16 Treatment of Facial Imperfections with Dermal Fillers
Key Concepts
Numerous soft tissue filling agents are available for facial rejuvenation and scar revision.
Knowledge of the properties of individual soft tissue fillers will guide the aesthetic physician in selecting the appropriate agent for a particular patient and anatomical location.
It is critical that the aesthetic practitioner recognize an adverse event and initiate appropriate action at the first sign of a complication from a soft tissue filler.
Introduction
As the demand for facial rejuvenation continues to rise, the goal for physicians performing aesthetic procedures is to optimize outcomes while minimizing downtime. It has become recognized that successful rejuvenation of most cosmetic patients requires addressing facial volume deficiencies. Therefore, the armamentarium of soft tissue fillers continues to grow, making it more important than ever that the aesthetic practitioner choose the best soft tissue filler for each patient and anatomical location.
This chapter serves as a practical guide for injecting dermal fillers for all practitioners regardless of experience. Agents discussed include the hyaluronic acids: Restylane/Perlane (Medicis Pharmaceutical, Scottsdale, AZ), Juvederm Ultra/Juvederm Ultra Plus (Allergan, Irvine, CA), Prevelle Silk (Mentor, Santa Barbara, CA), and Belotero (Merz Aesthetics, San Mateo, CA); calcium hydroxylapatite (Radiesse, Merz Aesthetics, San Mateo, CA); poly-L-lactic acid (PLLA, Sculptra Aesthetic, Sanofi-Aventis U.S., Bridgewater, NJ); polymethyl methacrylate (PMMA, Artefill, Suneva Medical, San Diego, CA); and the technique of autologous fat transfer. Silicone injections are not addressed in this chapter.
Background: Basic Science of Procedure
Special factors to take into consideration when selecting the appropriate agent for a patient include longevity of the filler, the anatomical area to be treated, the depth of the defect, the risk of allergic reaction, the risk of delayed (granulomatous) reactions, and the risk of infection.
Hyaluronic Acid Fillers
Hyaluronic acid (HA) is a ubiquitous component of connective and synovial tissues. It is a glycosaminoglycan biopolymer composed of alternating residues of D-glucuronic acid and N-acetyl-D-glycosamine that can bind up to 10,000 times its weight in water. Its affinity for water molecules gives it a unique ability to add to skin turgor and volume. However, injection of unmodified HA results in almost immediate degradation. Therefore, for HA to be a clinically useful injectable filler, it must undergo cross-linking to confer longevity. Currently available HA fillers will vary with regard to particle size and the type of cross-linking used. For example, Restylane has 100,000 particles/mL, and Perlane has 10,000 particles/mL. Although Perlane has fewer particles per milliliter, the particles are much larger than thaose of Restylane.
HA fillers continue to gain popularity among both practitioners and patients due to ease of use, lower rates of hypersensitivity reactions, longer-lasting effects than traditional human and bovine collagen fillers, and reversibility upon injection with hyaluronidase.1 Several agents are available, including Restylane and Perlane, Juvederm Ultra/Juvederm Ultra Plus, Prevelle Silk, and Belotero. Although Hylaform (no longer available) is avian derived, the others are derived from Streptococcus fermentation.2,3
Restylane, Juvederm Ultra, and Prevelle Silk are injected into the mid-dermis. Perlane, Belotero, Juvederm Ultra Plus, and Juvederm Ultra Plus XC are injected into the deep dermis/subcutaneous junction. Mid-dermal injections can be accomplished with the use of a 30-gauge half-inch needle, which allows for easy flow of the filler and minimizes bruising.
Deeper filler placement into the dermal/subcutaneous junction can be accomplished with a 29-gauge, thin-walled needle (Terumo Medical, Somerset, NJ) or a 27-gauge needle. The 29- gauge thin-walled needles have replaced the need for 27-gauge needles in the authors’ practice. These needles have a 29-gauge outside diameter and an inside diameter equivalent to a regular 27-gauge needle, thereby allowing the treatment to proceed with less bruising.
Studies indicate that the effects of products such as Restylane last approximately 6 months.4 Juvederm has been reported in the literature to last for 12 to 15 months in the deeper folds such as the melolabial region.5 In the authors’ experience, Juvederm has proven to last for a shorter duration of approximately 6 months in most patients and up to 18 months in a small subset of patients.
Calcium Hydroxylapatite
Calcium hydroxylapatite (CaHA), which is naturally found in bone, had been used in orthopedics, dentistry, and urology prior to its use as a semipermanent soft tissue filler.6,7 The CaHA microspheres measure 25 to 45 µm, are suspended in a carboxymethylcellulose gel base, and are capable of augmenting bone structure.
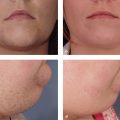
CaHA is a volumizing filler, making it not ideal for finer lines but useful for elevating soft tissue, such as in the malar, buccal, and mandibular regions. The risk of visible nodules makes it unsuitable for augmentation of the periorbital region (unless cautiously placed deep along the periosteum) and the lips.
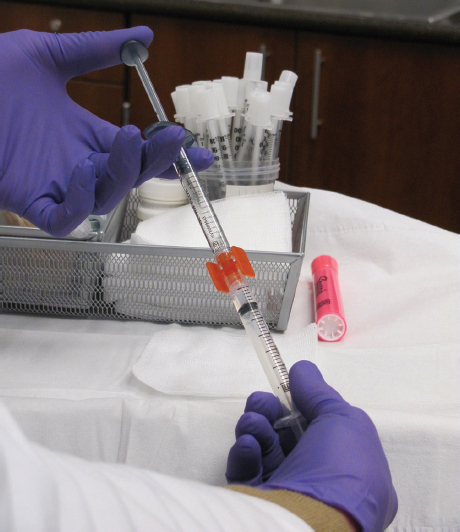
Prior to injecting, the addition of 0.2 mL sodium bicarbonate and 0.3 mL lidocaine with epinephrine has shown to improve patient comfort. The vasoconstrictive effects of lidocaine with epinephrine help to reduce bruising. The dilution is best achieved with a female-to-female Luer Lock-to-Luer Lock adapter (Sterile Rapid-Fill Connector, Baxter, Englewood, CO) ( Fig. 16.1 ).8 The use of a 28-gauge three-quarter-inch needle and a retrograde injection is the preferred technique. The product is placed into the deep subcutaneous plane or at the level of the periosteum. The material is then massaged immediately after injection to avoid palpable nodule formation. Typically, the effects of CaHA last for at least 1 year and possibly longer.4
Poly-L-Lactic Acid
PLLA was originally used for manufacturing absorbable suture material (Vicryl, Dexon) prior to its use as a soft tissue filling agent. Sculptra (Sanofi-Aventis), the powdered form, consists of 40 to 63 µm microspheres2 suspended in a nonpyrogenic mannitol and sodium cellulose preparation.9 Placement of PLLA into the deep dermal–subcutaneous plane yields dermal fibroplasia and cutaneous thickening over the subsequent months.9 Previously published reports indicate that higher dilutions may result in a more uniform mixture with decreased incidence of nodule formation.9,10,11 The manufacturer suggests diluting PLLA with 5 mL sterile water for injection at least 2 hours prior to planned injection and keeping the reconstituted product for no more than 72 hours.12 More recently, the authors favor reconstitution with 6 mL bacteriostatic water 7 days prior to the planned injection. On the day of the procedure, 3 mL lidocaine with epinephrine is added to aid in the reduction of bruising. A gentle rolling motion of the vial prior to injection will ensure an even suspension. The PLLA is transferred into 1 mL syringes via a 21-gauge needle immediately prior to injection. The areas to be treated are outlined with a marking pen prior to the injection of PLLA. The authors use a depot (0.1 to 0.2 mL aliquots) and fanning technique with a 25-gauge 1-inch needle for soft tissue augmentation at the level of the upper subcutaneous fat and along the periosteum in certain areas. PLLA has been reported to have its greatest effect at approximately 28 months posttreatment.4 Bauer has reported the effects of PLLA lasting up to 40 months in some individuals.13
Polymethyl Methacrylate
PMMA consists of microspheres suspended in a collagen matrix with 0.3% lidocaine.14 The 30 to 40 µm microspheres are suspended in bovine collagen derived from a closed herd in the United States.14 The bovine collagen serves to deliver the microspheres to the soft tissue. The microspheres are left behind to stimulate the deposition of the patient′s own collagen in place of the bovine collagen.14 It is recommended that this product be injected into the deep dermis/subcutaneous junction to prevent surface irregularities. It is important to perform skin testing prior to its injection given the bovine collagen contents. This agent has been shown to provide correction in the highly mobile region of the nasolabial folds for as long as 5 years postinjection.14 Due to the need for skin testing, patients may find the product less convenient.
Autologous Fat Augmentation
Any discussion of soft tissue filling agents would not be complete without the mention of autologous fat transfer. In most patients of normal to increased body mass index, the donor site is plentiful and holds virtually no risk of immunogenicity.15 The authors favor a modification of a technique developed by Dr. Sidney Coleman, in which adipocytes are harvested in manual suction via 10 mL syringes.16 Minute fat parcels (0.05 to 0.1 mL) are placed along multiple facial places using blunt-tip cannulas for a long-lasting restoration of facial volume. The small depot technique is designed to maximize surface area exposure of individual adipocytes and may improve the vascular supply to the newly grafted tissue.15 Care must be taken to ensure that the fat is harvested, prepared, and infiltrated in a sterile fashion to minimize complications, including infection.15 The patient is advised that the excess harvested fat will be frozen for 1 year and can be used for touch-ups within that time period.15
Technical Aspects of Procedure
Autologous fat transfer is the authors’ preferred method for full-face soft tissue augmentation. However, not all patients have adequate fat donor sites or are willing to tolerate the 7-day recovery period associated with the procedure. The authors consider the following synthetic filling agents to be the products of choice when autologous fat cannot be utilized. The approach to the patient is broken down by anatomical region, with certain fillers having better filling properties than others for a given area Video 16.1.
Periorbital Region
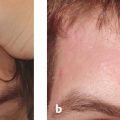
The orbital region tends to naturally decrease in volume with age.17 In addition, some patients have undergone aggressive blepharoplasty procedures that have further exacerbated the orbital hollowing. More attention is being placed on volume restoration of the periorbital region as part of full facial rejuvenation.18 Patients may find the concept of adding volume to the brow in an effort to augment their appearance difficult to visualize; Lambros proposed injecting a dilute local anesthetic into the brow region to offer the patient a preview of the outcome.19
The periorbital region consists of the thinnest skin on the face. Thus care should be taken to use a filler that will result in smooth contour enhancement. Certain large-particle fillers can result in visible nodules in this area and are best avoided.
The authors favor the use of a small-particle HA, such as Restylane, in the periorbital region for brow and tear-trough augmentation. This filler has a very smooth consistency upon injection, can be easily massaged into place, and has the least likelihood of edema. Additionally, the use of an HA filler in the periorbital region has the added advantage of being reversible with hyaluronidase should the patient not like the final outcome.
Although fillers can be purchased with lidocaine premixed in them, the authors prefer to mix the filler with 0.3 mL of 1% lidocaine plus epinephrine and 0.2 mL of sodium bicarbonate. A female-to-female Luer Lock adapter allows for the addition of lidocaine with epinephrine ( Fig. 16.1 ).8 The epinephrine induces vasoconstriction and decreased bruising. The mixing technique also allows for ease of injection and improves malleability of the product.
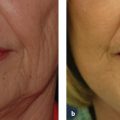
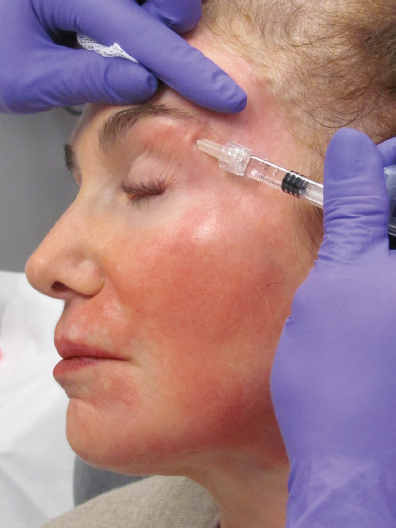
Brow augmentation is used for patients who have lost volume in the brow fat pad. The deflation of the brow appears as dermatochalasis of the upper eyelid and a bony appearance of the brow. A 30-gauge half-inch needle is used to inject this area in a linear, retrograde fashion along the contour of the upper orbital rim ( Fig. 16.2 ). The area is then massaged in a linear fashion to smooth out the contour and to identify “skip” areas.
The tear-trough deformity poses a challenge because it may be due to (1) loss of volume along the lid–cheek junction, (2) loss of volume and increased lower eyelid edema in patients with a history of seasonal allergies, or (3) iatrogenic hollowing following lower lid fat resection. The goal of treatment is to blend the lid–cheek junction for a smoother appearance to the area. However, patients with thinner skin should be counseled that it is best to perform this treatment conservatively over a couple of treatment sessions so that one does not create visible nodules.
For tear-trough augmentation, use a 30-gauge half-inch needle to inject small (0.1 mL), submuscular depots of HAs (mixed with lidocaine plus epinephrine) over the bony orbit, followed by massage in the direction of the deformity (i.e., massage toward the nose if trying to fill the medial aspect of the tear-trough deformity). Injection in the submuscular plane will help to avoid the Tyndall effect (or visible blue hue). In thicker-skinned patients, it is adequate to inject submuscularly. However, in thinner-skinned individuals, one may have to inject at the level of the periosteum to avoid visible filler or nodule formation. The area is then massaged along the entire lower orbital rim to ensure a smooth contour and to avoid skip areas. Lambros has reported that the effects of HAs in the lower lids and brows can exceed the 2 year mark.19 The use of a larger particle size of HA, such as Juvederm Ultra Plus, in the lower eyelid can result in prolonged posttreatment edema in some patients and should thus be undertaken cautiously.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree