22 Enucleation and Evisceration
Abstract
“Enucleation and Evisceration” discusses the removal of an eyeball (enucleation) or of the contents of the eyeball (evisceration) and the reconstruction of the anophthalmic socket, which still pose a considerable challenge for ophthalmic surgeons despite many advances in orbital implant materials. Good results from such surgery are not easy to achieve consistently, and a poor result can have profound psychological implications for the patient for the rest of his or her life. The preoperative counseling of a patient who requires an enucleation or evisceration demands time and considerable compassion on the part of the ophthalmic surgeon. Close collaboration between the ophthalmic surgeon and the ocularist is essential and should commence preoperatively whenever possible. The goals of an enucleation or an evisceration are to obtain a healthy and comfortable socket free of discharge that can be fitted with a stable ocular prosthesis that mimics the fellow eye in appearance and movement; a symmetrical appearance without enophthalmos or upper eyelid sulcus deformity; no upper or lower eyelid malposition; and normal eyelid closure over the ocular prosthesis. To achieve these goals, the surgery must be approached in the same aseptic manner as any intraocular procedure and must be performed meticulously with strict attention to hemostasis.
22.1 Introduction
The removal of an eye and the subsequent management of the anophthalmic socket still pose a considerable challenge for ophthalmic surgeons despite many advances in orbital implant materials. Good results from such surgery are not easy to achieve consistently, and a poor result can have profound psychological implications for the patient for the rest of his or her life. The preoperative counseling of a patient who requires an enucleation demands time and considerable compassion on the part of the ophthalmic surgeon. Close collaboration between the ophthalmic surgeon and the ocularist is essential and should commence preoperatively whenever possible.
The goals of an enucleation or an evisceration are to achieve the following:
A healthy and comfortable socket free of discharge that can be fitted with a stable ocular prosthesis that mimics the fellow eye in appearance and movement.
A symmetrical appearance without enophthalmos or an upper eyelid sulcus deformity.
An absence of an upper or a lower eyelid malposition.
Normal eyelid closure over the ocular prosthesis.
To achieve these goals, the enucleation must be approached in the same aseptic manner as any intraocular procedure and must be performed meticulously with strict attention to hemostasis.
22.2 Indications for Enucleation
There are a number of indications for an enucleation:
A blind painful eye, such as after failed retinal reattachment surgery or with rubeotic glaucoma.
A blind unsightly eye.
An intraocular tumor, such as a large choroidal melanoma.
Severe irreparable ocular trauma and a high risk of sympathetic ophthalmia.
It is important to consider alternatives to enucleation. The movement of a blind (or partially sighted) eye or of a microphthalmic or phthisical eye can be more natural than that of an orbital implant, and such an eye may tolerate a cosmetic shell or cosmetic contact lens. A painful eye may respond to simple surgery to relieve the pain, such as cyclodestructive procedures or a conjunctival flap, and it may also then tolerate the fitting of a cosmetic shell or cosmetic contact lens.
22.3 Preoperative Preparation
Most patients undergo an enucleation as an elective procedure. The operation is rarely performed as an emergency. Preoperatively the patient should be advised about the following:
The advantages, disadvantages, risks, and potential complications of an enucleation procedure.
The advantages, disadvantages, risks, and potential complications of the use of any orbital implant.
The implant options.
The options regarding implant wrapping materials.
The choice of anesthesia.
Postoperative pain and its management.
The use of a postoperative compressive dressing.
The use of a temporary suture tarsorrhaphy.
The use of a postoperative conformer.
The likelihood of a temporary postoperative ptosis.
The role of the ocularist and the timing of the fitting of the ocular prosthesis.
Informed consent should be obtained after the patient has had time to consider the options.
The patient should discontinue aspirin and any other antiplatelet drugs, if medically permissible, at least 2 weeks preoperatively. Likewise, anticoagulants should only be altered or discontinued after discussion with the patient’s hematologist. Any bacterial conjunctivitis should be treated preoperatively and topical steroids used to reduce any conjunctival inflammation.
Key Point
Computed tomography (CT) of the orbits and paranasal sinuses should be performed if a patient has previously experienced orbital trauma to exclude the possibility of a missed orbital wall blowout fracture. If a fracture is present and is of significant size, this should be repaired at the time of the enucleation and before the placement of the orbital implant.
22.4 Orbital Implant Materials
Enucleation (or evisceration) of an eye creates an orbital volume deficit that varies from patient to patient. This necessitates the replacement of the equivalent spherical volume of approximately 6 to 7 mL (depending on the size of the globe). This can be partially compensated for by the placement of an orbital implant. An 18-mm spherical implant has a volume of only 3.1 mL. The average ocular prosthesis (artificial eye) must then be larger than 2 mL to make up the difference. The advantage of using a larger orbital implant is to keep the ocular prosthesis as light as possible. This will reduce the incidence of inferior displacement of the lower eyelid by gravitational force on the ocular prosthesis. This advantage, however, must be balanced against the disadvantages of the use of a larger implant:
Increased pressure on the conjunctival wound with a risk of dehiscence and implant exposure or extrusion.
A longer period required for vascularization of a porous implant.
Insufficient room for placement of a motility peg in the case of a porous implant.
Insufficient room for the ocularist to fit an ocular prosthesis with sufficient thickness to adequately mimic the presence of an anterior chamber.
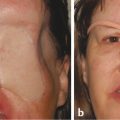
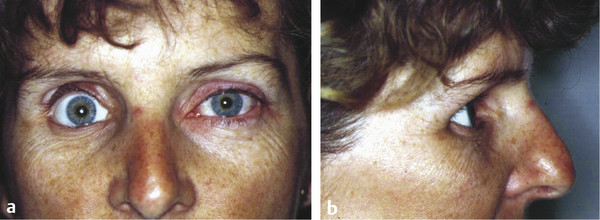
If no orbital implant is placed, or if the implant is of insufficient size, the ocular prosthesis will have to be made larger than is desirable to reduce the volume deficit, which manifests itself by an enophthalmic appearance and an upper eyelid sulcus deformity. The lower lid eventually becomes stretched, the ocular prosthesis becomes inferiorly and posteriorly displaced, the levator palpebrae superioris muscle loses its fulcrum of action, and the upper eyelid sulcus deformity becomes more exaggerated. The patient then exhibits features of postenucleation socket syndrome (PESS) (Fig. 22‑1). In the absence of an orbital implant, a posterior rotation of the levator muscle and superior rectus complex and the superior orbital fat occurs, with an anterior rotation of the inferior rectus muscle and the inferior orbital fat. This may result in a retraction of the upper lid in some patients. It is also responsible for the backward tilt seen with the ocular prosthesis.
22.4.1 Primary Orbital Implant
The ideal time for orbital implantation is at the time of enucleation or evisceration unless primary implantation is contraindicated, such as in the case of severe ocular and orbital trauma or infection. The overall results of primary enucleation or evisceration are superior, and there is a much-reduced need for subsequent surgical procedures.
Controversy has raged about the use of evisceration versus enucleation. Some ophthalmologists prefer evisceration to enucleation because, in the hands of a general ophthalmologist, it may offer a more functional and cosmetically acceptable orbit compared with enucleation. Evisceration tends to produce less disruption of the orbital tissues and the physiological dynamics of muscle function, and the orbital volume can be maintained very close to its original state. It has the advantage that it can be performed very quickly under local anesthesia with sedation and is therefore ideal for elderly patients with blind painful eyes. It should be noted, however, that evisceration, although a faster and simpler surgical procedure, has the following potential disadvantages:
Dissemination of an unsuspected intraocular tumor is possible (all globes with opaque media should be subjected to an ultrasound examination before surgery).
An inadequate pathological specimen is provided.
A phthisical globe will not accept an adequately sized implant.
There is concern about the possible risk, albeit small, of sympathetic ophthalmia.
Key Point
All globes with opaque media should be subjected to an ultrasound examination before undergoing an evisceration.
22.4.2 Secondary Orbital Implant
An implant can be inserted secondarily into the anophthalmic socket, but the surgical procedure is more difficult and the results are less predictable. Patients undergoing secondary orbital implantation are more likely to require additional surgical procedures to address a residual volume deficit, conjunctival adhesions or cysts, and eyelid malpositions. Similarly, the removal of an extruding, exposed, or migrated implant with an implant exchange can be difficult.
22.4.3 Choices of Orbital Implant
Over the years, many different implant materials have been used, the first being glass. Many materials have followed, including cartilage, fat, bone, cork, aluminum, wood, silk, ivory, and paraffin. Many of these orbital implants were associated with numerous problems and have been abandoned. Until the arrival of hydroxyapatite, bioceramic, and porous polyethylene, the most commonly used materials were acrylic and silicone, with the most common configuration being spherical.
Orbital implants can be classified as nonintegrated or integrated.
Non-Integrated Implants
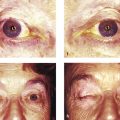
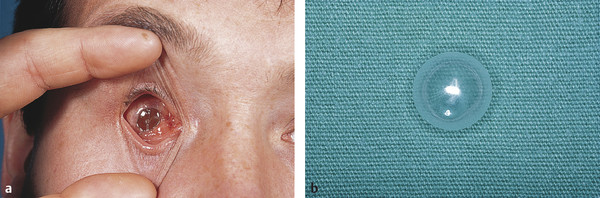
Nonintegrated implants have no direct attachments to the extraocular muscles and are usually single spheres of inert material (silicone or acrylic) buried beneath the conjunctiva and Tenon’s capsule in the muscle cone. The rectus muscles may or may not be incorporated into the soft tissue closure anterior to the implant. Such implants may be inserted behind the posterior layer of Tenon’s capsule within the intraconal fat space. Such implants may migrate within the orbit, causing secondary problems with fitting and stability of the ocular prosthesis, and can extrude many years after their insertion into the socket (Fig. 22‑2).
Integrated Implants
Integrated orbital implants may be further classified as buried or exposed.
Buried Integrated Implants
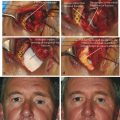
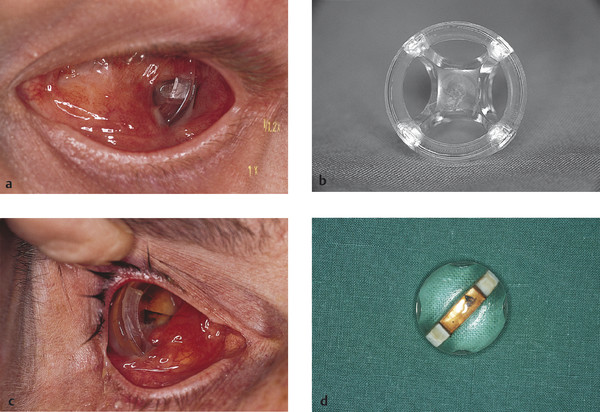
Buried integrated implants may have either a spherical or an irregular shape. The spherical implants may be wrapped in donor sclera, autogenous fascia lata or temporalis fascia, bovine pericardium, or Vicryl or Mersilene mesh, to which the extraocular muscles can be sutured. Some spherical implants may be left unwrapped, and the muscles sutured directly to the implant material, such as the porous polyethylene (Medpor) implant. In buried integrated orbital implants with an irregular surface, muscle attachment is achieved by passing the muscles through tunnels in the implant (Allen or Castroviejo implants; Fig. 22‑3a,b) or through grooves in the implant created by mounds on the anterior aspect (Iowa and Universal implants). The Roper Hall implant has a magnetic strip incorporated into the center of the implant to enable a magnetic coupling to occur with a metallic strip in the prosthesis (Fig. 22‑3c,d). Although this implant is no longer used, patients who were implanted with this device in the 1970s and 1980s are still seen in clinics.
Exposed Integrated Implants
Exposed integrated implants have the muscles directly attached to the implant, and a portion of the implant is exposed to the outside environment (an Arruga implant). The exposed portion is in the form of a projection or an indentation that permits the implant to be directly coupled to a prosthetic eye with its posterior projection (Fig. 22‑4).
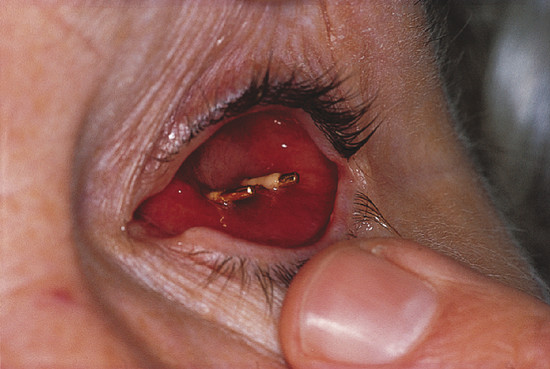
Although these implants provided excellent motility, this benefit was outweighed by their disadvantage of chronic infections and extrusion. Such implants have been abandoned.
Current Orbital Implants
Eight orbital implants are currently used:
Hydroxyapatite implant.
Porous polyethylene implant (Medpor).
Bioceramic implant.
“Baseball” implant.
Simple acrylic sphere implant.
Simple silicone sphere implant.
Universal implant.
Dermis fat graft.
The most popular orbital implants available today are the hydroxyapatite implant, the porous polyethylene (Medpor) implant, and the aluminum oxide (bioceramic) implant. Hydroxyapatite is an inorganic salt of calcium phosphate similar to the inorganic portion of normal human bone. The hydroxyapatite implant gained enormous popularity following its introduction in the 1990s and rapidly became the implant of choice among many leading oculoplastic surgeons worldwide (Fig. 22‑5).
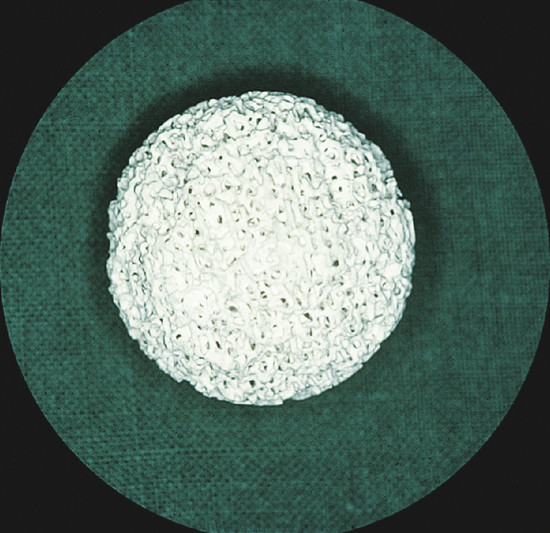
A synthetic form of hydroxyapatite is also available. Medpor has gained in popularity along with the synthetic bioceramic implant (Al2O3), which is also purported to offer yet further advantages over other porous implants, with even better biocompatibility. These implants are all available in 14 to 22 mm sizes.
These materials have the capacity to develop complete fibrovascular ingrowth. The time taken for this to occur depends on a number of the following factors:
The size of the implant.
The use of the implant—as a primary, secondary, or exchange implant.
The wrapping material used.
The porous hydroxyapatite and bioceramic implants can be directly coupled with the ocular prosthesis to improve movement by means of a small methylmethacrylate peg, which fits into a hole drilled into the buried implant. The conjunctiva will grow down the sides of this drilled hole only if the implant is fully vascularized. Alternatively, a titanium-sleeved peg can now be screwed into the implant after creating a central guide hole with a series of free needles of increasing size. Likewise, a titanium motility peg system is available for the Medpor implant. The use of motility pegs has decreased dramatically over recent years, because most surgeons have experienced problems with their use, including foreign body granulomas, recurrent inflammation, and discharge.
The time to complete vascularization varies from 4 to 12 months. The vascularization of the implant may be determined by CT with contrast, magnetic resonance imaging (MRI) with gadolinium, or a technetium-99 m bone scan. These are not accurate, however, and it is preferable to observe a minimum safe waiting period before proceeding with the insertion of a motility peg: that is, 6 months for primary implants and 12 months for secondary implants.
These implants have been heavily marketed on the basis of a number of purported advantages over other implants.
Advantages
Once the implant has become vascularized, it has a reduced risk of extrusion.
The risk of migration of the implant within the orbit is reduced.
The implant permits better movement of the ocular prosthesis.
The motility peg allows the weight of the ocular prosthesis to be borne by the implant rather than by the lower eyelid, reducing the chances of instability of the artificial eye and the need for lower eyelid tightening procedures later.
The implant is quickly and easily inserted and has a low complication rate in the hands of an experienced surgeon.
These purported advantages are not, however, completely borne out in clinical practice.
Disadvantages
The implant has a number of disadvantages. These include the additional costs involved: the cost of the implant, the requirement and expense of the second stage procedure to place a motility peg, and the required modifications to the ocular prosthesis. Additional expense may also be incurred if scans are used to determine whether the implant is vascularized and safe to drill. A small proportion of patients will require management of minor complications, such as removal of foreign body granulomas associated with the motility peg.
22.4.4 Results
The results of primary implantation tend to be very good, and an implant exposure rate of less than 1% can be achieved with meticulous surgery. The results of secondary implantation are less predictable, because the socket anatomy has been disrupted and the eye muscles are retracted, scarred, and difficult to locate at the time of surgery. The results of implant exchange surgery vary depending on the type of implant that has to be removed and whether this has become exposed. In experienced hands, however, the results are usually good, but the degree of movement is very variable from patient to patient.
22.4.5 Rationale for Implant Use
Hydroxyapatite is a relatively expensive implant material. The successful use of this material in most surgeons’ hands appears to require the use of a wrapping material (unless used in conjunction with an evisceration) that has several disadvantages:
Adds expense (Vicryl mesh).
Carries a risk for viral disease transmission (donor sclera).
Incurs further surgical morbidity (autologous fascia).
The same applies to the bioceramic implant.
The early complication rate of hydroxyapatite implants was higher than that of other materials. Particular problems with implant exposure have been the subject of a number of publications, and the potential advantages with regard to late complications, such as migration and extrusion, await the test of time. Their main advantage lies in their ability to accept a motility peg to enhance movement of the ocular prosthesis. However, the use of a motility peg has greatly diminished because of a high incidence of complications. The assumption is, therefore, that most patients are satisfied with the initial motility achieved. Nonetheless, there is no evidence that when similar techniques are used, unpegged hydroxyapatite implants have superior motility to acrylic sphere implants. Furthermore, there is no anatomical basis for this to be so. Although the superior motility of the pegged hydroxyapatite implant is continually alluded to, there has yet to be a double-blind study to confirm this. There is little evidence to support the use of this more expensive implant in patients who do not wish to undergo the extra time and expense required to fit a motility peg. It seems reasonable to reserve the use of the hydroxyapatite implant or other porous implants for patients who desire the enhanced motility the peg offers (in full knowledge of what is involved—including the increased incidence of severe postoperative pain).
Concern has also been raised as to whether the hydroxyapatite system of motility enhancement will stand the test of time, and we can only wait and see whether such a system is sustainable over a lifetime.
Similar criticism can be leveled at the Medpor implant, although this implant does offer the advantage that it can readily accept direct suturing, which obviates the need for a wrapping material. The implant is smooth, in contrast to the rough brittle surface of the hydroxyapatite implant, although the hydroxyapatite implant is now available with a smooth surface coating. Synthetic hydroxyapatite implants have also been developed with a smooth anterior surface, with preformed drill holes and suture posts to enable direct suturing, and synthetic hydroxyapatite implants are also available prewrapped in Vicryl mesh.
The ability of the Medpor implant to accept direct suturing without the necessity for a wrapping material is certainly an attractive quality. Long-term results of the placement of motility pegs in Medpor implants are not yet available, however, and concerns have been raised about the implant’s pore size and completely porous nature as well as its hydrophobic surface characteristics. There are reports of implant exposure requiring further surgery, which therefore raises concerns about the wisdom of using the implant without a wrapping material as a preventative measure. Modifications of the implant shape have been described to reduce the problems of residual upper lid sulcus deformity. Some surgeons have placed this implant with a motility peg already inserted. This is then “externalized” later. This fails to recognize, however, that the implant can alter position postoperatively and that the peg placement should be determined by the ocularist and not by the surgeon.
22.4.6 Alternative Implants
There are several alternative implant types:
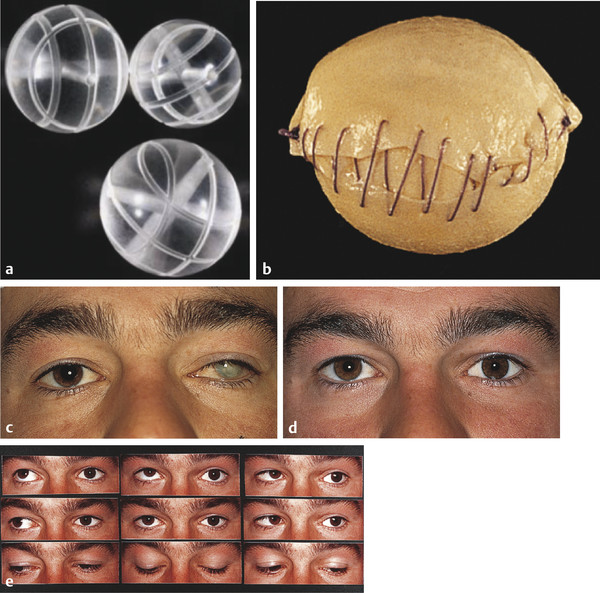
Baseball implant. This remains a simple, inexpensive, but effective primary implant. The acrylic sphere is wrapped in donor sclera (or autogenous fascia or Mersilene mesh) and buried behind posterior Tenon’s fascia (Fig. 22‑6a–e). It is of particular use in patients who do not wish to take advantage of the second stage motility peg placement of porous implants.
Simple acrylic sphere implant. This is easy to place at the time of an evisceration in the elderly, although the use of a subconjunctival patch of sclera or autogenous fascia will reduce the risk of implant exposure and/or extrusion.
Simple silicone sphere implant. Silicone has achieved a certain notoriety with patients and is rarely used. It is used in the same way as a simple acrylic sphere implant.
Universal implant. This remains a primary implant of choice in many centers in the United States. It has a good track record, with few complications, although its placement is not as simple to perform as a spherical porous implant.
Castroviejo implant. This implant is still used in a small number of centers in the United Kingdom but is associated with long-term problems with posterior tilting and extrusion of the implant. Its use is not recommended.
Dermis fat graft. This autogenous implant is still useful for the surgical rehabilitation of sockets which have a conjunctival lining problem in addition to a volume deficit, although some surgeons use such implants for primary cases in which other implants would pose a higher risk of exposure or extrusion, e.g. in acute severe trauma. It is more commonly used as an exchange implant for one that has extruded.
The search for the ideal implant material and design will continue. A surgeon faced with a patient requiring an enucleation or evisceration, or a secondary or exchange implant reconstruction of an anophthalmic socket, currently has a number of implants to choose from and should select the implant most appropriate to the individual patient. The choice will be influenced by a number of factors:
The age and general health of the patient.
Consideration of cost.
The motivation of the patient to undergo a second stage motility peg procedure.
The relative expertise of the surgeon.
The risk factors for implant extrusion.
Key Point
It should be recognized that the following situations represent a higher risk of implant exposure or extrusion:
Previous failed retinal reattachment surgery
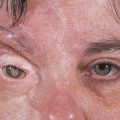
Previous failed glaucoma surgery, with or without the use of antimetabolites (Fig. 22‑7)
Acute trauma
Cicatrization of the eyelids and conjunctiva
A history of previous orbital radiotherapy
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree