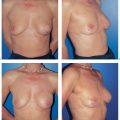
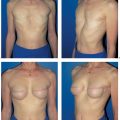
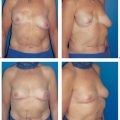
6.4 SGAP Flap
It is well accepted that autogenous breast reconstruction can provide the most natural and long-lasting results. Nowadays, the abdomen constitutes the primary donor site, as abundant and well-vascularized tissue is often available here. However, it may sometimes not be available—for example, when excessive abdominal scarring is present, or in very thin patients. In these cases, the gluteal region offers a valuable alternative source of autogenous tissue.
The microsurgical transfer of a gluteal myocutaneous flap for breast reconstruction was first described by Fujino et al. in 1975. Following initial enthusiasm, however, the flap was only rarely used, due to the difficulty of the dissection, the shortness of the pedicle, and substantial donor-site morbidity. The advent of perforator flaps in the early 1990 s marked a major breakthrough in reconstructive surgery. Koshima initially described a pedicled gluteal perforator flap in 1993, but this was based on short parasacral perforating vessels. Shortly after, Allen described the use of a superior gluteal artery perforator (SGAP) flap as a free tissue transfer for breast reconstruction. Blondeel further refined the operation in 1999 by demonstrating the use of the SGAP as a sensate flap. In comparison with the myocutaneous flap, the perforator flap allows minimal donor-site morbidity by avoiding resection of the gluteus maximus. In addition, the pedicle length can be extended, resulting in greater flexibility in flap insetting.
The SGAP flap thus represents the best further development of the gluteal myocutaneous flap and constitutes the second choice for autogenous breast reconstruction after the DIEP flap.
Regional Anatomy
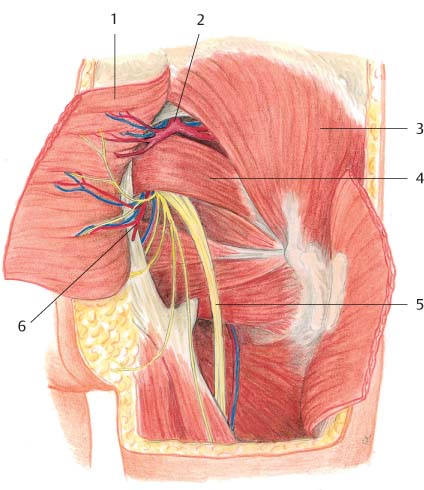
The gluteus maximus is a broad muscle involved in hip extension and lateral rotation of the thigh. It originates from the posterior surface of the ilium, sacrum, coccyx, and sacrotuberous ligament. Its fibers run down inferolaterally to insert into the iliotibial band and the gluteal tuberosity of the femur. It is a type III muscle with dual vascularization by the superior and inferior gluteal vessels ( Fig. 6.54 ). These are terminal branches of the internal iliac vessels and emerge from the pelvis above and below the piriformis muscle. The superior gluteal vessels divide into a superficial branch to the gluteus maximus and a deep branch running between the gluteus medius and minimus. The superficial branch gives off several perforators through the muscle to supply the overlying fat and skin.
Two motor nerves accompany the superior and inferior gluteal vessels around the piriformis. The superior gluteal nerve runs with the deep branch of the superior gluteal vessels to supply the gluteus medius, minimus, and tensor fascia lata. The inferior gluteal nerve exits under piriformis and enters the gluteus maximus to supply it. The cutaneous supply to the buttock is provided by the superior clunial nerves. These originate from the dorsal branches of the first three lumbar nerves, pierce the deep fascia above the iliac crest, and then course into the subcutaneous fat to supply the buttock skin.
Preoperative Preparation
When contemplating elective microsurgical breast reconstruction with perforator flaps, appropriate patient selection is the priority in order to ensure a satisfactory outcome. Patients should be motivated and well informed about the procedure. In order to withstand the prolonged anesthesia associated with free tissue transfer, the patient should be in a good general state of health. Smokers are asked to refrain from using nicotine for at least 3 months before the operation. Aspirin, nonsteroidal anti-inflammatory drugs, and herbal medications should be avoided for 3 weeks before the operation.
For the flap harvest to be as safe as possible and more time-efficient, it is crucial for the preoperative investigation to include a proper examination of the gluteal vascular anatomy. This is especially important in cases in which alterations in the blood supply to the buttock may be present—for example, after liposuction in the gluteal region. Unidirectional Doppler flowmetry can easily be used to detect and map important musculocutaneous perforators before the operation. Usually, four or five adequately sized perforators can be found in the territory of the superior gluteal artery (SGA). A large SGAP flap can normally be perfectly perfused by harvesting only one good perforator. The Doppler device has the advantages of being ubiquitous, portable, inexpensive, and straightforward for the surgeon to use. However, it only provides approximate information about the location of the perforators, with subjective evaluation of the vessel size. It also tends to generate false-positive and false-negative signals, thereby reducing its reliability.
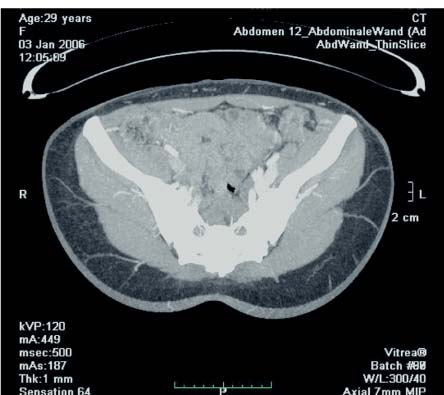
We recently started using multidetector computed tomography (CT) during the preoperative planning of perforator flaps ( Fig. 6.55 ). This modality has clear advantages over Doppler and duplex imaging. It can provide a highly detailed depiction of the perforating vessels, including their intramuscular portion. The caliber can be assessed, as well as the subcutaneous branching patterns. A precise surgical plan can therefore be devised, taking into account the best perforator and alternatives, reducing the risk of intraoperative errors and minimizing operating time. Choosing the most remotely located perforator from the SGA pelvic exit point will provide a longer pedicle, making the insetting and shaping of the flap easier.
Preoperative Marking
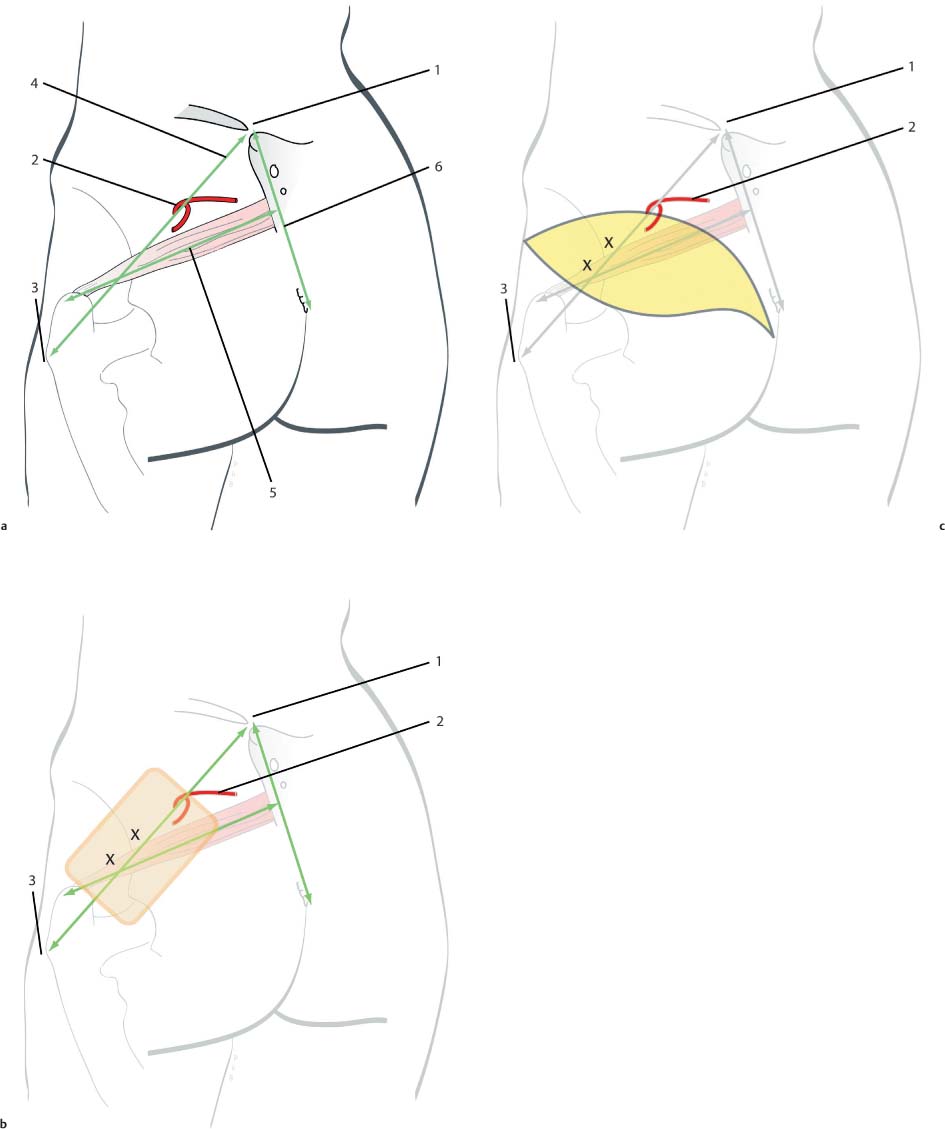
The patient is placed in the ventral decubitus position. The following marks are important ( Fig. 6.56 ):
The site at which the SGA enters the buttock is identified at the junction of the proximal and middle thirds of a line connecting the posterior superior iliac spine (PSIS) to the apex of the greater trochanter of the femur. A line is then drawn between the PSIS and the coccyx.
The position of the piriformis is located by joining the middle of the PSIS–coccyx line to the superior edge of the greater trochanter.
As the SGA supplies the suprapiriform portion of the gluteus maximus, only perforators located cranial to the piriformis are taken into consideration. The position of the relevant perforators is identified and marked on the skin.
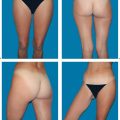
Next, the flap design is drawn. It should be centered over the selected perforator as much as possible. Although the skin island can be oriented in any way, we use a fusiform shape oriented parallel to the bikini line. The incision ends medially with a fishtail design to avoid dog-ear formation. The oblique orientation of the flap results in a well-concealed scar and minimizes deformity after closure of the donor site. The flap can be up to 30 cm long and 13 cm wide. If a large volume of tissue is needed, it is preferable to extend the flap medially, to avoid lateral contour deformities.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree