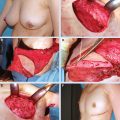
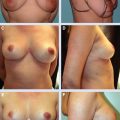
16 Reduction and Mastopexy Techniques With Parenchymal Autoaugmentation
Mammaplasty reduction techniques have become a popular option for the management of partial mastectomy defects, as described in Chapter 15. Plastic surgeons are familiar with these techniques, making the incorporation of this approach into their reconstructive practice an easy addition. 1 – 3 They are probably the most common oncoplastic techniques used by plastic surgeons in the United States. In Europe, they became popular for reconstructing quadrantectomy defects in the lower pole, improving the poor cosmetic results associated with defects in this location. 4
In the United States, the popularity of this technique probably evolved out of the frustration of managing breast cancer patients with macromastia. 5 Large, pendulous breasts are often more difficult to reconstruct after a total mastectomy because of the patient’s body habitus, any associated morbidities, and the inherent difficulties associated with skin envelope reduction. These women are often deemed poor candidates for reconstruction because of the increased potential for complications and unfavorable cosmetic results. Breast-conserving therapy (BCT) was also seen as less desirable for women with large breasts. Macromastia initially was believed to be a relative contraindication to BCT because of poor cosmetic results and less effective radiotherapy. 6 Radiation-induced fibrosis is a greater problem in women with larger breasts because of the dosing inhomogeneity. 7 , 8 In addition, late-radiation fibrosis has been demonstrated in 36% of patients with larger breasts, compared with 3.6% of patients with smaller breasts. 8 Higher doses of radiotherapy are often necessary in women with larger breasts, which contributes to morbidity and adversely affects the appearance of the breast.
For these reasons, reduction mammaplasty techniques were a welcome addition to the reconstructive armamentarium for partial mastectomy defects in patients with macromastia. Suddenly, BCT became a viable option for women with large breasts, simply by adding reduction techniques. These techniques are relatively easy to learn and produce predictable results for patients in whom a total mastectomy and reconstruction can be extremely challenging. This chapter expands on the use of simple reduction mammaplasty techniques for reconstructing partial mastectomy defects.
The Decision-Making Process
Indications
The indications for oncoplastic techniques are covered in detail in Chapter 11. These techniques minimize the potential for a BCT deformity and broaden the indications for BCT in select patients. Traditionally, reduction techniques were used either in women with large breasts in whom sufficient breast tissue was available to reshape the breast using almost any pedicle type (glandular displacement) or in women in whom the partial mastectomy defect could be incorporated into the tissue being removed in a standard reduction technique. An example of the latter is the removal of a lower pole tumor with the dermatoglandular Wise pattern using a superior pedicle reduction. However, as familiarity with BCT grew, the indications for its use broadened.
This chapter demonstrates how glandular flaps or autoaugmentation from within the breast mound—together with standard reduction or mastopexy techniques—can be used to reconstruct lumpectomy defects in almost any location, as long as sufficient breast tissue remains after the resection. This technique suddenly becomes a reasonable option for women with smaller, ptotic breasts, even when the defect lies outside of the typical reduction specimen. When the defect is small enough, the indications have evolved into mastopexy-type techniques in women with moderate-sized or ptotic breasts.
Understanding the Defect
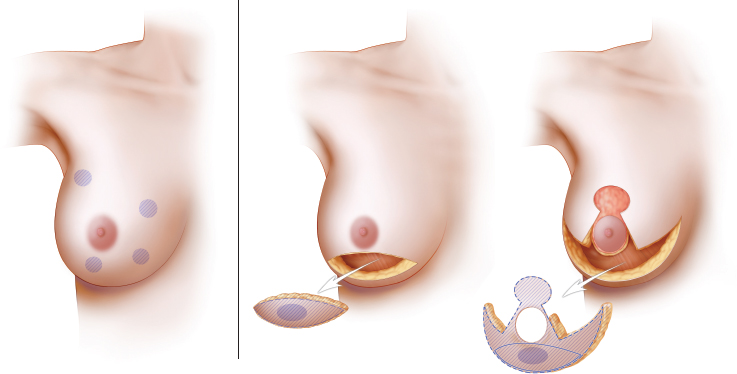
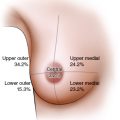
The type of defect is important when considering the reduction technique. This approach is reasonable even for a quadrantectomy-type defect involving skin and parenchyma, but only when the skin resection is within the Wise pattern or vertical take-out pattern. In some situations, a medial or lateral resection is required outside the Wise pattern markings, either to resect some skin involvement or to resect a previous scar. In these cases, as long as the defect is low and small enough, it can be “plugged” with a small skin island from the deepithelialized dermatoglandular pedicle. Any skin defect in the upper half of the breast typically requires a flap reconstruction. Lumpectomy-type defects involving only parenchyma are easier to reconstruct, because the issue is mainly a volume void. In moderate or large breasts with or without ptosis, a lumpectomy defect in almost any location within the breast mound can be reconstructed using reduction or mastopexy techniques. This is especially true for larger breasts in which a simple reduction pattern is usually sufficient. In smaller breasts, when the defect is not in an area that would ordinarily have been resected with a standard mastopexy (that is, the lower pole), glandular flaps are required to fill the defect. One example is a medial defect in a cosmetically sensitive location in smaller-breasted women in whom the volume of the pedicle or remaining breast parenchyma is not sufficient to fill the defect without autoaugmentation. The remaining tumors discussed in the chapter are lumpectomy-type defects, unless otherwise specified.
No-Man’s Land
Reduction and mastopexy techniques essentially involve creating a smaller breast mound and positioning the areola at the center of the mound. However, in some situations, mobilizing the flap carrying the areola to reshape the breast mound is limited by where the nipple needs to be positioned. For aesthetic reasons the nipple cannot be placed higher than 16 cm from the sternal notch or closer than 7 cm from the midline. A woman with smaller breasts and a tumor in this location—referred to as no-man’s land—cannot rely on filling the defect with the nipple pedicle, and additional measures are required. 9 However, reconstruction of these defects is now possible by using reduction techniques and either extended dermatoglandular or secondary pedicles.
Surgical Technique
Surgical Plan
The decision to proceed with the oncoplastic breast conservation is discussed with the team, and the choice between an immediate or delayed-immediate procedure is made.
The patient is marked preoperatively.
Wire is placed for tumor localization, if necessary.
The resective and reconstructive surgeons discuss the final plan after the wire has been placed and the patient has been marked.
The tumor is excised, with or without lymph node sampling.
Mammographic confirmation with frozen section analysis is performed, if appropriate.
Separate cavity sampling is performed.
The cavity is clipped for orientation.
The residual breast tissue and the size and location of the deformity are evaluated.
The intraoperative reconstructive goals are as follows:
Preserve nipple viability and reposition the nipple on the breast mound.
Eliminate dead space.
Resect excess breast parenchyma and skin.
Reshape the breast mound to preserve its shape.
Perform a contralateral procedure for symmetry, if necessary.
As the indications for therapeutic mammaplasty become more liberal, we cannot forget the importance of patient selection for optimal cosmetic outcomes and oncologic safety.
Patient Selection
Patient selection is multifactorial; however, it is usually initiated by the resective surgeon, who is concerned either about a potential deformity or asymmetry using the standard excision or about not being able to obtain clear margins without a wide excision. This process may also begin with the patient’s wish to have smaller breasts.
The ideal patient for this oncoplastic technique is one whose tumor can be widely excised within the reduction specimen and for whom a smaller breast is viewed as a positive outcome. The aim of this approach is threefold: a high survival rate, a good cosmetic outcome, and improved patient satisfaction. Patient selection is also critical in terms of margin status, because some women (younger patients with ductal carcinoma in situ [DCIS]) are at a higher risk for positive margins, which influences the timing of partial breast reconstruction.
Preoperative Planning
Once it is established that the patient is a candidate for BCT and would benefit from a simultaneous reduction, the preoperative planning phase can begin. If a two-team approach is used, communication between the teams is essential. The resective surgeon must understand the importance of the blood supply to the nipple, placement of skin incisions, and breast aesthetics. It is equally vital that the reconstructive surgeon understand the size and location of the tumor and the margin status and locoregional control. They should review the radiographic imaging and discuss the anticipated defect location and size, as well as whether the resection should include skin. This discussion helps to determine the most appropriate glandular pedicle to use to maintain nipple viability and to reshape the mound. There should always be a backup plan, because the defect occasionally is different from what was anticipated.
Preoperative Marking
Preoperative marking must be relatively conservative on both sides. If a Wise pattern is drawn, its vertical limbs are slightly longer than normal, and the angle is smaller to ensure minimal tension on the incisions and to reduce the incidence of healing problems. If radiographically placed wires are used for the lumpectomy, these should be examined and the radiographs reviewed.
The team should discuss possible access incisions on the mound for tumor resection. Poorly placed incisions could interfere with the viability of the skin flaps and lead to less than optimal results.
Tumor Resection
The tumor resection is performed in the usual fashion using safe and effective oncologic principles (see Chapter 5). The intraoperative margin assessment could include radiographic imaging, macroscopic assessment, frozen section analysis, or touch cytology. We have found that patient selection, cavity sampling, and generous resection further reduce the incidence of positive margins. The cavity is clipped for postoperative surveillance and guidance for radiation boosts to the tumor bed, if required.
Partial Mastectomy Reconstruction
Partial mastectomy reconstruction typically is performed before radiotherapy. It can be delayed until negative margins are confirmed in patients who are at an increased risk of having positive margins (that is, patients younger than 40 years of age and patients with extensive ductal carcinoma in situ [DCIS], lobular carcinoma in situ [LCIS], or prior chemotherapy). As with any oncoplastic procedure, the technique selection depends on the breast size and shape and the defect size and location. Numerous options exist, depending on these variables.
On examination of the partial mastectomy defect, we are faced with trying to determine the following:
How to keep the nipple viable and positioned appropriately on the mound
How to fill the dead space
Where to resect excess breast tissue when necessary
How to reshape the breast mound using the pedicles and remaining breast tissue
When deciding on reconstructive options, one aspect that is almost more important than the size of the defect is evaluating the remaining breast tissue and determining where it is in relation to the defect, the nipple, and the breast mound. The first decision that should be addressed is establishing how the nipple will be kept alive. Typically, the shortest pedicle maximizes the nipple viability and allows additional glandular manipulation, without compromising the nipple. Many options exist for nipple pedicles, and most surgeons have a favorite. For example, if the superomedial pedicle is the procedure of choice for standard breast reductions, then this technique could be used for most oncoplastic defects—provided the patient is a candidate and the defect location is not medial to the nipple. As a general rule, if the pedicle points to or can be rotated into the defect, it can be used. Occasionally, it is not possible to preserve the nipple because of the breast size or the tumor location. Options include amputation and a free nipple graft or nipple reconstruction at a later date.
Once the nipple preservation has been determined, the pedicle is deepithelialized and dissected using the cautery unit, enough to allow rotation into the proposed nipple position. A second decision is then made about how to fill the dead space. At this point, the glandular resection has not yet been performed. If the defect is removed as part of a reduction specimen and is adequately filled through glandular displacement with the pedicle and/or remaining glandular tissue, then autoaugmentation is not required. If it is believed that additional glandular flaps are required to fill the dead space, then a decision is made based on what tissue is available and where it is in relation to the nipple pedicle. If possible, filling the defect by rotating an extended portion of the original nipple pedicle is often the technique of choice. This single-pedicle autoaugmentation approach works well for smaller defects in women with small- or moderate-sized breasts or when tissue can be taken with the pedicle from a less cosmetically sensitive area and rotated to fill a defect.
Patient Example
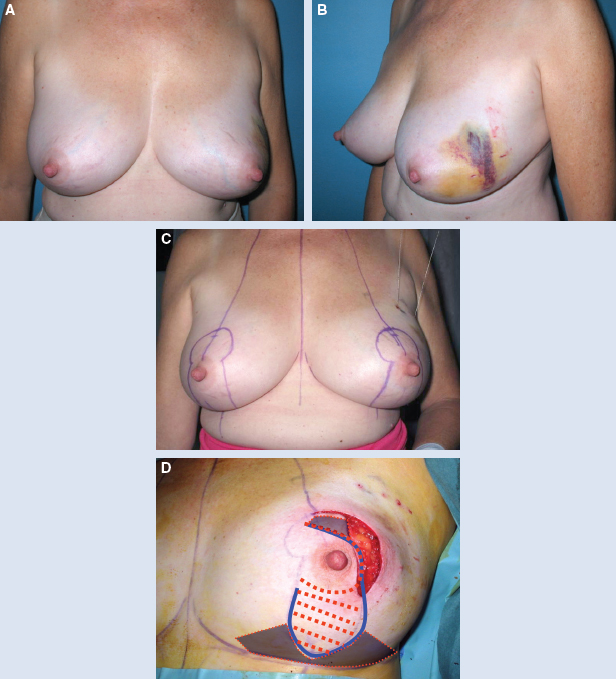
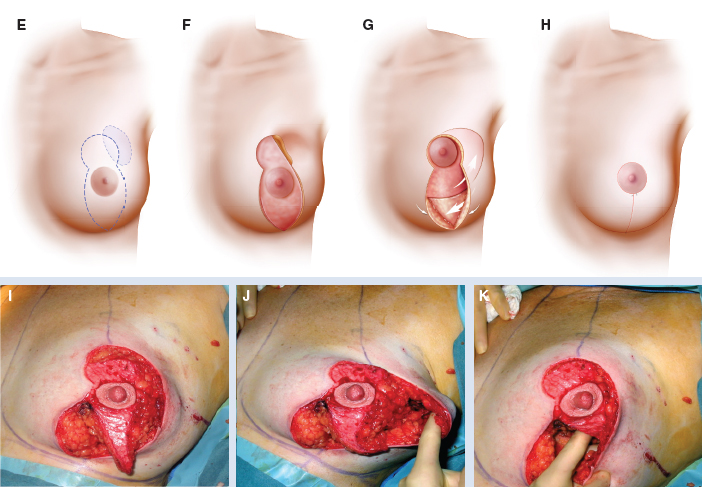
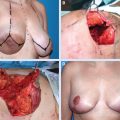
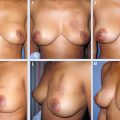
This 42-year-old woman had moderate-sized breasts with minimal ptosis and some asymmetry (A and B). Her breasts were marked preoperatively (C). A 92 g DCIS was resected from the lateral aspect of her left breast, leaving a defect slightly above the proposed nipple position (D). This was filled with an extended superomedial dermatoglandular pedicle (blue line) that was rotated to fill the defect. An additional 22 g was resected from above the nipple and at the inframammary fold (gray shading).
The medial and lateral pillars were plicated in the usual vertical mastopexy fashion, and a contralateral symmetry procedure was performed (total resected 102 g) (E through K).
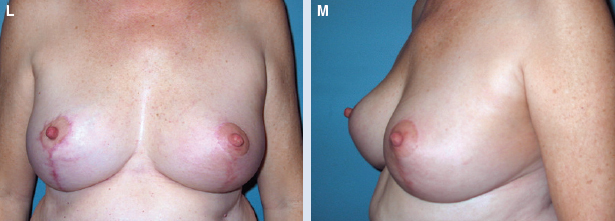
She had good contour laterally 1 year after radiotherapy (L and M).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree