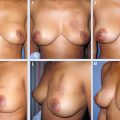
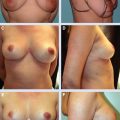
19 Latissimus Dorsi Miniflap Reconstruction
Until recently, breast-conserving surgery (BCS) and mastectomy have been the only two surgical choices for women with breast cancer. Survival rates from these techniques are similar, 1 although local recurrence rates are higher when the breast is conserved. 2 A meta-analysis by Clarke et al 3 demonstrating the negative impact of local recurrence on long-term survival after BCS highlighted the importance of thorough local excision in preventing local recurrence. 4
An extensive resection of parenchymal tissue around the tumor can reduce the risk of local recurrence but increases the risk of an unacceptable cosmetic result and the associated psychological distress. 5 Recent data suggest that resecting much more than 15% or 20% of the breast volume, particularly from central, medial, or inferior locations, is likely to lead to a poor cosmetic result. 6 Cosmetic failure can be caused by poor surgical technique, such as inappropriate incisions, or complications like hematoma formation, infection, and necrosis; however, volume loss underlies most of the more visible and distressing examples of poor cosmetic outcome.
Volume replacement methods using latissimus dorsi muscle can overcome volume loss by providing immediate volume replacement using living autologous tissue at the time of BCS. Although these techniques can be used to reconstruct resection defects in women with any breast size, they are best applied in women with small or medium breasts. This particular group of patients often cannot afford the loss of breast volume associated with therapeutic mammaplasty techniques, which are detailed in Chapters 15 and 17. Tissue replacement is also suitable for women with larger breasts who may prefer to avoid a breast reduction and the need for contralateral surgery to achieve asymmetry.
Evolution of the Technique
The latissimus dorsi muscle has been used regularly for breast reconstruction after a total mastectomy since the early 1970s, but in 1985 Pearl and Wisnicki 7 were the first to describe how a myocutaneous flap can be used to reconstruct the breast to correct a deformity after a partial mastectomy and radiotherapy. The following year, Santi et al 8 described using a myocutaneous latissimus dorsi flap for a technique to transpose the entire latissimus dorsi muscle through small incisions to correct a number of deformities, including those following a quadrantectomy. In 1988, deepithelialized flaps of latissimus dorsi were used by Papp et al 9 to correct iatrogenic and congenital volume loss.
Noguchi et al 10 were the first to describe immediate reconstruction of quadrantectomy defects with a myosubcutaneous latissimus dorsi flap in small-breasted Japanese women in 1990. In 1992 Slavin et al 11 used myocutaneous latissimus dorsi flaps as a secondary procedure to correct cosmetic deformities in a variety of breast locations after BCS. A year later, Zoetmulder et al 12 described the use of similar flaps for the immediate reconstruction of big resection defects resulting from the excision of large T2 and T3 breast cancers. In 1994 Rainsbury 13 described a modification of Noguchi’s technique and called this the latissimus dorsi miniflap. This technique allowed a quadrantectomy, axillary dissection, flap harvest, and reconstruction of the resection defect to be performed through a single lateral retromammary incision, thereby avoiding a frontal scar. Eaves et al 14 described an endoscopic latissimus dorsi flap harvest in 1995, and Audretsch et al 15 played a central role in popularizing the use of the latissimus dorsi muscle in mainland Europe to reconstruct the partial mastectomy defect.
Indications And Assessment
Patient Factors | Tumor Factors |
Small or medium breast | Any breast location |
20% to 70% volume loss | In situ disease |
Mastectomy declined | Invasive disease |
Contralateral surgery declined | Segmental disease |
Radiotherapy anticipated | Not locally advanced |
Limited comorbidity | Typically 100-350 g specimen |
Functioning, intact latissimus dorsi muscle | Clear margins |
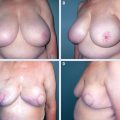
Volume replacement with a latissimus dorsi flap is a very useful alternative to a wide excision or a mastectomy when a patient wishes to conserve her breast and preserve its size and shape; it is also useful if an unfavorable cosmetic result with resection alone is a concern. A poor cosmetic outcome may result after resecting a 15 mm tumor from a small breast, whereas resecting a 45 mm tumor from a large, pendulous breast may result in little or no deformity.
Numerous factors must be considered before performing a volume replacement procedure. For example, the surgeon must determine the expected volume loss, which can be estimated in a number of ways. One simple technique that provides a good estimate of the percentage of breast tissue to be excised involves calculating the volume of the resection specimen (by assuming it is spherical and use the Pythagorean formula to find the volume of a sphere); this figure is then divided by the volume of the breast, which can be estimated from the craniocaudal mammogram. 6
An estimated resection volume of 10% to 15% is unlikely to have an adverse aesthetic outcome, and the patient can be treated with conventional BCS.
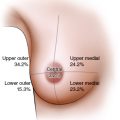
Resecting a larger proportion of the breast, even up to 70%, requires reconstructing the resection defect to avoid an inevitable cosmetic deformity. Patients with tumors in upper inner, central, and lower locations where the estimated resection volume is greater than 20% are particularly vulnerable to these deformities and should be advised against BCS without reconstruction. 6
An estimation of the tumor type and extent is an essential adjunct to the clinical evaluation. This is true for tumors in any location for in situ and invasive disease (see the table). By definition, to achieve the complete local excision of a breast cancer, the tumor in its entirety must be located within a breast segment and must be excised with tumor-free margins. The minimum preoperative workup should therefore include mammography and ultrasound, with MRI if a multifocal disease is suspected. In a small number of women with very large breasts, it may be impossible to harvest sufficient volume to reconstruct 300 to 400 g resection defects. In this clinical setting, either a therapeutic reduction mammaplasty or a total mastectomy and immediate breast reconstruction would be a more appropriate alternative.
The Decision-Making Process
Treatment Decision
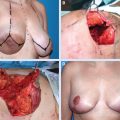
As with any relatively new and emerging technique, decisions about the patient selection, technique selection, and overall management undergo a continuous process of analysis and refinement. A partial mastectomy with immediate latissimus dorsi miniflap reconstruction was developed at the Winchester Breast Unit, 16 and more than 270 patients have undergone this operation at this facility.
Drawing on this experience, the following four key questions should be answered to help assess if this approach is suitable for your patient:
Is a partial mastectomy with oncoplastic reconstruction a reasonable treatment option?
Will the patient lose more than 15% to 20% of her breast volume?
Is the tumor in the upper pole, the upper inner quadrant, the subareolar region, or the lower pole, thus increasing the risk of cosmetic failure?
Does imaging confirm suitability for resection by segmental resection?
Is the patient prepared to have adjuvant breast irradiation?
Is the patient prepared to accept the risks of the procedure?
Is volume replacement preferable to therapeutic mammaplasty?
Does the patient have small to medium breasts (150 to 500 g)?
Does the patient prefer that her breasts remain the same size?
Does the patient prefer to avoid contralateral surgery, even if she has a larger breast size?
Is the patient prepared for more complex surgery and a longer convalescence?
Is the patient aware of the potential complications?
If she subsequently needs a mastectomy, is the patient aware that one of her options for full reconstruction will have already been “spent”?
Should the operation be carried out in one or two stages?
If a one-stage procedure is selected:
Is the pathologist prepared to perform an intraoperative frozen section analysis?
Are you confident using this method of analysis?
Does your operative schedule allow for any delays related to waiting for the frozen section examination?
Is your patient fully informed about the risk of a false-negative report and the subsequent sequelae?
Is your patient prepared to lose her nipple in the event of a positive subareolar biopsy?
If a two-stage procedure is selected:
When will the second stage be scheduled?
Can you accommodate both procedures in your timetable?
Will you delay any axillary surgery as well?
If not, are you skilled at identifying the thoracodorsal trunk in a recently dissected axilla?
A two-stage procedure has certain advantages. When access to pathologists with sufficient experience in frozen section margin analysis is limited, a detailed examination of all specimens, including sentinel nodes (SNs), can be performed without the time constraints of intraoperative analysis. In our experience, it should be considered in those situations where rapid margin analysis may be difficult, even for the most experienced pathologist. Interpretation is particularly challenging in patients following neoadjuvant chemotherapy and previous radiotherapy (RT), and also when resecting lobular carcinoma and low/intermediate grade DCIS.
The benefits of postmastectomy RT in patients with one to three positive nodes 17 and the significant risks of RT after postmastectomy reconstruction 18 will influence decisions about the timing and type of reconstruction in this group of patients.
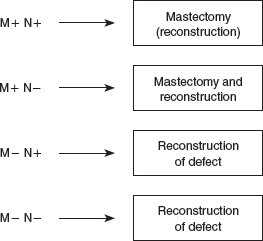
Delayed margin analysis informs decision-making and generates a number of different scenarios, depending on the final nodal and margin status. (M+, positive margin; M−, negative margin; N+, positive sentinel node; N−, negative sentinel node.)
In our experience, patients fall into four broad groups. Group 1 consists of patients with extensive margin positivity and positive SNs: conversion to mastectomy is recommended, delaying reconstruction until completion of chest wall RT. Group 2 has extensively involved margins but negative SNs, without other pathologic features indicating the need for RT (such as multicentricity, close margins, and T3/T4 tumors). Again, conversion to mastectomy is recommended, combined with reconstruction if desired. If the margin involvement is limited, an early reexcision may achieve clear margins, allowing reconstruction of the defect in this group of patients. Patients in groups 3 and 4 have clear margins, with or without positive SNs. They can be advised that reconstruction of the partial mastectomy defect followed by RT is a safe option that will result in outcomes similar to WLE and RT. 19
How are you going to manage the axilla? There is a range of alternatives based around the following three main options:
The SN is biopsied preoperatively, clearing the axilla at the time of reconstruction if positive or the area around the vascular pedicle if negative.
The SN is biopsied and assessed perioperatively, using the same approach that was followed for the preoperative SN biopsy specimen.
A “lateral” axillary clearance is undertaken, because the lateral aspect of the axilla has to be cleared anyway to identify and skeletonize the deep subscapular and thoracodorsal vascular pedicles.
Rationale for Choosing the Latissimus Dorsi Miniflap
The main alternatives to volume replacement with latissimus dorsi miniflaps are discussed in detail in Chapter 18. The rationale for selecting this approach over other techniques can be summarized as follows:
Use of implants to reconstruct resection defects has not been widely adopted. 20 , 21 Problems include interference with mammographic surveillance and capsular contraction leading to pain and local distortion. These features are not reported after latissimus dorsi miniflap reconstruction.
Lateral thoracic and other adipose tissue flaps have been reported mainly for the reconstruction of upper outer quadrant tumors. 22 , 23 They provide a relatively limited amount of volume for reconstruction. The latissimus dorsi miniflap can be used to reconstruct any part of the breast, and it provides a very significant amount of tissue for volume replacement.
Perforator flaps are more complex, and there are little data on their long-term performance. Experience using the pedicled latissimus dorsi flap is considerable and supports their reliability and durability.
Therapeutic mammaplasty techniques offer an excellent alternative in patients with medium or large breasts, when volume loss associated with these techniques is acceptable or beneficial. 24 , 25 There are potential problems with the margin orientation and analysis and with fat and parenchymal necrosis, which may delay or disrupt radiotherapy. Therapeutic mammoplasty excises tumors that are often situated in the center of a very large breast specimen. The surface area of this specimen is therefore extensive, increasing the time required for com prehensive pathologic examination.
Preoperative Planning
As with all oncoplastic and cosmetic breast surgeries, careful preoperative planning is vital to a good oncologic and cosmetic outcome. It should be carried out on the ward, with the patient relaxed and with good lighting. Before surgery, the patient must be made aware of the potential that a conversion to a completion mastectomy and reconstruction may prove necessary if negative margins are not achieved, and consent for this should be obtained in advance.
Choice of Incision
The choice of incisions on the breast, in the axilla, and over the latissimus dorsi muscle is influenced by oncologic considerations and by the type of latissimus dorsi miniflap selected for reconstruction.
Incisions on the Breast
There are four types of breast incisions that can be used: (1) a radial or circumferential incision over the tumor-bearing quadrant, (2) a circumareolar incision for subareolar and central tumors, (3) an elliptical incision, mainly for lower pole tumors, and (4) lateral S-shaped or inframammary fold incisions.
Axillary Incisions
The two choices of axillary incision are (1) a lateral S-shaped incision, which can be extended for access to the breast or the latissimus dorsi muscle and (2) a transverse incision, extending posteriorly for access to the latissimus dorsi muscle.
Incisions on the Back
The choice of incision on the back depends on whether a myocutaneous or myosubcutaneous (myofascial) latissimus dorsi miniflap has been selected. A myosubcutaneous latissimus dorsi miniflap is usually performed without a back incision, but a counterincision over the lower border of the flap has been described. A myocutaneous latissimus dorsi miniflap can be raised through an elliptical transverse or oblique incision, depending on the surgeon’s preference.
Choice of Technique
Myocutaneous | Myosubcutaneous | |
Resection site | Central and lower pole | Central and upper pole |
Volume loss | Up to 70% | Up to 50% |
Weight of flap | Up to 350 g | Up to 250 g |
Skin replacement | Yes* | No |
Body habitus | Any | Average or above |
*Including the nipple-areola complex.
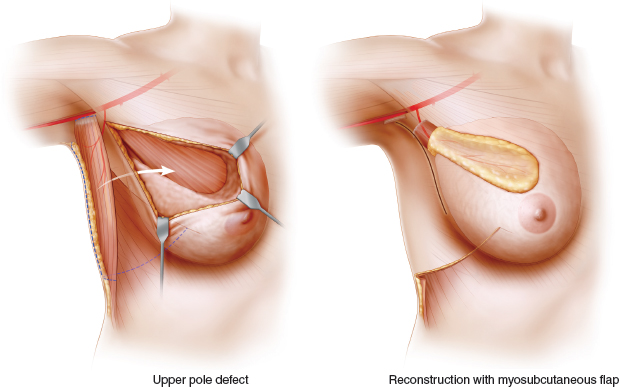
When planning a partial mastectomy, either a myosubcutaneous or myocutaneous flap is chosen for the latissimus dorsi miniflap reconstruction. The myosubcutaneous flap is best suited for reconstructing resection defects in the center and upper half of the breast, where scarring and 20% to 50% volume loss can lead to major local deformity. Most subcutaneous flaps weigh 150 to 250 g, and a myocutaneous flap should be considered when more extensive volume replacement is anticipated.
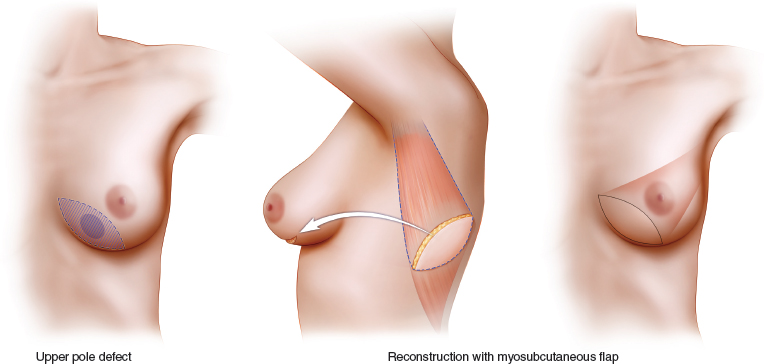
Lower pole and central defects are most easily reconstructed with a myocutaneous flap. This approach overcomes the difficulties encountered in recruiting and transposing a sufficient volume of flap to reconstruct these more remote locations. It is most appropriate when reconstructing large central and lower pole defects either by deepithelializing and burying the whole skin island and the flap, or by using a small island of skin to reconstruct the nipple-areola complex or the skin envelope of the lower pole of the breast. This approach is also useful in very slim patients in whom the volume of a myosubcutaneous flap is inadequate.
The added volume provided by the deepithelialized skin island allows volume replacement when 70% or even 80% of the breast is resected.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree