of the laminin subunits with which they associate. In terms of laminin α3A subunit, the most abundant and most studied isoform is laminin 332, comprising laminin α3A, β3, and γ2 (LM332, formerly known as “laminin 5/kalinin/epiligrin/ladsin” ( Fig. 1 C)). In addition, laminin α3A associates with laminin β1 and γ1 forming LM311 (laminin 6, k-laminin (see Fig. 1 C)) and, from co-immunoprecipitation data, with β2 and γ1 to form LM321 (laminin 7 (see Fig. 1 C)). The expression profile LM332, as expected, roughly matches that of its constituent mRNAs. Immunofluorescence staining for LM332 in adult tissues gives positive results in the BM of glomeruli and tubuli in kidney, the BM of alveoli, bronchioli and bronchi in lung, in the dermal-epidermal junction of skin, corneal BM, and in the enteric BM zone of the small intestine under the intestinal epithelium.
Laminin heterotrimer formation proceeds by a βγ dimer stage and seems to be dependent on sequences toward the C-terminus of the LCC. In theory, the laminin α3B subunit should be capable of associating with the same repertoire of β and γ laminin subunits as α3A. Although immunohistologic analyses have suggested the presence of LM3B11 in the BM of blood vessels, only LM3B32 has been studied in any detail to date (see Fig. 1 C). Interestingly, although βγ dimers require α laminin subunit incorporation to drive secretion, there is evidence that laminin α subunits can be secreted independently of trimerization; the functional significance of this observation is yet to be established.
Human LM332 is secreted as a 460-kDa species that is subsequently processed to a predominant 440-kDa form in keratinocytes maintained in low calcium medium (0.035 mM) and to a predominant 400-kDa form in keratinocytes maintained at higher concentrations of calcium (1 mM). These size shifts are caused by processing of the C-terminus of the α3A subunit, which removes LG domains 4 and 5 and converts it from approximately 190 kDa to 165 kDa, and processing of the γ2 subunit toward its N-terminus, converting it from a 155-kDa form to 105 kDa. LM332 containing the 165-kDa α3A and 105-kDa γ2 processed subunits is sometimes termed “mature” (matLM332). Processing of the laminin α3B subunit converts it from approximately 325 kDa to an approximately 280-kDa mature form. An additional minor product of 145 kDa, which is recognized by laminin α3 antibodies, has also been identified in extracts from human amnion, with the secondary processing occurring at the N-terminus, just before the LCC. Interestingly, in vitro studies have demonstrated that laminin α3A in LM311 is processed at a much lower rate than when it is incorporated into LM332, which may be relevant with regards to some of the functional differences between these heterotrimers (discussed later).
The function of the laminin α3 subunit in cellular adhesion
In epithelial cells LM332 is able to interact with two integrins, α6β4 and α3β1, and thereby is of central importance in the function of the two major forms of dermal-epidermal junctions: hemidesmosomes and focal adhesions.
Hemidesmosome Formation
Hemidesmosomes are specialized adhesion structures that provide linkage from LM332 to the intermediate filament cytoskeleton. This linkage is established through the association of the extracellular domains of α6β4 integrin with the laminin α3 subunit and through binding of the intracellular tail of β4 integrin to the plakin molecule plectin (HD1). Plectin, in turn, interacts with the keratin cytoskeleton. Adhesion is further strengthened through the association with the transmembrane protein, bullous pemphigoid antigen 2 (collagen XVIII), which also interacts with LM332 and by binding to β4 integrin of a second plakin molecule termed BPAG1e (BP230), which acts to strengthen the link to the keratins.
Carboxy terminal processing of laminin α3A may regulate the assembly of hemidesomsomes. Specifically, in tissue culture, only the matrix of cell lines that contain a C-terminally processed form of laminin α3A supports formation of hemidesmosomes. Furthermore, treatment with plasmin of an extracellular matrix rich in LM332, but containing an unprocessed α3A laminin subunit that fails to support hemidesmosome formation, results in laminin α3A processing and conversion of that matrix to one that is competent to induce HD assembly. In addition to plasmin, all of the bone morphogenetic protein-1 isoenzymes (mammalian tolloid, mammalian tolloid–like-1 and -2) have been shown to be capable of processing the laminin α3A subunit to 165 kDa and the laminin γ2 subunit to 105 kDa in vitro. The skin of mice deficient for mammalian tolloid–bone morphogenetic protein-1 exhibits defects in hemidesomosomes, suggesting the importance of such processing for hemidesmosome formation.
The importance in vivo of laminin α3 in dermal-epidermal adhesion is dramatically exemplified by skin blistering at the dermal-epidermal junction from mutation of the LAMA3 gene in patients with junctional epidermolysis bullosa, in which hemidesmosomes are either entirely undetectable ultrastructurally or are reduced in number and aberrant (see discussion of junctional epidermolysis bullosa elsewhere in this issue).
Focal Contact Formation
In contrast to hemidesmosomes, which provide a link from LM332 to the keratin cytoskeleton, focal adhesions provide a link from LM332 to the actin cytoskeleton through interaction of the laminin α3 subunit with α3β1 integrin. α3β1 Integrin, in turn, interacts with a number of linker molecules, which mediate the association of the actin cytoskeleton with the cell surface. Moreover, α3β1 integrin also interacts with molecules involved in signal transduction. In cultured epidermal cells, α3β1 integrin is found clustered at the site of focal adhesions. In intact skin, focal adhesions are not obvious and are likely transient matrix adhesion points that are assembled by actively moving cells. The identification of mutations in FERMT1 , a gene encoding a focal contact protein termed “Kindlin-1,” have recently been demonstrated as pathogenic in another form of epidermolysis bullosa associated with photosensitivity, the Kindler syndrome subtype. These data suggest that the LM332-α3β1-actin linkage may also be required for maintenance of epithelia-dermal attachment integrity; however, whether the skin fragility of Kindler syndrome is a direct result of loss of LM332-α3β1 integrin linkage or indirect, caused by disruption of HDs in Kindlin-1–deficient skin, requires further investigation.
The laminin α3 subunit in cell migration and wound healing
There is considerable evidence that LM332 is an important regulator of cell migration. Histologically, LM332 is deposited into the provisional BM of healing wound beds within 8 hours of wounding. Moreover, in squamous cell carcinoma (SCC) an upregulation of LM332 correlates with poor prognosis as a result of increased metastatic potential. The precise mechanisms through which laminins with an α3 subunit regulate cell migration is controversial, particularly with respect to the roles of α3β1 and α6β4 integrin, and the functional significance of LM332 proteolytic processing.
Historically, α3β1 integrin has been thought to promote cellular migration, whereas α6β4 integrin, because of its ability to nucleate hemidesmosome formation, has long been believed to retard migration by promoting stable adhesion. A recent paper has suggested, however, that the α3β1 integrin–LM332 interaction may actually slow wound healing rates, specifically that α3 integrin–deficient keratinocytes migrate with increased velocity and persistence relative to controls. Furthermore, there are accumulating data suggesting that α6β4 integrin positively regulates skin cell migration.
Processing and Regulation of Motility and Proliferation
The role of proteolytic processing of LM332 in regulating its function requires further clarification. The processed form of LM332 is known to be present in mature, unwounded skin, whereas the unprocessed form is deposited at the leading edge of acute wounds or in culture equivalents. As previously described, laminin α3 subunit processing is required for hemidesmosome formation. Similarly, using an antibody to LG4/5 domain of laminin α3, Frank and Carter showed that migrating keratinocytes deposit unprocessed laminin α3 in a linear trail that marks the path of migration.
Interestingly, the presence of the released LG4/5 region also seems to aid deposition of LM332 or its incorporation into the BM. Processing could drive a localized increase in LM332 concentration, which in turn may enhance integrin clustering and signaling activities. Consistent with this, the level of LM332 deposition in SCCs correlates well with their invasive potential. Given that the unprocessed form of the laminin α3A subunit is predominantly found in SCC, whereas only the mature, processed subunit is present in unwounded skin, the Marinkovich group has generated an antibody specific for the LG4/5 region of the laminin α3A subunit that might specifically target SCC cells therapeutically. Indeed, in a mouse model of humanized SCC, treatment with LG4/5 antibodies induced a significant decrease in tumor volume without causing skin fragility.
The C-terminus of the laminin α3 subunit may also activate cell proliferative responses. Function-inhibiting antibodies to the laminin α3 LG domain inhibit proliferation of epithelial cells and decrease the level of p42/p44 MAPK activity. Ligation of either of its integrin-binding partners may be responsible for initiation of this response. Ligation of α6β4 integrin by LM332 induces phosphorylation of the β4 cytoplasmic domain. The Shc adaptor protein binds to these phosphorylated tyrosines and is subsequently tyrosine phosphorylated. On phosphorylation, Shc recruits Grb2 (which is stably associated with the Ras-GTP exhange factor mSOS) and this leads to activation of the Ras-Erk and Rac-Jnk MAPK pathways. Similarly, function-blocking antibodies to integrin α3 and β1 also block proliferation and MAPK phosphorylation. Further, laminin α3 subunit antibody-induced inhibition of proliferation can be rescued through treatment with β1 activating antibodies, indicating that α3β1 integrin likely mediates signals initiated by LM332 that control growth and drive proliferation.
Laminin γ2 subunit processing is also an important regulator of LM332 function. The second stretch of LE repeats in the laminin γ2 subunit has been shown to be capable of interacting with epidermal growth factor and it has been proposed that the amino terminal processing of the laminin γ2 subunit exposes this region and allows this interaction to occur, thereby triggering cell motility.
LM332 Deposition
A critical aspect of appropriate cell migration is the ability to move in a polarized manner and this ability is dependent on the exogenous ligand presented to cells; in one study approximately 50% of cultured epithelial cells displayed a polarized phenotype when plated on LM332, compared with only approximately 11% when plated onto collagen. The precise way LM332 is deposited, rather than deposition alone, however, is most important in supporting directed keratinocyte migration.
The involvement of both α3β1 integrin and α6β4 integrin in LM332 deposition has been made apparent through analyses of LM332 matrix patterns in keratinocytes deficient in either α3 integrin or β4 integrin. Specifically, keratinocytes derived from α3 integrin null mice deposit LM332 into spikes and arrowhead patterns, compared with more diffuse arcs in wild-type keratinocytes. Furthermore, α3 integrin deficient keratinocytes are unable to reorganize precoated LM332 into ring structures in the same way as do wild-type cells. In comparison, migrating human cells deficient in integrin β4 deposit LM332 in circular arrays, as compared with the linear trails deposited by migrating wild-type keratinocytes. Moreover, the precise way LM332 is deposited into the matrix is dominant with regards to motile behavior, because plating β4-deficient cells onto the LM332 trails deposited by wild-type cells restores their migration patterns, whereas plating wild-type cells onto the circular tracks laid down by β4-deficient cells leads to a circular motility phenotype.
Multiple further studies have implicated a role for the actin cytoskeleton in determining the specific arrangement of LM332 in the matrix of cultured keratinocytes. Inhibition of actomyosin contraction in wild-type cells, either through drug treatment or through introduction of dominant negative forms of the Rac, Rho, and Cdc42 small GTPases, leads to an aberrant organization of LM332. It has also been observed that reorganization of precoated LM332 occurs in regions that have been extended over by filopodia and lamellipodia. The different ability of α3β1 and α6β4 integrins to activate RhoGTPase family members plays a role in their mediating deposition of LM332. Specifically, α3 integrin has been implicated in the activation of RhoA, whereas β4 integrin regulates Rac activity. In the case of α6β4 this regulation likely is caused by formation of a complex with Rac, because Rac can be co-immunoprecipiated with β4 integrin and the activity level of Rac is decreased in β4-deficient cells. Downstream of Rac, activity of the actin severing and remodeling protein cofilin is also reduced in β4-deficient cells. Rac and cofilin activity are intrinsically linked to directed migration through their ability to nucleate and drive extension of lamellipodia; the α6β4 integrin-Rac association may provide a means of spatially restricting this signaling. Intriguingly, recent data indicate that the interaction of β4 integrin with Rac, and Rac activation, is dependent on BPAG1e and further, that BPAG1e knockdown cells show a loss of front-rear polarity. These results are somewhat surprising because both α6β4 integrin and BPAG1e are hemidesmosomal components and have been thought to be primarily involved in stable adhesion rather than migration.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree