24 Orbital Exenteration
Abstract
“Orbital Exenteration” describes the surgical removal of all the soft tissue contents of the orbit (in contrast to enucleation, which removes only the eyeball, leaving the extraocular muscles and other tissues within the orbit). An exenteration is classified as total, subtotal, or extended. In a total exenteration, all of the soft tissues of the orbit and periocular adnexa are removed. In a subtotal exenteration, the eyelid skin is preserved. An extended exenteration involves resection of adjacent structures, including the paranasal sinuses, usually for the management of a sino-orbital malignant tumor. The extent of the surgical resection is dictated by the extent of the disease process. This mutilating procedure is used for the management of a number of benign as well as malignant conditions that are not amenable to other treatment modalities, such as life-threatening infections, severe nonspecific orbital inflammatory disease with intractable pain and blindness, or severe orbital deformities caused by disorders such as neurofibromatosis.
24.1 Introduction
Orbital exenteration is a surgical procedure that involves the removal of all the soft tissue contents of the orbit. An exenteration is classified as one of the following:
Total.
Subtotal.
Extended.
In a total exenteration, all of the soft tissues of the orbit and periocular adnexa are removed. In a subtotal exenteration the eyelid skin is preserved. An extended exenteration involves resection of adjacent structures, including the paranasal sinuses, for the management of a sino-orbital malignant tumor. The extent of the surgical resection is dictated by the extent of the disease process.
This mutilating procedure is used for the management of a number of benign as well as malignant conditions that are not amenable to other treatment modalities.
24.2 Classification
24.2.1 Malignant Disorders
Malignant eyelid tumors with orbital involvement (e.g., basal cell or squamous cell carcinoma)
Malignant eyelid tumors beyond simple surgical excision (e.g., sebaceous carcinoma with extensive conjunctival involvement)
Malignant conjunctival lesions (e.g., extensive conjunctival melanoma)
Orbital invasion by malignant paranasal sinus tumors
Primary malignant orbital tumors (e.g., lacrimal gland carcinomas)
24.2.2 Non-malignant Disorders
Benign orbital tumors (e.g., aggressive orbital meningioma)
Life-threatening infection (e.g., sino-orbital mucormycosis)
Severe nonspecific orbital inflammatory disease with intractable pain and blindness
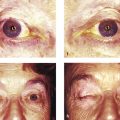
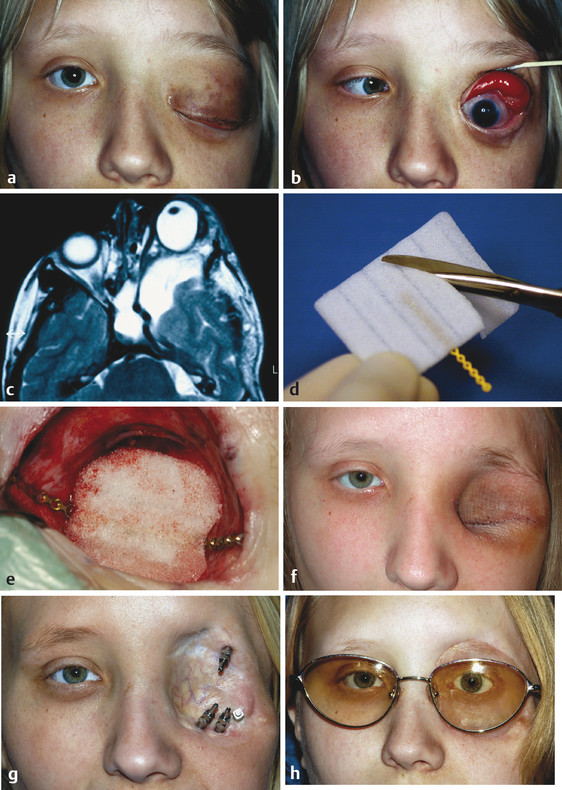
Severe orbital deformity (e.g., neurofibromatosis) (Fig. 24‑1)
End-stage socket contracture
Patients who are to undergo an orbital exenteration for malignant disease should be managed by a multidisciplinary team, which may include the following specialists:
Orbital surgeon.
Ear, nose, and throat (ENT) surgeon.
Plastic surgeon.
Neurosurgeon.
Radiologist.
Radiotherapist.
Oncologist.
Anesthetist.
Mohs’ micrographic surgeon.
Pathologist.
Ocularist.
Psychologist.
Oculoplastic nurse practitioner.
24.3 Preoperative Evaluation
The preoperative evaluation comprises the following:
A review of paraffin-fixed histological sections.
A thorough ophthalmic examination of both eyes.
A general physical examination of the patient.
A review of radiological imaging.
Mohs’ micrographic surgery may be warranted to gain clearance of the skin margins in basal cell carcinomas (BCCs) and squamous cell carcinomas (SCCs).
Because the operation may be associated with significant blood loss, typed and cross-matched blood should be made available. The patient should have a full blood count, platelet count, and coagulation profile performed preoperatively. All antiplatelet agents should be discontinued 2 weeks before surgery. The assistance of a hematologist should be sought for the management of any patient who takes anticoagulants.
24.4 Preoperative Patient Preparation
Most orbital exenterations are performed as elective procedures. The patient should receive appropriate counseling by senior, experienced members of the oculoplastic team about the diagnosis; the prognosis; the nature of the surgery; and its goals, risks, and potential complications. The patient should be warned about inevitable and permanent anesthesia of the forehead, temple, and lateral canthus postoperatively. The options for surgical reconstruction of the exenterated socket should be discussed and determined according to the patient’s wishes as well as the patient’s age and general health. In determining the method of orbital reconstruction in the case of patients with a malignancy, the likelihood of recurrent disease must be taken into consideration.
The patient must be carefully prepared for the ensuing cosmetic deformity. Photographs of other patients who have undergone a similar exenteration may be helpful to use along with samples of typical orbital prostheses or methods of cosmetic camouflage.
The oculoplastic nurse practitioner should be on hand to explain his or her role in postoperative wound care and in coordinating aftercare in the community after discharge from hospital. The pathologist should be consulted well in advance preoperatively and the possible need for frozen-section control of the resection margins discussed and organized.
24.5 Anesthesia
General anesthesia is preferable for this procedure, although local anesthesia with intravenous sedation can be used for patients who are medically unfit. For local anesthesia, 5 mL of 0.5% bupivacaine with 1:200,000 units of adrenaline are given with hyaluronidase as a retrobulbar injection. In addition, an additional 10 to 12 mL of 0.5% bupivacaine with 1:200,000 units of adrenaline are given as a series of subcutaneous injections around the orbital margin and as specific nerve blocks around the supratrochlear, supraorbital, infratrochlear, anterior ethmoidal, infraorbital, zygomaticofacial, and zygomaticotemporal nerves (Fig. 24‑2). If the patient is under general anesthesia, 10 to 12 mL of 0.5% bupivacaine with 1:200,000 units of adrenaline are given with hyaluronidase as a series of subcutaneous injections around the orbital margin, and the same retrobulbar injection is given to try to block the oculocardiac reflex.
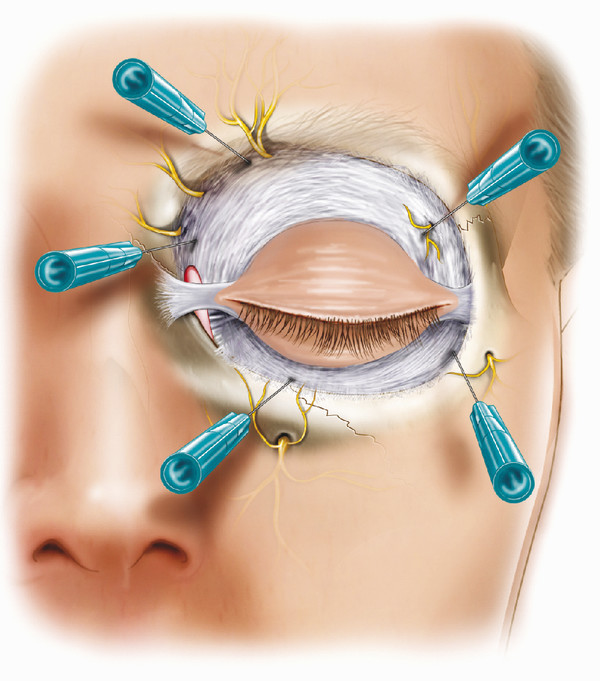
The anesthetist should be warned that the dissection of the socket can provoke the oculocardiac reflex, inducing a severe bradycardia and occasionally asystole. The anesthetist may wish to use glycopyrrolate or atropine before the dissection.
24.6 Applied Surgical Anatomy
The surgeon must have a thorough knowledge of orbital anatomy to facilitate an expeditious exenteration that avoids excessive hemorrhage and other potentially serious complications. In particular, the surgeon should be aware of the following:
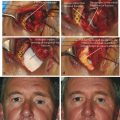
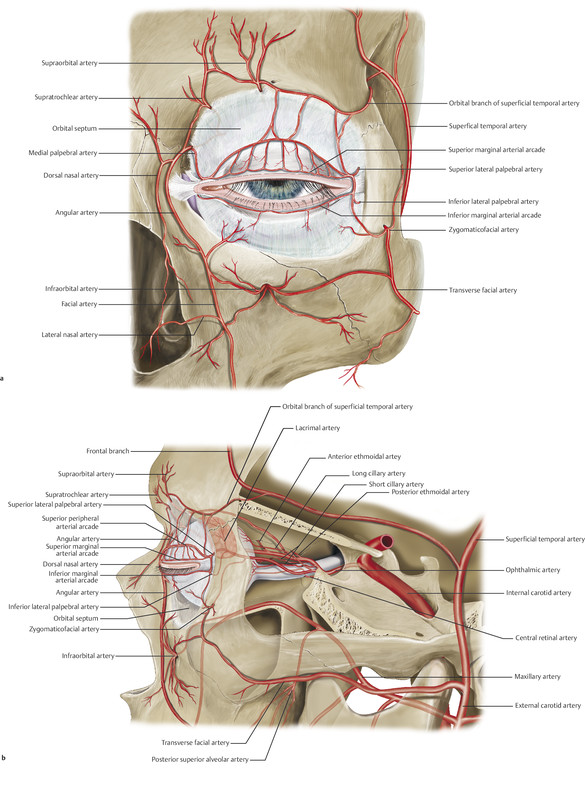
The anatomical position of all the major orbital blood vessels (Fig. 24‑3a,b).
The points of increased periosteal attachment within the orbit.
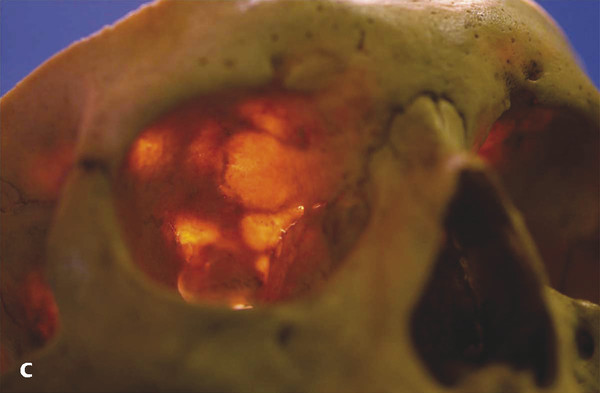
The potential weak areas in the bony orbital walls (e.g., the orbital roof in the elderly) (Fig. 24‑3c).
The position of the superior and inferior orbital fissures.
For more detail, review Chapter 2.
24.7 Exenteration
The patient’s computed tomography (CT) and/or magnetic resonance imaging (MRI) scans should be placed on the viewing screen in the operating room to be referred to if necessary during the course of the surgery.
Key Point
The surgeon must identify the correct side to be exenterated before the induction of general anesthesia. The surgeon must mark the correct side and be personally responsible for the prepping and draping of the patient. The surgeon must also ensure that the fellow eye is instilled with a preservative-free ointment (e.g., Lacri-Lube or Xailin Night ointment) and is taped closed and fully protected from inadvertent injury during the course of the surgery.
24.7.1 Total Exenteration
Surgical Procedure
The proposed skin incision is marked with gentian violet around the orbital margin. The eyebrow is normally preserved unless involved in the malignant tumor.
The eyelids are sutured together with two 2–0 silk sutures and the sutures held with an artery clip.
Next, the skin and superficial subcutaneous tissues are incised with a Colorado needle.
The skin edges are firmly retracted, and the dissection is carried down to the periosteum of the orbital rim using the Colorado needle. Careful attention is paid to hemostasis with additional use of bipolar cautery.
The periosteum at the orbital margin is then incised with the Colorado needle.
The periosteum is elevated from the margins of the orbit and into the orbit with a Freer periosteal elevator.
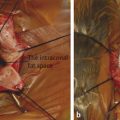
Jaffe retractors are placed around the orbit to retract the soft tissues (Fig. 24‑4a).
The periosteum is elevated from the orbital walls, beginning superotemporally. Care is taken to keep the periosteum intact to avoid a prolapse of orbital fat.
The Freer elevator should be used with great care along the orbital roof, because dehiscences in the bone are common in the elderly. This dissection must not be performed blind, because this risks damage to the dura mater with a subsequent cerebrospinal fluid (CSF) leak. It is important to avoid the use of monopolar cautery along the orbital roof, because this also risks causing a CSF leak in the presence of any bony defects.
The dissection is continued inferotemporally.
The lateral canthal tendon is incised with the Colorado needle.
The zygomaticotemporal and zygomaticofacial vessels are cauterized as they are encountered using bipolar cautery. Any bleeding from the bone is managed with bone wax.
The dissection is then continued across the floor of the orbit. A constant vessel is encountered approximately 8 mm posterior to the inferior orbital margin. This is cauterized.
Great care is taken to avoid inadvertent injury to the infraorbital neurovascular bundle. The infraorbital nerve may lie exposed along the floor of the orbit.
The Colorado needle is used to dissect tissue across the anterior portion of the inferior orbital fissure.
The dissection then continues from the superolateral orbit to the superomedial orbit. The supraorbital and supratrochlear vessels are cauterized using bipolar cautery and transected.
As the dissection approaches the medial canthus, the angular vessels are cauterized using bipolar cautery.
The anterior limb of the medial canthal tendon is incised with the Colorado needle and reflected off the underlying bone with a Freer periosteal elevator.
The lacrimal sac is rotated posteriorly and laterally, and the posterior limb of the medial canthal tendon is incised with the sharp end of the Freer periosteal elevator.
The nasolacrimal duct is severed using the Colorado needle.
The periorbita is raised from the medial orbital wall with the Freer elevator, taking great care not to fracture the lamina papyracea.
The anterior and posterior ethmoidal vessels are identified as they pass through the periosteum to their respective foramina and are cauterized.
Superomedially, the trochlea is elevated along with the periosteum.
Once the dissection has approached the apex of the orbit, the periosteum is incised with the Colorado needle medially and laterally.
Two large curved artery clips are applied across the apical orbital tissues, one medially and one laterally.
Curved enucleation scissors are used to excise the tissues anterior to the clips while pulling anteriorly on the traction sutures. Alternatively, a snare may be used (Fig. 24‑4b).
The extent of the exenteration performed is dictated by the clinical requirements. The exenteration can be modified to a less extensive procedure or to a more radical resection that involves the removal of orbital walls and paranasal sinuses.
Bipolar cautery is applied to the stump of tissue at the orbital apex (Fig. 24‑4c,d).
Bone wax may be required for any bleeding vessels that perforate the orbital walls.
Gelfoam soaked with thrombin is applied to the socket, and swabs moistened with 1:1,000 adrenaline are placed over this. Pressure is applied for 5 minutes. If excessive bleeding occurs, Floseal, a very effective hemostatic sealant, may be applied to the apex of the orbit. (Its use must, however, be avoided in patients who have a known allergy to materials of bovine origin.)
If the indication for exenteration is a malignancy, the socket is carefully inspected for any residual tumor tissue. The adequacy of resection may be judged with the aid of frozen-section control. The resection of additional orbital apical tissue may be required.
The stump of the nasolacrimal duct is oversewn with interrupted 5–0 Vicryl sutures if possible. Alternatively, the stump is thoroughly cauterized.
If the socket is to be left to heal by secondary intention or if a split-thickness skin graft is used (see below), antibiotic ointment is instilled, and either an Aquacel dressing or an Allevyn Cavity dressing is placed into the socket and an occlusive secondary dressing comprising sterile eye pads and Opsite Flexifix is applied along with a pressure bandage.
Key Point
The subperiosteal dissection along the orbital roof must not be performed blind, because this risks damage to the dura mater with a subsequent CSF leak. It is important to avoid the use of monopolar cautery along the orbital roof, because this also risks causing a CSF leak in the presence of any bony defects.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree