The Oncologic Outcome and Immediate Surgical Complications of Lipofilling in Breast Cancer Patients: A Multicenter Study—Milan-Paris-Lyon Experience of 646 Lipofilling Procedures
Jean Yves Petit, M.D.
Visnu Lohsiriwat, M.D.
Krishna B. Clough, M.D.
Isabelle Sarfati, M.D.
Tarik Ihrai, M.D.
Mario Rietjens, M.D.
Paolo Veronesi, M.D.
Fabio Rossetto, B.S.
Anna Scevola, M.D.
Emmanuel Delay, M.D.
Milan, Italy; Paris and Lyon, France; and Bangkok, Thailand
From the Division of Plastic and Reconstructive Surgery, European Institute of Oncology; the Paris Breast Center; the Leon Berard Center; and the Department of Surgery, Siriraj Hospital, Mahidol University.
Received for publication October 21, 2010; accepted January 14, 2011.
Copyright ©2011 by the American Society of Plastic Surgeons
DOI: 10.1097/PRS.0b013e31821e713c
Disclosure: The authors have no conflict of interest to report. No outside funding was received.
Background: Lipofilling is now performed to improve the breast contour, after both breast-conserving surgery and breast reconstruction. However, injection of fat into a previous tumor site may create a new environment for cancer and adjacent cells. There is also no international agreement regarding lipofilling after breast cancer treatment.
Methods: The authors included three institutions specializing in both breast cancer treatment and breast reconstruction (European Institute of Oncology, Milan, Italy; Paris Breast Center, Paris, France; and Leon Berard Centre, Lyon, France) for a multicenter study. A collective chart review of all lipofilling procedures after breast cancer treatment was performed.
Results: From 2000 to 2010, the authors reviewed 646 lipofilling procedures from 513 patients. There were 370 mastectomy patients and 143 breast-conserving surgery patients. There were 405 patients (78.9 percent) with invasive carcinoma and 108 (21.1 percent) with carcinoma in situ. The average interval between oncologic surgical interventions and lipofilling was 39.7 months. Average follow-up after lipofilling was 19.2 months. The authors observed a complication rate of 2.8 percent (liponecrosis, 2.0 percent). Twelve radiologic images appeared after lipofilling in 119 breast-conserving surgery cases (10.1 percent). The overall oncologic event rate was 5.6 percent (3.6 percent per year). The locoregional event rate was 2.4 percent (1.5 percent per year).
Conclusions: Lipofilling after breast cancer treatment leads to a low complication rate and does not affect radiologic follow-up after breast-conserving surgery. A prospective clinical registry including high-volume multicenter data with a long follow-up is warranted to demonstrate the oncologic safety. Until then, lipofilling should be performed in experienced hands, and a cautious oncologic follow-up protocol is advised. (Plast. Reconstr. Surg. 128: 341, 2011.)
CLINICAL QUESTION/LEVEL OF EVIDENCE: Therapeutic, II.
Breast reconstruction is included in the treatment plans for each individual breast cancer patient to improve quality of life.1–4 To obtain satisfactory results, surgeons focus on cosmetic issues and, even more importantly, on oncologic safety.5 Despite various
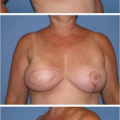
techniques of reconstruction with autologous tissues or prostheses, adipocyte tissue has been used more frequently in the recent era.6–9 The indications for lipofilling include micromastia, tuberous breasts, Poland syndrome, postlumpectomy deformity, postmastectomy deformity, sequelae of postradiotherapy, secondary reconstruction after flap or prosthesis reconstruction, and nipple reconstruction.10–13
techniques of reconstruction with autologous tissues or prostheses, adipocyte tissue has been used more frequently in the recent era.6–9 The indications for lipofilling include micromastia, tuberous breasts, Poland syndrome, postlumpectomy deformity, postmastectomy deformity, sequelae of postradiotherapy, secondary reconstruction after flap or prosthesis reconstruction, and nipple reconstruction.10–13
Recently, a number of new techniques of fat preparation have been described, with the ultimate goal of improving adipocyte purification and stem cell selection.14–16 However, many fundamental studies have suggested that adipocytes and preadipocytes and their products are involved in the tumor cell cycle through autocrine, paracrine, and exocrine/endocrine secretions.17,18 Such secretions delivered on tumor sites may play a role in tumorigenesis, tumor progression, and tumor recurrence or metastasis. In contrast, some of them could have an inhibitory effect on some particular stages of the tumor cycle. Breast cancer cells might still be present in the residual mammary parenchyma after conservative treatment or in the subcutaneous tissue after mastectomy. Therefore, based on these contradictory studies, a highly relevant clinical question is whether the lipofilling technique is a safe procedure after breast cancer treatment, especially in breast-conserving protocols.19–24
The review article by Lohsiriwat et al.25 on experimental studies found that adipocyte, preadipocyte, and progenitor cell secretions can stimulate angiogenesis and cell growth. They also emphasized that the “tumor-stroma interaction” can potentially induce cancer reappearance by “fueling” dormant breast cancer cells in the tumor bed. However, there is a lack of translational research that proves this concern clinically. No study on the effects of lipotransfer on human cancer breast cells in vivo is available. The direct and indirect effects of lipotransfer in breast cancer patients, highlighting pro and con–related issues, remain unclear. Today, no informed consent can be given to our patients stating that lipofilling does not stimulate fueling of dormant cancer cells or eventually induce new cancer cells.26
There is no international agreement for lipofilling after breast cancer treatment. Depending on country, the safety of lipofilling is still a controversial issue. In 2007, the French Society of Plastic Reconstructive and Aesthetic Surgery (Société Française de Chirurgie Plastique Reconstructrice et Esthétique) addressed the question of cancer safety for lipofilling technique in breast cancer patients.27 The Society sent a “recommendation to the French plastic surgeons to postpone the lipofilling in the breast with or without breast cancer history unless it is performed under prospective controlled protocol.” In 2009, the American Society of Plastic Surgeons Fat Graft Task Force concluded that no reliable studies can confirm definitely the oncologic safety of lipofilling in breast cancer patients.13 The Italian Society of Plastic Surgery in 2010 advised surgeons to perform lipofilling with caution and with a precise consent form but did not impose any restrictions regarding lipotransfer indications. Our purpose was to design a multicenter observational study on lipofilling after breast cancer with a significant number of cases to check the complication rate of the technique, the risk of modification of mammography and ultrasound, and the occurrence of a suspicious image, and to record oncologic follow-up.
Patients and Methods
We set up a multicenter study gathering information from three institutions with a large experience of breast cancer treatment and reconstruction (European Institute of Oncology, Milan, Italy; Paris Breast Center, Paris, France, and Leon Berard Centre, Lyon, France). A collective chart review of lipofilling procedures was performed for the registration of the cases.
We included all breast cancer patients with lipofilling after treatment of breast cancer. Patients could have undergone breast-conserving surgery or mastectomy, regardless of preoperative and postoperative treatment (i.e., radiotherapy, chemotherapy, or hormonal treatment). Both carcinoma in situ and invasive carcinoma were included. The exclusion criteria were lack of histology or lack of complete operative data, follow-up less than 6 months, and treatment for local or distant recurrence between the primary cancer treatment and the lipofilling procedure.
The patients’ data from each institution were collected and registered systematically on the same case record form. All patients’ preoperative clinical and radiologic data were registered. The techniques of lipofilling procedures were recorded. The missing data were completed by telephone call and/or appointment. Patient age, date and indication for oncologic surgery, radiotherapy, tumor histopathology and staging, indication for lipofilling, type of anesthesia, donor sites of fat harvesting, volume of preparation and injection, complications, and clinical and radiologic follow-up data before and after lipofilling were registered.
Surgical Technique
All three institutes performed lipofilling using the same technique. The procedure was performed under local or general anesthesia, depending on quantities of fat required and the patient’s clinical conditions. The
selected donor site was infiltrated with Klein’s solution, which consists of 1 cc of epinephrine diluted in 500 cc of 0.001% lactated Ringer’s solution; 50 cc of mepivacaine is added to the solution if the surgeon is operating using local anesthesia. For harvesting, we use a Coleman blunt-tip cannula attached to a 10-cc Luer-Lok syringe or a controlled pressure vacuum machine. A combination of slight negative pressure and the curetting action of the cannula through the tissues allows the fat harvesting. Then, we centrifuge the fat at 3000 rpm for 3 minutes until the serum and oily components are separated from the adipose tissue. The cellular component is transferred immediately to a 1-cc Luer-Lok syringe and prepared for injection.
selected donor site was infiltrated with Klein’s solution, which consists of 1 cc of epinephrine diluted in 500 cc of 0.001% lactated Ringer’s solution; 50 cc of mepivacaine is added to the solution if the surgeon is operating using local anesthesia. For harvesting, we use a Coleman blunt-tip cannula attached to a 10-cc Luer-Lok syringe or a controlled pressure vacuum machine. A combination of slight negative pressure and the curetting action of the cannula through the tissues allows the fat harvesting. Then, we centrifuge the fat at 3000 rpm for 3 minutes until the serum and oily components are separated from the adipose tissue. The cellular component is transferred immediately to a 1-cc Luer-Lok syringe and prepared for injection.
Results
From December of 2000 to March of 2010, we reviewed 646 lipofilling procedures from 513 patients. There were 370 mastectomy patients (476 lipofilling procedures) and 143 breast-conserving surgery patients (170 lipofilling procedures). The average amount of lipofilling performed per session was 107.3 cc (range, 5 to 400 cc). The average number of sessions required was 1.25 (range, one to six sessions). Three patients had lipofilling immediately at the time of total reconstruction with prostheses. Thirty-seven underwent lipofilling within 6 months, and the rest underwent lipofilling more than 6 months after primary oncologic surgery.
There were 240 T1 tumors, 131 T2 tumors, 31 T3 tumors, and three T4 tumors.28 There were 405 invasive carcinomas (78.9 percent) and 108 carcinomas in situ (21.1 percent) (ductal carcinoma in situ, n = 101; lobular carcinoma in situ, n = 7).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree