Introduction
Chemical burns represent a small percentage of burn injuries yet up to one-third of burn-related deaths. Many common household and industrial compounds have the potential to induce severe chemical burns. The American Association of Poison Control Centers National Poison Data System 2014 annual report demonstrated 199,291 cases of exposure to cosmetic or personal care products; 198,018 household cleaning substances; 83,005 pesticides; 31,903 hydrocarbons; and 38,975 unspecified chemicals. Exposure to specific chemicals, including acids, alkalines, peroxides, bleaches, and phenols, in 2014 totaled 38,594, which was up from the 38,552 cases of exposure in 2013. The unfortunate reality concerning the ease of access to toxic products is evident in the presence of a rising number of pediatric exposures to chemical injuries. Most chemical burns involving children are secondary to common household products. Domestic chemical burn injuries are often caused by poor labeling and storage, and they are secondary to intentional assault and suicide attempts. The most commonly affected areas of the body are the face, eyes, and arms and legs. As a result, the length of hospital stay and duration of healing tend to be greater with chemical burns. The majority of these deaths are related to the ingestion of chemical substances. This chapter provides general principles for the treatment of chemical injuries.
Pathophysiology
All burn wounds, whether caused by chemical or thermal sources, have in common the denaturation of key structural and functional proteins. The structure of biologic proteins involves not only a specific amino acid sequence but also a three-dimensional structure depending on weak forces, such as hydrogen bonding or van der Waals forces. These three-dimensional structures impart biologic activity to the proteins and are easily disrupted by outside influences, specifically chemical and thermal energy sources. Weak bonds are impaired by heat energy sources degrading and denaturing proteins. Moreover, any variations in pH or dissolution of surrounding lipids may neutralize a protein and disrupt its function. Direct chemical effects on a reactive group in a protein will similarly render it inactive.
The severity of a chemical burn injury is determined by several factors :
-
■
Concentration of chemical in contact or ingested
-
■
Quantity of chemical agent
-
■
Manner and duration of contact (skin or ingestion)
-
■
Extent of penetration
-
■
Mechanism of action of the chemical
-
■
Physical state of agent (liquid, solid, gas)
There are six mechanisms of action for chemical agents in biologic systems, which are classified by how they denature and damage proteins: ,
-
1.
Reduction : Reducing agents act by binding free electrons in tissue proteins, causing denaturation. In general, they do so by reducing the amide link. Examples include hydrochloric acid, nitric acid, alkyl mercuric compounds, ferrous iron, and sulfite compounds. ,
-
2.
Oxidation : Oxidizing agents are oxidized on contact with tissue proteins. These agents cause destruction by inserting oxygen, sulfur, or halogen atoms to structural and functional proteins. Byproducts are often toxic and continue to react with the surrounding tissue. Examples of oxidizing agents are sodium hypochlorite, potassium permanganate, chromic acid, and peroxide.
-
3.
Corrosive agents : Corrosive substances denature tissue proteins on contact and form eschar and a shallow ulcer. Examples of corrosive agents include phenols, cresols, white phosphorus, dichromate salts, sodium metals, lyes, sulfuric acid, and hydrochloric acid.
-
4.
Protoplasmic poisons : These agents produce their effects by binding or inhibiting calcium or other organic ions necessary for tissue viability and function. These agents form esters with proteins and/or chelate calcium or other ions. Examples of protoplasmic poisons include alkaloidal acids, acetic acid, formic acid, and metabolic competitors and inhibitors such as oxalic acid, hydrofluoric acid, and hydrazoic acid.
-
5.
Vesicants : Vesicant agents produce ischemia with necrosis at the site of contact. There is associated tissue cytokine release and blister formation. Examples include cantharides, dimethyl sulfoxide (DMSO), mustard gas (sulfur and nitrogen), and lewisite.
-
6.
Desiccants : These substances cause damage by dehydrating tissues and exothermic reactions, causing the release of heat into the tissue. Examples include sulfuric acid, muriatic acid, calcium sulfate, and silica gel.
Chemical burns are often described as acidic or alkali. Acids act as proton donors in the biologic system, and strong acids have a pH less than 2. Alkali, or basic, materials capable of producing injury typically have a pH greater than 11.5. In general, alkaline materials cause more injury than acidic compounds. Whereas acids cause coagulation necrosis with precipitation of protein, the reaction to alkali is liquefaction necrosis, allowing the alkali to penetrate deeper into the injured tissue. The presence of hydroxyl ions within these tissues increases their solubility, allowing alkaline proteinases to form when the alkalis dissolve the proteins of the tissues. Organic solutions tend to dissolve the lipid membrane of cell walls and cause disruption of cellular architecture as their mechanism of action. Inorganic solutions tend more to remain on the exterior of cells but may act as transporters to carry the previously mentioned agents that denature proteins or form salts with proteins themselves.
General principles of management
The most important aspects of first aid for patients with chemical burns involve removal of the offending agent from contact with the patient—that is, stop the burn. This requires removal of all potentially contaminated clothing and copious irrigation. Irrigation of chemical burns requires protection of healthcare providers to prevent additional injuries and additional patients. In addition, the wounds should not be irrigated by placing the patient into a tub, thereby containing the chemical and spreading the injurious material. Irrigation should be a large-volume shower or decontamination station and drained out of an appropriate drain. Immediate copious irrigation has been shown to reduce the extent and depth of injury, especially to eyes. No measure of adequacy of lavage has been developed, but monitoring the pH from the effluent can provide quantifiable information as to adequacy of lavage. Thirty minutes to 2 hours of lavage is often necessary.
Safety data sheets are mandated to be available for all chemicals present in the workplace. These can be valuable resources for potential systemic toxicity and adverse effects of an agent. Further assistance is available from regional poison control centers for household chemicals or unidentified agents.
The use of neutralizing agents is generally contraindicated. Neutralizing agents cause exothermic reactions, producing a thermal component along with a chemical injury. When the chemical agent is known and an appropriate antidote to support the physiologic changes incited by the original agent is known, some benefit to its use has been documented but has not been found to be superior to water for irrigation. An example is calcium gluconate for hydrofluoric acid burns (discussed later in the chapter).
Treatment paradigms remain unchanged for burn and trauma patients with strict adherence to Advanced Trauma Life Support and Advanced Burn Life Support guidelines. After airway patency is assured, adequate air movement and hemodynamics should be maintained. Conventional thermal burn formulas are used for resuscitation at maintaining end-organ perfusion. Monitoring of urine output remains paramount to assessment of adequacy of end-organ perfusion and hence resuscitation. Systemic disturbances of pH are potential complications and must be monitored until acid-base disorder and electrolyte abnormalities are corrected.
The typical large-volume lavage required to adequately dilute chemical exposures puts the patient at potential risk for hypothermia, both from evaporative cooling losses and from the use of unwarmed lavage fluid. Principles of wound care for chemical burns are typically the same as for thermal burns. Early excision and grafting of obviously nonviable tissue is advocated, particularly in light of the observation that chemical burns tend to be deeper than they initially appear. As a result, they tend to heal more slowly.
Specific agents
Acids
Acetic acid
Acetic acid, also known as ethanoic acid, ethylic acid, and methane carboxylic acid, is a mild chelating agent. Solutions diluted to less than 40% concentrations, such as table vinegar and hair-wave neutralizing products, are usually harmless, but if used inappropriately they may cause injuries. Chemical exposures may cause symptoms of upper- and lower-airway irritation, including cough, tachypnea, wheezing, nose and throat irritation, and pharyngeal and pulmonary edema. Other symptoms found are tooth erosion, conjunctivitis, headache, nausea, vomiting, impaired vision, abdominal pain, eye pain, and whitish discoloration of the skin. In such cases, initial treatment involves irrigation.
Carbolic acid (phenol)
Carbolic acid is a hydrocarbon derived from coal tar, which acts to cause damage secondary to its ability to induce denaturation and necrosis. , The most common adverse effects are dermatitis, abnormal pigmentation, and burns to the skin. Concentrated amounts of phenol are caustic, therefore prolonged skin contact causes partial- or full-thickness burns. These burns tend to become extensive before detection, secondary to the local anesthetic properties of phenol. Ingestion of as little as 1000 mg may be fatal. Systemic effects include ventricular arrhythmias, pulmonary edema, stridor, and tachypnea. Locally, conjunctivitis, corneal edema or necrosis, and skin necrosis result.
Acute poisonings are potentially fatal, hence prompt action is necessary with copious irrigation. Polyethylene glycol (PEG; molecular weight 300 or 400 Da) has been shown to be of potential benefit, but large-volume lavage should not be delayed while PEG application is begun. Reports in the literature indicate that intravenous sodium bicarbonate may be of use to prevent some of the systemic effects of phenol.
Chromic acid
Chromic acid causes nonpainful but corrosive ulcers upon contact with the skin. Ulceration of the nasal septum and bronchospasm can occur with inhalation. This agent causes protein coagulation. Peak blood levels are thought to be achieved within 5 hours of exposure. Symptoms may occur with just 1% total body surface area (TBSA) burn, but a 10% burn or greater is often fatal owing to its systemic effects. Irrigation is the primary treatment for exposure, but in an industrial setting, washing with a dilute solution of sodium hyposulfite or water followed by rinsing in a buffered phosphate solution may be a more specific antidote. Dimercaprol may be used at 4 mg/kg intramuscularly every 4 hours for 2 days followed by 2 to 4 mg/kg/day for 7 days in total to treat the systemic effects. Dialysis in the first 24 hours is a reasonable means to remove circulating chromium and to address existing electrolyte imbalances. Exchange transfusion may be necessary. Various ointments containing products such as 10% calcium ethylenediaminetetraacetic acid or ascorbic acid are available for small superficial burns. , There have been case reports supporting the early excision of chromic acid burn to assist in preventing systemic toxicity. ,
Epichlorohydrin acid
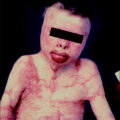
Epichlorohydrin acid is a rare, corrosive carcinogen that is colorless and exudes a garlic-like odor. It is used in the production of glues, plastic, glycerols, and resins, as well as in paper reinforcement and water purification. It can also be converted into a binder used in the production of explosives. These burns may demonstrate a rapid progression to a full-thickness wound within hours. Management commences with copious irrigation and hemodynamic monitoring ( Fig. 33.1 ).
Epichlorohydrin acid burn 3 days after admission.
Formic acid
Formic acid is a strong inorganic acid used by glue makers and tanning workers. After contact, it creates an eschar, which does not prevent systemic absorption. After it is absorbed, metabolic acidosis, intravascular hemolysis with hemoglobinuria, renal failure, pulmonary complications, and abdominal pain with necrotizing pancreatitis usually occur. All patients injured by formic acid should be hospitalized because of this multitude of potential systemic effects. Formic acid is the acid most commonly used in assaults, especially in developing countries because of its easy availability.
Hydrochloric acid, muriatic acid, and sulfuric acid
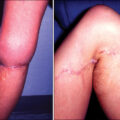
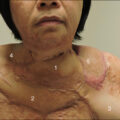
Hydrochloric acid is one of the most commonly treated chemical burns. Hydrochloric acid and sulfuric acid are proton donors, which cause the pH in local tissues to drop to zero as hydrogen ions disassociate. Coagulation necrosis and tissue ulceration occur, leading to consolidation of connective tissue and thrombosis of intramural vessels, ulceration, fibrosis, and hemolysis. Many household cleaners contain dilute hydrochloric acid (3%–6%) and sulfuric acid and its desiccant precursor (sulfur trioxide) in concentrations up to 80% to 99%. Muriatic acid is the commercial grade of concentrated hydrochloric acid. When in contact with the skin, it denatures proteins into their chloride salts. Copious irrigation and early excision are the treatments of choice. Hydrochloric acid fumes can cause inhalation injury with acute pulmonary edema ( Figs. 33.2 and 33.3 ). Other symptoms found to occur are white or grayish discoloration of the skin and exposed mucosa. Patients may have eye, mouth, throat, and abdominal pain and injuries. They may experience hematemes is, vomiting, dizziness, headache, dyspnea, cough, tachypnea, pneumonia, laryngospasm, and ultimately respiratory failure.
Hydrochloric acid burn to the hand before blister removal.
Hydrochloric acid burn to the hand after blister removal.
Hydrofluoric acid
Hydrofluoric acid is a corrosive that is commonly used in industrial applications and computer processing. It is used as a cleaning agent in the petroleum industry, in the production of high-octane fuel, glass etching, germicides, dyes, tanning, and fireproofing material, as well as in rust removal. Hydrofluoric acid is particularly lethal owing to its properties both as an acid and as a metabolic poison. The acid component causes coagulation necrosis and cellular death. Fluoride ions then gain a portal of entry that chelates positively charged ions such as calcium and magnesium, resulting in hypocalcemia and hypomagnesemia. This causes an efflux of intracellular calcium with resultant cell death. The fluoride ion remains active until it is completely neutralized by the bivalent cations. This may exceed the body’s ability to mobilize calcium and magnesium rapidly enough, causing muscle contraction and cellular dysfunction. Fluoride ion also acts as a metabolic poison by inhibiting Na-K ATPase, allowing efflux of potassium. Excess potassium subsequently causes shifts at nerve endings and is thought to be the cause of the extreme pain associated with hydrofluoric acid burns. Patients may present with self-limiting symptoms such as nausea; vomiting; fever; whitish tissue with surrounding erythema; immediate profound abdominal, mouth, and throat pain; skin edema; ulcers; and necrosis or stridor, laryngeal edema, wheezing, tachypnea, tetany, and potentially fatal cardiac arrhythmias.
Hydrofluoric acid burns are classified based on the concentration of the exposure. Concentrations greater than 50% cause immediate tissue destruction and pain. Concentrations of 20% to 50% result in a burn becoming apparent within several hours of exposure. Injuries from concentrations less than 20% may take up to 24 hours to become apparent.
Death from hydrofluoric acid exposure is usually secondary to systemic toxicity. Systemic symptoms are secondary to acidosis, hypocalcemia, hypomagnesemia, and hyperkalemia, which can lead to ventricular fibrillation. Thus electrolytes and cardiac rhythm should be monitored closely. When cardiac dysrhythmias develop, it is difficult to restore a normal rhythm. Compounding the problem of hypocalcemia, the fluoride ion may act as a metabolic poison in the myocardium to promote the irritability. The typical electrocardiographic change seen is QT interval prolongation. The fluoride ions can be removed by hemodialysis or cation exchange resins.
Treatments for hydrofluoric acid exposure are designed to neutralize the fluoride ion and prevent systemic toxicity. Primarily, the wound should be copiously irrigated for 30 minutes. If the concentration of exposure is less than 20% or the duration of exposure is minimal, this may serve as the extent of treatment. For more serious exposures, topical, subcutaneous, or intraarterial mixtures of calcium gluconate can be used as a first option. The topical gel is a mixture of 3.5 g of 2.5% calcium gluconate and 5 oz of water-soluble lubricant. It should be applied to the wound four to six times each day for 3 to 4 days. The mixture’s penetration into the dermis is limited by its calcium component. This is improved with the use of DMSO, which has its own associated toxicity. Calcium gluconate injections into the area of the wound (0.5 mL/cm 2 of 10% calcium gluconate subcutaneously or intradermally) have also been used with good results. Intraarterial injections into the radial artery (10 mL of 10% calcium gluconate and 40 mL of 5% dextrose in water infused over 2–4 hours) can be used for management of burns to the hand, but in severe cases palmar fasciotomy may be needed. , This injection should be performed within 6 hours of exposure to prevent tissue necrosis and minimize pain. It should continue until the patient is symptom free.
Nitric acid
Nitric acid is a strong oxidizing agent that can combine with organic proteins to produce organonitrates, which act as metabolic poisons. It is used in fertilizer management, casting iron and steel, and engraving. Upon skin contact, a yellow-brown stain develops on the skin and mucosa, with an eschar. Demarcation tends to occur slowly, causing difficulty in discerning burn depth. Initial treatment involves irrigation and the use of topical treatment. In addition, patients may present with whitish tinge of the teeth, eye pain, oropharyngeal pain, or abdominal pain. Patients may have dyspnea, hematemesis, dizziness, cough, tachypnea, and even develop pneumonia and laryngospasm.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree