6.3 TRAM/DIEP Flap
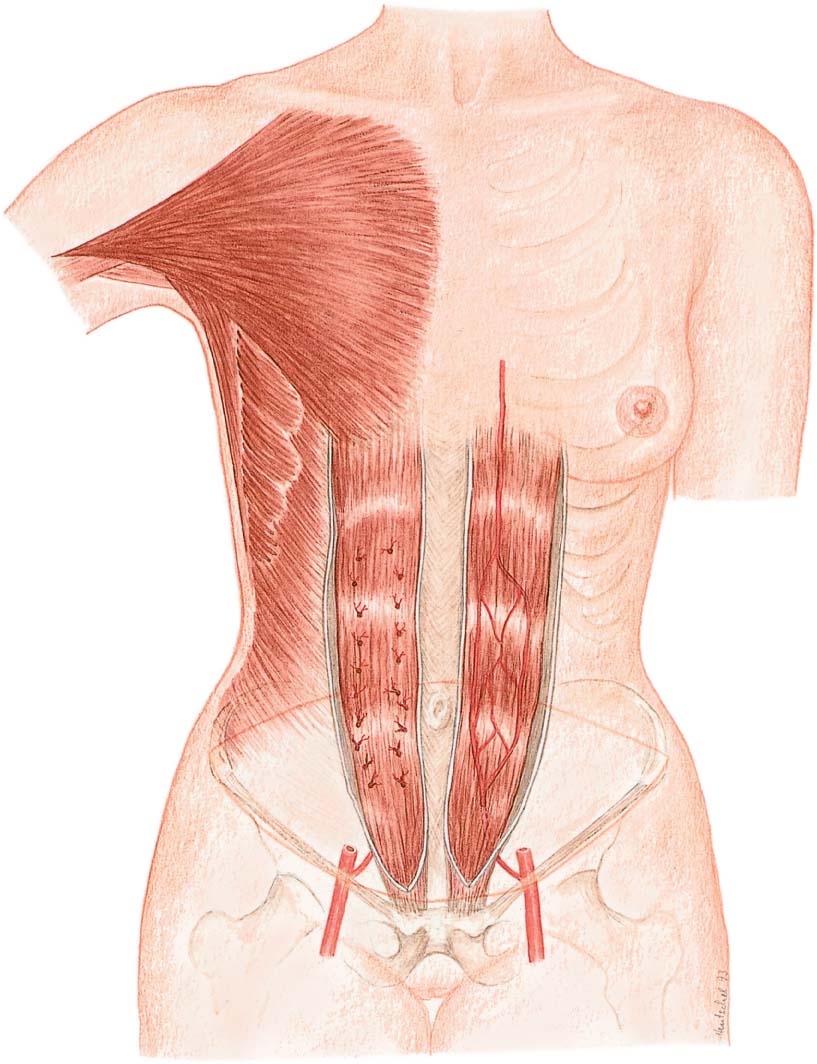
Anatomy of the Lower Abdominal Flap
The use of lower abdominal tissue in breast reconstruction became widespread with the development of the TRAM flap, which became synonymous with this technique. The advent of the perforator flap has changed this, however, and nowadays both procedures are more appropriately described as “lower abdominal flap” techniques. Surgical reconstruction of the breast using lower abdominal tissue is a challenging procedure. Thorough knowledge of the vascular anatomy, as well as of the proportions and relations of various tissue structures to each other, is essential. The illustrations provided in the text place particular emphasis on a lifelike representation of the relevant anatomy. Attention to every detail is crucial for successful surgery and good results, and detailed anatomical drawings are a better source of information than lengthy descriptions. Every effort has thus been made to highlight the special anatomical considerations related to this technique in the accompanying illustrations.
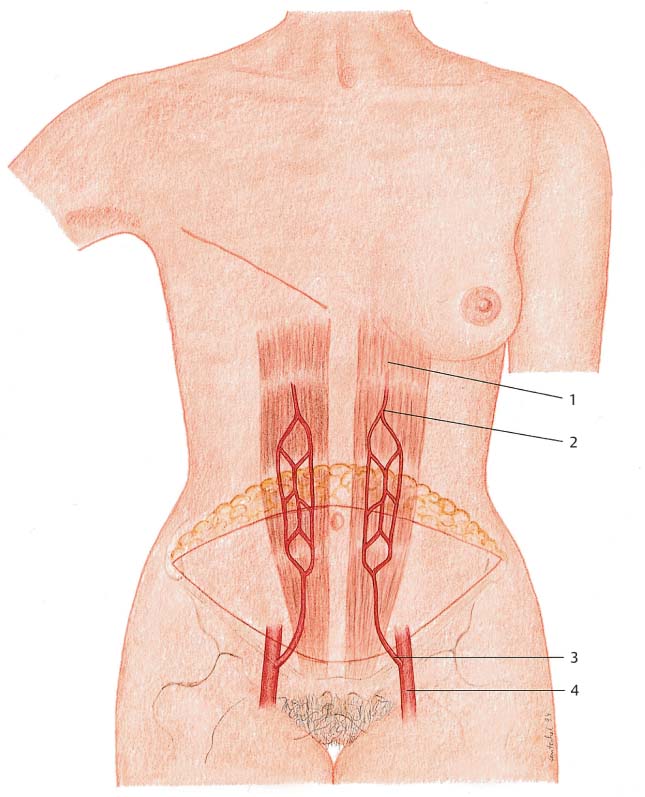
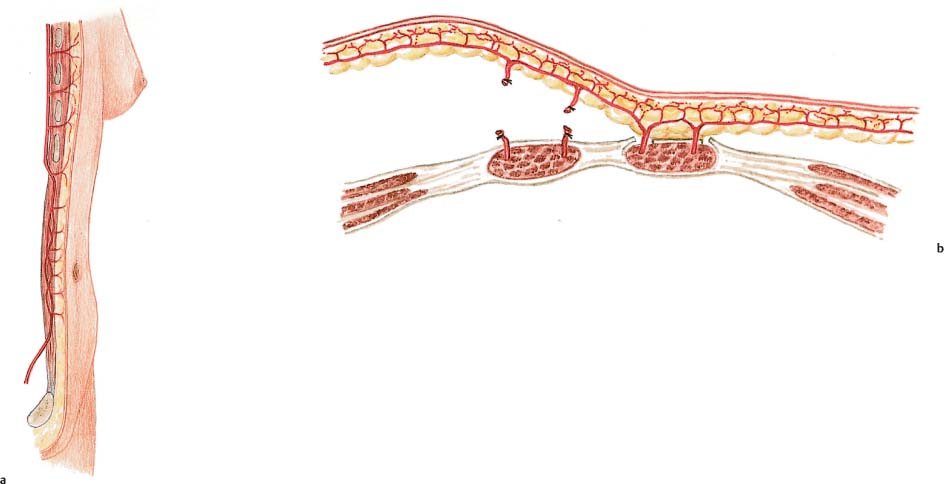
The rectus abdominis and overlying skin are supplied by the superior epigastric artery, which arises from the internal thoracic artery (also commonly known as the internal mammary artery), and the inferior epigastric artery, a branch of the external iliac artery. The superior and inferior epigastric arteries are connected by a complex vascular network. The inferior epigastric is the largest artery supplying the abdominal wall. Together with the somewhat smaller superior epigastric artery, it directly supplies the rectus abdominis. Via numerous perforating vessels, it also indirectly supplies the abdominal wall. The perforating vessels course in pairs along the axis of the rectus abdominis muscle, normally ascending diagonally to the overlying abdominal skin. They are arranged in two parallel rows, with the largest-caliber perforators located between the umbilicus and the arcuate line.
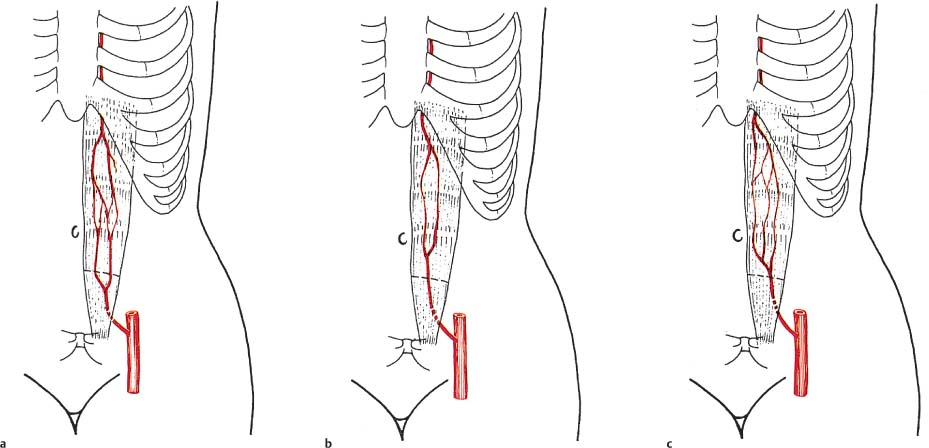
Moon and Taylor classified the branching patterns of the inferior epigastric vessels into three types. In most people, two parallel branches course along the axis of the rectus abdominis. In 14 % of patients, there are three parallel branches.
For perforator dissection, it is important to identify the largest perforators. On the other hand, it is also preferable for dissection to extend over only a short distance. A dissection study by Munoz showed that the lateral perforating vessels are short and pass in a straight line through the muscle in nearly 80 % of patients. The medial perforators are larger in caliber, but in 80 % of patients have a longer intramuscular course. This prolongs the time needed for dissection and increases muscle trauma. There are significantly fewer lateral perforators (34 %) than medial perforators (66 %); Blondeel has also reported similar findings. Isolation of the flap on a lateral perforator is preferable, for shorter and atraumatic dissection. Preoperative Doppler examination and intraoperative assessment are essential for choosing the most suitable perforator or perforators.
The deep inferior epigastric artery arises from the external iliac artery. It is usually accompanied by two veins. After penetrating the loose adipose tissue of the pelvis and the transversalis fascia, it usually gives off an anterior and a lateral branch before passing to the underside of the rectus muscle near the arcuate line. Its course beneath the muscle varies, but it is typically in the lateral third. After entering the rectus abdominis just below the arcuate line, it divides into a somewhat thinner medial and a thicker lateral branch. The lateral branch usually travels in the lateral third of the muscle.
Both branches distribute vessels, the medial and lateral perforators, which pierce the muscle and fascia to supply the over-lying subcutaneous layer and the skin.
In addition to the deep arterial system, the lower abdominal skin flap is also supplied by the superficial inferior epigastric artery (SIEA) and respective veins. Isolation of a flap around these vessels, which are branches of the great pelvic vessels, is also an option.
Innervation of the rectus abdominis is segmentally arranged. The nerve supply is from intercostal neurovascular bundles, which cross the lateral border of the muscle and pass diagonally along its underside. The main branch innervates the middle third of the muscle. Small branches penetrate the lateral third separately.
The aim in traditional pedicled TRAM flap surgery was to include the largest possible number of perforators in the flap. This has changed with the development of microsurgical techniques. Greater emphasis is now placed on leaving as much muscle in place as possible with intact innervation and ensuring flap vascularization with only one or two perforating vessels.
Further information can be found in the illustrations and detailed descriptions provided in the accompanying legends. These include specific anatomical considerations, which are relevant to all lower abdominal flap techniques used in breast reconstruction—i. e., pedicled TRAM, free TRAM, and DIEP flaps.
Principles of the TRAM/DIEP Flap Techniques
Techniques in autologous reconstruction using lower abdominal tissue have advanced so rapidly since the first edition of this book appeared that this section has had to completely rewritten. The earlier classification of lower abdominal flaps into unipedicled, bipedicled, and free TRAM flap procedures is now outdated as a result of research findings regarding functional deficits in the abdominal wall after bipedicled muscle transfer and the development of perforator techniques.
In addition, the use of autologous lower abdominal tissue has become the gold standard with regard to aesthetic outcomes after breast reconstruction. Less than 10 years ago, autologous tissue transfer was an exceptional procedure in selected patients, but it is now one of the main options available for reconstruction of the breast.
The quality of autologous reconstruction using lower abdominal tissue is superior to all other procedures with regard to:
Size and shape
Natural appearance and feel
Warmth
Natural aging of the reconstructed breast
The pedicled transversalis rectus abdominis flap came into widespread use following its description by Hartrampf, Schefan, and Black in the early 1980 s. Since then, this procedure has advanced more than almost any other in reconstructive surgery. From being an aggressive surgical technique associated with substantial blood loss, it has now become a highly refined microsurgical procedure that uses naturally occurring excess tissue located between the umbilicus and pubic symphysis to obtain excellent reconstruction results. Despite its enthusiastic reception, however, it should be remembered that this is a complex reconstructive procedure that requires a high degree of surgical expertise and technical skill. Well-organized and smoothly running perioperative management is necessary to avoid complications. The patient should also understand that she has to be medically fit to undergo the operation, which generally takes several hours, and that recovery can sometimes take several weeks.
Lower abdominal tissue transfer is a complex procedure. It is associated with a higher rate of complications than simpler breast reconstruction strategies.
Particularly among plastic surgeons, there is a trend toward using microsurgical tissue transfer. The unipedicled TRAM flap continues to be the standard procedure performed by gyneco-logical surgeons, however, and the present book therefore focuses on this procedure in greater detail.
The bipedicled rectus flap is not discussed in the present book. Available studies confirm our own experience that the associated abdominal wall morbidity is too great to justify its use. Even attempted muscle-sparing bipedicled techniques result in major functional deficits in the rectus abdominis. Two-thirds of patients are unable to sit up from a supine position without using their hands. Since microsurgical procedures involve a much lower rate of functional loss, bipedicled rectus flap procedures should no longer be used. If suitable expertise in the relevant microsurgical techniques is lacking, the patient should be given a referral.
Historical Development
Case reports of lower abdominal free tissue transfer were published as early as 1978 by Holström, but it was Hartrampf and colleagues who first provided the decisive stimulus for the use of the pedicled TRAM flap in breast reconstruction. The technique began to be used in Germany during the early 1980s.
Most early procedures used unipedicled rectus flaps. Unpredictable vascular complications were common, most often due to venous congestion. The bipedicled flap, which incorporated muscle pedicles from both rectus muscles, was designed to increase the reliability of the TRAM flap to the same level as with the latissimus dorsi flap, which was regarded as the standard of reliability in flap reconstruction.
The first edition of this book reported the general consensus that a unipedicled flap is appropriate if 50–60 % of flap tissue is needed for reconstruction. If all of the tissue is needed, a bipedicled flap was considered the appropriate choice.
In view of the literature reports now available regarding donor-site morbidity in the abdominal wall, the bipedicled TRAM flap can no longer be regarded as a viable reconstructive option.
During the early 1990s, microvascular TRAM flaps became increasingly common. This required resection of only a relatively small, periumbilical portion of the rectus abdominis and resulted in improved flap perfusion via the dominant vascular pedicle of the inferior epigastric vessels. There were also distinct advantages with regard to donor-site morbidity, although these turned out to be less significant than was initially hoped.
The much-awaited leap forward in minimizing the donor-site defect came with the development of the perforator flap, which was introduced into clinical practice during the late 1990s.
It is important to bear in mind the wide variety of surgical steps used when evaluating comparative studies of various lower abdominal flap techniques. In pedicled flap procedures, for instance, the entire rectus muscle may be dissected. In a muscle-sparing procedure, only half to two-thirds may be dissected. Either the medial or lateral part of the muscle can remain in place. Microsurgical free tissue transfer can either harvest the entire muscle width or use a muscle-sparing procedure—although the definition of “muscle-sparing” may vary. Perforator flaps may include one, two, or three perforators from various rows. The degree of muscle trauma resulting from transection of the corresponding motor nerves also varies. Damaged nerves may remain traumatized, or they can be repaired microsurgically. All of these differences should be taken into account in any critical analysis of the currently available literature.
Indications
The TRAM/DIEP flap is the method of choice in autologous breast reconstruction. Other techniques—such as latissimus dorsi, gluteal, and gracilis flaps—are less often indicated.
The Rubens flap (so called in reference to the curvaceous figures of the women painted by Peter Paul Rubens) is a myocutaneous flap taken from the flank region that includes portions of the external oblique and transversalis muscles. It is of as little relevance in autologous tissue reconstruction as the lateral trans-verse thigh flap.
The most common alternatives are currently the superior gluteal artery perforator (SGAP) flap (see section 6.4, pp. 167 ff.) and the transverse myocutaneous gracilis (TMG) flap (see section 6.5, pp. 179ff.). A latissimus dorsi flap may also be an alternative (see section 6.2, pp. 100 ff.).
Patient Suitability
In addition to the patient’s overall health, the risk of local tumor recurrence and systemic metastasis also needs to be taken into account during patient selection.
It is also imperative that the patient should expressly wish to undergo this complex surgical procedure. In the worst-case scenario, she will have to live with the results of a failed operation. Strong patient motivation is also important for accepting the considerably higher postoperative burden in comparison with other procedures.
From a technical standpoint, the patient’s physical build, breast size and shape, and the mastectomy defect are the key factors in the choice of procedure. The patient’s age, occupation, lifestyle, and the amount of time needed for surgery and recovery also need to be factored into the decision.
The best candidate for autologous reconstruction with lower abdominal tissue is a woman aged 40–60 with medium-sized or large breasts and a full figure, who has had one or two full-term pregnancies. She should also be healthy, medically fit for surgery, a nonsmoker, and should not have previously undergone any major abdominal surgery. The individual points should then be discussed once again on the basis of this “ideal candidate.”
What are the most important factors in determining whether a patient is a suitable candidate for autologous tissue reconstruction? In addition to the patient’s wishes and her overall and specific health status, the strongest determinant is the shape and size of the contralateral breast. Generally speaking, the larger the breast, the more appropriate is autologous tissue reconstruction. The mastectomy defect is also important. The poorer the local conditions—such as scarring, irradiated tissue, and the extent of skin and muscle resection—the greater the support for using a flap procedure. The deciding factor is then whether the physical requirements are present for removing a larger block of tissue from the lower abdomen. Most patients have sufficient lower abdominal tissue for recreating the breast, despite appendectomy, cholecystectomy, or cesarean section. Even a lower abdominal laparotomy though a midline incision is not an automatic exclusion criterion, although it considerably complicates surgical strategy (e. g., elevating the flap as a unipedicled flap on one side and as a perforator flap on the other).
Nature also plays a part in establishing the indication. While women with fuller breasts tend to have excess lower abdominal tissue, it is less common to see slim women with larger breasts.
In bilateral reconstruction, it can be more difficult to determine the best procedure, as the flap volume may not be sufficient. However, bilateral reconstruction has the advantage that the reconstructed breast does not have to match an existing breast.
Contraindications
The list of contraindications is relatively short. This is partly due to microsurgical techniques, which have eliminated many potential complications related to the highly tenuous blood supply in pedicled flaps. Nevertheless, careful assessment of contraindications is still imperative.
Smoking is still the primary contraindication. It has been shown that the microcirculation is reduced or even disturbed in smokers. The problems are less likely to involve flap hypoper-fusion than insufficient venous drainage. Limited respiratory function, which is common among smokers, can also lead to postoperative complications. The procedure is therefore not recommended for patients who smoke more than 20–30 cigarettes a day. Complete abstinence is required for at least 3 months before surgery, although this does not compensate for long-term effects related to smoking.
Obesity is also associated with hypoperfusion. This procedure is therefore contraindicated in patients with a body mass index of 30 or more (clinical obesity).
Prior chemotherapy can also lead to microcirculatory disorders and reduced stem cell function. This should at least be taken into account when planning autologous blood donation.
There is also a greater risk of thrombosis in this relatively long procedure, and it is therefore not advisable in patients in whom an increased risk of thrombosis is suspected. Hormone replacement and antihormone therapies should be discontinued perioperatively. To avoid complications related to thrombosis, compression stockings should be worn intraoperatively and briefly after surgery.
Parasternal radiotherapy is no longer regarded as a potential contraindication. Abdominal scarring also seldom contraindicates the use of lower abdominal tissue in autologous reconstruction.
An increased bleeding tendency or a history of clotting disorders are contraindications for undergoing this complex surgical procedure. Patients who are taking clotting agents (including vitamin C) should discontinue their use. The long half-life of aspirin should also be borne in mind.
The procedure is no longer contraindicated in women who wish to have children.
Relative contraindications include psychological instability or a lack of motivation to undergo this physically and psychologically taxing procedure.
Preoperative Preparation
Preoperative Marking
The marks are made with the patient in a standing position. The inframammary fold is drawn to match the level of the contralateral side, and it should be borne in mind that it will descend after closure of the lower abdominal wound. Next, using the contralateral side as a guide, the previous anatomical borders of the breast—the superior limit and the boundaries along the border of the sternum and near the anterior axillary line—are marked.
Correct drawing of the flap requires some experience. Sufficient tissue has to be obtained while avoiding tension along the wound margins, which would interfere with healing and the formation of a symmetrical scar. With the patient sitting and then standing, a pinch test is carried out to determine the amount of excess tissue on the upper and lower abdomen. Taking into account the periumbilical location of the perforators, the superior limit of the flap should be above the umbilicus (or at least at the same level). Any adjustments in flap length can thus only be made by altering the inferior border. In patients with a firm abdominal wall, a shorter flap length will have to be acceptable; in patients with looser abdominal skin, a more generously proportioned flap is possible.
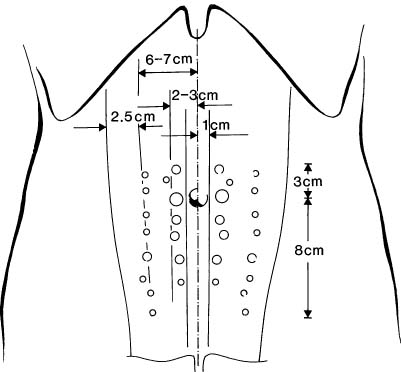
There were some early reports on the use of a middle abdominal flap with a scar near the center of the abdomen. The method currently preferred is a lower abdominal flap with a suprapubic scar. After determining the length of the flap in the midline (typically 13–18 cm), the lateral corners of the flap are marked symmetrically on the patient in a standing position. Depending on the preferences of the patient and surgeon, these can be at the level of the anterior iliac spines or higher, as in a high-tension lateral abdominoplasty using the Ted Lockwood technique. The outer limits of the flap are marked as symmetrically as possible about 15–20 cm from the midline. The final outline of the flap is then drawn with the patient standing or supine. Sufficient time should be taken to ensure that the marks are drawn symmetrically. The marks are then carefully rechecked with the patient in a standing position. A final check can be carried out on the operating table and any adjustments needed can be made.
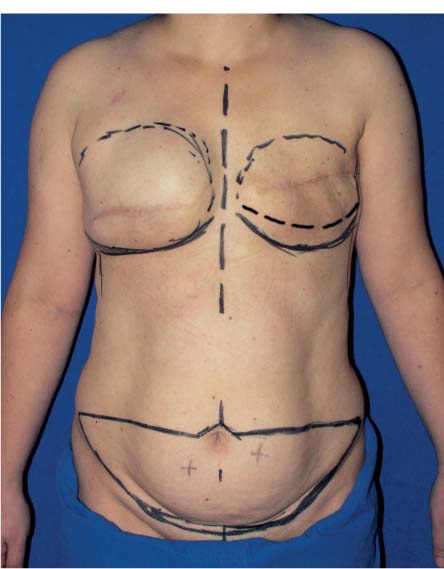
As the abdominal flap almost always provides excess skin, it is not necessary to place the skin island strategically, as described in the section on latissimus dorsi flaps.
Preoperative Measures
Although a blood transfusion is usually not necessary during the operation, we recommend storing two units of autologous blood in advance. First, this can make the postoperative period easier for the patient. Second, by banking her own blood the patient becomes psychologically aware that this is more than just a “minor” operation.
Bowel preparation is completed the day before surgery. This aids closure of the abdominal wound and facilitates recovery. Intestinal function should be monitored postoperatively.
For the operation, the patient is placed in the semisitting position with the arms abducted.
Compression stockings should be applied in order to ensure pump function in thew calf muscles during the long surgical procedure.
A solution of saline and epinephrine is injected along all of the planned lines of incision and the area of skin to be dissected in the mastectomy defect. To avoid unnecessary blood loss, intra-operative blood pressure should not exceed 100 mmHg. Intra-operative fluid replacement should be kept to a minimum.
Surgical Technique for Unipedicled Rectus Flaps
Preparing the Mastectomy Defect
It is useful to have two surgical teams, with one team preparing the mastectomy defect and the other preparing the flap dissection. More often, however, the two steps are carried out by a single surgeon.
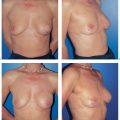
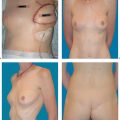
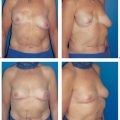
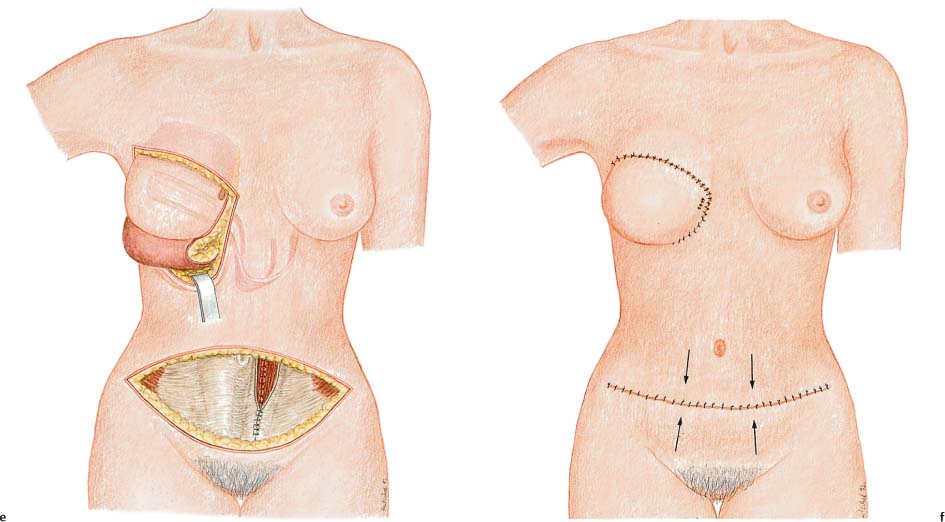
First, the mastectomy defect is prepared to receive the flap. For successful reconstruction, the flap should not be “inset” into the mastectomy scar. Instead, it should be used to create an aesthetic unit. Generously resecting the original breast skin, far into the inframammary fold, is thus preferable to producing a “patch” effect. In contrast to latissimus dorsi reconstruction, the amount of donor tissue allows an incision to be made around the mastectomy defect superiorly (preferably from superolateral to inferomedial, along the cleavage) and removal of breast skin all the way into the inframammary fold (which will be reconstructed). This conceals the inferior scar, while providing sufficient latitude for positioning the flap so as to achieve adequate ptosis.
With continuing dissection in the subcutaneous plane, the breast skin is completely freed off the pectoralis muscle along the preoperative markings, which have been drawn to match the boundaries of the contralateral breast. Dissection proceeds to 2 cm superior to the inframammary fold that has been marked, and it should be remembered that the wound margin will later descend with abdominal wound closure. Excessively wide dissection, beyond the anterior axillary line, has not proved useful, as it often produces exaggerated lateralization of the flap. This is a common risk, given that the postmastectomy scar extends far into the axilla. If the entire scar is excised, the axillary region is opened too widely. Surgeons often mistakenly believe that the breast can be restored to its original form by excising the mastectomy scar and dissecting the previous breast skin. This fails to take into account postoperative skin shrinkage, which completely alters the anatomical conditions. For tension-free placement of the abdominal flap and reconstruction of the breast as an aesthetic unit, it is absolutely imperative to preserve the anterior axillary line and excise the breast skin. It is only by doing this that a good aesthetic outcome can be achieved. Unfortunately, this also entails the risk that primary wound closure will be very difficult in cases of partial or complete flap necrosis.
In delayed reconstruction, the best aesthetic results are achieved by placing the inferior wound margin in the inframammary fold. Excess skin can be excised immediately at the start of the operation when dissecting the mastectomy defect, or later when shaping the flap. We prefer the latter, since shaping the flap is the final step in surgery and it is possible to assess flap perfusion at the same time.
In patients undergoing corrective surgery after implant reconstruction, we recommend removing the entire capsule with the implant and repositioning the pectoralis muscle on the chest wall. This allows anatomically correct positioning of the autologous tissue flap.
Dissection of the Unipedicled Rectus Flap
After the umbilicus has been grasped with two single-pronged hooks, an incision is made around its perimeter, and it is then dissected along its stalk as far as the abdominal wall. Next, the abdominal flap is incised along the preoperative marks. Depending on the amount of tissue needed for reconstruction, the superior incision may be slightly beveled to obtain more fatty tissue.
The first step in the pedicled flap operation is to dissect the portion of the abdominal wall superior to the flap off the anterior abdominal wall fascia, as far as the costal arches and into the epigastrium.
At the same time, a subcutaneous tunnel can be created between the mastectomy defect and the abdominal wall cavity. The tunnel should be located in a more suprasternal than parasternal position. This helps preserve or reconstruct the infra-mammary fold, a key element in achieving proper breast shape. It is impossible to completely avoid crossing the inframammary fold medially, however, as a certain amount of space is needed to transfer the flap and position the rectus muscle. This is a drawback of the pedicled flap.
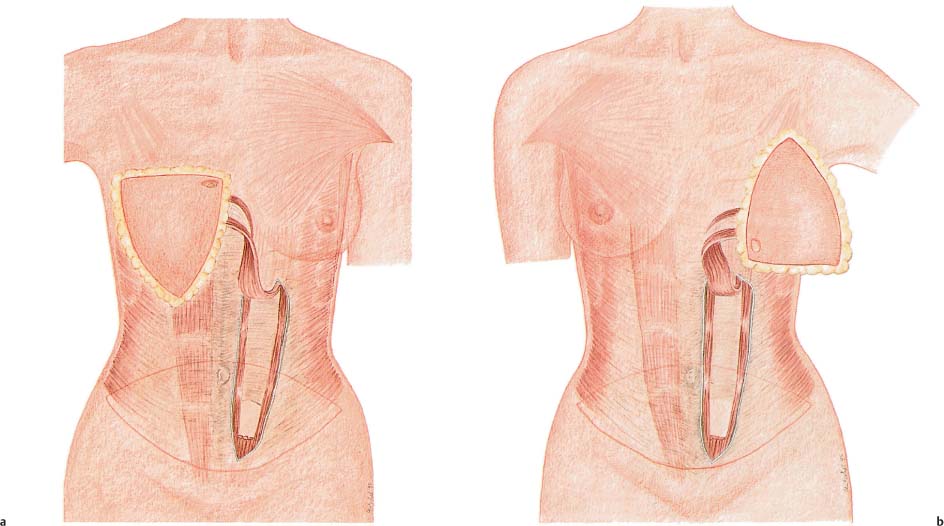
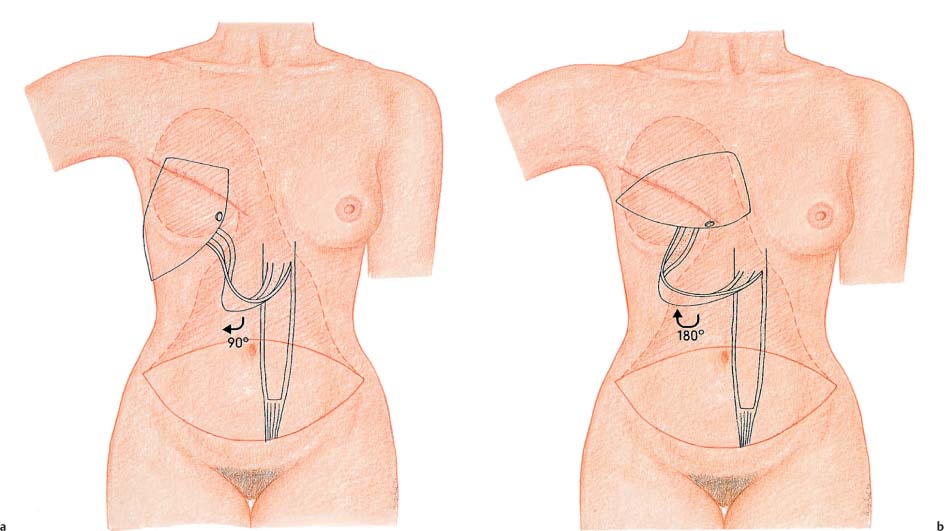
Now, at the latest, the decision has to be taken whether the flap will have an ipsilateral or a contralateral pedicle. There are pros and cons for each method. The contralateral pedicle often used to be preferred, as it was thought to avoid sharp folding of the pedicle. However, some authors have reported that the shorter path of an ipsilateral pedicle allows better draping of the flap. The present authors tend to recommend a contralateral pedicle. Adequate dissection of the rectus muscle to the costal arch provides a sufficiently long muscle pedicle with minimal disruption of the medial inframammary fold. Nevertheless, an ipsilateral pedicle may be preferable in certain situations—for instance, in patients with scarring in the flap region.
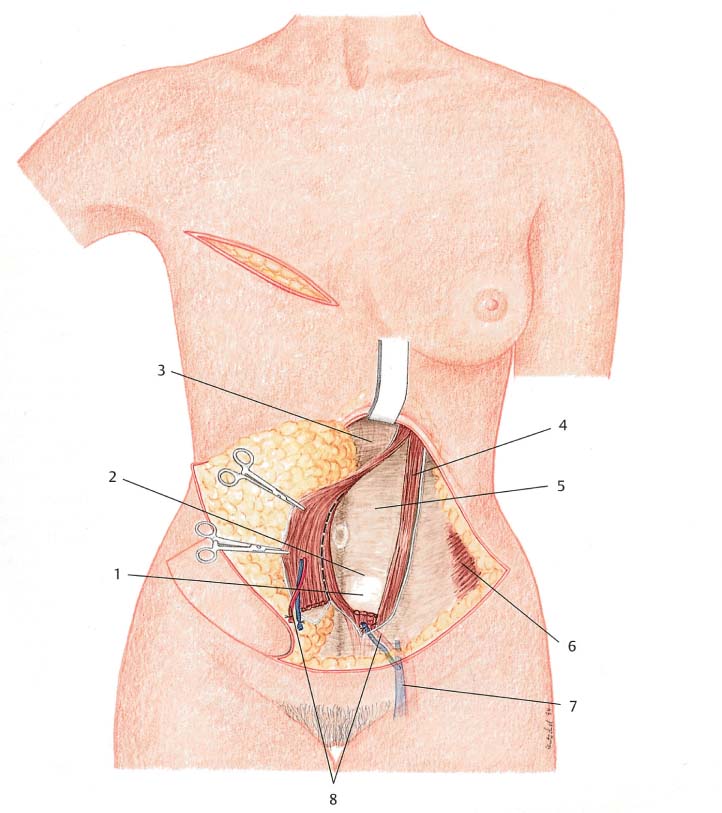
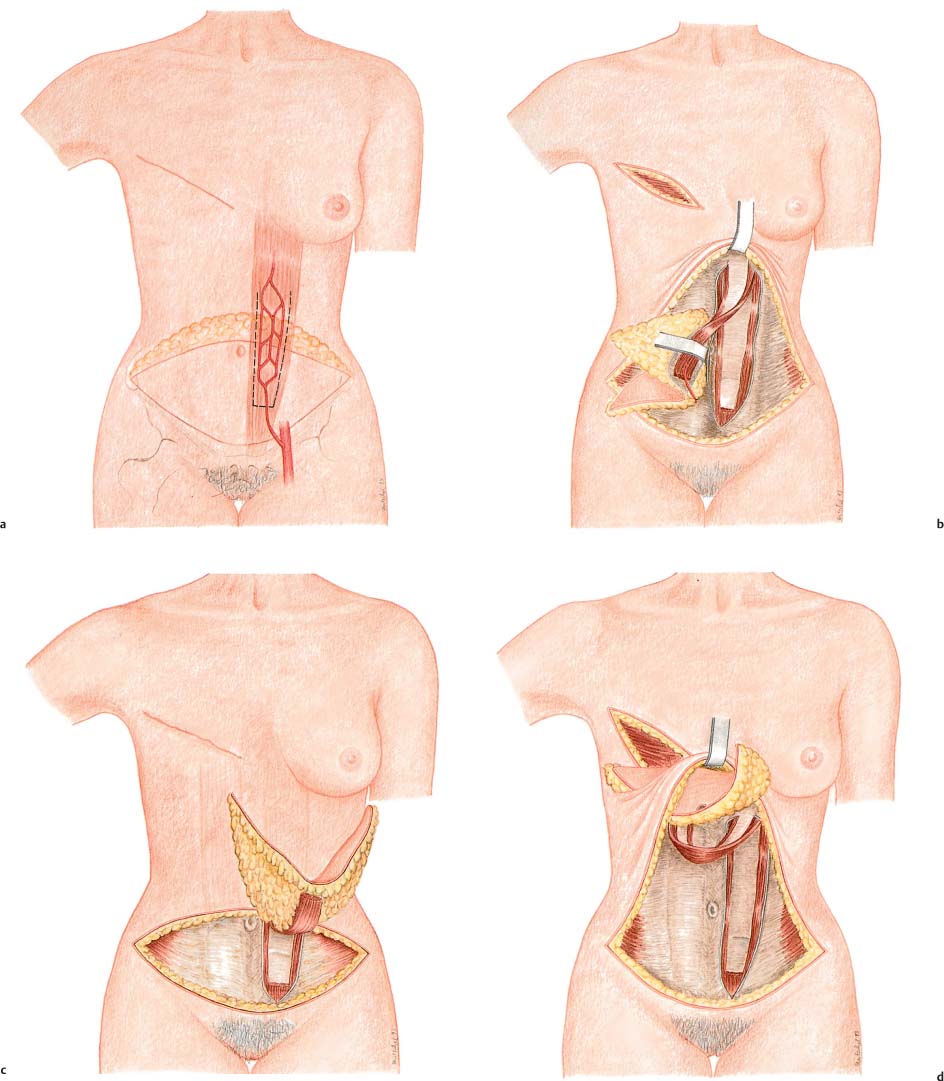
Flap harvesting starts with dissection of the nonpedicled side of the flap to beyond the midline. One can already obtain a rough idea of the position and number of perforators in the pedicled portion of the flap, although absolute symmetry is rare. Dissection of the medial border of the fascia is often carried too far medially, with the fascial incision being nearly at the midline. This can occur particularly in patients with diastasis recti. A safe and reliable method is to carry the dissection to 1.0–1.5 cm medial to the midline. This spares a few centimeters of valuable fascia and allows reliable abdominal closure.
Inferiorly, the strip of fascia is dissected to nearly the arcuate line. Inexperienced surgeons have a tendency to remove too much fascia, fearing that the flap may not have a sufficient number of perforators. It should be recalled that a single perforator is sufficient to supply an entire flap. Below the level of the arcuate line, the rectus sheath no longer consists of two layers, and preservation of the fascia is essential for ensuring abdominal wall stability.
The most technically challenging aspect is dissection from the lateral side and determining the lateral fascial incision, as the lateral perforators and the main vessel, which is located relatively far laterally, need to be included in the muscle flap that is being raised. This only applies, of course, to muscle-sparing techniques—i. e., when the aim is not to include the entire muscle width in the pedicle, but only the medial two-thirds, which is preferable in relation to stability.
Preoperative sonographic examination of the lateral row of perforators is often helpful. However, it is still necessary in actual practice to rely on intraoperative exposure of the lateral perforator row or on previous experience. Normally, the lateral border of the fascial incision is about 2 cm medial to the lateral border of the rectus abdominis. Perfect visualization of the muscle through the fascia is often impossible, however. To help estimate the portion of muscle to be harvested, after a bloodless field has been obtained, the limits of the muscle can be marked from the costal margin to the flap, and then lateral to the flap. Intra-operative Doppler assessment of the superior epigastric artery may be helpful in determining the necessary size of the muscle harvest. In our experience, however, it is not the quality of arterial perfusion, but rather of venous drainage that determines normal flap perfusion.
Inexperienced surgeons are therefore advised to avoid taking chances and instead to include the entire muscle in the flap dissection.
Postoperative abdominal wall instability is not caused by the width of the muscle pedicle superior to the flap, but by overly aggressive muscle harvesting around the perforator/fascial strip.
Superior to the skin island, the portion of fascia to be harvested with the muscle is marked. This strip of fascia, typically 2.0–2.5 cm wide, should ideally be located along the course of the superior epigastric artery. The parallel fascial incision on the rectus muscle is now carried from the costal margin to the superior border of the flap. Bleeding always results and should be controlled with bipolar forceps. The use of electrocautery scissors is also an option. Although damage to the artery by the scissors is hard to imagine, it is of course possible if the intramuscular dissection is carried too deep.
The next step is the most technically demanding one in flap dissection: freeing the rectus muscle from its sheath. Good exposure can be achieved by dissecting the muscle from medial to lateral. Starting at the medial fascial incision, the entire rectus muscle superior to the flap is dissected free. Dissection is more difficult near the tendinous intersections of the rectus abdominis, which adhere to the muscle. Medial dissection is now continued to the inferior border of the perforator/fascia strip.
Inferior to the flap and between the tendinous intersections, the rectus muscle can be bluntly dissected off the posterior fascia. In the lower portion of the muscle, the inferior epigastric artery and its two companion veins are easily visualized. In the superior portion, at the costal margin, it is often possible to identify the superior epigastric artery. The strip of fascia can now be adjusted if it the dissection of its lower portion appears to be too wide. Next, the inferior fascial incision is placed and carried along the lateral perforators until it joins the lateral incision of the rectus fascia superior to the flap. Via the generous medial access route, the inferior epigastric vessels are now dissected free over a distance of 4–5 cm and incorporated into the flap. In the event of venous congestion, this has the benefit of maintaining vessel patency with temporary drainage; or, if there are further complications, microvascular anastomosis is possible.
In order to preserve the intramuscular anastomotic network, which is mainly located superior to the umbilicus, the lateral portion of the rectus muscle is now dissected off the posterior rectus fascia. Mosquito clamps are placed at the lateral border of the rectus fascia, and—under tension on the fascia—the muscle is dissected to its lateral margin. Muscle-splitting dissection is carried out at the level of the fascia strip in order to preserve the muscle fibers located here.
Finally, the rectus muscle is divided at the inferior border of the fascia strip, with care being taken to avoid injuring the inferior vessels.
The flap is now attached by the rectus muscle only, which is dissected to the costal arch or above it (depending on level of experience) in order to achieve sufficient rotation. The difficulty of the dissection may vary, depending on the shape of the bony thorax. In patients with a low thorax and relatively high breasts, dissection of the pedicle has to be carried further superiorly than in the reverse situation.
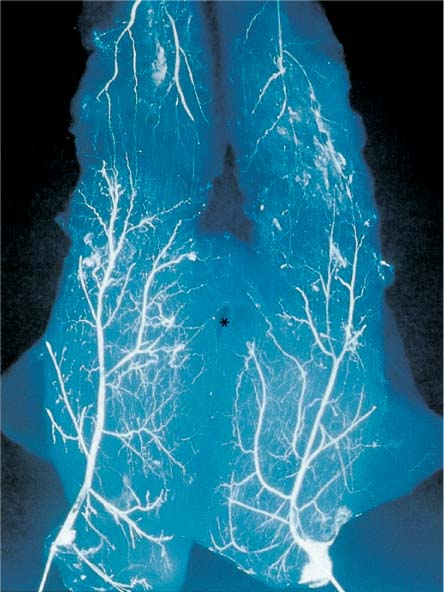
Experienced surgeons in particular tend to want to use a muscle-sparing procedure along the lateral muscle border to preserve the integrity of the abdominal wall. For anatomical reasons, this is not advisable, however. As the arteriography image in Fig. 6.26 demonstrates, the anastomotic networks are especially well developed in the lateral portion of the muscle. As the veins follow the course of the arteries, such a procedure would fail to include important flap drainage systems. To improve flap rotation, the muscle pedicle can be narrower at the costal arch, as the vessels from the anastomotic network rejoin here to form larger-caliber vessels.
During dissection of the superior portion of the muscle, the neurovascular bundle of the eighth intercostal nerve is transected. The resultant bleeding is rather heavy, but this should not lead to concern that the superior epigastric has inadvertently been cut, as this artery courses along the inferior border of the muscle. Transecting the eighth intercostal nerve is important for atrophy of the rectus muscle and can help eliminate the need for later correction.
Although it is not necessary to expose the entrance of the superior epigastric vessels into the rectus muscle, this can be safely done macroscopically or with a surgical loupe. Use of a head lamp can facilitate dissection. The entrance of the vessel is somewhat eccentric, in the medial portion of the muscle.
Dissection of the flap pedicle is now complete.
Next, the flap is tunneled to the mastectomy defect. From the mastectomy defect, the nonpedicled portion of the flap is grasped with ring forceps and advanced superiorly under simultaneous tension and pressure. The flap should not be squeezed through the tunnel. If more space is needed, the subcutaneous tunnel should be widened. Moistening the tissue can also facilitate advancement through the tunnel.
The flap is now temporarily placed in the mastectomy defect and the flap pedicle is checked. If there is excessive tension on the flap pedicle, dissection should continue to free it in order to relieve tension on the supplying vessels. Dissection of a tension-free flap pedicle is always preferable to accepting excessive tension for fear of vascular injury. Excessive tension on the pedicle can cause kinking of the vessels and subsequent flap demise.
When these steps have been carried out to the surgeon’s satisfaction, the skin island is temporarily secured in the mastectomy site.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree