32 Cancer Surveillance After Partial Breast Reconstruction
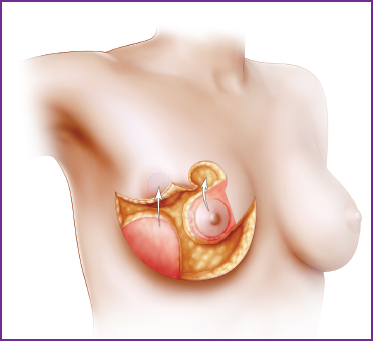
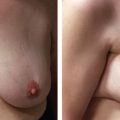
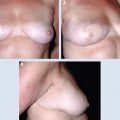
Partial breast reconstruction has been shown to improve cosmetic outcomes after BCT, and the indications for its use have broadened. However, many surgeons are concerned that these reconstructive procedures–whether through parenchymal rearrangement or flap reconstruction—alter the architecture and affect patterns of local recurrence. Because BCT does preserve the majority of breast tissue, the importance of accurate surveillance for recurrent cancer cannot be underestimated. The recurrence rate after these oncoplastic techniques has been discussed in previous chapters. The all-important question is: “Does partial breast reconstruction impair our ability to accurately screen for breast cancer recurrence in these patients?” It is safe to say that for a reconstructive procedure to be accepted by the medical community or by patients we need to demonstrate that it does not in any way impede the detection of a recurrent lesion. Despite the impact this information has on the acceptance of oncoplastic-type techniques, the actual data are limited.
The advantages of partial breast reconstruction are limited in terms of postoperative surveillance. One potential advantage is that the physical examination and mammography might be easier after oncoplastic reduction techniques, because the size of the breast is smaller. The potential difficulty associated with partial breast reconstruction is that additional surgeries might cause scarring, fat necrosis, inclusion cysts, or other findings, which could be misinterpreted as a recurrence on radiologic imaging or a physical examination. Changes caused by radiation further confound the situation. All of this information needs to be considered when managing these patients postoperatively.
One main concern with partial breast reconstruction is that it could change the patterns of recurrence or impair screening of the actual tumor bed. It is helpful for the medical team to be aware of what was done to the reconstructed breast so that they know where to look and what anatomic changes can be expected in that location. Another helpful addition is to place metal clips in the tumor bed to facilitate its identification during postoperative screenings. The team needs to be educated on the differences that might be present and develop an effective surveillance protocol.
Surveillance Options
The tools available for postoperative surveillance include the physical examination, radiologic imaging, and tissue sampling. About 30% to 50% of local recurrence is detected by mammography alone or a combination of mammography and the physical examination. Clinical and mammographic surveillance are complementary in helping to detect a local recurrence following BCT. When a local recurrence is detected, it is usually in the form of a mass, calcifications, or both. The tumor recurrence is typically found at the lumpectomy bed (true recurrence), adjacent to it (marginal miss), or elsewhere in the treated breast. 1 The greatest emphasis on surveillance is in the first 5 years, because the risk of recurrence is the highest during this time; however, the risk persists.
Physical Examination
It is important that the patient and physician are familiar with the various changes that might occur after partial breast reconstruction, and that both have an understanding of the reconstructive procedure performed. Regardless of which reconstructive technique was used, there is a potential for scarring, fat necrosis, partial flap loss, or radiation fibrosis, which might present as a palpable mass or thickening within the breast. Breast swelling, skin edema, and areas of firmness are considered normal in the early postoperative period. These should be followed closely and be accurately documented. They often improve with time as the swelling resolves and the breast takes on a more natural appearance and feel. Epidermal inclusion cysts and areas of fat necrosis may occur after reduction mammaplasty and need to be addressed. 2 Areas of firmness that persist are most likely fat necrosis or surgical scarring. However, it is safer to obtain a tissue diagnosis to rule out a recurrence. New masses need to be worked up appropriately. The patient needs to be educated on breast self-examination and report any suspicious findings or changes to the team.
Physical Examination Postoperative Surveillance
The physical examination should continue postoperatively as follows:
The patient needs to perform a monthly breast self-examination.
The physician needs to record the history and perform a physical examination every 3 to 6 months for the first 3 years; every 6 to 12 months for years 4 and 5; and annually thereafter.
Radiologic Imaging
Currently, no consensus is available regarding appropriate imaging protocols after oncoplastic procedures. Regardless of the imaging modality used, the main objectives are to (1) exclude residual disease, (2) rule out a recurrence, (3) establish a new baseline, and (4) evaluate for metachronous disease—all while minimizing misinterpretation. For nonpalpable lesions, imaging-directed preoperative localization is indicated.
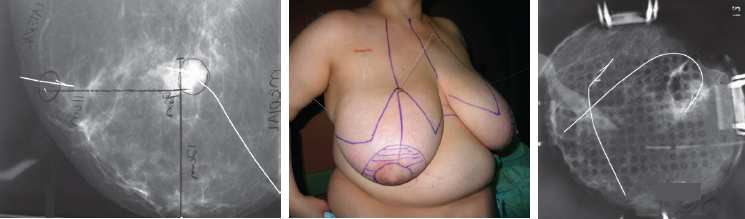
Specimen radiography can ensure the removal of the target lesion, confirm that the specimen includes calcifications, and aid in assessing the margin status. 3 Using a prearranged system of orienting the submitted specimen often allows the radiologist to not only comment on the apparent closeness of a margin, but to specify which margin is affected and to communicate this to the surgeon while the patient is still anesthetized, thus allowing reexcision of that margin.
Assessment of the margins remains a pathologic determination. If there is any question regarding the involvement of margins, the reconstruction should be delayed until the final margin status is determined to prevent having to address this issue after reconstruction.
If malignant calcifications are a component of the lesion, magnification mammography of the surgical bed can be performed a few weeks after surgery, before initiating radiotherapy, to establish a new baseline and to exclude residual malignant calcifications. Ill-defined masses or asymmetries, often seen when lobular carcinoma is the target lesion, may require full-breast mammography and breast MRI to ensure complete removal.
Mammography is less sensitive in the treated breast; it is about 55% to 68% accurate in patients who have undergone BCT. 4 – 7 The surveillance data on combined procedures are limited; however, we do have a reasonable understanding of the radiographic appearance of the conservatively treated breast. Mendelson 9 followed the mammographic changes over time in patients who underwent BCT, and the most frequent findings were skin thickening and breast edema, which occurred in almost 100% of the patients. Six months after treatment, other findings included scarring and fibrosis (50%), fluid collections/seromas (40%), and dystrophic calcifications (10%). Skin thickening, edema, and seromas tend to resolve with time. These probably represent the changes associated with radiation dermatitis and often peak about 6 months after radiotherapy and resolve over 2 to 3 years in most patients.
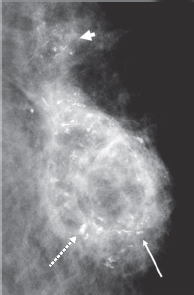
The true nature of calcifications is at times difficult to determine. Magnification of the mediolateral view of the left breast shows rim and lucent-centered calcifications (long arrows) around a low-density area of tissue superior to the lumpectomy site (surgical clip, dashed arrow). These changes are characteristic of evolving fat necrosis. Adjacent, more suspicious, linear and pleomorphic calcifications (arrowhead) required a core biopsy to exclude a recurrent ductal carcinoma in situ. The pathology results indicated fat necrosis with dystrophic calcifications.
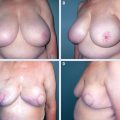
Typical findings after breast-reduction surgery are displaced parenchyma, architectural distortion, skin thickening, and dystrophic calcifications. We wanted to determine whether additional surgery (that is, reduction mammaplasty) at the time of a lumpectomy delayed mammographic stabilization or reduced the sensitivity of this screening tool. 8 The concept of mammographic stability was described by Mendelson 9 and is defined as the time when there is no significant change between two successive mammograms. We evaluated our patients at Emory University Hospital who underwent oncoplastic reduction procedures and compared them with a cohort of risk-matched women who underwent BCT without reconstruction. 10 The average follow-up was about 6 years. The mammographic findings were similar between the groups and included architectural distortion, cysts, and calcifications. There was a slight trend toward longer times to mammographic stabilization in the study group (21.2 months for the BCT group versus 25.6 months for the oncoplastic group, p = 0.23), which is expected given the additional scarring, inflammation, and parenchymal alteration associated with the reconstruction. The time to mammographic stability in the oncoplastic group demonstrated a 95% confidence interval between 20 and 30 months.
To confirm stability, we feel that, based on these data, biannual mammographic screening should be extended until the third postoperative year in patients who undergo partial breast reconstruction.
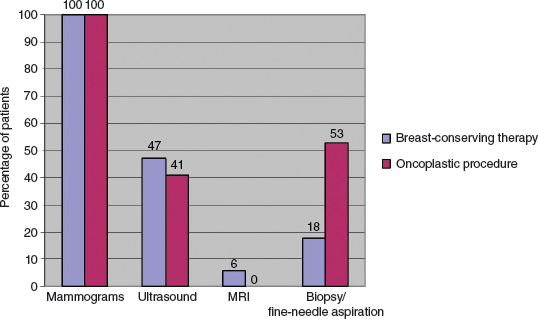
We also performed a greater number of postoperative mammograms and ultrasounds in the oncoplastic group when indexed per each follow-up year. The oncoplastic group averaged 1.28 mammograms per follow-up year, and the BCT-only group averaged 1.21 mammograms per follow-up year (p = 0.38). The oncoplastic group averaged 0.17 ultrasounds per follow-up year compared with 0.09 ultrasounds per follow-up year (p = 0.29) in the BCT-only group. A breast ultrasound examination was performed in around 45% of patients in both groups. The breast density, measured as a percentage (fatty to extremely dense), was used to equate the sensitivity of the test. A sensitivity value from 1 to 4 was assigned (4 = least sensitive in an extremely dense breast: parenchyma content 75% or greater). The average sensitivity value for the group that underwent BCT was 2.67, and for the group that also had reconstruction it was 2.36, showing that the reconstruction did not seem to reduce the sensitivity of a mammographic evaluation.
Surgical manipulation of the breast may also result in a hematoma or seroma, which may resorb with time and be replaced by a fibrotic scar (especially after radiotherapy). This process may result in an area of increased density with irregular and spiculated margins that may also mimic malignancy. The presence of entrapped fat, which often occurs with scar formation, can be evaluated based on clinical suspicion and serial imaging, which may help distinguish these changes from those of cancer.
Calcifications after breast conservation and reconstruction are relatively common and occur in one fourth to one third of patients. 9 , 11 Although the majority of these are benign, some represent a disease recurrence.
Patient Examples

In this 76-year-old woman there was progression of her postsurgical changes over time, with recurrence, after she underwent the oncoplastic removal of a left infiltrating ductal carcinoma. Left mediolateral oblique views were obtained from 3 consecutive years (A). The examination performed 1 year postoperatively (left) shows the lumpectomy site with the initial density and distortion (arrows) in the upper breast. This was less conspicuous in the views from the 2-year (center) and 3-year (right) examinations. Three years postoperatively (right), a 1 cm new focal asymmetry was present (arrowhead). This lesion was a solid, shadowing mass on ultrasound and was confirmed to be an infiltrating ductal carcinoma on the core biopsy (B).
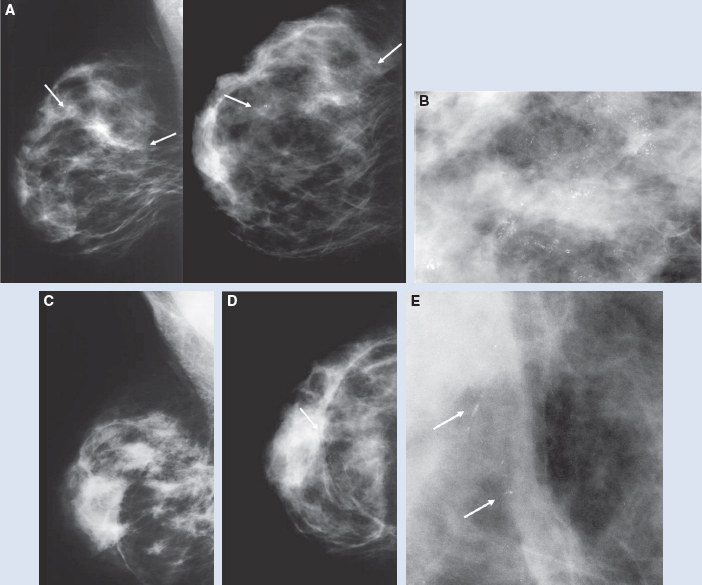
There is a subtle recurrence of ductal carcinoma in situ after an oncoplastic procedure. Mediolateral oblique (left) and craniocaudal (right) views of the right breast show segmentally distributed linear and pleomorphic calcifications in the upper, outer right breast (between the arrows) (A). Their highly suspicious nature was confirmed on a magnified craniocaudal view (B). A postsurgical mammogram obtained 1 year later shows postoncoplastic changes (decreased breast size, mild distortion, and scarring) and a few subtle calcifications (C and D; arrow). A magnified craniocaudal view shows the calcifications (arrows) (E). Ductal carcinoma in situ was confirmed on a core biopsy.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree