A Meta-Analysis of Human Acellular Dermis and Submuscular Tissue Expander Breast Reconstruction
John Y. S. Kim, M.D.
Armando A. Davila, B.S.
Scott Persing, B.A., M.P.H.
Caitlin M. Connor, B.A.
Borko Jovanovic, Ph.D.
Seema A. Khan, M.D.
Neil Fine, M.D.
Vinay Rawlani, M.D.
Chicago and Maywood, Ill.
From the Division of Plastic and Reconstructive Surgery, the Department of Preventative Medicine, and the Lynn Sage Breast Center, Northwestern University, Feinberg School of Medicine, and Loyola University Chicago, Stritch School of Medicine.
Received for publication March 21, 2011; accepted June 16, 2011.
Copyright © 2011 by the American Society of Plastic Surgeons
DOI: 10.1097/PRS.0b013e3182361fd6
Disclosure: Dr. Kim is a consultant for and receives research funding from Mentor and the Musculoskeletal Transplant Foundation. Dr. Fine receives research funding from Allergan, Inc. The remaining authors have no financial relationships to disclose.
Background: Human acellular dermal matrix has become an increasingly used adjunct to traditional submuscular tissue expander/implant breast reconstruction, but there is no strong consensus regarding complication outcomes. This study stratified outcomes based on a meta-analysis of complications.
Methods: A query of the MEDLINE database for articles on human acellular dermal matrix and submuscular tissue expander breast reconstruction yielded 901 citations. Two levels of screening identified 48 relevant studies. The DerSimonian and Laird random-effects model was used to perform the meta-analysis. Risk ratios and pooled complication rates were calculated for each outcome of interest.
Results: Nineteen studies reporting human acellular dermal matrix (n = 2037) and 35 reporting submuscular outcomes (n = 12,847) were used to estimate complication rates. Rates were generally higher in acellular dermis patients: total complications, 15.4 versus 14.0 percent; seroma, 4.8 versus 3.5 percent; infection, 5.3 versus 4.7 percent; and flap necrosis, 6.9 versus 4.9 percent. Six studies reporting both acellular dermis and submuscular outcomes were used to estimate relative risks. There was an increased risk of total complications (relative risk, 2.05; 95 percent CI, 1.55 to 2.70), seroma (relative risk, 2.73; 95 percent CI, 1.67 to 4.46), infection (relative risk, 2.47; 95 percent CI, 1.71 to 3.57), and reconstructive failure (relative risk, 2.80; 95 percent CI, 1.76 to 4.45) in acellular dermis patients.
Conclusions: The meta-analysis suggests that the use of human acellular dermal matrix increases complication rates vis-à-vis submuscular expander/implant reconstruction. This must be weighed against its reported advantages in enhancing cosmesis and ameliorating contracture. (Plast. Reconstr. Surg. 129: 28, 2012.)
CLINICAL QUESTION/LEVEL OF EVIDENCE: Therapeutic, III.
In the United States, approximately 57,000 tissue expander/implant–based reconstructions are performed annually, representing approximately 65 percent of all breast reconstructions.1 Acellular dermis has been an increasingly popular adjunct to traditional expander reconstruction with putative benefits including improved inframammary control, decreased incidence of migration, greater intraoperative fill (with decreased concomitant expansion time and number of postoperative visits), improved cosmesis, and amelioration of contracture.2–8 The basic technique involves releasing the pectoralis muscle along its inferior border and using the acellular dermis to reconstruct the ensuing lower pole defect.
The technique of using human acellular dermal matrix for soft-tissue reconstruction has existed since the early 1990s, and its use has been described in a myriad of clinical contexts from burn treatment and neourethra reconstruction to ameliorating contracture in hand and breast surgery.2,4,9–11 However, universal acceptance in these varying clinical indications has been tempered by concerns about surgical complications including seromas, infections, and loss of mechanical integrity.12–14 The complication profile of human acellular dermal matrix in breast reconstruction ranges widely throughout the 6 years of published reports (range, 3.2 to 48.7 percent), making it difficult to develop generalized outcomes from study to study.2,3,5,7,12,15–18 The central unknown remains regarding how outcomes of human acellular dermal matrix–assisted breast reconstruction compare with outcomes of traditional submuscular breast reconstruction. Our study attempts to collate recent studies in both treatment arms and determine general complication profiles and patterns by means of standard meta-analysis methodology.
Patients and Methods
Search Methods
A literature search was performed using PubMed to query the MEDLINE database (January 1, 2000, to February 6, 2011). Search terms included “tissue expander,” “implant,” “acellular dermis,” “acellular dermal matrix,” “AlloDerm” or “acellular matrix,” and “breast reconstruction.” Additional search methods included a manual review of reference lists of relevant studies.
Selection Criteria
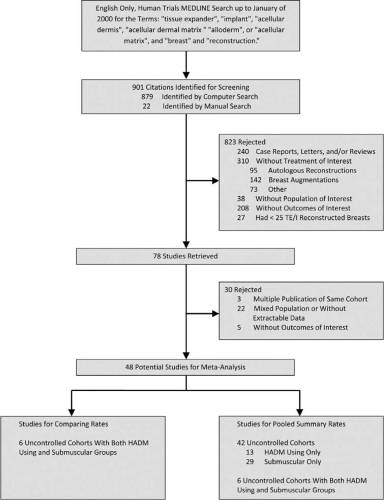
Selection criteria were defined a priori. Eligible studies were limited to English-only and had to examine breast reconstruction in human patients. Study selection underwent two levels of review by two independent researchers (Fig. 1). Each study was required to clearly indicate the number of reconstructions performed. Studies reporting fewer than 25 tissue expander/implant reconstructions in at least one cohort were excluded. Titles and abstracts were screened for the following exclusion criteria: publications of brief communications, correspondence, discussions, letters, conference/lecture manuscripts, case reports, and reviews; publications containing only abstracts; novel modifications of surgical technique; outcomes related to breast augmentation; outcomes related solely to autogenous reconstruction; and outcomes about only a specific high-risk population.
Full articles were then retrieved for all studies that met the first level of criteria. Studies needed to report or provide data to calculate a total complication rate and report or provide data to calculate at least one of the following postoperative complications: seroma, hematoma, infection, and flap necrosis. In any studies that reported similar or overlapping cohorts, the publication with the greatest number of reconstructions was included.
Data Collection and Analysis
Data collection and analysis was performed following the guidelines set forth by the Cochrane Handbook for Systematic Reviews of Interventions and the “Meta-Analysis of Observational Studies in Epidemiology.”19,20 Two independent reviewers extracted data from all selected studies by using a standardized data abstraction form. This electronic data form included the lead author, publication year, type of reconstructive procedure, number of patients, number of reconstructions, number of unilateral and bilateral reconstructions, average patient age and body mass index, percentage of smokers and diabetic patients, percentage of patients who received radiation therapy before surgery, percentage of patients who received postoperative radiation therapy, percentage of patients who received chemotherapy, average tissue expander intraoperative fill, and average follow-up. Complication data included number of reconstructions with seromas, hematomas, infection, flap necrosis, and explantation.
All rates used in the analysis were based on the number of reconstructions in each study. Two levels of analysis were performed. Cumulative pooled estimates were calculated from the standard error of complication rates based on the binomial distribution.21 In instances where a study specified a zero occurrence rate of an outcome, the standard error was estimated using a constant continuity correction.22 If a study did not specifically report an outcome, no correction was used, and the study was not included in the analysis of that outcome. Total complications for both analyses were retrieved for only defined postoperative complications (i.e., seroma, hematoma,
infection/cellulitis, and flap necrosis). Relative risks were calculated from studies that examined complications in both human acellular dermal matrix and submuscular cohorts specifically. For both analyses, the DerSimonian and Laird random-effects method was used based on interstudy heterogeneity.23,24 Heterogeneity was assessed using the Q statistic and the I2 statistic.25 Small Q statistic p values are indicative of statistically significant heterogeneity, whereas I2 percentages indicate the amount of heterogeneity between studies. For any heterogeneity of statistical significance, sources of heterogeneity were explored by means of an exclusion sensitivity analysis. Funnel plots, Egger’s regression test, and Begg’s rank correlation test were used to assess publication bias.26,27 Statistical analysis was performed using MIX 2.0, Professional Software for Meta-analysis plug-in for Microsoft Excel, version 2.0.1.2 (BiostatXL, Sunnyvale, Calif.), and corroborated with RevMan5.0 (Cochrane Collaboration Information Management System, Oxford, United Kingdom).28,29
infection/cellulitis, and flap necrosis). Relative risks were calculated from studies that examined complications in both human acellular dermal matrix and submuscular cohorts specifically. For both analyses, the DerSimonian and Laird random-effects method was used based on interstudy heterogeneity.23,24 Heterogeneity was assessed using the Q statistic and the I2 statistic.25 Small Q statistic p values are indicative of statistically significant heterogeneity, whereas I2 percentages indicate the amount of heterogeneity between studies. For any heterogeneity of statistical significance, sources of heterogeneity were explored by means of an exclusion sensitivity analysis. Funnel plots, Egger’s regression test, and Begg’s rank correlation test were used to assess publication bias.26,27 Statistical analysis was performed using MIX 2.0, Professional Software for Meta-analysis plug-in for Microsoft Excel, version 2.0.1.2 (BiostatXL, Sunnyvale, Calif.), and corroborated with RevMan5.0 (Cochrane Collaboration Information Management System, Oxford, United Kingdom).28,29
Results
Study Characteristics
An English-language PubMed search of MEDLINE identified 879 articles that were eligible for screening. An additional 22 articles were identified through a manual bibliography search, resulting in a total of 901 citations. Of these, 823 were rejected after a review of titles and abstracts (Fig. 1). After full-text review of the remaining 78 articles, 30 were rejected. All 48 articles that were included were uncontrolled cohort studies. These 48 studies were used to develop pooled summary complication rates. Thirteen studies had information only on human acellular dermal matrix–based reconstructions.2,3,5–7,15,18,30–35 Twenty-nine studies had information only on submuscular-based reconstructions.36–64 Six studies reported complications for both human acellular dermal matrix and submuscular techniques.12,13,16,17,65,66 In total, 2037 human acellular dermal matrix reconstructions and 12,847 submuscular reconstructions were included in the meta-analysis (Tables 1 and 2). Both cohorts had similar population demographics (Table 3). Human acellular dermal matrix reconstructions trended toward a higher average intraoperative fill (264.9 ml versus 187.1 ml; p = 0.10) when compared with submuscular reconstructions. The six studies used to calculate relative risks represented 877 human acellular dermal matrix reconstructions and 3464 submuscular reconstructions (Table 4).
Pooled Complication Rates
There was an increased rate of total complications, 15.4 percent (95 percent confidence interval, 9.3 to 21.4 percent) versus 14.0 percent (95 percent confidence interval, 11.7 to 16.3 percent); seroma, 4.8 percent (95 percent confidence interval, 2.8 to 6.9 percent) versus 3.5 percent (95 percent confidence interval, 2.6 to 4.4 percent); infection, 5.3 percent (95 percent confidence interval, 3.1 to 7.4 percent) versus 4.7 percent (95 percent confidence interval, 3.8 to 5.7 percent); and flap necrosis, 6.9 percent (95 percent confidence interval, 3.6 to 10.2 percent) versus 4.9 percent (95 percent confidence interval, 3.7 to 6.2 percent) in human acellular dermal matrix when compared with submuscular reconstructions (Fig. 2 and Table 5). However, the rate of hematoma was greater in the submuscular cohort (1.5 percent; 95 percent confidence interval, 1.0 to 2.0 percent) than in the human acellular dermal matrix cohort (1.0 percent; 95 percent confidence interval, 0.5 to 1.5 percent), and the rate of reconstructive failure was very similar in both cohorts, 3.8 percent (95 percent confidence interval, 2.3 to 5.4 percent) versus 3.8 percent (95 percent confidence interval, 2.9 to 4.7 percent).
Meta-Analysis of Comparative Studies
There was an increase in the risk of total complications (relative risk, 2.05; 95 percent confidence interval, 1.55 to 2.70), seroma (relative risk, 2.73; 95 percent confidence interval, 1.67 to 4.46), infection (relative risk, 2.47; 95 percent confidence interval, 1.71 to 3.57), and reconstructive failure (relative risk, 2.80; 95 percent confidence interval, 1.76 to 4.45) in the human acellular dermal matrix cohort (Figs. 3 through 5 and Table 6). There was a trend toward increased risk of hematoma (relative risk, 2.06; 95 percent confidence interval, 0.86 to 4.95) and flap necrosis (relative risk, 1.56; 95 percent confidence interval, 0.85 to 2.85) in the human acellular dermal matrix cohort, but the results were not statistically significant.
Study Heterogeneity
The majority of pooled complication analyses showed significant heterogeneity (Table 5). Heterogeneity values of the 19 studies in the human acellular dermal matrix cohort (Q statistic, p < 0.01; I2 = 95.4 percent) were similar to those of the submuscular cohort (Q statistic, p < 0.01; I2 = 94.0 percent). Relative risk analyses were generally more homogeneous, with only total complications and flap necrosis reporting statistically significant heterogeneity (Q statistic, p = 0.08; I2 = 49.0 percent; and p = 0.02; I2 = 68.3 percent). In analyses of seroma (Q statistic, p = 0.22; I2 = 29.3 percent), infection (Q statistic, p = 0.72; I2 = 0.0 percent), hematoma (Q statistic, p = 0.98; I2 = 0.0 percent), and reconstructive failure (Q statistic, p = 0.53; I2 = 0.0 percent), heterogeneity was not significant (Table 6).
Publication Bias
The funnel plot generated to test for publication bias in the relative risk analysis of total complications showed relative symmetry (Fig. 6). Egger’s weighted regression and Begg’s rank correlation test showed minimal evidence of bias (bias = 0.30; 95 percent confidence interval, -5.96 to 6.57; p = 0.90, and Kendall’s tau-b = -0.06; p = 0.85).
Discussion
The advent of any new technology—and specifically, its integration with a new technique—generates uncertainty about outcomes and questions about benefits vis-à-vis risks. With human acellular dermal matrix–assisted tissue expander breast reconstruction, the potential for improvement in cosmetic and reconstructive outcomes has been promulgated with little consensus on cumulative risks. The variance in total complication profiles reported in the literature (range, 3.2 to 48.7 percent)
suggests the need for pooled analysis.2,3,5,7,12,15–18 The aim of our meta-analysis was to focus on complication profiles and perform both a risk analysis and pooled summary estimates using the most recent literature. We did not specifically address the underlying rationale for using human acellular dermal matrix or validate the potential advantages of this technique, as these topics have been discussed elsewhere in the literature.2,4,5,6,15
suggests the need for pooled analysis.2,3,5,7,12,15–18 The aim of our meta-analysis was to focus on complication profiles and perform both a risk analysis and pooled summary estimates using the most recent literature. We did not specifically address the underlying rationale for using human acellular dermal matrix or validate the potential advantages of this technique, as these topics have been discussed elsewhere in the literature.2,4,5,6,15
Table 1. Characteristics of Study Populations Reporting Human Acellular Dermal Matrix Reconstruction Outcomes | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree