Anatomy
The abdominal wall is a multilayered structure that wraps around and protects the abdominal viscera. In addition, it is involved in numerous other functions including trunk movement, respiratory effort (active expiration), and an adjunctive force in micturition and defecation. Anatomically, it is bound by the xiphoid process and costal margin superiorly and the iliac crests and pubis inferiorly.
The abdominal wall consists of skin, superficial fat, muscles, transversalis fascia, and parietal peritoneum.
Skin, Subcutaneous Fat, and Fascia
The skin envelope of the abdomen is compliant and of intermediate thickness. Traditionally, two distinct layers of fascia are recognized – Camper’s fascia and Scarpa’s fascia. Camper’s fascia represents the superficial layer, which contains variable amounts of fat. In males it is continuous with dartos fascia in the penis. Scarpa’s fascia is deeper and more fibrous. Scarpa’s fascia merges with the deep thigh fascia and superficial perineal fascia to contribute to fascia lata and Colles’ fascia, respectively. In reality, it is more practical to think of three layers between the skin and abdominal musculature – superficial fat, membranous fascial layer (Scarpa’s), and deep fat. The superficial fat layer is evenly distributed throughout the abdomen and is more vascular. The deeper fat layer is of variable thickness, being thicker in the upper abdomen compared with the lower abdomen.
Muscles
The central abdominal wall is formed by the two paired rectus abdominis muscles and pyrimidalis muscles inferiorly.
The paired rectus abdominis muscles are long and flat muscles involved in trunk flexion, forced expiration, and in creating increased abdominal pressure such as in coughing. They originate from the pubic symphysis and pubic crest and insert into the xyphoid process and costal cartilages 5–7, interdigitating with the inferior fibers of pectoralis major. The two paired muscles are enclosed within the rectus sheath and separated from each other by a midline band of connective tissue, the linea alba. The vertical muscle fibers are separated by three or four tendinous interconnections, which are horizontal bands of connective tissue. It is a type III muscle with a dual co-dominant blood supply (deep inferior and superior epigastric arteries). It is segmentally innervated from the 7th to 11th intercostal nerves.
The pyrimidalis muscles originate from the pubis and insert into the linea alba.
The lateral abdominal wall muscles include three sets of paired muscles — external oblique, internal oblique, and transversus abdominis, which all contribute to the rectus sheath medially ( Fig. 40.1 ).
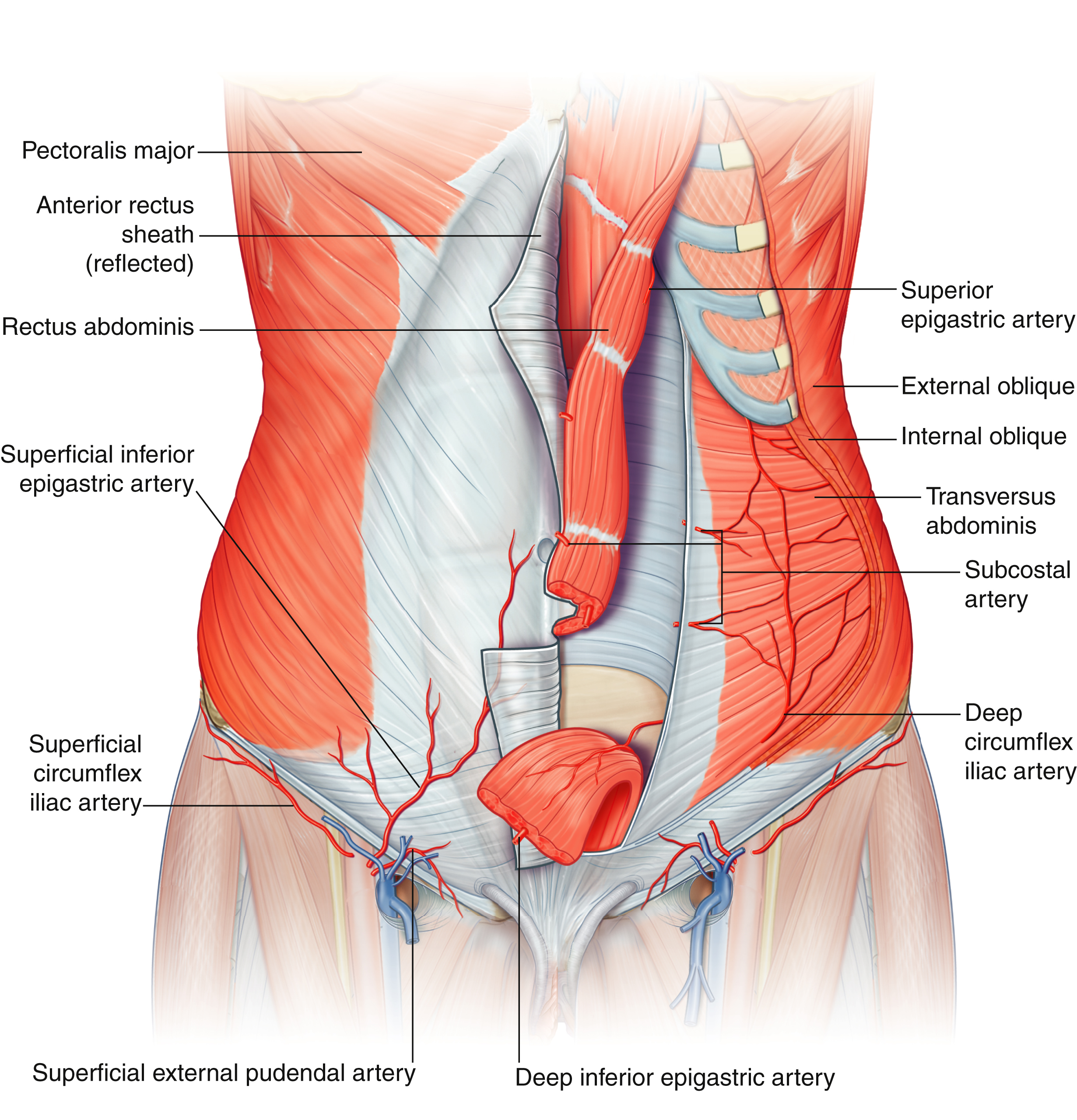
The external oblique is the most superficial and largest of the three lateral muscles. It originates from ribs 5–12 and runs in an inferiomedial direction to insert into the linea semilunaris, where it contributes to the anterior rectus sheath, and iliac crest. The aponeurosis of external oblique forms the inguinal ligament inferiorly. It is innervated and nourished by the intercostal neurovascular bundles (intercostal nerves 5–12).
The internal oblique originates from the thoracolumbar fascia and iliac crest and runs in a superomedial direction, perpendicular to the external oblique fibers. It becomes aponeurotic medially contributing to both anterior and posterior rectus sheath before inserting into the linea alba and ribs 10–12. It is innervated by the lower intercostal nerves and the first lumbar spinal nerve. There are multiple source vessels feeding the muscle, including the deep circumflex iliac artery, deep inferior epigastric artery and lower intercostal vessels.
The transversus abdominis is the deepest of the three lateral muscles and runs transversely towards the midline from the iliac crest and thoracolumbar fascia. The transversalis fascia is a continuous fascial sheet that lies on the deep surface of transversus abdominis.
Blood Supply
The blood supply of the abdominal wall is robust, with multiple independent sources and extensive collateralization. Huger considered the skin and subcutaneous components of the abdominal wall in terms of three zones of perfusion, as illustrated in Fig. 40.2 .
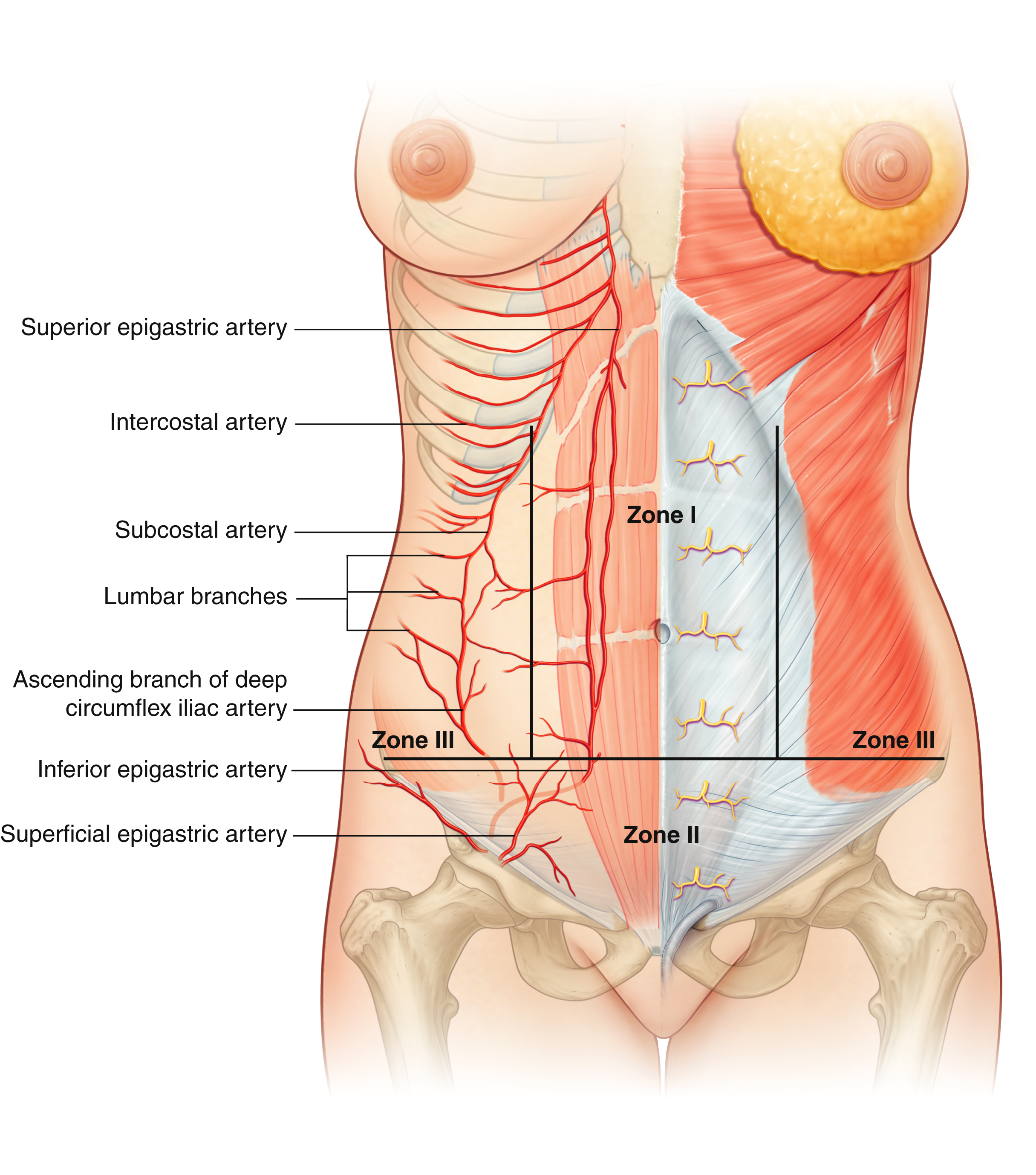
Zone I encompasses the central abdominal tissue overlying the paired rectus abdominis muscles. It is bound by the xyphoid process, pubic symphysis, and lateral edges of the rectus sheath. The skin and subcutaneous tissue of Zone I is perfused by musculocutaneous perforators from the deep inferior epigastric (branch of external iliac artery) and to a lesser extent the superior epigastric arteries (branch of internal mammary artery). The perforators are predominantly arranged in two rows – medial and lateral. The highest concentration of perforators is within 3 cm of the umbilicus. The medial row perforators provide greater perfusion across the midline than their lateral counterparts.
Zone II refers to the lower abdominal wall, below the line of the anterior superior iliac spines. Zone II is perfused by three separate branches of the common femoral artery – the superficial circumflex femoral artery, the superficial inferior epigastric artery, and the external pudendal artery. Zone III refers to the flank and abdominal wall lateral to the rectus muscles. It is supplied by the lower intercostal arteries and first lumbar artery – a robust blood supply that can perfuse the entire abdominal wall after division of Huger’s Zone 1 and II as demonstrated after an abdominoplasty.
Innervation
The motor and sensory innervation of the anterior abdominal wall is derived from the anterior rami of the 7th to 12th intercostals and first lumbar root. The intercostal neurovascular abdominal bundles pierce the transversus abdominis and travel in a plane deep to internal oblique. Preservation of the motor supply is essential for maintaining the integrity of the abdominal wall to prevent hernia formation or bulging.
Fascia
A detailed understanding of the fascia of the abdominal wall muscles is essential in abdominal wall surgery ( Fig. 40.3 ). The rectus sheath is formed laterally at the linea semilunaris by the condensation of the aponeuroses of external oblique, internal oblique, and transversus abdominis. The anterior rectus abdominis is covered by sheath throughout its course. Posteriorly, the rectus is covered by sheath only above the arcuate line, a transverse line at the level of the anterior superior iliac spine. Below the arcuate line, only transversalis fascia and parietal peritoneum lie deep to the rectus.
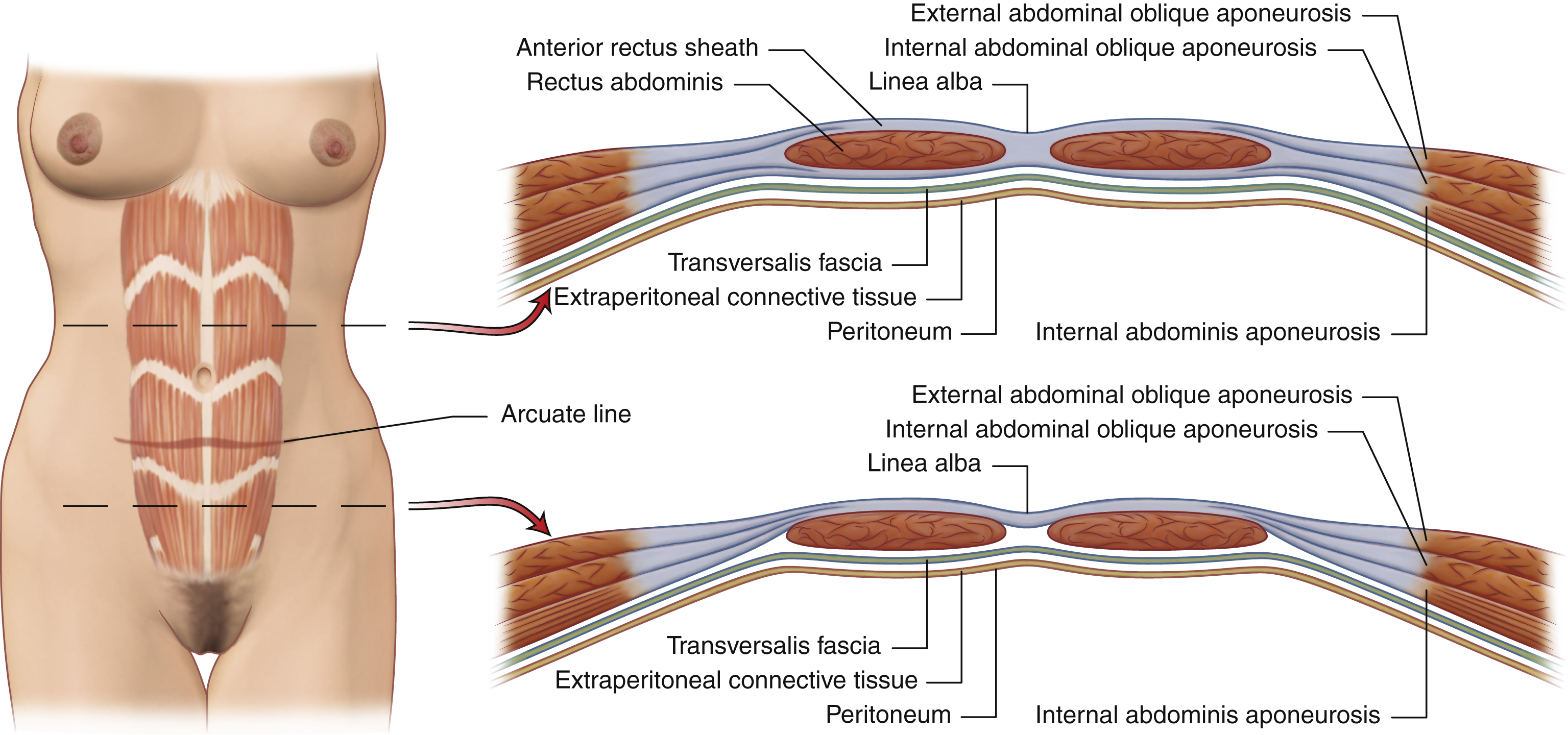
Above the arcuate line, the anterior rectus sheath is composed of external oblique and the superficial leaf of the internal oblique fascia, whereas the posterior rectus sheath is formed by the condensation of deep leaf of internal oblique and transversus abdominis fascia. Below the arcuate line, the transversus abdominis fascia and deep leaf of the internal oblique fascia also contribute to the anterior rectus sheath, whereas the posterior rectus sheath is devoid of muscular aponeurosis. The rectus abdominis muscle is tightly adherent to the sheath at the tendinous intersections.
Etiology and Classification of Abdominal Wall Hernia Defects
Abdominal wall hernias can be congenital or acquired. Congenital hernias include umbilical hernias, periumbilical hernias, Spigelian hernias (between rectus and linea semilunaris), lumbar hernais, and epigastric hernias. Gastroschisis and omphalocele represent two severe types of congenital abdominal wall defects that are commonly associated with other developmental anomalies. An omphalocele is a large defect where the abdominal contents protrude through the umbilicus and are maintained in a peritoneal sac. A gastroschisis is a smaller defect where the abdominal contents protrude freely without coverage from the peritoneum. Both conditions occur infrequently (1 in 5000 births) but are often detected antenatally by ultrasound.
Most acquired hernias are iatrogenic and arise in a previous surgical incision. Other acquired causes include infection, trauma, and tumors. There are a number of known risk factors for incisional hernias. Systemic factors include poor intrinsic wound healing, obesity, older age, comorbidities, and malignancy. Local factors include suboptimal wound closure, infection, ischemia, prior radiation, increased intraabdominal pressure and prolonged ventilation. Initial surgical technique plays an important role in the prevention of hernias with evidence suggesting that low dehiscence rates are best achieved by taking wide tissue bites (>1 cm from the fascia margin) with short stitch intervals (1 cm) and avoiding strangulating tension on the suture. However, even in the absence of identified risk factors, incisional hernias develop in 11% of primary laparotomies and may be as high as 33% in obese patients. Recurrent incisional hernias are especially challenging and the incidence of recurrent herniation after repair increases with each subsequent hernia repair operation.
The European Hernia Society has proposed a classification system of abdominal hernias to deliver conformity and allow ease of comparison in published clinical studies. This classification system distinguishes between primary and incisional ventral hernias. For primary ventral hernias, the two parameters are location and size (hernia diameter), as illustrated in Table 40.1 . The location is subdivided into midline defects (epigastric, umbilical) and lateral defects (Spigelian, lumbar). For incisional hernias, there are additional criteria, including whether the hernia is recurrent or not.
| Location | Diameter | |||
|---|---|---|---|---|
| Small <2 cm | Medium ≥2–4 cm | Large ≥4 cm | ||
| Midline | Epigastric | |||
| Umbilical | ||||
| Lateral | Spigelian | |||
| Lumbar | ||||
Assessment of the Abdominal Wound
Clinical
Initial assessment must start with an appropriately directed clinical history and physical examination. Local and systemic risk factors for impaired wound healing should be identified. A clear history and timeline of the wound must be elucidated to determine whether it is acute or chronic in nature. Any prior surgical interventions and their effects should be noted.
The examination should evaluate both the wound itself and the quality of the surrounding tissues. The abdomen should be inspected for scars, fistulas, sinuses, radiation injury or evidence active infection. The wound should be characterized in detail and include evaluation and documentation of the components that are absent. In chronic wounds the potential for malignant change should be considered and ruled-out with tissue biopsy where indicated.
Imaging
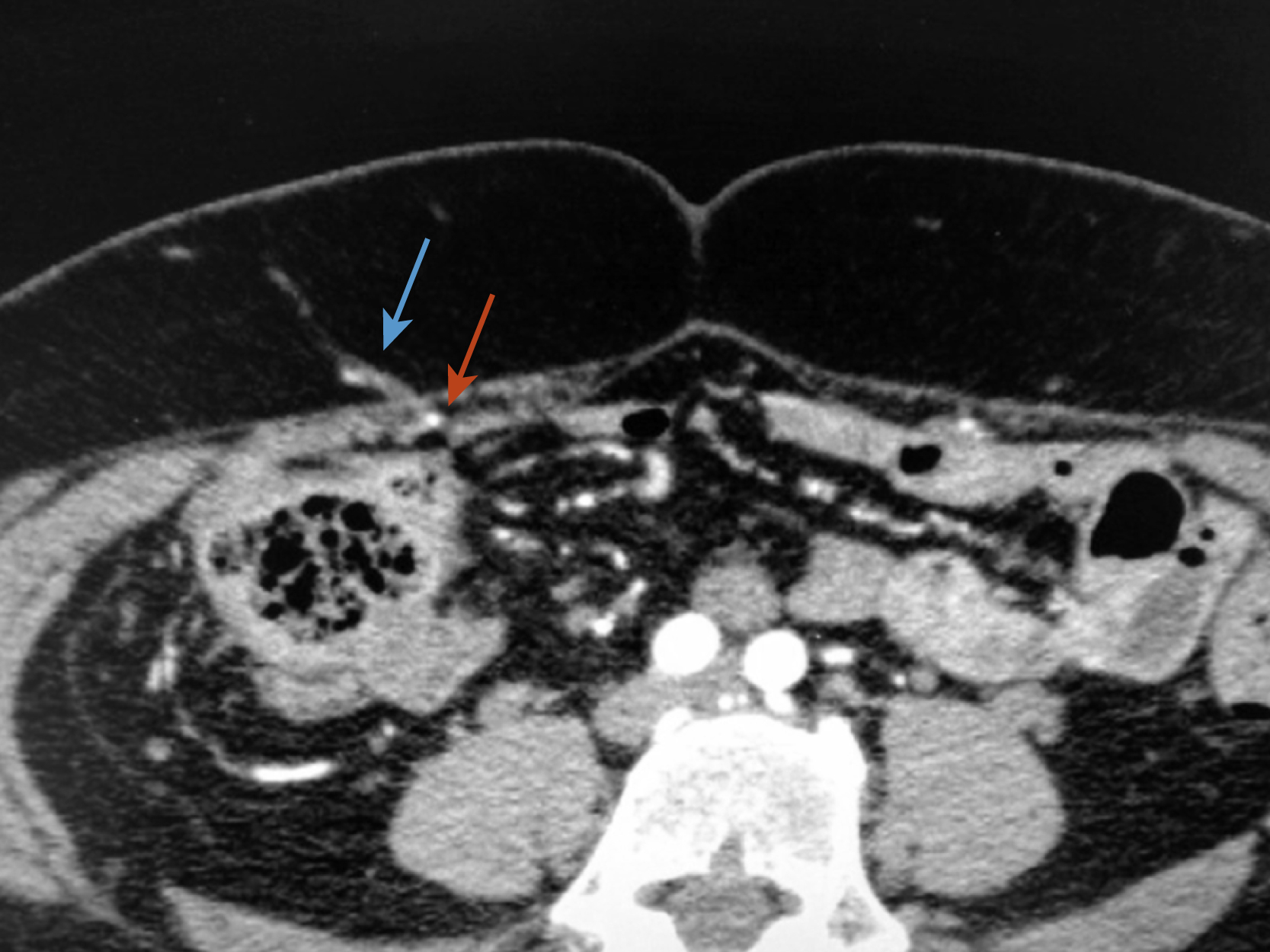
Preoperative imaging is very helpful in defining the pathology and in preoperative planning. Both abdominal computed tomography (CT) and magnetic resonance imaging (MRI) can successfully identify the extent of the abdominal wall defect and the integrity of adjacent musculofascial structures. However, CT angiography (i.e., with intravenous contrast) is superior at delineating the vascular anatomy, especially when planning pedicled flaps or free tissue transfer ( Fig. 40.4 ). An abdominal and pelvic CT scan with oral contrast enables detailed understanding of the wound and intraabdominal contents.
Goals of Reconstruction
The goal of abdominal wall reconstruction is twofold. The first goal is to restore function and integrity of the musculofascial abdominal wall to prevent visceral exposure and provide dynamic muscle support. The second is to provide skin resurfacing.
Management of Abdominal Wounds
Acute Wounds
Acute abdominal wounds may be the result of dehiscence post laparotomy, infection, trauma or neoplasms.
Most acute abdominal wounds following laparotomy can be managed with local wound care, including bedside debridement of necrotic tissue and daily dressing changes or application of negative-pressure wound therapy (NPWT). In select cases, secondary closure following wound excision may be feasible and has been shown to be superior to healing by secondary intention. Fascial dehiscence post laparotomy may occur in the acute setting without a skin defect. The typical history involves persistent drainage of serous fluid through the wound after 1 week. Fluid cultures are often positive. Fascial reclosure has a high chance of failure. Ideally, tension-free fascial closure is achieved and this may require use of a biologic or synthetic mesh with skin closure or NPWT.
In the acute trauma setting an abdominal wound may be left open because visceral edema makes it impossible to close, to facilitate further exploratory surgery or to prevent intraabdominal hypertension and compartment syndrome. In most cases NPWT is used as a temporizing measure. Although this approach may have benefits in selected cases, it may be associated with high rates of complications such as entero-atmospheric fistula or frozen abdomen. Early definitive closure should be performed as soon as possible, preferably within 7 days of the index surgery. In cases where this cannot be achieved, a “planned ventral hernia” procedure may be performed where the skin is closed or skin-grafted but the fascial defect is not addressed in the acute setting. Skin graft excision and formal hernia repair can be performed at a later date.
Chronic Wounds
Chronic uncomplicated wounds without fistula can be managed in a number of ways. Small wounds may be managed with simple dressings and closed directly at the time of hernia repair. Moderate to large wounds are probably best skin-grafted initially to allow a temporizing wound closure. The skin graft can be excised at the time of definitive hernia repair. In cases where there is exposed mesh, the mesh should be removed if grossly contaminated. However, exposed mesh can be readily salvaged with application of NPWT and subsequent split-thickness skin grafting once sufficient granulation tissue has developed.
Small sinuses with retained foreign material but no fistula (e.g., stitch abscess) should be managed by removal of the foreign body. Healing by secondary intention usually follows.
The presence of an enterocutaneous fistula, defined as an epithelial-lined connection between the skin and internal organ, poses an immense clinical challenge. Treatment must be individualized and despite involvement of experienced multidisciplinary teams, outcomes are often poor with a high morbidity, mortality and psychosocial dysfunction.
Prevention is key. Enterocutaneous fistulas most commonly develop in patients who have suffered a postoperative anastomotic leak. In wounds with exposed abdominal viscera, wet-to-dry dressings should be avoided as they can cause mechanical debridement of friable bowel mucosa and subsequent fistula development. Instead, the bowel should be covered with omentum where possible or absorbable mesh if omentum is unavailable.
Once a fistula is established, initial therapy should be directed at systemic measures to facilitate wound healing and maintain bowel function. These include aggressive treatment of sepsis, nutritional supplementation, and attention to fluid and electrolyte disturbances. Patients have high caloric and metabolic needs. In addition, vitamins and trace elements such as copper and zinc can be rapidly depleted through fistula-related losses. Total parenteral nutrition (TPN) can reduce enteric losses by as much as 50%, although enteral may also be used to provide trickle feeds. Reduction of fistula output may increase the chance of spontaneous closure.
Once systemic factors have been addressed adequately, a surgical closure should be carefully planned. Imaging is critical to determine the anatomy of the fistula. This may include CT, MRI, fistulograms, and small bowel imaging. In the early stages, the wound may be skin-grafted to convert the fistula into an ostomy, which can be managed more easily with a pouch system. Other techniques have been used to divert the fistula effluent from the wound bed in the early stages, such as NPWT and bridging dressings, which isolate the fistula. In either case, the periwound skin should be adequately protected with topical barrier ointments (containing zinc oxide, petrolatum, dimethicone or a combination) to prevent chemical denudation. A pouch system is used to collect the effluent and in addition may suppress odour.
The fistula can then be allowed to close spontaneously or be closed secondarily. Secondary fistula closure is best achieved by bowel resection and reanastomosis with abdominal wall reconstruction using acellular dermal matrices or components separation.
Management of Fascial Defects
Mesh reinforcement has been widely reported to improve outcomes following ventral hernia repair, with reduced recurrence rates compared to primary fascial closure. , Although an increasing number of synthetic and biological products are now available, none meets all the requirements of an ideal mesh and so the relevant risks and benefits must be considered in each individual clinical case. The ideal mesh would not adhere to the underlying bowel, would allow tissue ingrowth without shrinkage, prevent recurrence and fistula formation, and would not cause additional pain. The material should be inert, non-carcinogenic, sterile, resistant to infection and capable of withstanding significant changes in intraabdominal pressure that range from 1.55 mmHg in patients lying supine up to 150 mmHg maximum peak pressure in coughing patients.
Synthetic Mesh
Among the commonest meshes are permanent synthetic meshes that can be made from a variety of polymers, including polypropylene, polyester, and polytetrafluoroethylene (PTFE). Polypropylene is the most commonly used mesh due to its proven efficacy and relatively low cost. Individual filaments are knit together to form a flat sheet and the density of the filaments and the intervening pore size influence the clinical behavior of the material. Lighter weight meshes contain a lower density of polypropylene and are believed to be more favorable as they are associated with less foreign body response, superior compliance, increased tissue ingrowth, and less shrinkage of the material. The increased pore size is also believed to be beneficial in cases where bacterial contamination occurs, with salvage rates of up to 100% reported. Conversely, materials such as PTFE have very small pores, in the order of microns, that prevent macrophage penetration and make salvage very difficult in cases of infection. However, decreased pore size is associated with reduced adhesion formation and may therefore be preferable in some cases where the mesh is placed in the peritoneum. Meshes that combine PTFE and other non-adherent materials with other polymers such as polypropylene have been developed for use when mesh is in contact with bowel.
A range of resorbable meshes are also available and commonly comprise a formulation of polyglycolic acid (PGA), polylactic acid (PLA), trimethyl carbonate (TMC), or poly-4-hydroxybuturate (P4HB). These components are frequently combined to exploit their respective properties. These meshes aim to support the abdominal wall repair temporarily while wound healing occurs and then degrade completely leaving no permanent foreign material. Composite meshes have also been developed that combine permanent and resorbable polymers. These products offer some long-term tensile strength while reducing the amount of permanent foreign materials and its associated complications.
Biologic Mesh
A wide range of biologic meshes is now available, originating from different species (human, porcine, bovine) and anatomical locations (skin, intestine, pericardium). Tissues are processed and sterilized using a variety of techniques that remove the cellular component while preserving the extracellular matrix. The method of decellularization and sterilization may affect the efficacy of the mesh as any injury to the material may result in increased host inflammatory response and resorption. The mesh may also be crosslinked to varying degrees to strengthen and stabilize the collagen network but excessive crosslinking will impede recellularization and neovascularization.
Biologic meshes are far more expensive than synthetic products but may have advantages in certain clinical scenarios as they have the potential for revascularization and superior integration. They are more resistant to infection and can therefore be used in contaminated fields. In addition, biologic meshes can be placed directly on the abdominal viscera as they have a low rate of adhesion formation.
Although biologic meshes have a number of theoretical advantages they are not without associated complications. Among the most significant concerns is hernia recurrence, with rates as high as 80% frequently reported. Although the source and composition of the mesh is likely to influence recurrence rates there is a lack of well-designed studies that directly compare products. Some studies suggest that porcine products may have lower recurrence rates. The method of mesh inset is also important, with interpositional placement associated with higher recurrence rates. Repairs are more successful in cases where fascial approximation can be achieved.
Other complications included infection, seroma, and skin necrosis. Although relatively common, infection rarely leads to matrix extrusion and can often be managed with targeted antibiotics as early capillary ingrowth allows effective delivery of antimicrobial agents and innate host defenses. Even when implanted in a contaminated field, infection has been demonstrated to be as low as 16%. Seromas are common but rarely affect outcome. Strategies to reduce seroma formation include avoiding wide undermining of abdominal skin over fascia, preservation of suprafascial fat, use of quilting sutures, and abdominal binders.
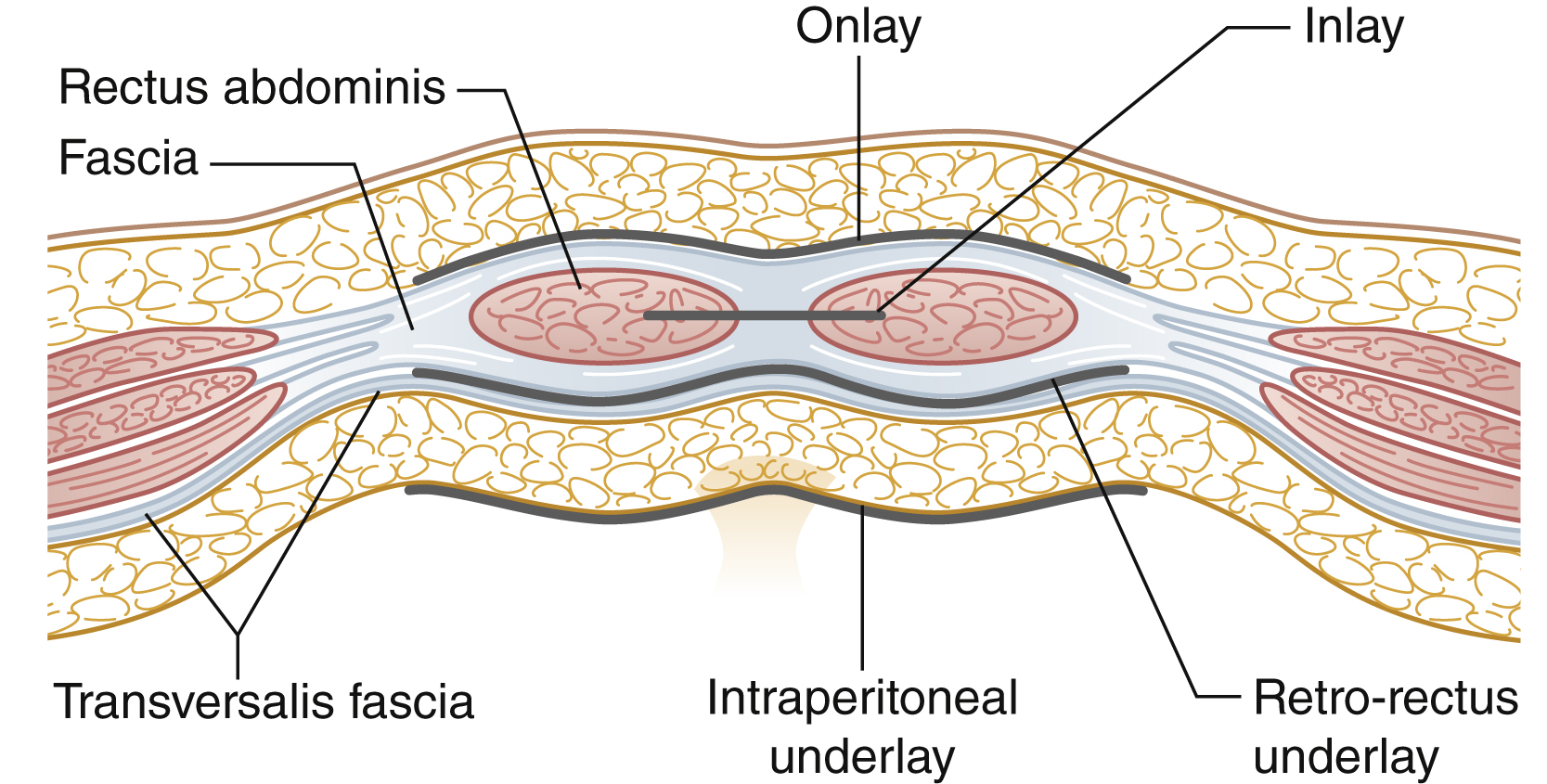
Surgical Techniques for Mesh Placement
The general principles of a successful mesh repair technique involve wide coverage with adequate overlap and secure suture fixation to the fascia. The mesh may be inserted above the fascia (onlay or overlay), underneath the fascia (inlay) or deep to the rectus (underlay, Fig. 40.5 ).