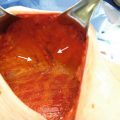
Fig. 13.1
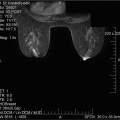
The partial subpectoral approach depicted and is facilitated using ADM. The ADM is sutured to the inferior edge of the pectoralis major muscle and to the inframammary fold (Image courtesy of Acelity—A LifeCell company)
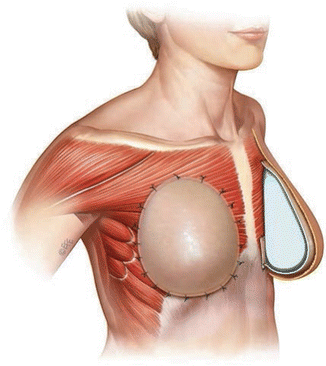
Fig. 13.2
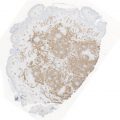
The prepectoral approach is depicted using ADM. The ADM is sutured circumferentially around the prosthetic device (Image courtesy of Acelity—A LifeCell company)
This chapter will examine the controversy as to whether or not acellular dermal matrices provide a substantial benefit to surgical and aesthetic outcomes in the setting of nipple-sparing mastectomy and prosthetic breast reconstruction. A review of the various components of this operation is provided.
Nipple-Sparing Mastectomy
Nipple-sparing mastectomy can be performed in the setting of therapeutic or prophylactic mastectomy [6, 7]. Classic indications are based on breast and tumor characteristics that include mild to moderate breast volume and tumors with a diameter of less than 3 cm that are located at least 4 cm from the nipple areolar complex [8]. However, these parameters are being challenged, as strategies for nipple areolar preservation in women with large or ptotic breasts are now possible [9, 10]. In addition, larger tumors in excess of 3 cm may no longer be a contraindication as long as adequate margins are obtained [11].
Preservation of the entire skin envelope of the breast provides the plastic surgeon with a variety of reconstructive options. These will be dependent on the quality and viability of the mastectomy skin flaps . Assessment of the perfusion to the mastectomy skin flap and nipple areolar complex is achieved using clinical observation; however, fluorescent angiography can provide additional useful information [12]. When tissue perfusion is sufficient and skin thickness is adequate, either a direct-to-implant or two-stage reconstruction can be performed. The two-stage reconstruction involves placement of a tissue expander followed by replacement with a permanent implant. When tissue perfusion is compromised, a two-stage or delayed reconstruction is considered. The rationale for this is to minimize any pressure placed on the nipple areolar complex. Direct to implant reconstruction is usually performed in the setting of partial muscle coverage in order to release the inferior insertion of the pectoralis major muscle and to permit a hand-in-glove fit of the permanent implant within the skin envelope. Direct-to-implant reconstruction is not commonly performed in the setting of total muscle coverage because of the challenges associated with placement of a prefilled silicone gel implant under the pectoralis major muscle following mastectomy. Two-stage reconstruction can be considered with either partial or total muscle coverage techniques. With the direct-to-implant and two-stage procedures in the setting of partial muscle coverage, the use of acellular dermal matrix is often considered. With total muscle coverage, ADM is usually not necessary because the device is already completely covered. The purpose of ADM will be further described.
Acellular Dermal Matrix
Acellular dermal matrix is cadaveric skin derived from human, porcine, or bovine sources [13]. It is processed to remove all cellular elements such as fibroblasts, erythrocytes, leukocytes, and bacteria and to retain noncellular elements such as collagen, hyaline, elastin, and fibronectin. ADM has the capacity to recellularize and revascularize when in contact with adjacent host tissues [14]. The role of ADM in the setting of prosthetic devices is to provide coverage of the implant or tissue expander. This is achieved in two ways. The first is in the setting of partial muscle coverage by maintaining coverage of the device from the divided inferior edge of the pectoralis major muscle to the inframammary fold [15–18]. The second is in the setting of prepectoral coverage where ADM is used to line the subcutaneous mastectomy skin envelope and to compartmentalize the device [19]. The primary function of the ADM in both settings is to provide additional soft tissue support, compartmentalization, and prevention of window-shading or upward movement of the pectoralis major muscle . It is beneficial in the setting of immediate direct-to-implant as well as two-stage prosthetic reconstruction.
Acellular dermal matrices come in a variety of shapes and sizes and are manufactured by a variety of companies (Fig. 13.3). Not all ADMs are created or perform equally. The processing and sterilization methods vary amongst the different companies (Table 13.1). A thorough review is not possible within the context of this chapter; however, when selecting an ADM, surgeons should review the relevant data and literature, study the performance profile, and be aware of the mechanism of action. Readers are encouraged to review the referenced manuscripts for a complete breakdown of the various ADMs [13, 20–24].
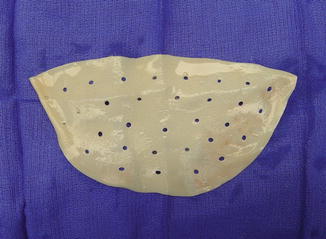

Fig. 13.3
Acellular dermal matrix (AlloDerm) in its typical form that is contoured, perforated, and ready to use (LifeCell Corporation, Bridgewater, NJ)
Table 13.1
The characteristics of various acellular dermal matrices are listed
AlloDerm (RTU) | Flex HD | DermaMatrix | AlloMax | |
|---|---|---|---|---|
Source | Human | Human | Human | Human |
Cross-linking | None | None | None | None |
Decellularization and processing | Proprietary | Hypertonic bath | Hypertonic bath | Acetone, hypo/hypertonic bath, H2O2 |
Sterilization (SAL) | Radiation (10−3) | Detergents (aseptic) | Detergents (sterile) | Radiation (10−6) |
The essential component of ADM is the extracellular matrix. The ability of an ADM to revascularize and recellularize when adjacent to vascularized tissue is dependent upon the integrity of the extracellular matrix. The integration of ADM to the mastectomy skin flap requires a certain degree of inflammation for wound healing to occur. Once integrated, the ADM will provide long-term tissue support without resorption or conversion to scar or capsule. Histologic examination of ADM following implantation into the breast demonstrates repopulation with red blood cells and fibroblasts with retention of the elastin [25, 26]. There is a clear line of demarcation between scar tissue and the ADM.
A common clinical observation following the use of prosthetic devices and ADM is that the amount of scar tissue formed along the surface of the ADM adjacent to the prosthetic device is reduced compared to when ADM is not used [2]. The physiologic basis for this is that there are specific cell-signaling mechanisms that limit the amount of collagen deposited on the surface of the ADM adjacent to the device. The host response to the ADM is that of “host recognition” because of the intact extracellular matrix that allows for incorporation and regeneration. Thus, capsular contracture appears to occur less frequently when ADM is used. The mechanics can be explained based on principles regulating wound contraction given that it is reduced in the setting of a full thickness skin graft. Although ADM is not a skin graft in the true sense because it lacks the cellular elements of a full thickness skin graft, it does possess the mechanical properties of a full thickness skin graft. When a full thickness skin graft is placed on a raw skin surface, it will inhibit wound contraction by inhibiting the activity of myofibroblasts that are necessary for contraction to occur. When ADM is used, collagen deposition occurs at the junction of the graft and the tissue surface and is not exaggerated [25, 26]. For capsular contracture to occur around a breast implant , a spherical capsule must be present. When ADM is used, the spherical nature of the capsule is disrupted, thus capsular contracture is rarely observed in nonradiated patients.
Several experimental and clinical studies have attempted to explain this phenomenon of reduced scar formation. In an experimental study, silicone sheets were implanted into the dorsum of 20 rabbits [27]. In ten rabbits, the silicone sheets were wrapped in ADM and in another ten rabbits, silicone sheets were not wrapped in ADM. The periprosthetic tissue was analyzed at 13 weeks. The ADM cohort demonstrated thin capsule with minimal cellularity or inflammatory infiltrates , whereas in the non-ADM cohort, the capsule was thick with increased cellularity. In another experimental study, implanted devices that were partially wrapped in ADM demonstrated less capsule formation than devices that were not wrapped in ADM [28]. These two studies suggest that capsule formation is minimized in the setting of ADM. The effect of radiation therapy on ADM incorporation has also been studied [29]. Komorowska-Timek demonstrated that ADM (AlloDerm, LifeCell Corporation, Bridgewater, NJ) decreases radiation-related inflammation in a murine model and may lessen the progression to capsule formation and ultimately capsular contracture.
In a clinical study, the histology of implanted ADM and native capsule was studied in 23 women following prosthetic reconstruction [25]. ADM histology demonstrated a lack of granulation tissue and vascular proliferation as well as a mild increase in collagen and inflammatory infiltrates . Capsule histology demonstrated abundant granulation with mild vascular proliferation as well as a moderate increase in collagen and inflammatory infiltrates. This study demonstrated that the presence of ADM appeared to attenuate the process of fibrosis . Explanations are debatable but are most likely related to the diminished inflammatory response. The association of prolonged inflammation leading to increased fibrosis is well known. Other studies have demonstrated that in women with silicone gel implants and capsular contracture, the capsules are characterized by vascular proliferation and the presence of a lymphocytic and mononuclear infiltrate as well as silicone particles [26]. This was not seen in the setting of ADM and silicone gel implants.
Based on the observation that periprosthetic fibrosis or capsular contracture may be minimized in the setting of ADM, some surgeons are advocating prepectoral placement of the device with total anterior coverage using ADM. The rationale is that the device will remain covered in the event of delayed healing and that the operation would be simplified by not having to elevate the pectoralis major muscle . The implications of this technique are that tissue support would be provided by the ADM, capsular contracture would be minimized because of the ADM, and that breast animation would be eliminated because the device is positioned above the pectoralis major muscle .
Surgical Techniques
Nipple-Sparing Mastectomy and ADM-Assisted Reconstruction
Nipple-sparing mastectomy with ADM can be performed through an inframammary, vertical infra-areolar or lateral incision. The choice between the various incisions is based on surgeon preference, tumor location, and nipple position. Laterally based incisions are usually designed in a lazy S fashion to prevent lateralization of the NAC that sometimes occurs with scar contracture. Inframammary incisions are made from the midline and extend laterally to a length of 6–8 cm. Vertical infra-areolar incisions are recommended when a mild to moderate amount of nipple elevation is desired. All approaches will provide ample access to the breast parenchyma to ensure that an adequate mastectomy has been performed.
Patients are marked preoperatively while standing. Following completion of the mastectomy, the skin flaps are assessed for viability and perfusion. Tissue perfusion is assessed using direct observation for arterial and venous bleeding from the cut edges as well as color and capillary refill of the mastectomy skin flaps. When questionable, fluorescent angiography can be considered to assess perfusion using infrared spectroscopy and indocyanine green. The decision between DTI and 2-stage reconstruction is made based on volume requirements, tissue thickness and perfusion, and patient/surgeon preference .
Partial Subpectoral/DTI
Direct-to-implant (DTI) reconstruction is often considered for women with mild to moderate breast volume following nipple-sparing mastectomy [2, 3]. The argument for DTI is that no skin has been excised; therefore, there is no need for tissue expansion. Partial subpectoral coverage is often performed because the divided and elevated inferior edge of the pectoralis major muscle allows for excellent projection of the permanent implant. Acellular dermal matrices play a critical role in these cases in order to adequately compartmentalize the implant, control the position of the pectoralis major muscle, and to provide additional tissue support.
There are several technical variations in the setting of partial subpectoral coverage and direct to implant reconstruction using ADM. The described technique is one that is commonly performed by the author. Figures 13.4, 13.5, 13.6, and 13.7 illustrate a patient having nipple-sparing mastectomy and direct-to-implant reconstruction using an ADM (AlloDerm—LifeCell Corporation, Bridgewater, NJ). A range of permanent implants is considered; however, only 1–3 are typically selected based on bio-dimensional planning. Following completion of the nipple-sparing mastectomy, the inferior border of the pectoralis major muscle is divided and the subpectoral space is created. The superior edge of the ADM is sutured to the inferior border of the pectoralis major muscle. A template of the implant is used to determine where to suture the lateral edge of the ADM to the chest wall. Once determined, the implant is inserted and positioned along the medial cul-de-sac and the desired inframammary fold. The ADM is trimmed so that it will lie along the inferior and lateral mastectomy skin flaps and adequately compartmentalize the implant. The inferior and lateral edges of the ADM are sutured to the chest wall using absorbable sutures.
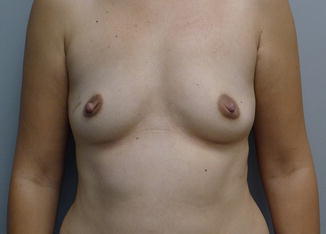
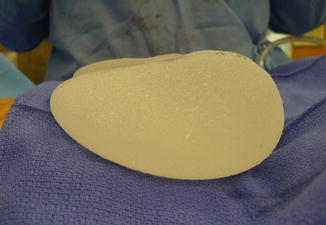
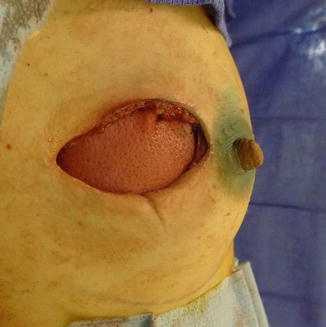
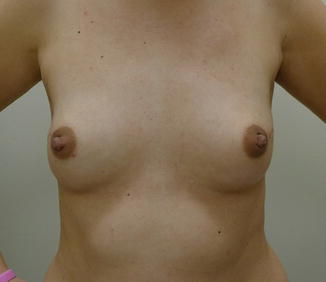

Fig. 13.4
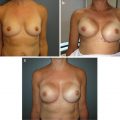
Preoperative view of a woman with mild breast volume prior to nipple-sparing mastectomy

Fig. 13.5
A shaped permanent silicone gel implant is selected based on biodimensional planning

Fig. 13.6
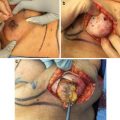
The bilateral nipple-sparing mastectomy is performed through a lateral areolar incision . The ADM and the shaped silicone gel implant are inserted, oriented, and compartmentalized

Fig. 13.7
Postoperative view at 6 months demonstrating excellent symmetry, volume, and contour
Partial Subpectoral/2-Stage
Two-stage reconstruction is defined as placement of a temporary tissue expander followed several months later by a permanent implant [17, 18, 30, 31]. A commonly asked question is why use a tissue expander in the setting of nipple-sparing mastectomy because the skin envelope is intact. There are several explanations; the first being that the mastectomy skin flaps may be thin and that perfusion to the skin and nipple areolar complex is questionable or uncertain. Placing a prefilled implant may cause additional pressure on the nipple areolar complex further compromising perfusion and possibly resulting in nipple areolar necrosis. A tissue expander is useful because it can be minimally filled avoiding undo pressure on the compromised skin flaps. Another reason is that selection of an optimal permanent implant is challenging given that there is an assortment of shapes, volumes, fillers, and textures. Selection of the optimal permanent implant is best made during the second stage.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree