19 Facial Liposculpture and Fat Transfer
Key Concepts
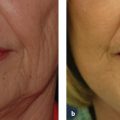
Volume loss contributes significantly to the aged appearance of the face, and autogenous fat is an attractive option for volume replacement.
Adipocyte-derived stem cells carried with the fat grafts seem to improve the quality and texture of the overlying skin.
Atraumatic transfer of fat is critical for success. Use of gentle harvest techniques with low negative pressures and gentle handling, and transplant of small aliquots of adipose tissue ensure optimum survival of the graft.
Use of small, blunt cannulas is essential to avoid embolization and arterial occlusion.
Introduction
Historically, it was believed that facial fat was a contiguous sheet of tissue and that facial aging was the result of the relentless downward pull on the facial skin and soft tissue. However, over the past 5 decades, several investigators have changed our understanding of facial aging. In 1965, Gonzalez-Ulloa and Flores proposed that facial aging involved changes in muscle and bone, as well as skin and fat.1 Further, recent work has demonstrated that facial fat is not a continuous sheet of tissue but rather is compartmentalized throughout the face.2 This discovery has allowed the evolution of improved techniques for facial rejuvenation.
For optimal results, the face should be considered in three dimensions, and the accepted surgical modalities of tightening and resurfacing of the skin are combined with the addition of volume. These principles should form the three limbs of the tripod for facial rejuvenation. Consideration of an individual′s appearance based on systematic mapping and a three-dimensional evaluation of the four levels of facial structure (bone, muscle, fat, and skin) will help the clinician to choose the most appropriate modalities for facial rejuvenation.
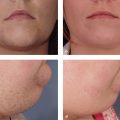
Autologous fat transplantation can be used to address all four levels of volume deficiency in the face and can provide a stable and potentially long-lasting alternative to off-the-shelf fillers. Transplanted fat can survive well in the face with meticulous grafting technique, although more than one injection in some areas may be required to achieve optimal outcomes. Appropriate counseling, realistic expectations, and exacting technique make fat transfer an effective method of soft tissue augmentation.
Background: Basic Science of Procedure
Autologous fat has been used for aesthetic facial surgery for over 100 years.3–6 In the 1950s, however, Peer demonstrated a graft survival rate of only 50% at 1 year.7 Enthusiasm subsequently waned for the technique while dermal adipose grafts and artificial materials gained in popularity.8 A new resurgence of interest occurred when Ellenbogen described free pearl fat grafts to the face.9 The development of liposuction by Illouz then provided a large supply of autologous fat in an injectable form, which appeared to survive when reinjected.10–12 This readily available material further bolstered the application of autologous fat transplantation for facial aesthetics. Refinements in technique have now produced more reliable results, and the concept of three-dimensional facial sculpting has led to the widespread application of autologous fat grafting to the face.13–15
Currently, there are two major theories describing the fate of grafted fat. Peer and Paddock proposed the host cell replacement theory, whereby host histiocytes phagocytize free fat and become adipocytes. The graft is thus replaced by host cells.16
This idea has been replaced by the cell survival theory.7,17 In this paradigm, circulation is restored to the grafted fat cells in a manner similar to the revascularization of a skin graft. Up to postoperative day 4, host cells such as polymorphonuclear leukocytes and lymphocytes infiltrate the graft. Within the vessels of the graft, blood cells are clumped and the graft survives by plasma imbibition. On or about the fourth day, neovascularization becomes evident. Host histiocytes act to remove fat from nonsurviving cells, and these scavenged cells tend to be the more mature adipocytes.
Some researchers believe centrifugation removes some of the mature, or “weaker” adipocytes, leaving more preadipocytes and resulting in a fat graft far more likely to be revascularized.18,19 Rigotti believes most mature fat cells are disrupted, and it is the adipose-derived stem cells carried with the graft that repopulate the recipient area. Removal of disrupted cells thus explains the loss of volume in the grafted areas.20,21 Most likely, the combination of cell revascularization and some stem cell differentiation accounts for treatment successes, but more study is required for definitive answers.
Technical Aspects of Procedure
The technique for autologous fat transfer can be divided into three parts: harvesting the graft, processing the graft, and reintroduction of the graft. Each is equally important to the success of the procedure.
Harvesting
Donor Site
Different donor sites have been advocated for the harvest of autogenous fat.22–26 We prefer the abdomen because it is easily accessible and stab incisions can be hidden within the umbilicus or in the hair-bearing skin of the pubic area. Most studies have failed to show an advantage of one donor site over another.25–27 Other common donor sites include the lumbar and trochanteric areas, the thighs including the knees, the arms, and the para-axillary area.
Tumescent Solution
Sterile technique is employed for fat harvest and injection. We recommend prophylactic antibiotics. Local anesthesia is used to anesthetize the site for a small stab incision. Through this incision, tumescent fluid is introduced into the region. The same incision is used for harvesting.
The choice of tumescent fluid varies. Our solution consists of 1 mg of epinephrine, 200 mg of lidocaine, and 5 mEq of sodium bicarbonate in 1 L of normal saline. Dosages of lidocaine up to 35 mg/kg can be used, although usually less is required for fat harvesting. Recently, a study on the effects of local anesthetics on preadipocytes found the immediate viability and the ability to differentiate were affected, with lidocaine demonstrating one of the strongest effects.28 One study noted that lidocaine potently inhibited glucose transport in adipocytes as well as their growth in culture. This effect, however, was only noted as long as the lidocaine was present. Adipocytes resumed normal metabolism and growth once they were washed.29
The β-adrenergic effects of catecholamines increase lipolysis in adipocytes, but it is unclear if there is a clinical effect of epinephrine in the tumescent solution used in fat transplantation.30
As an alternative, fat harvesting can be accomplished with minimal infiltration of lidocaine or “dry” without tumescent solution, thereby increasing the proportion of fat to residual fluid per harvested syringe.
Cannula and Negative Pressure
Atraumatic transfer of fat is a key principle for success. Trauma to fat in the process of harvesting or reintroduction affects the survival of the graft. Although a nonviable graft will initially appear to have corrected the problem, eventual resorption of the tissue negates the result.
Fat consists of adipose cells, which have thin cell membranes enmeshed in a fibrous network. Without the supporting fibers, the cells tend to collapse. An additional supporting network of connective tissue creates the lobules of fat, which can be observed grossly. Harvesting fat while retaining the supporting elements preserves the structural integrity of the tissue and helps the grafted fat survive in the transplanted site. The primary innovation of the Coleman technique was the use of a blunt-tipped cannula with dull distal openings, which harvests parcels of fat with intact architecture. These are small enough to pass through a 17-gauge injection cannula. The small parcels of fat placed in this manner allow maximal surface area contact with the recipient area.31
For harvesting, a 10 mL Luer Lock syringe is attached to the harvesting cannula, and the plunger is gently withdrawn to provide 1 to 2 mL of negative pressure. Larger cannulas combined with low aspiration pressure reduce trauma to the adipose tissue. Another commonly utilized technique designed to harvest the fat in parcels of readily transferable size uses the Lipivage system (Genesis Biosystems, Lewisville, TX). The aspiration cannula is connected to a low-suction (< 15 mm Hg) aspiration system used to harvest the fat. Traditional vacuum or syringe aspiration may be used as long as low pressure and large cannulas (4 or 5 mm) are utilized. A gentle passing motion for aspiration further limits trauma to the adipocytes ( Fig. 19.1 ) Video 19.1.
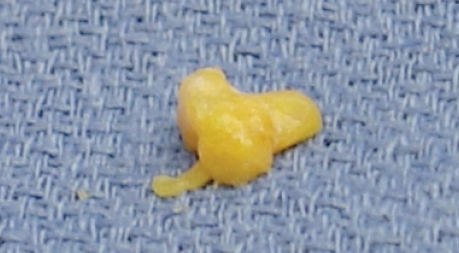
An alternative technique involves the use of a single-use disposable aspiration and injection kit, the Viafill System (Lipose, Maitland, FL). The kit includes a centrifuge, aspiration and injection syringes and cannulas, and centrifuge tubes. Fat aspiration is performed using a low negative pressure technique. The aspiration cannula has a diameter of 14 French and measures 12 cm in length. Centrifugation is performed at a low force of 50 g for a brief 2 minutes.
There is some controversy regarding the effect of negative pressure on the adipocyte.32–37 Despite a wide range of results, the fact remains that many groups have demonstrated either structural or metabolic derangements of fat cells after harvest with high negative pressures.32–34 Therefore, avoidance of trauma to the adipocytes is essential. The keys to atraumatic harvest are large cannulas, low negative pressures, and gentle passage of the cannula.
Processing
The goal of fat cell processing is to create the isolation and preparation of the best cells for reinjection while minimizing potential damage. The stromal vascular fraction remaining in the harvested fat can facilitate rapid degradation and must be removed. Strategies include systems such as Lipivage or Viafill, centrifuge, or gravity separation. There are advantages and disadvantages to all techniques. The Lipivage system is a closed filtration system, avoids exposure to excessive g force, and provides sterility and lack of exposure to air.
An alternative is the use of the centrifuge. Some investigators have demonstrated deleterious effects of a centrifuge on adipocytes, whereas others have had favorable results.38,39 After centrifugation, those fat cells surviving the process tend to be younger and tougher and are found in the compacted region of viable adipocytes.19,21,40 In addition, the layers of the compacted region differ with regard to the number of viable adipocytes. The bottom portion of the fat layer demonstrates 250% more viable cells when compared with the top layer and 140% more viable cells when compared with the middle layer.40,41 Therefore, using the bottom layer of the centrifuged cells is recommended.
Gravity is also an alternative for separation and is used by some to produce the least trauma to the adipocyte. One disadvantage is the time required. Another is the possibility that some viable cells are not included in the fraction that is grafted.39 Our technique utilizes both filtration and gravity separation. After the fat has been gently harvested and filtered by the Lipivage system, it is transferred to 30 or 60 mL syringes via a locking system and allowed to separate by gravity for 30 minutes ( Fig. 19.2 ). The excess fluids, including the upper layer of oil from disrupted fat cells and the lower layer of blood and lidocaine, are then removed and the fat transferred to 1 mL syringes for injection. The technique minimizes trauma and obtains a large number of viable cells. An alternative is the Viafill system using a sterile closed system and a low-g-force centrifuge Video 19.1 .
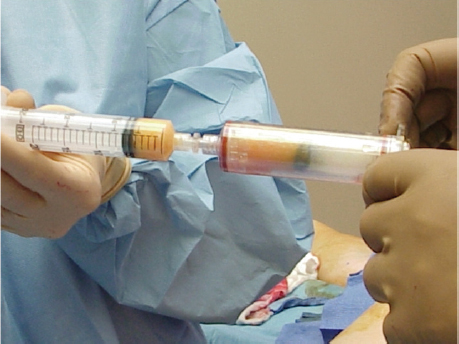
Grafting
The goal with any grafting procedure is to maximize graft survival by gentle reintroduction of fat cells into a well-vascularized bed. A Coleman Type II (Byron Medical, Tucson, AZ) 7 cm straight injecting cannula is inserted using a 2 mm stab incision made with an no. 11 blade knife and is used with the compacted fat cells in a 1 mL syringe. Small tunnels are created by advancing the cannula, and the fat is injected 0.1 to 0.2 mL at a time, only while withdrawing the injecting cannula. To reduce embolization, never inject with a sharp needle nor when advancing the injection cannula. The creation of small tunnels helps keep the grafted fat adherent to the recipient site, whereas independent tunnels prevent fat from leaking out. Care should be taken to avoid overfilling the tunnels because this can adversely affect graft survival. Do not use force; this creates an uncontrolled graft unlikely to survive. However, it is important to feel a small amount of resistance while injecting. If the injection goes too easily, it is likely that fat has already been placed in this location, and further injection will reduce graft survival.
Slight overcorrection is important to compensate for absorption of the liquid carrier. A general recommendation is for 30% overcorrection. After the fat is deposited, gentle digital pressure is used to check for placement and contour regularity. Additional facial surgical procedures can be completed at the time of lipoinjection if the procedures do not involve the graft recipient areas. Operating on a freshly injected area will disrupt the fat placement.
Patient Preparation
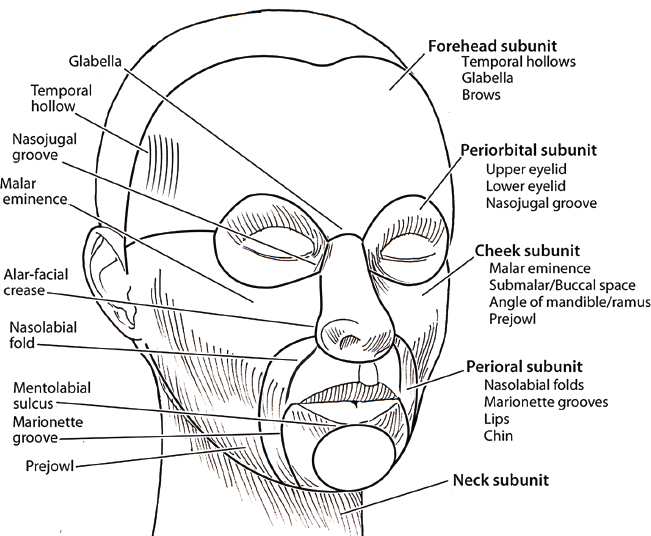
The face is divided into forehead, cheek, periorbital, nasal, perioral, and neck subunits ( Fig. 19.3 ). Each subunit is examined for three-dimensional (3-D) volume loss and is then broken down into grafting units. Preoperative photographs are taken with and without a flash. Photographs taken without flash will often accentuate areas of volume loss not apparent in standard flash photos. It is helpful to compare preoperative photographs to pictures taken of patients in their early twenties, preferably full face, nonsmiling views to better appreciate the degree of volume loss present.
The patient is marked preoperatively sitting upright in an area with direct overhead lighting, often supplemented with side lighting as needed to identify the areas of volume deficiency to be grafted.
Injection
By 3-D analysis of each subunit, an estimate of grafting volume as well as planes of injection can be formulated in a preoperative plan ( Table 19.1 ).
The Forehead Subunit
Fat injections to the forehead should only be performed using a blunt injection cannula. Injections are only made parallel to the lines for wrinkles.
Temporal Hollows
The temporal hollows are marked pretreatment to accentuate the posterior aspect of the lateral orbital rim just above the zygomatic arch. This area is usually the deepest part of the hollow and is often asymmetric between sides. Access points are made at the temporal hairline and work best with two different vectors. Special care must be taken when injecting the temporal hollow to avoid injury to the frontal branch of the facial nerve.
Units | Subunits |
Forehead | Forehead |
Brows | |
Glabella | |
Temporal hollows | |
Supraorbital units | |
Periorbital | Lower eyelids |
Upper eyelids | |
Lateral orbital rim | |
Nasojugal groove | |
Cheek | Malar eminence |
Submalar area | |
Mandible angle | |
Mandible ramus | |
Prejowl area | |
Perioral | Nasolabial folds |
Lips | |
Labiomental crease | |
Marionette lines | |
Chin | |
Philtrum | |
Neck | Submentum |
Neck | |
Suprahyoid | |
Subjowl |
The most visually important part of the temporal hollow is adjacent to the lateral orbit and the tail of the brow. The key to a good result is depth of injection, which should be in the plane immediately adjacent to the deep temporal fascia. This plane is deep to visible vessels and the frontal branch of the facial nerve. Injection should be performed while one is withdrawing the cannula, and care should be taken to avoid the sentinel vein.
The goal is expansion of the temporal hollow. Placement of 4 to 10 mL of fat in each hollow is done as evenly as possible, expecting ~ 30% resorption. Most of the fat should be placed anteriorly. It is not unusual to have asymmetry between hollows, but care must be taken not to overfill because the goal is to level the temporal hollow, not make it convex. With the temporal hollow leveled, the tail of the brow rotates anteriorly, giving the impression of rising and lengthening.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree