15 Cleft Palate
Summary
Cleft palate is a congenital craniofacial anomaly that affects feeding, hearing, and speech. The hard palate gives structural support, and the soft palate provides velopharyngeal competence. The aim of cleft palate repair is to close the oronasal fistula and to construct a dynamic velar sling, thereby establishing oronasal competence and enabling control of airflow for intelligible speech. With this goal in mind, palate anatomy, perioperative concerns, surgical technique, postoperative care, and palatoplasty complications are described in this chapter.
15.1 Introduction
Detailed understanding of palate development, normal and cleft palate anatomy, and epidemiology of clefting is essential to the comprehensive care of children with cleft palate.
15.1.1 Embryology
Palatal processes form from the maxillary prominences during the seventh week postconception. These vertically oriented shelves elevate to a horizontal position by the ninth week postconception. They meet with each other to form the secondary palate and also fuse with the primary palate. Failure of elevation and fusion of these shelves result in a cleft palate.
15.1.2 Anatomy
Normal Palate Anatomy
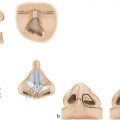
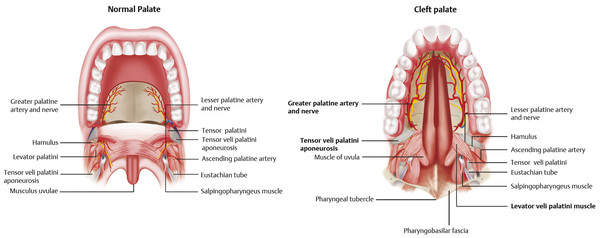
The incisive foramen is located at the midline, posterior to the alveolus, at the junction of the primary and secondary palate. Structures anterior to the incisive foramen are considered the primary palate, and structures posterior to the incisive foramen are considered the secondary palate. The secondary palate comprises both hard and soft palate. The hard palate is the growth center for the maxilla and is formed by paired palatine processes of the maxilla and palatine bones, articulating with the vomer. The soft palate is a muscular sling formed by the paired levator veli palatini, palatopharyngeus, palatoglossus, and uvular muscles (innervated by the pharyngeal branch of the vagus nerves) and is reinforced by the broad aponeurosis of the tensor veli palatini (innervated by the medial pterygoid branch of the mandibular nerves). These muscles work with the superior constrictor to form the velopharyngeal sphincter.
Palatal and velopharyngeal blood supply comes from branches of the paired internal maxillary arteries, recurrent pharyngeal arteries, ascending pharyngeal arteries, and ascending palatine arteries. The soft palate and velopharyngeal sphincter muscles have a rich blood supply from branches of these arteries. The greater palatine branches of the internal maxillary artery provide the major blood supply to mucoperiosteum of the hard palate (Fig. 15‑1).
Cleft Palate Anatomy
Isolated cleft palate involves secondary palatal structures. Maxillary and palatine bones are separated at the midline, resulting in an oronasal fistula. The tensor veli palatini aponeurosis, levator veli palatini muscles, and palatopharyngeus muscles are disoriented parallel to the cleft, inserting on the posterior hard palate (Fig. 15‑1). As a result, function of the velopharyngeal sphincter is compromised. These muscles normally participate in ventilation and pressure equalization of the middle ear. Altered anatomy of these muscles results in Eustachian tube dysfunction, frequently causing chronic middle ear effusion, hearing loss, and otitis media.
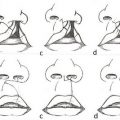
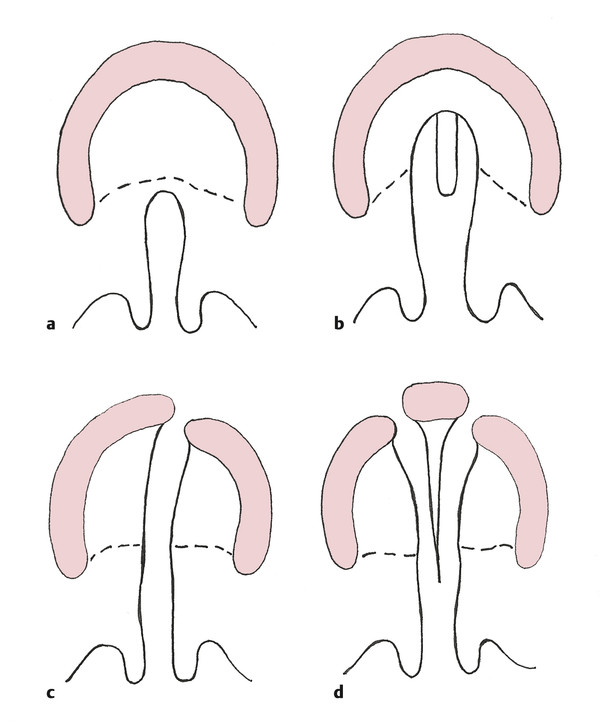
In cleft lip and palate, the defect extends along the junction of the primary and secondary palate, resulting in an alveolar gap. When the alveolus is cleft, lateral incisors are frequently absent and other dental anomalies, such as supernumerary teeth, are common. The palatal component of orofacial clefts is often described using the Veau classification (Fig. 15‑2).
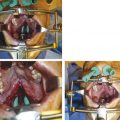
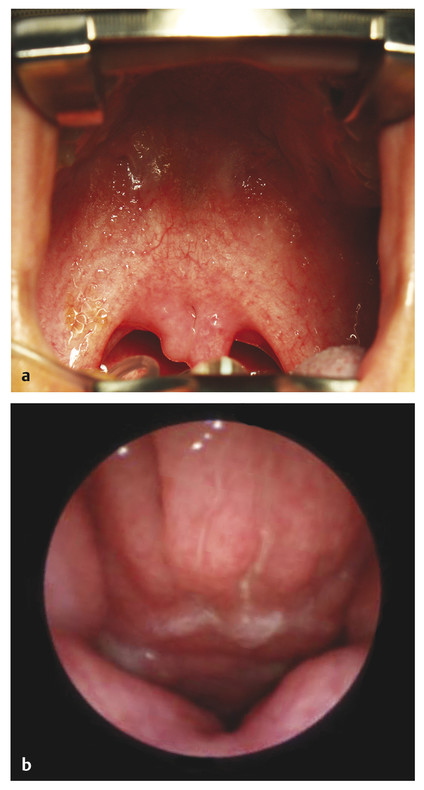
Submucous cleft palate is a lesser form of cleft palate involving muscular diastasis of the soft palate, with intact palatal mucosa. The diastasis may be visualized as a bluish “zona pellucida,” or furrow, in the soft palate. Other findings may include a bifid uvula and/or a notch in the posterior hard palate (Fig. 15‑3). These three findings constitute Calnan’s triad. This type of cleft is often diagnosed during evaluation of abnormal speech.
15.1.3 Epidemiology
Traditionally, cleft lip with or without cleft palate (CL/P) and isolated cleft palate (CP), also known as cleft palate only, have been considered separate etiopathogenetic entities; however, emerging evidence suggests that the delineation is less distinct. In the United States, recent data from the National Institute of Dental and Craniofacial Research estimate the incidence of CL/P to be approximately 1 in 940 live births. The condition is twice as common in male patients compared with female patients and has ethnic heterogeneity, with Asians and Hispanics demonstrating the highest incidence, Caucasians demonstrating intermediate incidence, and Africans manifesting the lowest incidence. Estimated incidence of CP is approximately 1 in 1,570 live births. The condition is twice as common in female patients and does not have the same ethnic variation that CL/P has.
Clefts of the lip and/or palate result from a combination of genetic and environmental factors. Environmental factors, including maternal smoking, folate deficiency, alcohol consumption, advanced maternal age, and retinoid use, are associated with increased risk. Mutations in multiple candidate genes, such as IRF6, Wnt signaling, MSX1, and BMP signaling factors, have also been associated with clefting. Cleft lip or palate is infrequently (10–14%) associated with a recognized syndrome, but 29 to 45% of patients with a cleft lip have associated anomalies, suggesting possible underdetection. Among the syndromes associated with CL/P are Van der Woude syndrome, oral-facial-digital syndrome, branchio-oculo-facial syndrome, and Opitz G/BBB syndrome. However, isolated CP is often (12–54%) associated with an underlying syndrome, and as many as 47 to 72% of patients have associated anomalies. Among more than 250 syndromes associated with CP are Stickler’s syndrome, 22q11 deletion spectrum, Cornelia de Lange syndrome, Emanuel syndrome, trisomy 13, Treacher Collins syndrome, and Apert’s syndrome.
15.2 Diagnosis
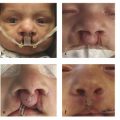
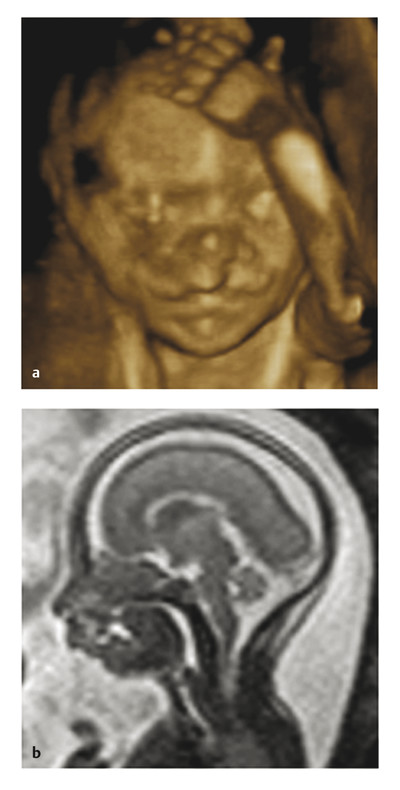
Clefts of the lip and palate are increasingly diagnosed prenatally. Cleft lip is often noted on routine prenatal examination, because the primary palate is well visualized on sonography (Fig. 15‑4a). Isolated CP is rarely diagnosed with sonography but may be incidentally detected with fetal magnetic resonance imaging (MRI) (Fig. 15‑4b) performed as a part of the evaluation of other congenital anomalies. In most cases, diagnosis of CP is made by physical examination postnatally. As soon as the diagnosis is made, the expectant mother or the newborn should be referred to a cleft team for evaluation and counseling.
15.3 Nonoperative Management
The concept of team care is one of the most important characteristics of modern cleft care. The American Cleft Palate-Craniofacial Association and Cleft Palate Foundation set standards for designated cleft centers. A cleft team must include a speech and language pathologist, a surgeon trained in cleft lip and palate repair, and an orthodontist. In addition, to best serve the unique needs of children with a cleft lip or palate, the team should have access to practitioners in psychology, social work, psychiatry, audiology, genetics, general and pediatric dentistry, otolaryngology, and pediatrics or primary care.
Infants with CP often have feeding difficulty early in life. When a cleft palate is present, infants are not able to generate suction due to the large oronasal fistula. Feeding instruction by a speech and language pathologist or a nurse skilled in assessing and feeding infants with cleft palate is initiated as soon as the diagnosis is made, either prenatally or shortly after birth. Infants with a cleft palate are best fed in semi-upright position, using cleft feeders. No one feeding system is suited to every infant. The two most commonly used at our center are Medela SpecialNeeds (Haberman) Feeder and Dr. Brown’s Specialty Feeding System. Other popular cleft bottles include the Mead Johnson’s cleft lip/palate nurser and Pigeon cleft palate nipple. The early management of infants with cleft palate is focused on feeding, weight gain, and nutrition.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree