2 Applied Anatomy
Abstract
“Applied Anatomy” presents detailed and practical anatomical descriptions as they apply directly to the surgical procedures described throughout this book and to a variety of clinical problems. A sound working knowledge of eyelid, orbital, nasal, and facial anatomy and the underlying neurovascular structures is essential before a surgeon can embark on oculoplastic, oculofacial, orbital, or lacrimal surgery. The procedures in this book are much easier to understand and much more likely to be successful when the surgeon has a good working knowledge of this anatomy.
2.1 Introduction
A sound working knowledge of eyelid, orbital, nasal, and facial anatomy is essential before a surgeon can embark on oculoplastic, oculofacial, orbital, and lacrimal surgery. The surgical procedures described throughout this book are much easier to understand and much more likely to be successful when a good working knowledge of this anatomy has been acquired. This chapter presents detailed and practical anatomical descriptions as they apply directly to these surgical procedures and to a variety of clinical problems.
2.2 The Eyelids
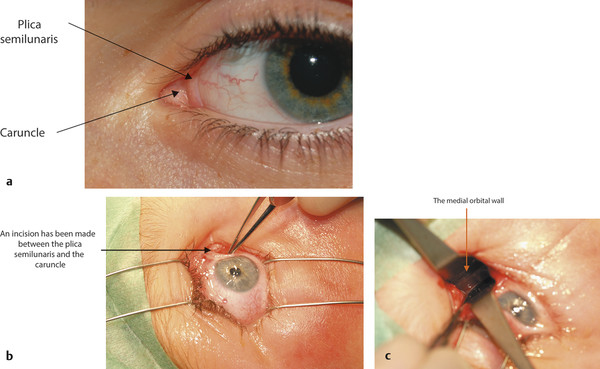
In adults the normal interpalpebral fissure varies in vertical height from 8 to 11 mm. The peak of the upper eyelid usually lies just nasal to the midline of the pupil. The horizontal length of the interpalpebral fissure measures approximately 30 to 32 mm. The angle between the upper and lower eyelids medially and laterally measures approximately 60 degrees. Laterally the eyelids should be in contact with the globe, but medially the eyelids are displaced away from the globe, creating a space—the lacus lacrimalis. Within the lacus lacrimalis lies the caruncle, and immediately lateral to the caruncle lies the plica semilunaris (Fig. 2‑1a). The plane between the caruncle and the plica semilunaris is commonly used to gain access to the medial orbital wall (e.g., to drain a subperiosteal abscess). This is referred to as a transcaruncular approach (Fig. 2‑1b,c).
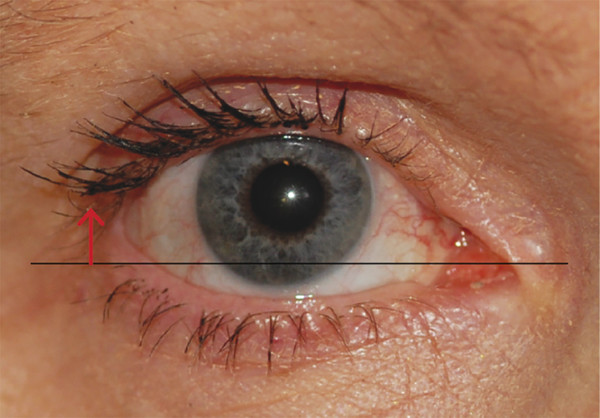
The lateral canthal angle usually lies approximately 2 to 3 mm higher than the medial canthal angle (Fig. 2‑2). This angle and its location should be considered when surgery is undertaken on the lateral canthus (e.g., a lateral eyelid tightening procedure performed with a lower eyelid blepharoplasty).
The eyelid margin is covered by conjunctival epithelium along the posterior half and meets the anterior margin, which is covered with cutaneous epidermis, at the gray line. The meibomian gland orifices lie within the conjunctival epithelium, and the eyelashes emerge within the cutaneous epidermis.
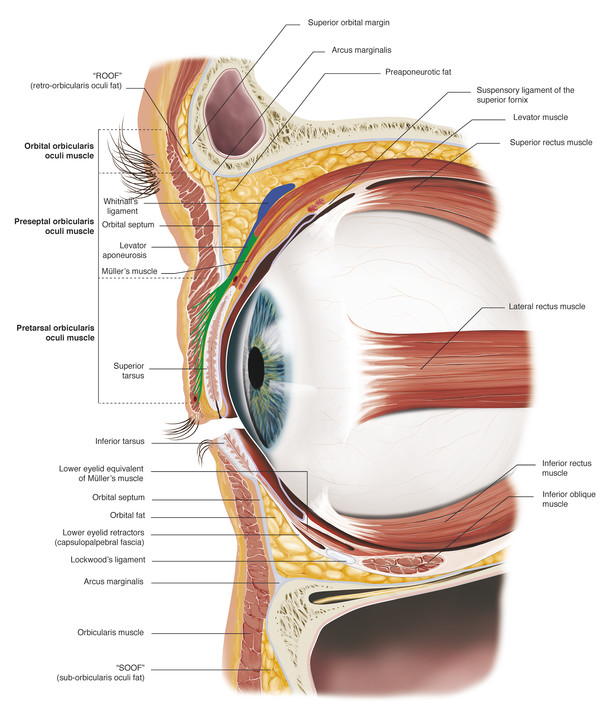
The eyelid can be divided into five structural planes:
The eyelid skin and subcutaneous fascia.
The orbicularis oculi.
The orbital septum.
The eyelid retractor muscles.
The tarsal plates and conjunctiva.
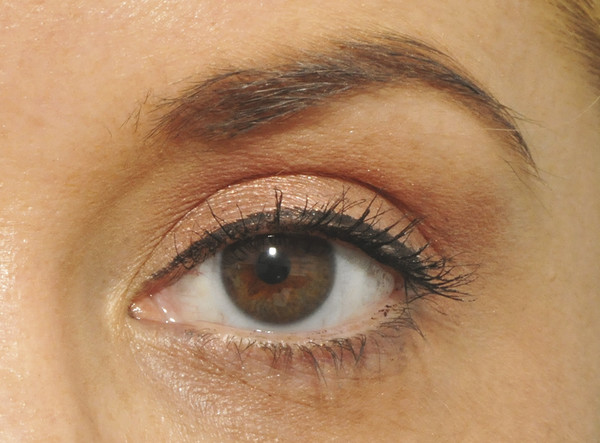
The upper eyelid skin crease is created by attachments from the superficial aspect of the levator aponeurosis into the orbicularis muscle and the subcutaneous tissue (Fig. 2‑3).
The skin crease varies in height and tends to lie 5 to 6 mm above the eyelid margin centrally in men and 7 to 8 mm above the eyelid margin centrally in women. Medially the crease extends to within 3 to 4 mm of the eyelid margin; laterally it lies approximately 5 to 6 mm above the eyelid margin (Fig. 2‑4). This should be considered when marking the desired position of the upper lid skin crease for ptosis surgery or an upper lid blepharoplasty.
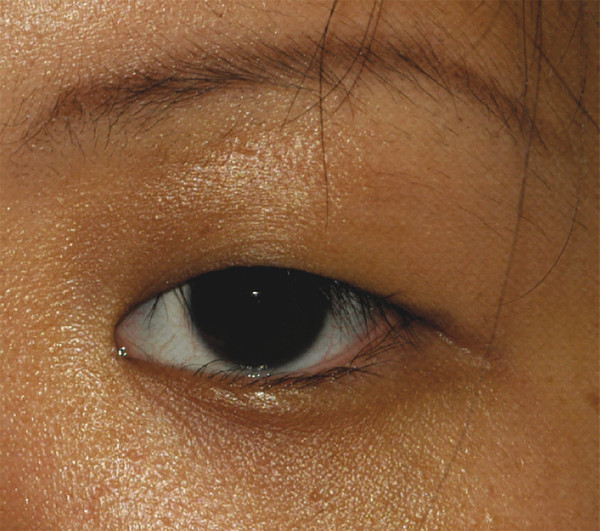
In patients of Asian descent, the upper lid skin crease is typically poorly developed or absent, because the orbital septum inserts onto the levator aponeurosis in a lower position (Fig. 2‑5). This anatomical arrangement allows the preaponeurotic fat to extend further into the eyelid.
2.2.1 The Eyelid Skin
The eyelid skin is the thinnest in the body and unique in having no subcutaneous fat (Fig. 2‑6).
This, along with the color match, makes the upper eyelid an ideal donor site for a small full-thickness skin graft to be used for lower eyelid reconstruction. The skin can be removed easily and quickly and does not need to be thinned, unlike skin harvested from other donor sites. The surgeon must be careful to avoid causing lagophthalmos when using the upper eyelid as a skin graft donor site.
2.2.2 The Orbicularis Oculi Muscle
The orbicularis oculi muscle is divided anatomically into three parts (Fig. 2‑7):
Orbital
Preseptal
Pretarsal
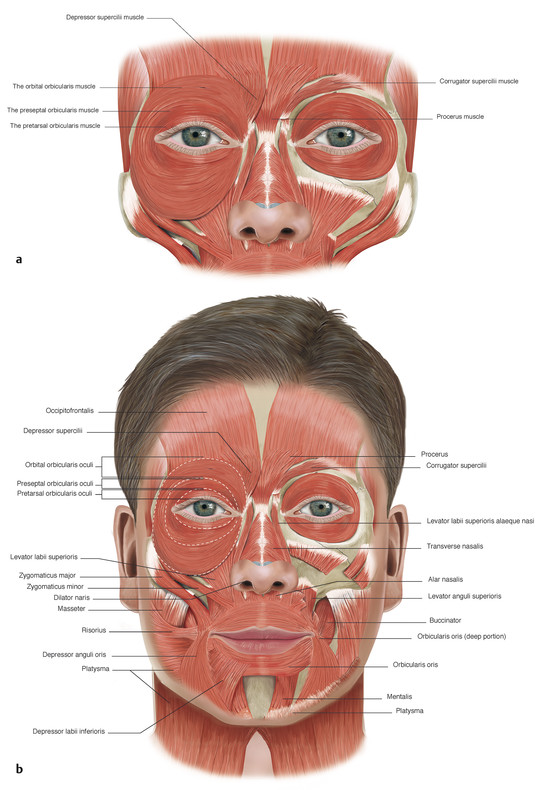
The orbital portion of the orbicularis muscle lies over the bony orbital margins. It arises from the frontal process of the maxillary bone in front of the anterior lacrimal crest, from the orbital process of the frontal bone, and from the medial canthal tendon. The fibers of the orbicularis pass around the orbital margins without interruption at the lateral canthus, and the bony insertions lie just below the points of origin.
The palpebral part of the orbicularis muscle extends from the orbital margins to the margins of the eyelids. The muscle fibers pass circumferentially around the eyelids and are fixed medially and laterally at the medial and lateral canthal tendons. The palpebral part of the orbicularis muscle is further subdivided into the preseptal and pretarsal orbicularis muscles.
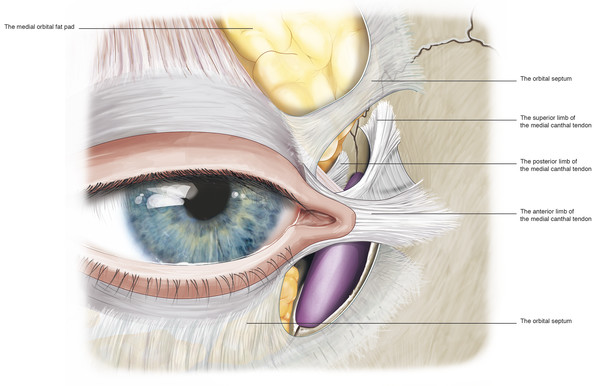
The preseptal part of the orbicularis muscle lies over the orbital septum in the upper and lower eyelids. Its muscle fibers arise in a perpendicular fashion from the upper and lower borders of the medial canthal tendon. In the upper lid, the muscle arises by anterior and posterior heads. The anterior head is a broad extension from the superior surface of the common part of the medial canthal tendon. The posterior head arises from the superior arm and from the posterior arm of the medial canthal tendon. The inferior preseptal muscle arises as a single head from the whole length of the common medial canthal tendon.
The superior limb of the medial canthal tendon fuses to the fundus of the lacrimal sac by a layer of fibrovascular fascia (Fig. 2‑8). When the deep head of the preseptal muscle pulls on the lacrimal sac via this fascia, it contributes to the lacrimal pump mechanism. The preseptal muscles extend in an arc around the eyelids and insert along the lateral horizontal raphe.
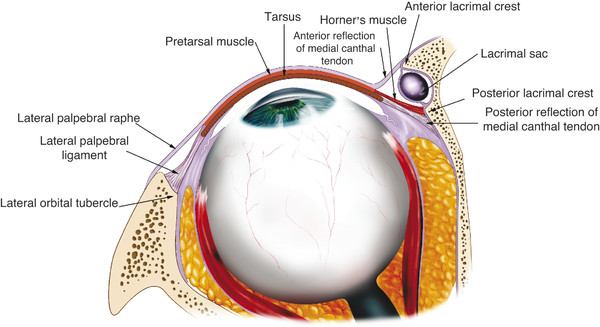
The pretarsal orbicularis muscle lies over the tarsal plates. The fibers of the muscle in both the upper and lower eyelids arise from the medial canthal tendon by means of superficial and deep heads. The superficial heads extend from the eyelid and continue anterior to the upper and lower limbs of the medial canthal tendons. The muscles thicken and surround the limbs of the medial canthal tendon and the canaliculi superiorly, anteriorly, and inferiorly. Contraction of these muscle fibers also contributes to the lacrimal pump mechanism.
Close to the common canaliculus, the deep heads of the pretarsal orbicularis muscle fuse together to form a prominent bundle of muscle fibers referred to as Horner’s muscle, which extends just behind the posterior limb of the medial canthal tendon (Fig. 2‑8). Horner’s muscle continues posteriorly to insert onto the posterior lacrimal crest immediately behind the posterior limb of the medial canthal tendon. A few fibers continue more posteriorly 3 to 5 mm along the medial orbital wall. As Horner’s muscle passes to the posterior lacrimal crest, it is joined by the medial horn of the levator aponeurosis, the posterior layer of the orbital septum, and the medial rectus check ligament.
Key Point
The function of Horner’s muscle is to maintain the posterior position of the medial canthal angle and to tighten the eyelids against the globe during eyelid closure. It is believed that it also contributes to the lacrimal pump mechanism.
The pretarsal orbicularis muscle fibers join together along the surface of the lateral canthal tendon and the lateral horizontal raphé. Additional bundles of thin muscle fibers run along the upper and lower eyelid margins. These are referred to as muscles of Riolan. These are firmly fixated to the tarsus. Medially these muscles insert into the puncta and ampullae of the lacrimal drainage system. Some deeper fibers pass posterior to the canaliculi and merge into the deep heads of the pretarsal orbicularis muscle. These muscle fibers run in various directions, unlike the rest of the orbicularis muscle.
Key Point
A detailed working knowledge of the anatomical arrangements of the orbicularis muscle is the key to the success of a conservative or radical orbicularis myectomy in the surgical management of a patient with essential blepharospasm.
Lying between the orbicularis muscle and the orbital septum–levator aponeurosis fascial complex is an avascular fascial plane composed of loose areolar tissue. This fascial plane extends to the margin of the eyelid as the gray line. The gray line itself marks the anatomical separation of the anterior skin muscle lamella from the posterior tarsoconjunctival lamella.
Key Point
The postorbicularis fascial plane is surgically important, because it allows a bloodless dissection, permitting careful identification of the underlying orbital septum during the initial phase of ptosis surgery. It also permits a bloodless approach to the superior and inferior orbital margins (Fig. 2‑6).
The postorbicularis fascial plane allows slight movement between the orbicularis muscle and the underlying orbital septum while maintaining an integrated lamellar structure. Disruption of the fibrous connections within the postorbicularis fascial plane during upper lid surgery results in an inferior slippage of the anterior lamella after anterior-approach levator advancement surgery or upper lid blepharoplasty surgery. This inferior slippage of the anterior lamella can be prevented by fixating the orbicularis muscle and skin edges to the levator aponeurosis with interrupted 7–0 Vicryl sutures during the skin closure at the end of the procedure.
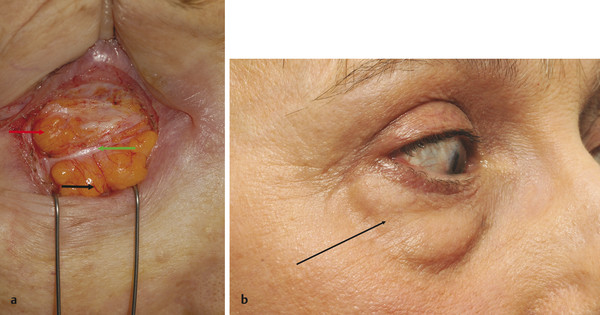
A deep plane of fat lies posterior to the orbital part of the orbicularis muscle superiorly and inferiorly. The suborbicularis oculi fat (SOOF) lies over the inferior orbital margin. The retro-orbicularis oculi fat (ROOF) lies over the superior orbital margin (Fig. 2‑9).
Key Point
A SOOF lift can be undertaken to enhance the result of a lower lid blepharoplasty in a patient with mild malar mounds. It is important to recognize the contribution a descent of the ROOF makes to the appearance of a brow ptosis.
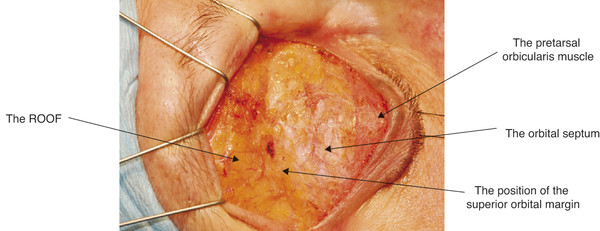
2.2.3 The Orbital Septum
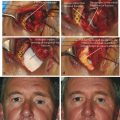
The orbital septum is a fibrous multilayered membrane of variable thickness that arises from the arcus marginalis, a line of periosteal condensation, along the superior and inferior orbital margins. This is demonstrated in Video 2.1 with a lower lid transconjunctival blepharoplasty. In young patients, the orbital septum is usually relatively thick and quite easily identified at surgery (Fig. 2‑10).
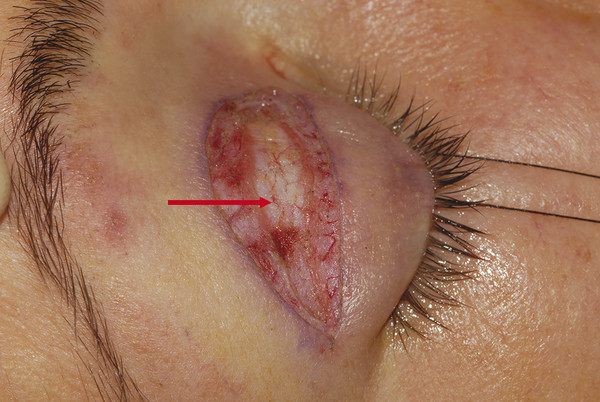
In contrast, the orbital septum may be very thin and dehiscent in older patients. The orbital septum is continuous with the inner layers of the periorbita. Within the orbit the periorbita lines the bony orbital walls and is composed of an outer layer of periosteum and an inner layer continuous with the orbital fascia. These layers separate at the arcus marginalis, with the outer periosteum continuing over the orbital margins as the periosteum of the forehead and facial bones. In the upper eyelid the inner fascial layer separates into two further layers at the arcus marginalis. The superficial layer continues over the brow, where it is continuous with the deep fascia over the frontalis muscle and the galea aponeurotica above the eyebrow. The deep layer becomes the orbital septum in the upper eyelid. In the lower eyelid the inner fascial layer extends into the eyelid as the orbital septum (Fig. 2‑11).
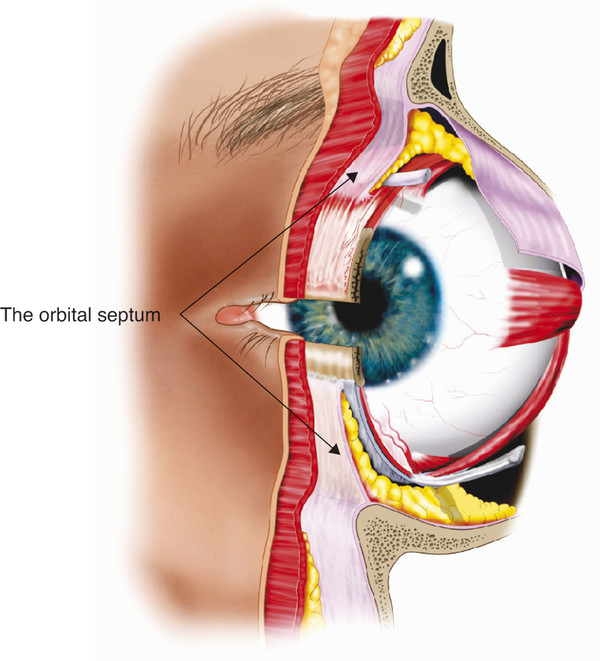
Laterally the orbital septum inserts into the lateral canthal tendon and also passes behind the tendon inserting onto the lateral retinaculum together with the lateral horn of the levator aponeurosis. In the superomedial aspect of the orbit the orbital septum extends inferiorly and posteriorly along the posterior lacrimal crest. Here it inserts just posterior to Horner’s muscle and the posterior limb of the medial canthal tendon. The orbital septum passes around the lip of the lacrimal foramen, where it fuses to the lacrimal sac fascia (Fig. 2‑12). The inferior orbital septum joins the superior orbital septum medially at the anterior lacrimal crest.
Superomedially at the orbital margin the orbital septum thins and becomes dehiscent, allowing for the passage of the infratrochlear neurovascular bundle and branches of the superior ophthalmic vein. In the central aspect of the upper lid, the inferior aspect of the orbital septum fuses with the anterior surface of the levator aponeurosis. The point at which the orbital septum fuses with the levator aponeurosis varies from a position approximately 3 to 5 mm above the superior aspect of the tarsal plate to as far as 10 to 15 mm above the tarsal plate. In the lower eyelid, the orbital septum fuses with the capsulopalpebral fascia a few millimeters below the inferior aspect of the tarsal plate. This common fascial sheet then inserts into the lower edge of the inferior tarsal plate.
The orbital septum can be readily identified during surgery by noting the firm resistance to traction applied to it because of its attachment to the arcus marginalis. The preaponeurotic fat pads lie immediately posterior to the orbital septum in the upper and lower eyelids. It is extremely important not to confuse an extension of the brow fat pad into the postorbicularis fascial plane of the upper eyelid with the preaponeurotic fat pads. The orbital septum may be incorrectly identified as the levator aponeurosis in such patients, and an inadvertent advancement of this septal layer will result in marked lagophthalmos. Likewise, it is important not to advance the capsulopalpebral fascia in the lower eyelid without first separating the orbital septum to prevent a resultant lower eyelid retraction. The orbital septum must not be sutured after eyelid surgery; this would result in inadvertent shortening, resulting in lagophthalmos or eyelid retraction.
Key Point
It is extremely important not to confuse an extension of the brow fat pad into the postorbicularis fascial plane of the upper eyelid with the preaponeurotic fat pads.
If the capsulopalpebral fascia is advanced in the lower eyelid without first opening the orbital septum, lower eyelid retraction may result.
2.2.4 Tarsal Plates
The tarsal plates are made up of dense fibrous tissue that provides the main structural integrity to the eyelids. The upper and lower tarsal plates are approximately 25 mm in length horizontally and are gently curved. They are approximately 1 to 1.5 mm thick; centrally the vertical height of the tarsal plate varies from 8 to 12 mm in the upper lid and 3.5 to 4 mm in the lower eyelid. The medial and lateral aspects of the tarsal plates taper to 2 mm high as they pass into the medial and lateral canthal tendons. A row of approximately 25 meibomian glands lie within the upper lid tarsus, and approximately 20 are positioned in the lower tarsus. The meibomian glands are multilobulated holocrine sebaceous glands that empty into a tiny central duct. The duct opens onto the posterior eyelid margin behind the gray line. These glands produce the lipid layer of the precorneal tear film.
2.2.5 Canthal Tendons
The tarsal plates continue into fibrous bands medially and form the crura of the medial canthal tendon. These crura lie between the orbicularis muscle anteriorly and the conjunctiva posteriorly. The superior and inferior crura fuse to form a common medial canthal tendon. This tendon inserts onto the adjacent bones by means of three arms: the anterior, posterior, and superior arms. The anterior arm inserts onto the orbital process of the maxillary bone anterior and above the anterior lacrimal crest (Fig. 2‑12).
The width of the tendon on insertion is approximately 4 to 5 mm. The anterior arm provides the main support for the medial canthal angle. The posterior arm arises from the common tendon close to the junction of the superior and inferior crura and passes posteriorly between the superior and inferior canaliculi. The posterior arm continues along the posterolateral aspect of the lacrimal sac. It continues posteriorly, fanning out to form a broad sheet between 6 and 9 mm in vertical height. The posterior arm inserts onto the posterior lacrimal crest just anterior to Horner’s muscle. The posterior arm serves to direct the vector forces of the canthal angle posteriorly to maintain a close apposition of the lids to the globe.
Key Point
The posterior arm of the medial canthal tendon inserting into the posterior lacrimal crest is most important in directing the vector forces of the canthal angle to maintain a close apposition of the lids to the globe. This must be respected (e.g., when a lower eyelid has been avulsed) to prevent an anterior malposition of the lower eyelid.
The superior arm of the medial canthal tendon originates at the anterior and posterior arms and passes superiorly 7 to 10 mm, inserting onto the orbital process of the frontal bone (Fig. 2‑8). The superior arm may provide vertical support to the canthal angle and may also play a role in the lacrimal pump mechanism.
Laterally the tarsal plates extend into less-well-defined fibrous strands that become the superior and inferior crura of the lateral canthal tendon. The lateral canthal tendon measures approximately 5 to 7 mm long. It is approximately 3 mm wide at the point where the superior and inferior crura unite but broadens to approximately 6 to 7 mm as it inserts into the lateral orbital tubercle (Fig. 2‑13a) 1.5 mm inside the lateral orbital margin. The lateral horn of the levator aponeurosis blends with the superior border of the lateral canthal tendon. The lateral canthal tendon and the lateral horn of the levator aponeurosis extend superiorly to a position 4 to 5 mm below the frontozygomatic suture line, forming a broad tendon-like insertion (Fig. 2‑13b).
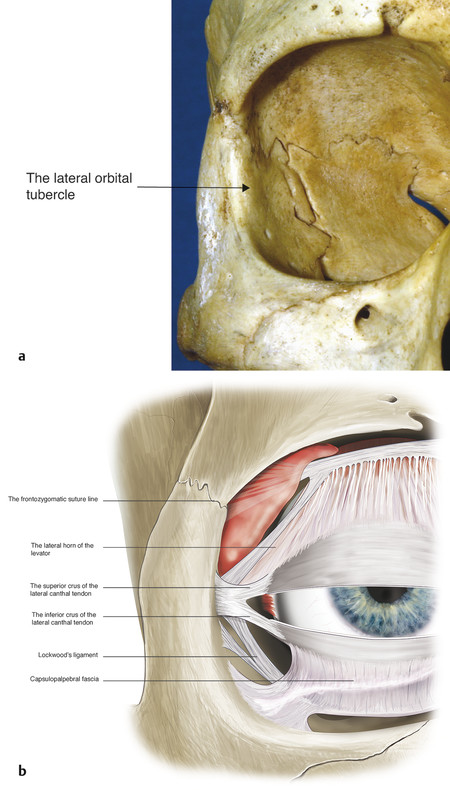
The inferior crus of the lateral canthal tendon is divided during the course of a lateral tarsal strip procedure and for a rapid orbital decompression for the management of an acute retrobulbar hemorrhage. Division of the superior crus of the lateral canthal tendon can be associated with significant morbidity because of the proximity of important adjacent structures (i.e., the lateral horn of the levator, Whitnall’s ligament, and the lacrimal gland; Fig. 2‑9).
Key Point
The superior crus of the lateral canthal tendon must be divided carefully, because the lateral horn of the levator and Whitnall’s ligament can be damaged, resulting in a lateral blepharoptosis, as can the lacrimal gland.
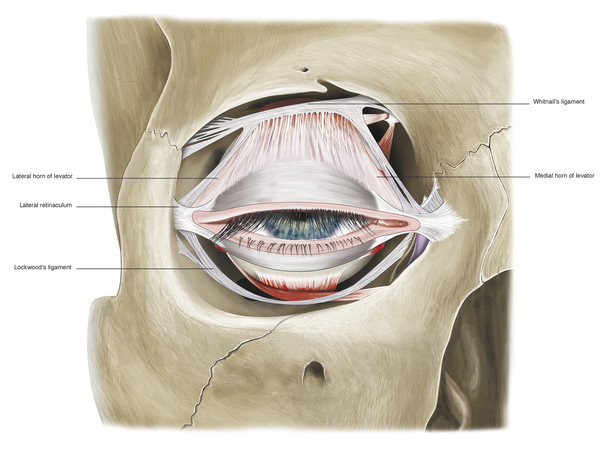
The fibers of the crura continue posteriorly along the lateral orbital wall, fusing with fibers of the lateral check ligament. Along with the lateral canthal tendon complex, the inferolateral fibers of Whitnall’s superior suspensory ligament and the lateral portion of Lockwood’s inferior suspensory ligament insert onto the lateral tubercle, forming the lateral retinaculum (Fig. 2‑14).
A small lobule from the precapsulopalpebral fat pad extends superiorly between the orbital septum and the lateral canthal tendon. This is referred to as Eisler’s pocket. This is believed to act like a bursa, allowing some independent movement of the lateral canthal tendon during eyelid movement, particularly on lateral gaze.
2.2.6 Preaponeurotic Fat Pads
The preaponeurotic fat pads in the upper eyelid and the precapsulopalpebral fat pads in the lower eyelid are anterior extensions of the extraconal orbital fat. The fat pads are enveloped by thin fibrous sheaths that represent forward continuations of the anterior orbital septal system. The individual lobules of fat within the fat pockets are surrounded by fine interlobular septa. The fibrous sheaths surrounding the fat pads are connected to the overlying orbital septum and the underlying levator aponeurosis by very fine fibrous bands. The eyelid fat pads are important surgical landmarks that assist in the identification of the eyelid retractors, which lie immediately posterior to the fat pads (Fig. 2‑2).
Key Point
The eyelid fat pads are very important surgical landmarks that assist in the identification of the eyelid retractors, which lie immediately beneath the fat pads.
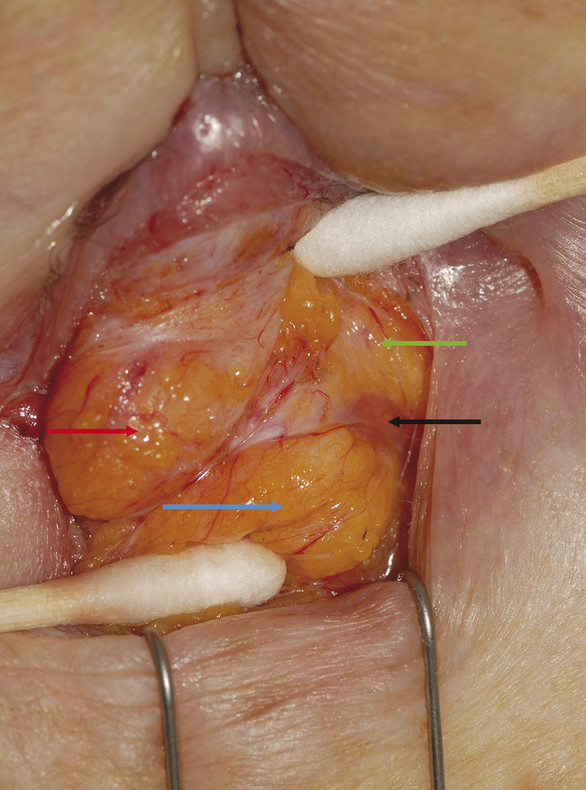
In the upper eyelid, there are two fat pads: one medially and one centrally (Fig. 2‑15). These are separated by fine fascial bands, which are continuous with the trochlea. The medial fat pad is paler than the central fat pad and contains thicker and more abundant interlobular septa. In the lateral aspect of the upper eyelid, the lacrimal gland is located in the lacrimal gland fossa just posterior to the orbital margin (Fig. 2‑13b).
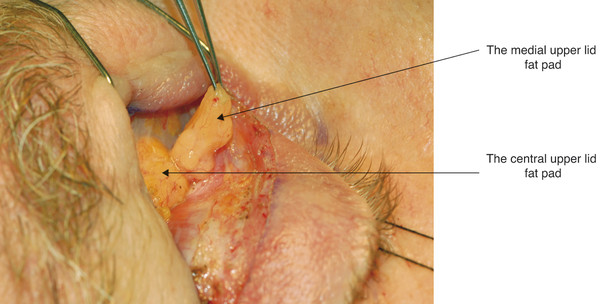
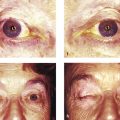
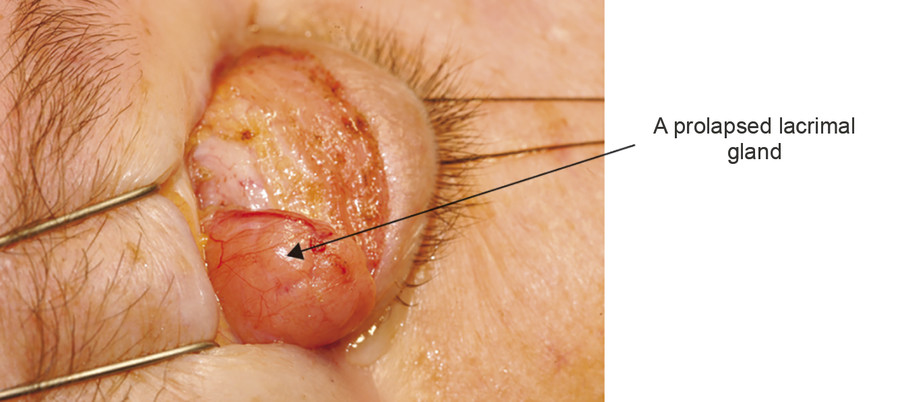
The lacrimal gland is isolated within its own fascial compartment. If this fascial support system of the lacrimal gland becomes loose, the gland may prolapse into the lateral aspect of the eyelid, where it must not be mistaken for preaponeurotic fat (Fig. 2‑16).
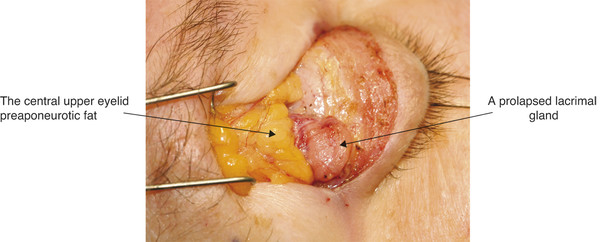
The appearance of the lacrimal gland is a quite distinctive pink color (Fig. 2‑17). It is also firm, unlike preaponeurotic fat.
There are three fat pads in the lower eyelid, each of which is separated by fibrous septa, which are continuous with the orbital connective tissue system (Fig. 2‑18).
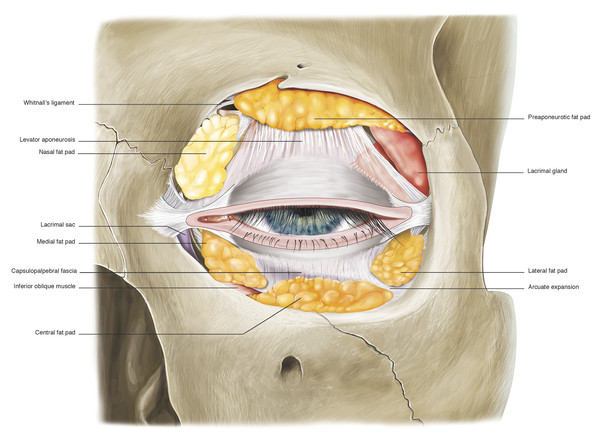
The central and lateral fat pads are separated by a connective tissue band referred to as the arcuate expansion, which extends from Lockwood’s ligament to the inferolateral orbital margin (Fig. 2‑19).
The inferior oblique muscle runs between the medial and central fat pads (Fig. 2‑20).
2.2.7 Eyelid Retractors
The levator palpebrae superioris muscle arises from the lesser wing of the sphenoid bone just above the annulus of Zinn and superolateral to the optic canal (Fig. 2‑21).
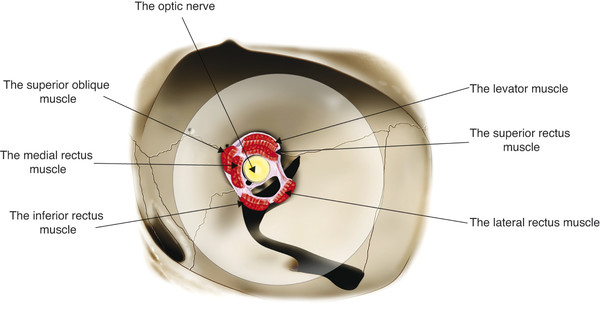
The muscle is approximately 35 mm long. At its origin it is approximately 4 mm wide, broadening to approximately 8 mm in the midsection of the orbit. As the muscle passes forward, it lies in intimate contact with the superior rectus muscle (see Fig. 2‑2). The levator and the superior rectus muscles are connected by fine fibrous strands of the superior orbital fascial system. Extensive fibrous tissue strands connect the sheath of the levator muscle with the superior rectus muscle below and with the superior conjunctival fornix and Tenon’s capsule just behind Whitnall’s ligament.
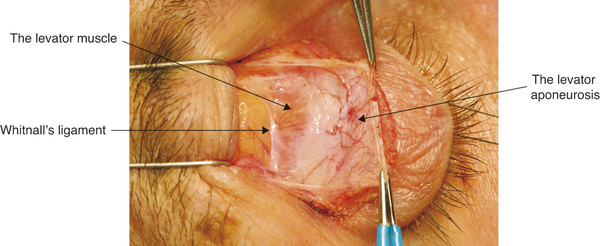
These fibrous tissue strands function as check ligaments but permit coordinated movement of the upper eyelid with alterations in the vertical ocular gaze position. The levator muscle widens to approximately 18 mm just posterior to the superior orbital margin. In this location, a condensation of variable thickness and density is seen running along the muscle sheath in a horizontal direction. This is Whitnall’s superior transverse ligament (Fig. 2‑22).
Whitnall’s ligament attaches to the fascia adjacent to the trochlea medially and laterally inserts into the lacrimal gland capsule and the periorbita of the lower aspect of the frontal bone (Fig. 2‑14).
The ligament is firmly attached to the levator muscle sheath medially and laterally but only very loosely attached to the levator muscle centrally. A very thin sheet of fascia passes from Whitnall’s ligament to insert into the superior orbital margin.
Whitnall’s ligament appears to function as a check ligament against posterior movement of the levator and superior rectus muscles. Whitnall’s ligament forms a circumorbital fascial ring in conjunction with Lockwood’s ligament. During ptosis surgery Whitnall’s ligament should be maintained intact wherever possible. If Whitnall’s ligament is severed, the levator muscle prolapses. This loss of support results in the need for a greater resection than would otherwise be required. The ligament can also be used as an internal sling during ptosis surgery.
Key Point
During ptosis surgery Whitnall’s ligament should be maintained intact wherever possible.
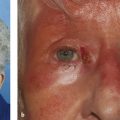
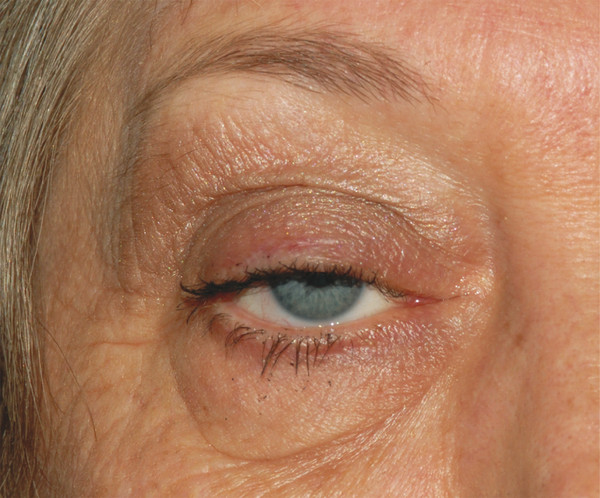
The levator muscle attaches to the fibrous levator aponeurosis in close proximity to Whitnall’s ligament (Fig. 2‑2). The levator aponeurosis continues inferiorly approximately 15 to 20 mm to inserts onto the superior two thirds of the anterior tarsal surface (Fig. 2‑2). Delicate fibers also continue forward from the levator aponeurosis to insert onto the septum of the pretarsal orbicularis muscle and the skin (Fig. 2‑2). The delicate fibers from the levator aponeurosis retract the pretarsal skin and muscle and orbicularis muscle, preventing an overhang of these structures on elevation of the eyelid. The superior limits of the attachment of these layers are marked by the upper lid skin crease. This relationship is disturbed by a dehiscence of the levator aponeurosis, which results in an apparent superior displacement of the upper lid skin crease (Fig. 2‑23).
The levator aponeurosis widens to form the medial and lateral levator horns as it passes inferiorly from Whitnall’s ligament (Fig. 2‑24).
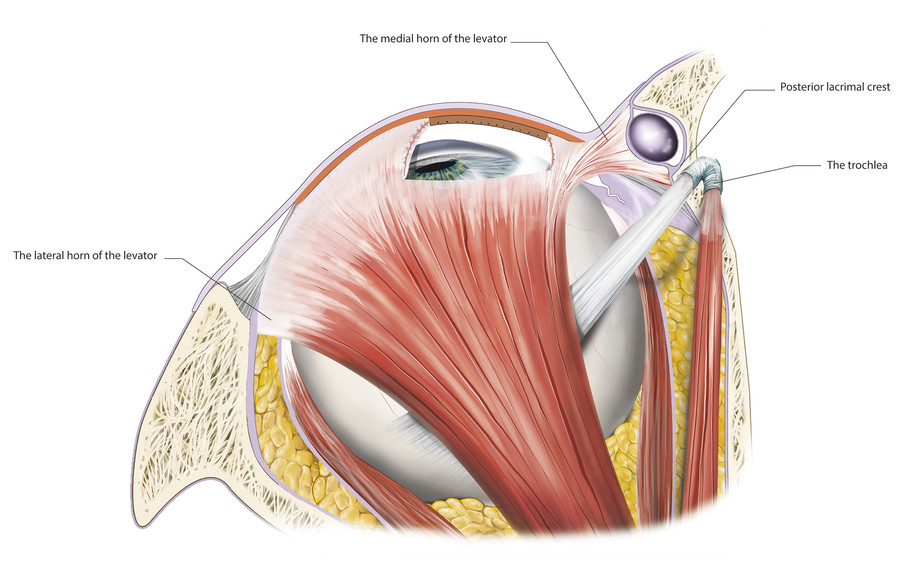
The medial horn is more tenuous and accounts for a lateral shift of the superior tarsus in older patients. The surgeon must be careful to avoid causing further intraoperative damage to the medial horn, which will result in a further lateral shift of the superior tarsus and an unsatisfactory temporal displacement of the peak of the eyelid. The lateral horn, which is a better-defined structure, separates the lacrimal gland into orbital and palpebral lobes (Fig. 2‑25).
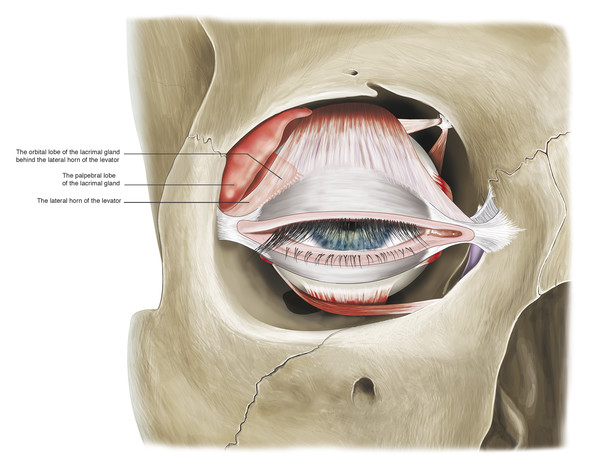
The lateral levator horn inserts onto the lateral orbital tubercle of the zygomatic bone at the level of the lateral retinaculum (Fig. 2‑14). It also joins with fibers of the capsulopalpebral fascia of the lower eyelid.
The medial horn of the levator aponeurosis inserts onto the posterior limb of the medial canthal tendon and the posterior lacrimal crest (Fig. 2‑24). The medial and lateral levator horns help to distribute the forces generated by the levator muscle along the levator aponeurosis, allowing the central aspect of the eyelid to elevate to the greatest extent. The lateral levator aponeurosis horn is severed during levator aponeurosis recession procedures undertaken for the management of thyroid-related eyelid retraction. In contrast, during surgery to advance to the levator aponeurosis for the management of a blepharoptosis, the levator horns should be preserved wherever possible.
Key Point
During a levator aponeurosis advancement procedure, the medial and lateral horns of the levator muscle should be preserved. Damage to the medial levator horn will result in a lateral shift of the tarsus, a problem more commonly encountered in older patients.
Müller’s muscle is joined to the overlying levator aponeurosis by a very loose connective tissue layer. This avascular plane can usually be dissected easily during ptosis surgery procedures (Fig. 2‑26).
Müller’s muscle originates from the undersurface of the levator muscle at a point just distal to Whitnall’s ligament (Fig. 2‑2). Müller’s muscle runs inferiorly posterior to the levator aponeurosis. It is approximately 8 to 12 mm long and has a variable thickness of 0.1 to 0.5 mm. Müller’s muscle inserts onto the anterior edge of the superior border of the tarsus (Fig. 2‑2). There is a small fat pad within the medial aspect of the muscle that should not be mistaken for the fatty degeneration of the muscle seen with increased age and in patients with thyroid eye disease (Fig. 2‑27).
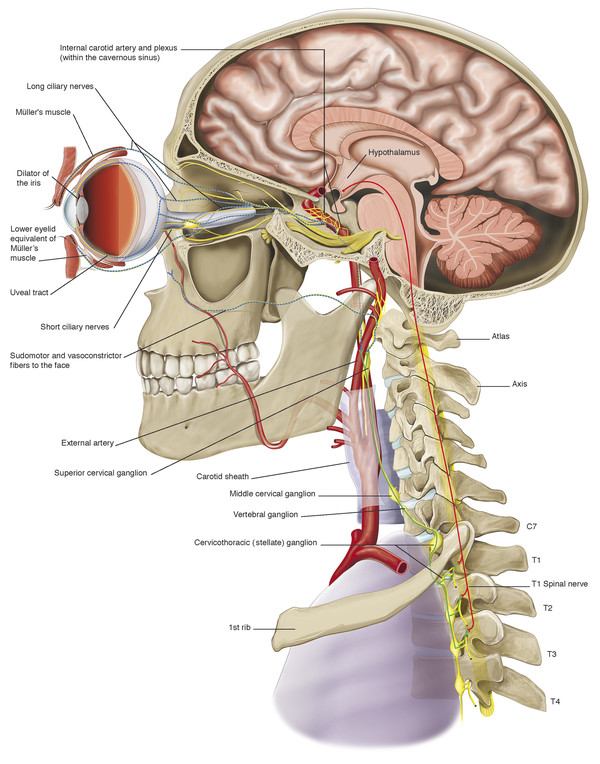
Müller’s muscle is innervated by sympathetic nerve fibers, which pass to the orbit via the internal carotid plexus. These sympathetic nerve fibers originate in the hypothalamus (Fig. 2‑28). Disruption of the sympathetic innervation of Müller’s muscle results in the ptosis seen in Horner’s syndrome.
In the lower eyelid, the capsulopalpebral fascia is a fibrous sheet that arises from Lockwood’s ligament and the inferior rectus and inferior oblique muscle sheaths (Fig. 2‑2). As it passes superiorly the fascia fuses with the orbital septum approximately 4 to 5 mm below the inferior border of the tarsal plate (Fig. 2‑2). From this position, a common fascial sheet passes superiorly and inserts onto the inferior border of the tarsus (Fig. 2‑2). Fine fibers pass anteriorly from this fascial sheet to the orbital septum and the skin creating the lower eyelid crease in a manner analogous to that of the upper eyelid.
Smooth muscle fibers analogous to Müller’s muscle in the upper eyelid lie posterior to the capsulopalpebral fascia, forming a very thin muscular layer (Fig. 2‑2). These fibers are also innervated by the sympathetic nervous system. For this reason, disruption of the sympathetic innervation of these muscle fibers results in the slight rise in the lower eyelid seen in Horner’s syndrome (Fig. 2‑29). A combination of the blepharoptosis and the rise in the lower eyelid is responsible for the apparent enophthalmos seen in patients with Horner’s syndrome.
2.2.8 Conjunctiva
The conjunctiva is a mucous membrane that can be arbitrarily divided into palpebral, forniceal, and bulbar portions. The palpebral portion is very closely attached to the posterior surface of the tarsus in the upper and lower eyelids, from which it cannot be separated. This portion is also in close approximation to Müller’s muscle in the upper lid and the equivalent smooth muscle fibers in the lower lid (Fig. 2‑3).
The palpebral portion continues into the forniceal portion. The superior fornix is situated approximately 10 mm above the superior corneal limbus. The superior fornix is supported by a fine fibrous suspensory ligament, which arises from the fascia of the levator muscle and the superior rectus muscle. The inferior fornix is situated approximately 8 mm below the inferior corneal limbus and is supported by a fine suspensory ligament that arises from Lockwood’s ligament. This is situated just posterior to the capsulopalpebral fascia and functions to retract the fornix inferiorly during downgaze.
The conjunctiva contains a series of small accessory lacrimal glands. Lymphatic blood vessels and sensory nerves run in the conjunctival submucosa. A small, elevated structure consisting of sebaceous glands, sweat glands, and fine hairs lies within the medial canthal angle. This is the caruncle. A vertical fold of conjunctiva, the plica semilunaris, lies just lateral to the caruncle. The medial wall of the orbit can be accessed via an incision made between these two structures (e.g., a transcaruncular approach to the drainage of a medial subperiosteal orbital abscess complicating orbital cellulitis).
2.2.9 Sensory Nerve Supply of the Eyelids
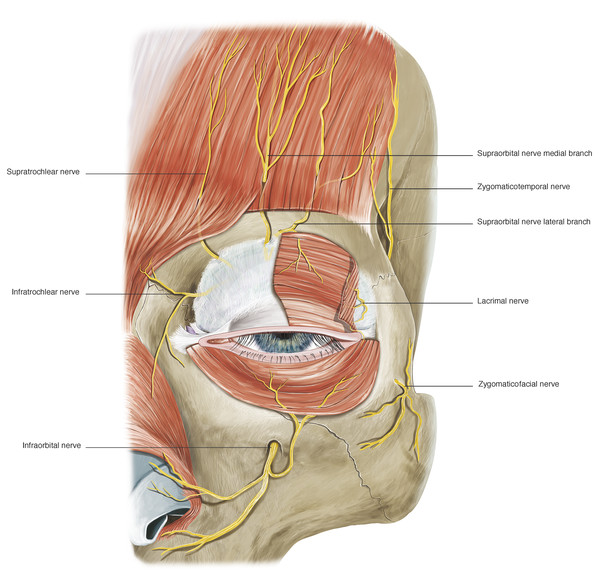
The sensory innervation of the eyelids is provided by branches of the ophthalmic and maxillary divisions of the trigeminal nerve. The upper eyelid is innervated by the supraorbital, supratrochlear, and lacrimal nerves (Fig. 2‑30).
The medial aspect of the upper and lower eyelids is supplied by the infratrochlear nerve. Branches of the infratrochlear nerve also supply parts of the adjacent eyebrow, forehead, and nose (Fig. 2‑30). The lateral aspect of the upper eyelid and temple is supplied by the zygomatico-temporal branch of the maxillary nerve (Fig. 2‑30). The central aspect of the lower eyelid is supplied by infraorbital nerve, a branch of the maxillary nerve, and the lateral aspect of the lower eyelid is supplied by the zygomaticofacial branch of the maxillary nerve. (A more detailed description of the periorbital sensory nerve supply is outlined later in this chapter.)
2.2.10 Vascular Supply to the Eyelids
The eyelids have a very rich blood supply with multiple anastomoses (Fig. 2‑31a).
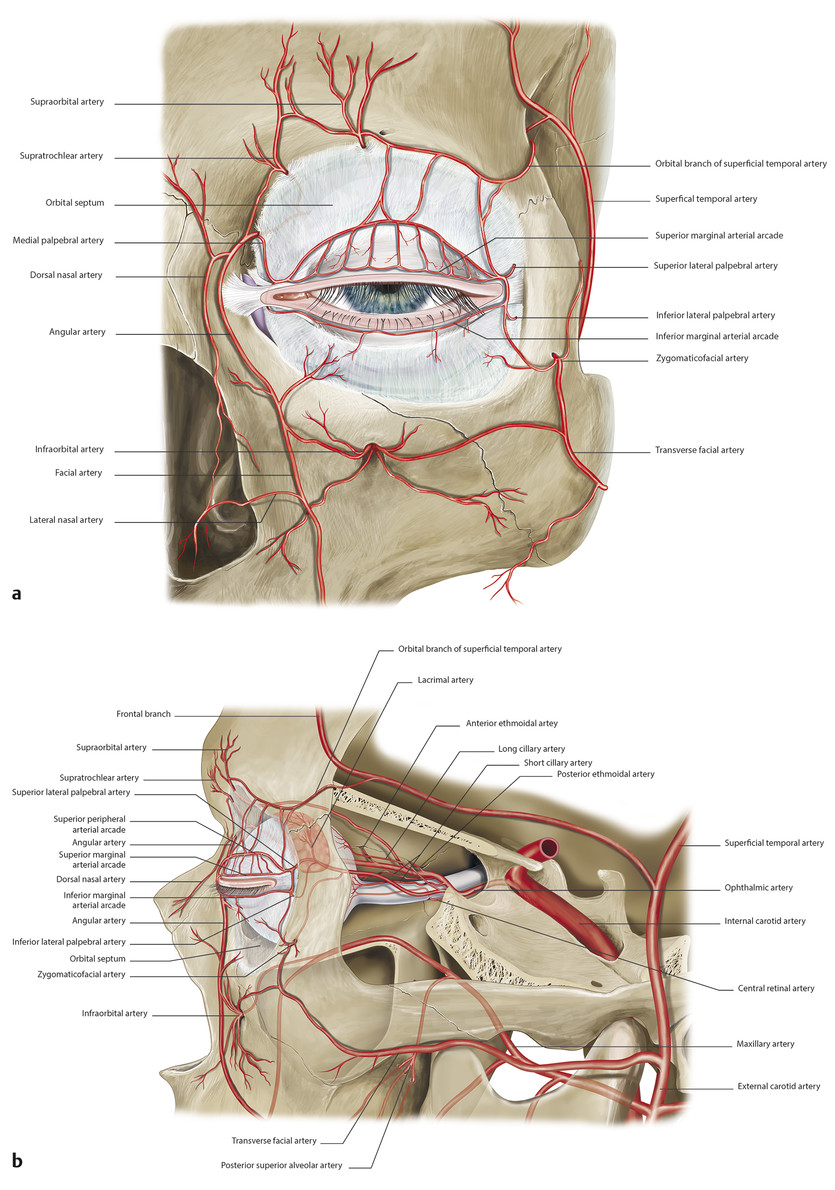
The anterior lamellae of the eyelids are supplied by branches of the external carotid artery, namely the transverse facial, superficial temporal, and angular arteries in conjunction with multiple anastomoses with vessels passing through the orbit (Fig. 2‑31). The posterior lamellae of the eyelids are supplied by vascular arcades. In the upper eyelid, a marginal arcade runs horizontally about 2 mm above the eyelid margin (Fig. 2‑31). A more peripheral arcade runs along the superior border of the tarsus between the levator aponeurosis and Müller’s muscle (Fig. 2‑26 ; Fig. 2‑31b).
These arcades are supplied medially by the superior medial palpebral artery, a terminal branch of the ophthalmic artery; laterally the arcades are fed by the superior lateral palpebral artery, a branch of the lacrimal artery (Fig. 2‑31). (A more detailed description of the periorbital vascular supply is outlined later in this chapter.)
2.2.11 The Lymphatic Channels in the Eyelids
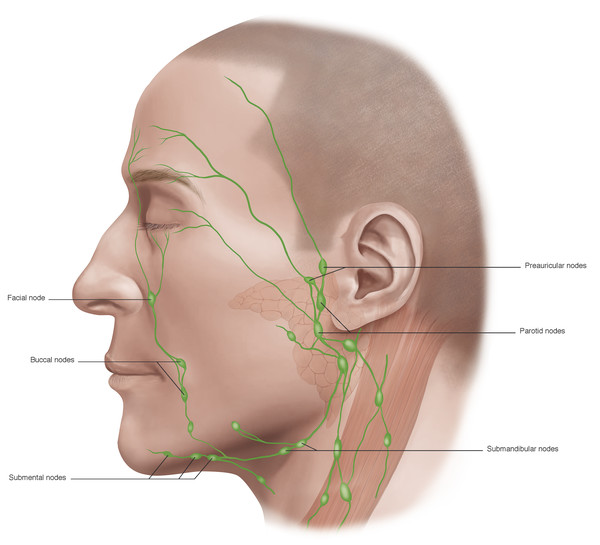
The lymphatic drainage channels are extensive and well developed and lie anterior to the orbital septum. Lymph from the lateral two thirds of the upper eyelid and the lateral one third of the lower eyelid drains laterally and inferiorly into the deep and superficial parotid and submandibular lymph nodes. Lymph from the medial one third of the upper eyelid and the medial two thirds of the lower eyelid drains medially and inferiorly into the anterior cervical lymph nodes (Fig. 2‑32). Extensive disruption of these lymphatic channels results in lymphedema.
2.3 The Eyebrows
The eyebrows consist of thickened skin overlying the supra-orbital prominences from which the eyebrows are separated by the brow fat pads (Fig. 2‑3). The eyebrows contain coarse hairs that emerge from the skin surface at an oblique angle. The eyebrows are raised by the frontalis muscle and are depressed by the actions of the procerus, corrugator and depressor supercilii muscles, and the orbicularis oculi muscles (Fig. 2‑4 ; Fig. 2‑33 ; Fig. 2‑87).
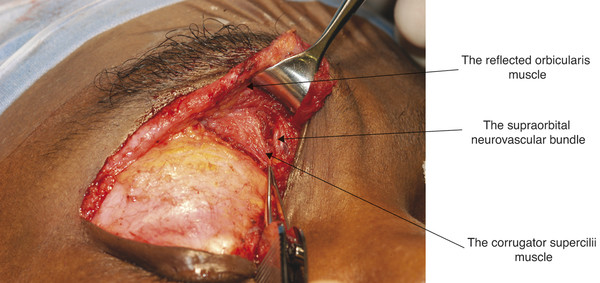
The brow elevator muscle and the brow depressor muscles are all innervated by branches of the facial nerve.
The eyebrows tend to be heavier and flatter in males and higher and more arched in females, although there are great interindividual variations. The brows tend to become progressively ptotic with age, particularly in the lateral aspect.
The muscle fibers of the frontalis muscle lie vertically on the forehead and form the anterior belly of the occipitofrontalis muscular fascial complex. The occipital and frontalis muscles are separated by the galea aponeurotica (Fig. 2‑34 ; Fig. 2‑87).
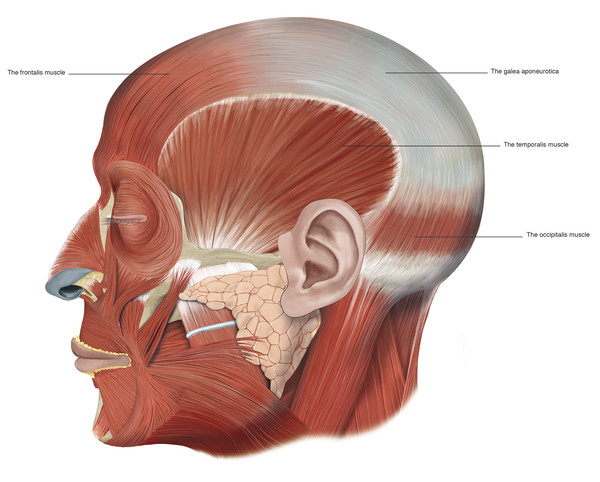
The frontalis muscle connects to the galea aponeurotica in the region of the coronal suture line and runs inferiorly to the superior orbital margin. The galea aponeurotica forms a narrow extension that runs between the bellies of the frontalis muscle onto the bridge of the nose. The frontalis muscle has no bony attachments at all. Medially the muscle fibers interdigitate with those of the procerus muscle, and laterally the frontalis muscle fibers interdigitate with the corrugator supercilii muscles and the orbital portion of the orbicularis oculi muscles. In the region of the superior orbital margin, a deep fat pad separates the frontalis muscle from the underlying deep fascia. This brow fat pad continues into the upper eyelid, where it fuses with the layer of fascia lying posterior to the orbicularis oculi muscle. In some patients, particularly those older than 50 years, globules of fat may descend into the eyelid; these should not be confused with the central preaponeurotic fat pads.
The frontalis muscle acts as an elevator of the brow and also serves as an accessory retractor of the upper eyelid. Overaction of the frontalis muscle is seen in patients with a blepharoptosis. Therefore the frontalis muscle should be firmly immobilized when measuring a patient’s levator function.
Key Point
The frontalis muscle acts as an elevator of the brow and also serves as an accessory retractor of the upper eyelid. Overaction of the frontalis muscle is seen in patients with a blepharoptosis. Therefore the frontalis muscle should be firmly immobilized when measuring a patient’s levator function. Botulinum toxin given into the frontalis muscle to reduce horizontal forehead lines may unmask a previously unsuspected blepharoptosis.
The corrugator supercilii muscle has a pyramidal shape and lies medially beneath the frontalis muscle and the orbital portion of the orbicularis oculi muscle (Fig. 2‑4). It arises from the medial end of the frontal bone at the superior orbital margin. It extends obliquely superiorly and laterally. The muscle inserts into the deep fascia of the frontalis muscle along the lateral third of the eyebrow by means of a series of slips. In this area, the deep fascia comprises several distinct layers, and fibers of the corrugator supercilii muscle interdigitate among these. Contraction of the corrugator supercilii muscles causes the brow to move in a medial and downward direction and creates vertical folds in the glabella.
The depressor supercilii muscle is distinct from the corrugator supercilii muscle and the medial head of the orbital portion of the orbicularis oculi muscle. It arises from most individuals as two distinct heads from the frontal process of the maxilla approximately 1 cm above the medial canthal tendon, with the angular vessels passing between the two muscle heads. The muscle inserts into the dermis approximately 13 to 14 mm above the medial canthal tendon (Fig. 2‑4).
The procerus muscle has a pyramidal shape. It arises from the fascia covering the lower part of the nasal bone and upper lateral nasal cartilage. It extends vertically between the eyebrows and inserts into the skin lying over the lower aspect of the forehead (Fig. 2‑4). The fibers of the procerus muscle merge with those of the medial aspect of the frontalis muscle. Contraction of the procerus muscle pulls the medial aspect of the brows in a downward direction and produces transverse lines over the bridge of the nose.
The orbital portion of the orbicularis muscle runs in a circular direction and overlies the bony orbital margins (Fig. 2‑4). Within the eyebrow the orbicularis muscle fibers interdigitate with those of the frontalis muscle and subcutaneous fascia. Contraction of the orbital portion of the orbicularis oculi muscle draws the brows downward and produces the lateral rhytids of the lateral canthi.
Key Point
The corrugator supercilii, the procerus, the depressor supercilii muscles, and the orbital portion of the orbicularis oculi muscle act as the brow depressor muscles and are targeted in the injection of botulinum toxin, both to reduce frown lines and lateral canthal rhytids, but also to effect a brow lift. This is achieved by the unopposed action of the frontalis muscle.
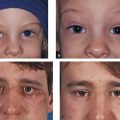
The frontalis muscle is responsible for the appearance of horizontal creases in the forehead skin. These creases can be reduced for aesthetic reasons with the use of botulinum toxin injections, but great care should be taken in administering such injections, because weakening the action of the frontalis muscle can result in an unsatisfactory brow ptosis. The corrugator and depressor supercilii muscles are responsible for the appearance of frown lines in the glabellar region. The injection of botulinum toxin into these muscles can remove or soften the appearance of such lines, which can be responsible for facial miscues, giving the unwanted impression of anger. This is particularly noticeable in patients with thyroid eye disease (Fig. 2‑35).
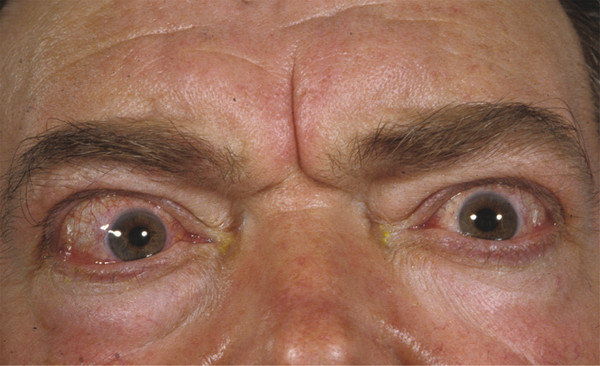
The superolateral part of the orbicularis oculi muscle is responsible for the appearance of lateral canthal rhytids and for depression of the temporal aspect of the eyebrow. The injection of botulinum toxin into this part of the muscle can remove or soften the appearance of these lines and can also result in a temporal brow lift resulting from the unopposed action of the frontalis muscle. In some individuals, this action can, however, result in an unwanted peaking of the temporal brow.
2.4 The Facial Nerve
A very good understanding of the relationship of the facial nerve and its branches to the fascial planes of the face is essential when undertaking facial surgery. The facial nerve exits the stylomastoid foramen and gives off the postauricular nerve and the branch to the posterior belly of the digastric and stylohyoid muscles. The main trunk then usually bifurcates into the upper (temporozygomatic) and lower (cervicofacial) branches as it approaches the posteromedial border of the parotid gland. As it passes forward within the substance of the parotid gland, it lies between the superficial and deep lobes. The nerve divides in a variable pattern into the main distal branches (the temporal or frontal, zygomatic, buccal, mandibular, and cervical branches) (Fig. 2‑36).
At the anterior border of the parotid gland, these branches lie on the surface of the masseter muscle deep to the parotidomasseteric fascia. The temporal nerve is the smallest of the branches and has the least number of interconnections. In most individuals, the temporal branch is a terminal branch. It is the branch that typically shows the least degree of recovery after a facial palsy.
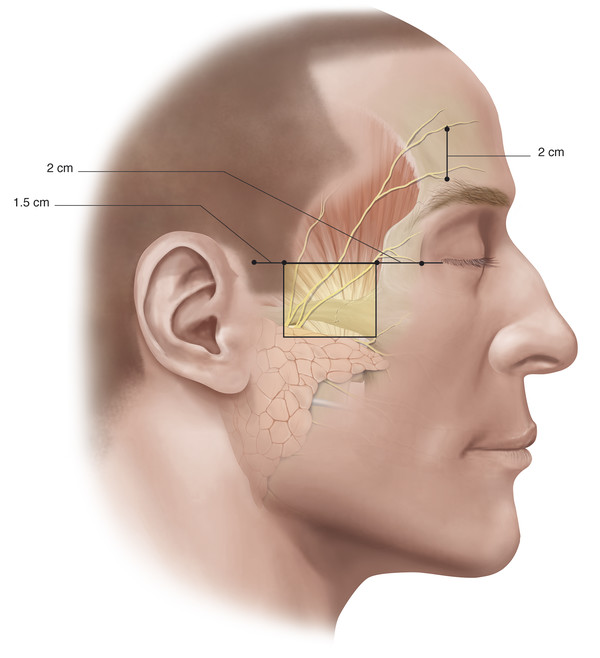
The temporal branch becomes superficial once it crosses the zygomatic arch where the nerve is most vulnerable to injury. The approximate position of this branch (branches) is indicated in Fig. 2‑37.
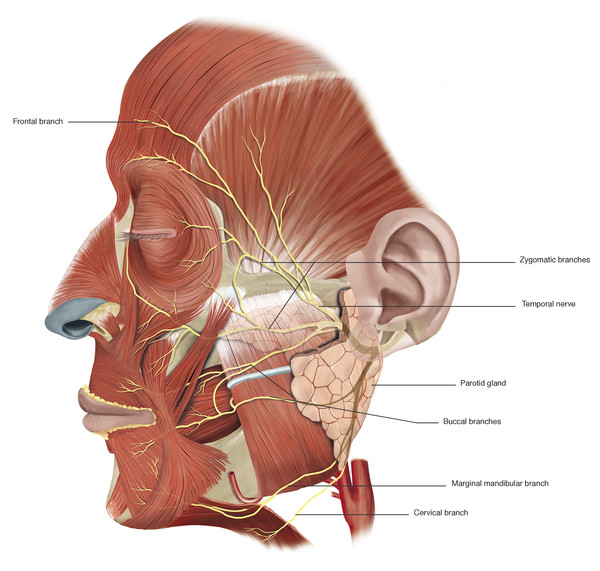
Key Point
In most individuals, the temporal branch is a terminal branch. It is the branch that typically shows the least degree of recovery after a facial palsy.
Three to five branches cross the zygomatic arch. The most posterior branch is always anterior to the superficial temporal vessels. The anterior hairline at the level of the lateral canthus represents the junction of the posterior and middle branches of the nerve. The most anterior branch crosses the zygomatic arch approximately 2 cm posterior to the posterior aspect of the lateral orbital margin (Fig. 2‑36). In the brow region, the frontal branch runs approximately 2 cm above the brow. In the temple the nerve runs within the superficial temporoparietal fascia and supplies the frontalis muscle, the superior fibers of the orbicularis muscle, and the procerus and corrugator supercilii muscles. It is important to stay on the glistening fibers of the deep temporal fascia deep to the temporoparietal fascia to avoid injury to the nerve during surgical dissections in this area (e.g., when undertaking an endoscopic brow lift).
The zygomatic and buccal branches of the facial nerve pass medially across the surface of the masseter muscle under the parotidomasseteric fascia, where they are relatively well protected. At the anterior border of the masseter muscle, the branches pass over the buccal fat pad and course forward to supply the inferior fibers of the orbicularis oculi muscle, the zygomaticus major and minor muscles, and the levator labii muscle by accessing the muscles from their deep surface.
Key Point
It is extremely important to understand the different anatomical planes that are traversed by branches of the facial nerve to avoid intraoperative damage to them. The safe planes of dissection are the following:
Preauricular area: superficial to the parotid gland.
Over the zygomatic arch: in the subcutaneous fat.
1 cm above the zygomatic arch: in the subcutaneous fat or deep to the superficial layer of the deep temporal fascia.
In the temple: just superficial to the deep layer of the temporal fascia.
Over the frontalis muscle: in the subcutaneous fat or deep to the galea aponeurotica.
In the midface: in the subcutaneous fat.
In the lower face: in the subcutaneous fat.
The marginal mandibular nerve passes from the lower pole of the parotid gland and descends below the mandible in the area posterior to the facial artery lying 1 to 2 cm inferior to its lower border (Fig. 2‑36). In older individuals and with extension of the neck, it can lie even lower. Like the frontal branch of the facial nerve, the marginal mandibular branch is a terminal branch and is most liable to injury. Its injury causes an obvious clinical deficit related to paralysis of the depressor labii inferioris muscle. It enters the platysma muscle along its deep surface and is liable to injury in operations requiring elevation and division of the platysma in the upper neck. In 10 to 15% of patients, there are connections between this nerve and the buccal branches. In these patients, considerable recovery of function will take place even if this nerve is transected.
The cervical branch is the most inferior of the facial nerve branches. Below the parotid gland, it passes deep to the platysma muscle which it innervates.
2.5 The Bony Facial Skeleton
The facial skeleton consists of (Fig. 2‑38) the following:
The frontal bone superiorly.
The bones of the midface: the maxillae; the zygomatic, palatine, and nasal bones; the zygomatic processes of the temporal bones; the lacrimal and ethmoid bones; and the turbinates.
The mandible inferiorly.
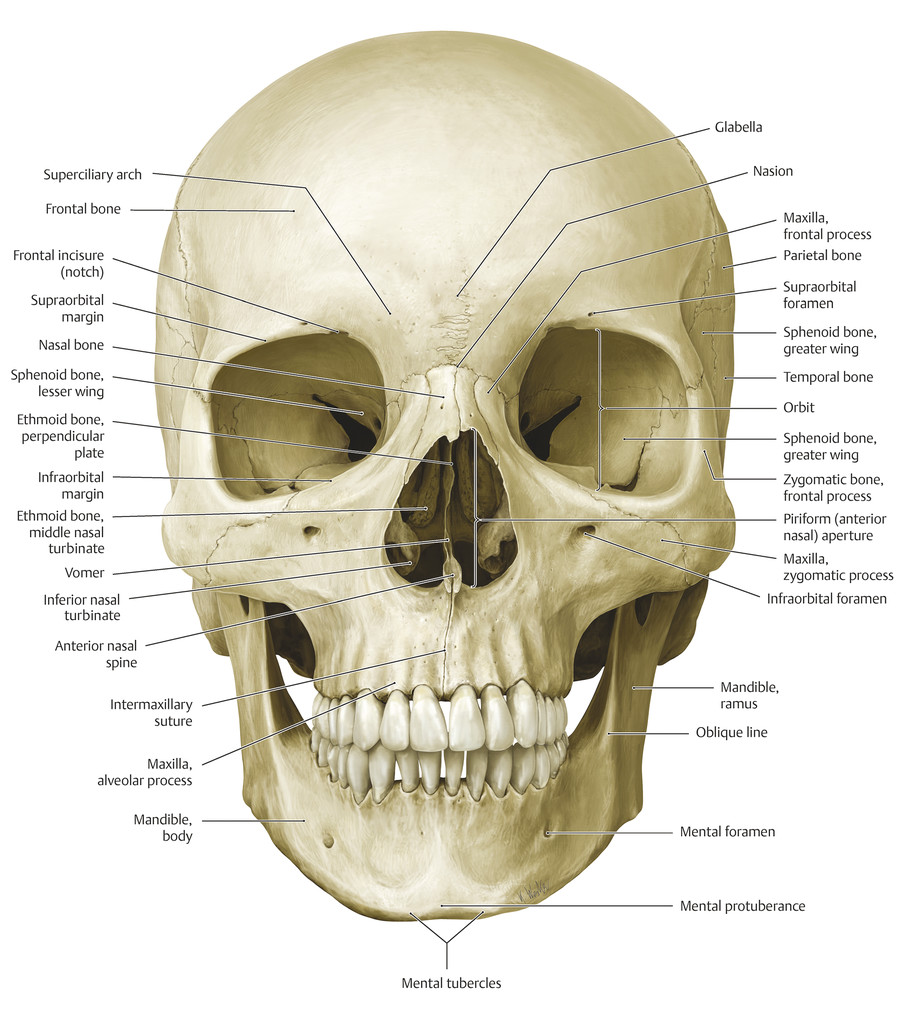
The maxillary bones contribute to the nasal aperture, bridge of the nose, floor of the orbits, and the cheekbones.
The mandible forms the lower part of the face. In the midline, the mental protuberance gives anterior projection to the overlying soft tissues. Laterally, the ramus of the mandible underlies the masseter muscle and continues superiorly to articulate with the cranium through the coronoid process and condylar process of the mandible. The mental nerve emerges from the mental foramen in the body of the mandible in line vertically with the infraorbital and supraorbital nerves.
As well as providing structural support, projection, and protection of sensory organs, including the globes, the facial skeleton also provides areas of attachment for the muscles of facial expression and the muscles of mastication.
2.5.1 The Bony Orbit
In an adult, the bony orbit is pyramidal in shape. The volume of the orbit in the average adult is approximately 30 cm3. The globe contributes approximately 7.5 cm3. The anterior entry to the orbit forms an approximate rectangle 4 cm in width by 3.5 cm in height. The widest dimension of the orbit is attained approximately 1 cm posterior to the anterior orbital margin. The anteroposterior axes of the orbits form a 45-degree angle between them. The lateral orbital walls form a 90-degree angle between them (Fig. 2‑39).
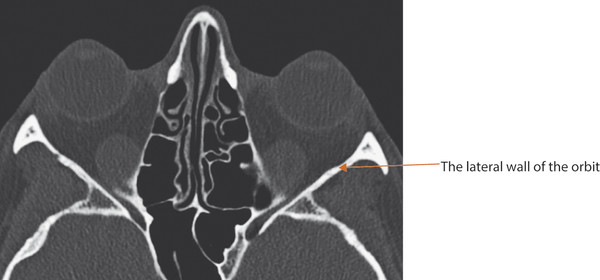
The dimensions of the orbit vary from patient to patient. For this reason, a surgeon cannot rely on any specific measurements as a guide to the precise location of the optic canal or of the superior orbital fissure. Great care should therefore be taken when injecting fat, for example, into the posterior orbit of an anophthalmic patient, because the cannula could penetrate the intracranial cavity.
The precise location of the anterior and posterior ethmoidal foramina cannot be determined accurately before surgical dissection. Therefore great care must be taken in undertaking dissections in the medial and posterior orbit during an orbital decompression procedure or during the exploration of a medial orbital wall blowout fracture. The inferior orbital fissure may lie within 10 to 15 mm of the anterior orbital margin. It should be noted that the floor of the orbit ends at the posterior wall of the maxillary sinus and does not extend to the apex of the orbit (Fig. 2‑40).
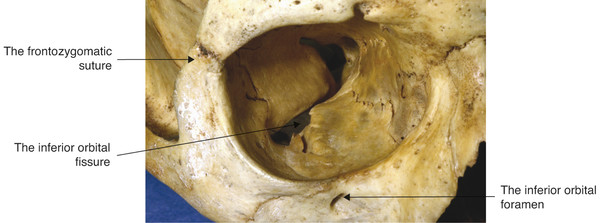
Key Point
The dimensions of the orbit vary from patient to patient. Therefore a surgeon cannot rely on any specific measurements as a guide to the precise location of the optic canal or of the superior orbital fissure.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree