Introduction
Wound healing occurs via a common pathway of four interdependent phases: hemostasis, inflammation, proliferation, and remodeling ( Fig. 43.1 ). Although these phases are sequential, significant temporal and functional overlaps exist. When controlled, a closed wound is the predictable outcome. When dysregulated, however, wound healing is delayed. Understanding the stepwise process of wound healing is critical, both to patient care and to the development of future targeted therapies.
Phases of wound healing.
ECM , Extracellular matrix.
Hemostasis
Hemostasis is the first response to tissue injury. Defined by three distinct phases, it both addresses acute blood loss and creates a stable microenvironment for subsequent wound healing.
Vasoconstriction
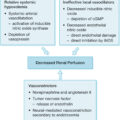
Vasoconstriction reduces local blood flow and is the first step in achieving hemostasis. Systemic vasoconstrictors such as epinephrine and norepinephrine are upregulated following trauma. Endothelial injury results in the release of local vasoconstrictors such as endothelin. Platelet activation and secretion of platelet-derived growth factor (PDGF) increase vascular smooth muscle contractility. Unfortunately, vasoconstriction alone is insufficient to produce durable hemostasis, as tissue hypoxia creates reflexive vasodilatation and restoration of blood flow. Primary and secondary hemostasis, then, are critical.
Primary hemostasis
In their native state, platelets are kept quiescent by the antithrombotic properties of vascular endothelium. Following injury, however, subendothelial matrix is exposed, activating integrins on the cell surface. These proteins increase adhesion to other platelets and to extracellular matrix (ECM), forming an early scaffold (plug) in the wound. Platelet-bound fibrin and fibronectin in the ECM attract inflammatory and mesenchymal cells. , Cytoskeletal rearrangement in the activated platelets causes contraction of the plug, obstructing vascular lumens and releasing granules of growth factors. , Recent interest in the utility of these growth factors for wound treatment has resulted in the development of products such as platelet-rich plasma, which has limited support in the literature.
Secondary hemostasis
The platelet plug serves as the scaffold upon which secondary hemostasis occurs. Intrinsic and extrinsic pathways of coagulation activate thrombin, cleave fibrinogen into fibrin, and cross-link the clot to create stable, durable hemostasis in the wound.
Inflammation
Early inflammation is characterized by the release of transcription-independent molecules that attract inflammatory cells to the wound bed. Hydrogen peroxide reduces bacterial burden, recruits neutrophils, and contributes to future angiogenesis. Stored arachidonic acid is released, which serves as a precursor for prostaglandins. Simultaneously, mast cells release stored histamine, which increases vascular permeability and stimulates prostaglandin. Prostaglandins work in concert with complement to cause vasodilation and increase vascular permeability. These and other similar molecules ultimately result in the migration of inflammatory cells to the site of tissue injury, adhesion to and diapedesis between vascular endothelial cells, and entry into the wound bed. This also permits fluid to enter the extravascular space, accounting for the insensible fluid loss seen in burn injury and trauma.
Neutrophils
Neutrophils are the predominant cell type in the wound in the first 24 hours. In their native state, they are not commonly found in the skin but are recruited from the bone marrow following injury. On activation, these cells release several products into the surrounding tissue. Matrix metalloprotease (MMP), collagenase, and elastase, in combination with other proteases, break down the ECM, permitting passage of cells and removal of debris. These enzymes also work in concert with the release of an oxidative burst and phagocytosis to combat early infection. Proinflammatory cytokines released by the neutrophils, such as tumor necrosis factor α (TNF-α) and interleukin 1 (IL-1), activate keratinocytes and fibroblasts, priming the early wound for subsequent phases of healing.
Macrophages
Macrophages are the predominant inflammatory cell in the wound 24 to 48 hours after injury following local activation and recruitment of precursors from the bone marrow. Once in the tissue, macrophages appear to exist as two distinct phenotypes: proinflammatory (M1) and antiinflammatory (M2). Like neutrophils, M1 macrophages express proinflammatory cytokines and MMPs to further degrade the surrounding ECM. They also clear neutrophils from the wound bed to control the early inflammatory response. As inflammation subsides, these cells transition to the M2 phenotype, promoting angiogenesis with the release of vascular endothelial growth factor (VEGF), stimulating fibroblasts via transforming growth factor β (TGF-β), and contributing directly to ECM formation by differentiating into fibrocytes.
Other cell types
Langerhans cells are dendritic cells found in the skin that bind antigen, migrate to regional lymph nodes, and activate the T-cell response. T lymphocytes both migrate to sites of tissue injury and exist in the skin as dendritic epidermal T cells. These cells secrete proinflammatory cytokines to enhance immune function and exhibit memory, persisting in the skin long after the resolution of an insult.
Proliferation and synthesis
Hemostasis and inflammation are typically complete by the third day after wounding. Growth factors secreted by these inflammatory cells recruit and stimulate cells responsible for ECM deposition, epithelialization, and angiogenesis.
Matrix synthesis
Fibroblasts
Fibroblasts are recruited to the wound bed by PDGF and TGF-β and adhere to fibronectin. , They then proliferate and begin synthesizing ECM components. Some fibroblasts undergo dedifferentiation into multipotent precursor cells, redifferentiating into terminal cell lineages in the healing wound based on the demands of the surrounding microenvironment. ,
The extracellular matrix
The ECM serves as a scaffold for wound healing, but also modulates cellular proliferation, signaling, and activity. It can be subdivided into three categories: fiber-forming structural molecules, non–fiber-forming structural molecules, and matricellular proteins. Fiber-forming molecules provide structure, whereas non–fiber-forming molecules contribute osmotic and electric potential to the ECM. Matricellular proteins do not have structural function but instead are involved in cell-ECM interaction.
Fiber-forming structural molecules in the ECM of the skin include collagen, fibronectin, fibrin, and elastin. Fibroblasts, under the regulation of TGF-β, produce 28 subtypes of collagen. , All subtypes consist of three polypeptide chains (α chains) in a triple-helix conformation. Fibrillar collagens (types I–III and V) form a scaffold for cells in the wound. Nonfibrillar collagens (types IV, VI–VIII, and XIV) create a reticular network connecting cells and other collagens to the basement membrane. Type VII collagen, in particular, is a critical component of cell anchoring during the reepithelialization phase of wound healing. Disruption of this subtype results in chronic wound formation, while topical application promotes wound closure. , Together, these collagen subtypes confer substantial strength and flexibility to the skin. The presence or absence of these subtypes, as well as the ratio between them, varies over the course of wound healing and can result in a wide spectrum of outcomes, from scarless wound healing to fibrosis. , Early wounds have a 2:1 ratio of type I to III collagen, gradually remodeled to the 4:1 ratio found in normal skin.
Fibronectin and fibrin are deposited into the early wound during the hemostasis phase. Soluble fibronectin circulating in the plasma is modified into an insoluble form in the ECM. Using multiple binding domains, fibronectin has a dynamic relationship with many of the components in wound healing. It is required for the migration of fibroblasts into the provisional platelet plug, as well as the future deposition of collagen type I. , Fibrin, bound by fibronectin, is first contracted by fibroblasts, then used as a substrate for provisional granulation tissue. It also modulates fibroblast response to TGF-β and mechanical stress, altering collagen production and ECM morphology. ,
Elastin, as a component of elastic fibers, is responsible for tissue elasticity and recoil. Elastic fibers have a half-life approximating the human lifespan, and production largely ceases in maturity. After injury, MMPs released by neutrophils liberate it from the surrounding wound bed for reorganization in the healing wound. This free elastin has been shown to promote fibroblast proliferation and subsequent collagen type I production. Production of new elastic fibers is limited, however, conferring mechanical limitations to the healed scar.
Glycosaminoglycans (GAGs) are linear polysaccharides of variable length that when bound to a protein become proteoglycans (PGs). Both are non–fiber-forming molecules in the ECM that fill the interstitial space and are negatively charged. Hyaluronic acid (HA) is the most common GAG in the skin and contributes to the maintenance of tissue hydration. In the wound-healing milieu, it binds both fibroblasts and TGF-β and stimulates fibroblast proliferation and ECM synthesis. , Dermatan and chondroitin sulfates are the most common GAGs attached to PGs and are in high concentrations in acute wounds. These molecules are required for CD44-mediated fibroblast binding and migration, and like HA bind growth factors that stimulate fibroblast proliferation and function. PGs function to maintain ECM hydration and organization by permitting entry of only certain molecules.
Epithelialization
Having created a stable scaffold in the wound, reepithelialization of the defect becomes possible. The epidermis is a complex structure formed from multiple layers of keratinocytes. These cells are joined together via desmosomes and are adherent to the underlying basement membrane through hemidesmosomes and focal adhesions. The most superficial layer, the stratum corneum, is impermeable to reduce fluid loss and sheds constantly. The basal layer, the stratum basale, is composed of keratinocytes with high-integrin expression to increase their adhesiveness to each other and to the basement membrane.
Following injury, keratinocytes at the leading edge of the wound express integrins and MMPs, dissolve adhesions to each other and the basement membrane, and begin migrating into the defect by binding to fibronectin in the ECM. These migrating cells are stimulated by epidermal growth factor (EGF) and cytokines like TNF-α, IL-1, and IL-6. They also stimulate continued ECM deposition and angiogenesis by producing cytokines and VEGF. , In addition to peripheral reepithelialization, stem cells in the interfollicular epidermis and stratum basale proliferate and differentiate into terminal skin-cell types. , After reconstitution of the epidermis, proliferation continues until native morphology is reestablished. When compared with uninjured skin, however, the neoepidermis has fewer basal cells and lacks rete pegs. Stem cells in the bulge of the hair follicle, when preserved in the wound, differentiate into pigment-producing melanocytes, which migrate into the epidermis to repigment the healed wound.
Angiogenesis
The newly synthesized tissues require new blood vessels to provide sufficient nutrition for homeostasis. Without angiogenesis, restoration of normal skin physiology would be impossible. In response to tissue hypoxia and growth factors such as VEGF, fibroblast growth factor (FGF), and EGF, vascular endothelial cells migrate into the wound from terminal capillaries. These cells release MMPs and collagenase, permitting entry into the perivascular space. Expression of the α v β 3 integrin allows them to bind the ECM and migrate through the wound. Fibrin serves as a critical promoter of endothelial migration, proliferation, and tubule formation. Early after wounding, it binds and enhances the effect of FGF-2, upregulating the expression of the α v β 3 integrin and localizing MMP-2 to the endothelial cells. Later, as VEGF is released by proliferating macrophages and keratinocytes, fibrin again binds and localizes the growth factor to create a positive feedback loop on vascular endothelium. VEGF, potentiated by local tissue hypoxia in the wound, creates the gradient along which endothelial cell differentiation occurs, forming the tips and stalks of new vessels.
Remodeling
Remodeling converts the reepithelialized but weak wound into a well-healed scar. This phase involves reorganization of the ECM, maturation of the vascular network, controlled cellular apoptosis, and wound contraction.
Much like the proliferative phase, fibroblasts are central to remodeling. These cells continue producing MMPs that degrade collagen type III in the wound bed while they shift production to the stronger collagen type I. They also secrete tissue inhibitors of metalloproteinases, diminishing MMP activity to prevent uncontrolled ECM degradation. In response to the changing mechanical microenvironment of a healing wound, some fibroblasts will migrate to the center and change their gene expression. These cells, termed myofibroblasts, bind to ECM proteins via integrins and pull perpendicular to the wound edge using organized actin microfilaments. This shift is stimulated by the presence of collagen type III in the provisional ECM and in response to shear stress. In addition to collagen type I synthesis, myofibroblast cells gradually shrink the diameter of the wound under the stimulation of TGF-β and PDGF. As collagen type III levels and shear stress diminish, fibroblasts and myofibroblasts undergo apoptosis to return ECM remodeling to normal homeostatic levels.
Early vascular networks created under the direction of VEGF are highly permeable with poor cell-cell adhesion. Angiopoietins (e.g., Ang-1) are produced by fibroblasts and myofibroblasts in the wound and are critical for vessel maturation. Immediately after injury, Ang-1 levels are downregulated so that capillaries may enter a more indeterminate state. As the wound matures and fibroblasts proliferate, however, Ang-1 levels increase, and endothelial cells create tight cell-cell junctions. PDGF attracts pericytes and smooth muscle cells to the new capillaries. The vascular endothelium then changes receptor expression to bind chemokines expressed in late-stage wound healing, resulting in the cessation of new tubule formation. As the wound heals and local hypoxia resolves, the antiapoptotic effects of VEGF are diminished, promoting cell-mediated apoptosis of residual disordered endothelium.
Remodeling gradually restores skin ductility and strength. By week 3, the healed wound has 15% strength when compared with normal skin. By week 6, 80% to 90% strength has been achieved. By 6 months, it has reached homeostasis at 90% strength and will not return to normal levels. The scar texture, thickness, and color will remain in a dynamic state for 12 to 18 months.
Growth factors and cytokines
Growth factors are proteins present in small quantities that have a profound impact on cell migration, function, and lifespan ( Table 43.1 ). They exert these effects through endocrine, paracrine, and autocrine signaling using cell surface receptors.
Table 43.1
Growth Factors in Wound Healing
| Growth Factor | Source | Target | Function |
|---|---|---|---|
| EGF | Platelets, macrophages | Keratinocytes | Epithelialization |
| FGF | Mast cells, endothelial cells, keratinocytes, fibroblasts | Fibroblasts, endothelial cells | ECM production, VEGF potentiation |
| PDGF | Platelets, macrophages, fibroblasts, keratinocytes, endothelial cells | Inflammatory cells, endothelial cells, vascular pericytes and smooth muscle cells, fibroblasts | Inflammation, granulation tissue formation, VEGF potentiation, vessel maturation, ECM remodeling |
| TGF-β | Platelets, macrophages, fibroblasts, keratinocytes | Inflammatory cells, fibroblasts, keratinocytes | Inflammation, granulation tissue formation, epithelialization, ECM production, wound contraction, ECM remodeling |
| VEGF | Platelets, neutrophils, macrophages, fibroblasts, keratinocytes, endothelial cells | Endothelial cells | Angiogenesis |
| CTGF | Fibroblasts | Fibroblasts | ECM production, remodeling |
| Cytokines | Neutrophils, macrophages | Keratinocytes, fibroblasts | Granulation tissue formation, epithelialization, FGF potentiation |
EGF, Endothelial growth factor; FGF, fibroblast growth factor; PDGF, platelet-derived growth factor; TGF, transforming growth factor; VEGF, vascular endothelial growth factor; CTGF, connective tissue growth factor; ECM, extracellular matrix.
Epidermal growth factor
EGF is a family of proteins that bind the EGF receptor on target cells. This receptor is present throughout the epidermis, though it is most prominent in the stratum basale. EGF is initially released by platelets and macrophages and later by fibroblasts, and results in keratinocyte proliferation and migration. It increases the speed of reepithelialization and tensile strength of the healed wound. , It has similar effects on angiogenesis.
Fibroblast growth factor
The FGF family includes over 23 individual growth factors, though FGF-2, FGF-7, and FGF-10 are most implicated in wound healing. FGFs are produced by mast cells, endothelial cells, keratinocytes, and fibroblasts. These growth factors must first bind to PG in the ECM for activation. FGF-2 promotes fibroblast migration and ECM production and keratinocyte migration. FGF-7 and FGF-10 promote keratinocyte and endothelial cell proliferation, potentiate the effects of VEGF, and are involved in antioxidant pathways.
Platelet-derived growth factor
The PDGF family consists of dimeric proteins that are released by platelets, macrophages, fibroblasts, keratinocytes, and vascular endothelial cells during each stage of wound healing. On injury, platelet degranulation causes an initial influx of PDGF, serving as a chemoattractant and mitogen for inflammatory cells and fibroblasts. In concert with TGF-β, PDGF also promotes macrophage function and granulation tissue formation. During the proliferation phase, it promotes fibroblast production of ECM and keratinocyte migration. It also increases the expression of VEGF resulting in a modest impact on angiogenesis. During remodeling it is involved in vessel maturation by recruiting pericytes and smooth muscle cells to the new capillaries. PDGF also upregulates MMP expression to assist in the breakdown of collagen, though it is also susceptible to degradation by the enzyme with low levels found in the proteolytic environment of chronic wounds.
Transforming growth factor β
The TGF-β family includes TGF-β1, TGF-β2, and bone morphogenic proteins. These proteins are produced by platelets, macrophages, fibroblasts, and keratinocytes. The TGF-β family has a profound impact on both pro- and antiinflammatory pathways and must be tightly regulated for successful wound healing.
TGF-β1 is upregulated in early wound healing. During the inflammatory phase, it promotes recruitment of inflammatory cells and potentiates macrophage function. During proliferation, it also stimulates granulation tissue formation by increasing ECM production and VEGF release. By altering integrin expression in keratinocytes, TGF-β1 permits their migration across the surface of the wound bed. Finally, during remodeling, the growth factor stimulates myofibroblast contraction and collagen production and inhibits MMPs from further collagen degradation. ,
TGF-β2 and TGF-β3 are also implicated in wound healing. Similar to the β1 isoform, they are involved in inflammatory cell and fibroblast recruitment, ECM synthesis and remodeling, and angiogenesis. Unlike the β1 isoform, however, TGF-β3 appears to be involved in suppressing keratinocyte function, resulting in better organized collagen and reducing scarring.
Vascular endothelial growth factor
The VEGF family of proteins binds and activates cell surface receptors on vascular endothelium. They are produced by platelets, neutrophils, macrophages, fibroblasts, keratinocytes, and endothelial cells in response to wounding and tissue hypoxia. VEGF-A is crucial during early angiogenesis, stimulating proliferation and migration of endothelial cells in the wound bed. This effect is potentiated by several other growth factors, including TGF-β, EGF, and PDGF. Although important for the formation of immature endothelial tubules, additional growth factors, such as Ang-1 and PDGF, are required for maturation.
Connective tissue growth factor
Connective tissue growth factor, such as FGF, requires an associated PG to exert its effects. It is produced by fibroblasts and stimulates proliferation and migration of these cells in an autocrine feedback loop, contributing to granulation tissue formation, ECM deposition, and remodeling.
Cytokines
Cytokines are small proteins involved in modulating the immune system. IL-1, IL-6, and TNF-α are secreted from inflammatory cells and keratinocytes during the inflammatory phase of wound healing. The interleukins trigger proliferation and migration of keratinocytes and fibroblasts. , TNF-α is more complex with effects that appear to be dose dependent. At low concentrations, it promotes wound healing by stimulating the production of FGF-7. At high levels, however, it has deleterious effects on wound healing.
Abnormal wound healing
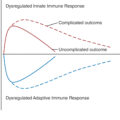
Normal wound healing requires tight control of each phase. Dysregulation as a result of intrinsic or extrinsic factors can produce an unfavorable outcome, ranging from a chronic nonhealing wound to a fibrotic scar.
Many chronic wounds exist in a perpetual inflammatory state. During controlled inflammation, timely neutrophil clearance by macrophages and neutrophil reverse migration help wounds progress to subsequent phases. , When this does not occur, neutrophils continue producing TNF-α and MMP, suppressing fibroblast ECM synthesis and degrading the provisional matrix in the early wound. , , An imbalance between the M1 and M2 macrophage phenotypes exerts a similar effect. MMP is central to this process, as many common pathways ultimately result in its unregulated activity in the wound bed. ,
Loss of control during synthesis or remodeling leads to poor scarring. Under normal conditions, TGF-β stimulates fibroblast proliferation, myofibroblast contractility, and ECM production. , As wound healing progresses, downstream inhibitors create a negative feedback loop, diminishing the production and release of the growth factor. Under these circumstances, fibroblasts undergo apoptosis or exit the wound. When this fails, however, overexpression of TGF-β results in collagen deposition and wound contraction beyond physiologic need. This derangement has been strongly linked to both hypertrophic and keloid scars. , Shear stress on the wound produces similar results, again preventing fibroblast apoptosis and stimulating differentiation into the myofibroblast phenotype.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree