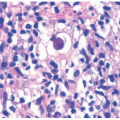
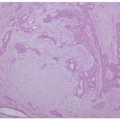
Vasculopathic, Vasculitic, and Neutrophilic Dermatosis
Ines Wu Soukoulis
Alejandro A. Gru
URTICARIA AND URTICARIAL VASCULITIS
Definition and Epidemiology
Urticaria generally represents a type 1 hypersensitivity reaction that can be triggered by a wide variety of factors, both immunologic and nonimmunologic. Acute and chronic forms exist, with the chronic form lasting more than 6 weeks. The great majority of urticaria cases resolve within a few weeks. This disease process can occur anytime from infancy to adulthood. Overall, it is estimated to occur in about 20% of people in their lifetime.1
Etiology
This type 1 hypersensitivity reaction can be caused by immunologic triggers or nonimmunologic factors that lead to mast cell release. Acute urticaria resolves within 6 weeks. In young children, common causes are infections such as a viral illness, medications such as anticonvulsants and antibiotics, and foods. Chronic urticaria recurs for more than 6 weeks and is less common in children than in adults; idiopathic and autoimmune causes are most common.2 Children with chronic urticaria may have a higher rate of autoimmune thyroid disease.3 Chronic urticaria can also be caused by foods, medications, and parasitic infections.4,5 Cold urticaria is a type of physical urticarial that is induced by cold exposure; in children, one-third of patients can have anaphylactic reactions.6 Cholinergic urticaria results from activities such as exercise and hot water that raise core body temperature.2
Clinical Presentation
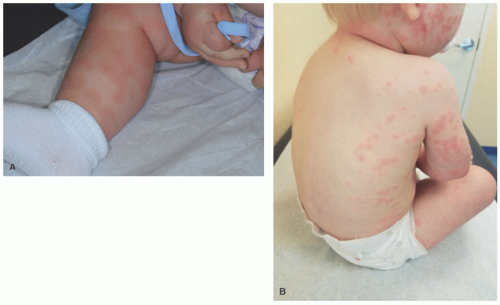
Lesions are erythematous, edematous wheals frequently with central clearing that are very pruritic. They range in size from pinpoint papules to large plaques (Figure 6-1).
They are characteristically transient, with each individual lesion resolving within 24 hours. Lesions resolve without dyspigmentation or scarring. Dermatographism is common (Figure 6-2).
They are characteristically transient, with each individual lesion resolving within 24 hours. Lesions resolve without dyspigmentation or scarring. Dermatographism is common (Figure 6-2).
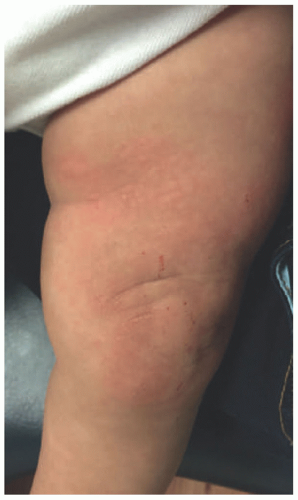 FIGURE 6-1. Acute urticarial papules and plaques with excoriation on the knee of a 7-month-old infant secondary to peanut allergy. |
Urticaria multiforme, also called “acute annular urticarial,” occurs in pediatric patients and presents with characteristic annular, serpiginous, and polycyclic urticarial plaques and is often associated with acral or facial edema (Figure 6-3).
Lesions can be centrally dusky and can resemble ecchymosis. This condition is more common in children ranging from ages 2 months to 4 years.7 Children may have fever but are overall well appearing; they can have symptoms of concurrent illness such as upper respiratory infection, otitis media, or other viral symptoms.7 This rash is self-limited and generally resolves in 1 to 2 weeks. Clinical findings can often be confused for erythema multiforme and serum sickness-like reactions (SSLR).7,8 Papular urticaria is typically seen in children, but can also be present in adolescents and young adults. It is characterized clinically by pruritic, edematous, and erythematous papules and wheals of variable size (3-10 mm). The lesions are located on the extremities and face, with sparing of the axillae and groin. The lesions can last for days to weeks, and leave residual hyperpigmentation at the affected sites. Recurrence following exposure to the causative agent is very frequent, although hyposensitization to the antigen can occur upon multiple reexposures.9
Histologic Findings
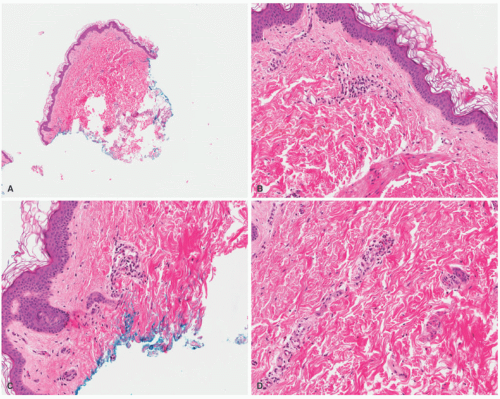
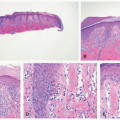
Urticaria is one of the conditions where the clinical finding can be quite drastic, but the histologic aspects are very underwhelming (Figure 6-4). Urticaria is one of the conditions that enter the pathologic diagnosis of “normal skin.” Early lesions of urticaria show intravascular clusters of neutrophils within the lumen, without much inflammation extravasated outside the vessels. Later on, there is a superficial and deep, or superficial to mid dermal, perivascular and interstitial mixed inflammatory infiltrate. The inflammatory cells include neutrophils, eosinophils, lymphocytes, and histiocytes. Papillary dermal edema and vascular congestion can
also be present. Chronic or resolving lesions of urticaria can show a slightly increased number of mast cells.10,11,12,13,14,15,16,17
also be present. Chronic or resolving lesions of urticaria can show a slightly increased number of mast cells.10,11,12,13,14,15,16,17
Papular urticaria is not a well-characterized pathologic term, but show features that are similar and indistinguishable from a dermal hypersensitivity reaction to a medication or arthropod bite reaction.9,18 In some cases, there might be a mild degree of spongiosis, but most of the time the epidermis is relatively unremarkable. There is a superficial and deep, perivascular and interstitial dense inflammatory infiltrate, composed of lymphocytes, histiocytes, eosinophils, and neutrophils. In cases where there is an abundance of eosinophils, degranulated cells and occasional “flame figures” can be seen. The inflammation can also extend into the subcutaneous tissue.
Differential Diagnosis
It is important when evaluating a biopsy for urticaria to obtain multiple levels of section to search for evidence of leukocytoclastic vasculitis (LCV), hence excluding the diagnosis of urticarial vasculitis. Frequently, early lesions of urticarial vasculitis show a brisk perivascular neutrophilic infiltrate with leukocytoclasis (nuclear dust), but without significant fibrinoid necrosis or endothelial swelling. Other disorders that present with a clinical and histologic urticarial pattern can also be considered in the differential diagnosis of urticaria. Bullous pemphigoid (BP), although more common in adults, has also been described in the pediatric population. The early phases of BP present with an urticarial pattern. One of the main differences between the urticarial BP and urticaria is the aggregation of eosinophils in the papillary dermis, near the dermal-epidermal junction. Additionally, eosinophilic spongiosis can be present, a feature that is not typical of urticaria. Later on, BP shows a subepidermal blister with eosinophils and neutrophils (in the classic form), and sometimes a pauci-inflammatory subepidermal bulla (in the cell poor variant). Invariably, direct immunofluorescence reveals linear deposits of C3 and IgG along the dermal-epidermal junction. Other considerations included
in the differential diagnosis are dermal hypersensitivity reactions (from a medication), telangiectasia macularis eruptive perstans (TMEP), arthropod bite reactions, and gyrate erythemas. Dermal hypersensitivity reactions typically lack the edema of urticaria and the vascular collections of neutrophils. TMEP is uncommonly seen in children and has a blaschkoid distribution. Performing special stains (Giemsa, Leder) or immunohistochemistry for mast cells (CD117, mast cell tryptase) is capital to establishing the diagnosis of this condition. Arthropod bite reactions and popular urticaria show identical findings. Gyrate erythemas can also have dermal edema, but the infiltrate is mostly lymphocytic, and the number of eosinophils is low, if present.
in the differential diagnosis are dermal hypersensitivity reactions (from a medication), telangiectasia macularis eruptive perstans (TMEP), arthropod bite reactions, and gyrate erythemas. Dermal hypersensitivity reactions typically lack the edema of urticaria and the vascular collections of neutrophils. TMEP is uncommonly seen in children and has a blaschkoid distribution. Performing special stains (Giemsa, Leder) or immunohistochemistry for mast cells (CD117, mast cell tryptase) is capital to establishing the diagnosis of this condition. Arthropod bite reactions and popular urticaria show identical findings. Gyrate erythemas can also have dermal edema, but the infiltrate is mostly lymphocytic, and the number of eosinophils is low, if present.
CAPSULE SUMMARY
URTICARIA
Urticaria generally represents a type 1 hypersensitivity reaction that can be triggered by a wide variety of factors, both immunologic and nonimmunologic. Acute and chronic forms exist, with the chronic form lasting more than 6 weeks. The great majority of urticaria cases resolve within a few weeks. Lesions are erythematous, edematous wheals frequently with central clearing that are very pruritic. They range in size from pinpoint papules to large plaques. Urticaria is one of the conditions where the clinical finding can be quite drastic, but the histologic aspects are very underwhelming. Urticaria is one of the conditions that enter the pathologic diagnosis of “normal skin.” Early lesions of urticaria show intravascular clusters of neutrophils within the lumen, without much inflammation extravasated outside the vessels. Later on, there is a superficial and deep, or superficial to mid dermal, perivascular and interstitial mixed inflammatory infiltrate with the presence of frequent eosinophils.
URTICARIAL VASCULITIS
Definition and Epidemiology
Urticarial vasculitis (UV) is a small-vessel vasculitis that is frequently associated with an underlying condition such as connective tissue disease. It is distinguished into two main groups: normocomplementemic and hypocomplementemic. Systemic disease is more commonly associated with the hypocomplementemic form.19 This condition is more commonly seen in adults and is rare in children.20
Etiology
Clinical Presentation
Clinical lesions of UV can be distinguished from urticaria by their persistence past 24 hours and their dusky and purpuric appearance (Figure 6-5).19 Lesions can be painful in addition to pruritic and can resolve with hyperpigmentation. They tend to be on the trunk and proximal extremities. Angioedema and joint pains can also occur.21 Confirmation of diagnosis often requires a biopsy, because it can present very similarly to urticaria. If confirmed, physical exam and labs are indicated to evaluate for underlying trigger. Hypocomplementemia, ANA, and anti-C1q antibodies are associated with a poor prognosis of the disease and with a systemic involvement. Nevertheless, these findings are exceptional in childhood.
Histologic Findings
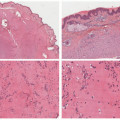
The pathologic criteria for the diagnosis of UV are less rigorous than for other LCV, because the infiltrates are sparser, and the vasculitic changes can be focal and subtle. Pathologically, there is a perivascular and interstitial neutrophilic and eosinophilic infiltrate, with either focal small-vessel neutrophilic vasculitis (evidence of fibrinoid necrosis) or focal perivascular leukocytoclasis without fibrin deposition, and with or without extravasated red blood cells (Figure 6-6). Therefore, the minimal histologic criteria for the diagnosis of UV include the presence
of perivascular leukocytoclasis, or fibrin deposits, with or without extravasated red blood cells. In hypocomplementemic UV, diffuse dermal neutrophilic infiltrates can be seen. Dermal eosinophils are more typical of the normocomplementemic forms of UV.22
of perivascular leukocytoclasis, or fibrin deposits, with or without extravasated red blood cells. In hypocomplementemic UV, diffuse dermal neutrophilic infiltrates can be seen. Dermal eosinophils are more typical of the normocomplementemic forms of UV.22
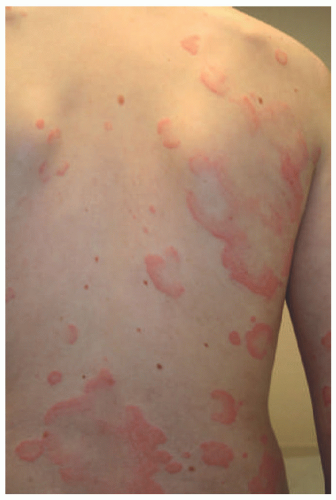
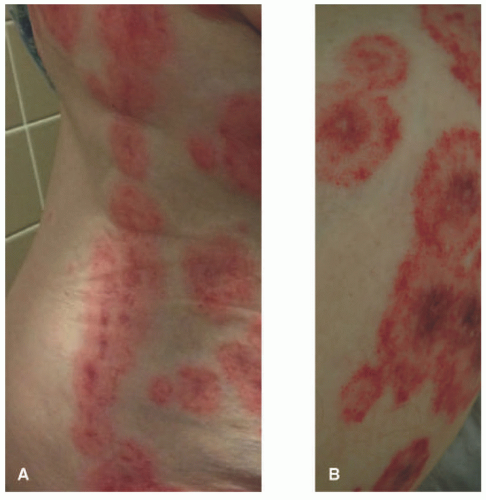 FIGURE 6-5. Urticarial vasculitis. Annular erythematous patches and plaques with dusky and purpuric appearance (A,B). |
CAPSULE SUMMARY
URTICARIAL VASCULITIS
UV is a small-vessel vasculitis that is frequently associated with an underlying condition such as connective tissue disease. It is distinguished into two main groups: normocomplementemic and hypocomplementemic. Systemic disease is more commonly associated with the hypocomplementemic form. This condition is more commonly seen in adults and is rare in children. Clinical lesions of UV can be distinguished from urticaria by their persistence past 24 hours and their dusky and purpuric appearance. The pathologic criteria for the diagnosis of UV are less rigorous than for other LCV, because the infiltrates are sparser, and the vasculitic changes can be focal and subtle. Pathologically, there is a perivascular and interstitial neutrophilic and eosinophilic infiltrate, with either focal small-vessel neutrophilic vasculitis (evidence of fibrinoid necrosis) or focal perivascular leukocytoclasis without fibrin deposition, and with or without extravasated red blood cells.
SERUM SICKNESS AND SERUM SICKNESS-LIKE REACTIONS
Definition and Epidemiology
True serum sickness (SS) is a type 3 hypersensitivity reaction that was more frequently seen in the past after the administration of horse or rabbit serum. SSLR are more common and can present with similar symptoms, but do not involve immune complex formation. SS is rarely seen in the pediatric population; in recent years, it has been reported in relation to rituximab.23 SSLR can occur occasionally in children. It is classically seen most often with cefaclor; however, other medications such as antibiotics, antifungals, psychiatric medications, and vaccinations have also been reported.24,25,26,27 Interestingly, children who develop SSLR to cefaclor usually do not have a similar reaction with other cephalosporin medications.28,29
Etiology
True SS involves deposition of immune complexes that are formed between host antibody and foreign protein, leading to an inflammatory cascade that includes the activation of complement. Deposition of these immune complexes in target tissues such as the vascular endothelial and glomerular basement membrane leads to damage of these areas.30,31 SSLR is thought to be caused by an inflammatory response to metabolites of the offending medication.30
Clinical Presentation
Classic symptoms of both diseases include fever, rash, and joint pains. The skin eruption in SS develops about one week after exposure and is generally morbilliform, with or without an urticarial component on the trunk before spreading to the extremities; the rash resolves within 2 weeks (Figure 6-7). A characteristic serpiginous erythema develops on the sides of the hands and feet.31,32 Patients can also have systemic manifestations including vasculitis, arthralgias/arthritis, gastrointestinal (GI) symptoms, nephritis causing proteinuria, and lymphadenopathy. SSLR presents with a rash about 1 to 2 weeks after the triggering medication that tends to be more urticarial and polycyclic; centrally, the lesions may have a violaceous discoloration. Joint swelling can occur, especially around the knees. Children can have facial swelling, myalgias, and headache. Nephritis and vasculitis do not happen because there are no immune complexes involved. This disease is generally selflimited and subsides within 2 to 3 weeks of discontinuing the medication.30,33
Histologic Findings
Because of the rare circumstances when a biopsy is performed, the pathologic findings are poorly characterized. Skin biopsies of these patients reveal limited lymphocytic and histiocytic perivascular infiltrates with only rare neutrophils. Fibrinoid necrosis is not typically seen, although there have been subsequent reports of true small-vessel vasculitis in SS. Some have documented the presence of abundant neutrophils and eosinophils in this condition.34,35
CAPSULE SUMMARY
SERUM SICKNESS AND SS-LIKE REACTIONS
True SS is a type 3 hypersensitivity reaction that was more frequently seen in the past after the administration of horse or rabbit serum. SS is rarely seen in the pediatric population; in recent years, it has been reported in relation to rituximab. Classic symptoms of both diseases include fever, rash, and joint pains. The skin eruption in SS develops about one week after exposure and is generally morbilliform, with or without an urticarial component on the trunk before spreading to the extremities; the rash resolves within 2 weeks.
ARTHROPOD BITE REACTIONS
Definition and Epidemiology
Arthropod reactions can occur in reaction to various bites, including by mites, ticks spiders, lice mosquitoes, fleas, bees, and bedbugs. This section focuses on two common distinct arthropod reactions in children: scabies and papular urticaria.
SCABIES
Scabies infestation is caused by the Sarcoptes scabiei mite and presents as a very pruritic rash. It is most often transmitted by direct human contact with an infested individual, although it can also be spread by contact with infested clothing and bedding. This infestation occurs in all ages, from infancy to adulthood, with the highest prevalence under age 2.36 Women and children appear to be infested more often, as well as immunocompromised individuals and those with mental or physical handicap.
Etiology
This infestation is caused by the S. scabiei mite, which is an obligated human parasite. The female mite burrows into the stratum corneum and causes a hypersensitivity reaction.
Clinical Presentation
The clinical presentation of scabies infestation varies according to age. Newborns and infants present shortly after infestation with very inflamed papulovesicles and nodules that can be crusted and serpiginous.36 Scabies nodules are primarily seen in infants. Lesions are reddish-brown and most commonly occur on the trunk, genital region, and axillae. They represent a hypersensitivity response and may persist for months.
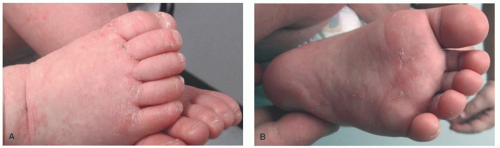
In infants, the scalp is frequently affected, which is rarely involved in older children and adults; the trunk is also more likely to be affected. In addition, infants and younger children are more likely to present with lesions that are vesiculated. In older children and adults, the incubation period is approximately 3 weeks.37 Skin lesions include papules, papulovesicles, and nodules and are most commonly seen in the web spaces of the hands and feet, wrists, ankles, axillae, inguinal areas, and palms and soles. There are frequently linear and curvilinear burrows on the palms and soles (Figure 6-8A and B). Excoriations are commonly seen, and secondary bacterial infection is common. In older children and adults who are healthy, the number of mites during an infestation averages around 10 to 12.37
Crusted or Norwegian scabies occurs primarily in immunocompromised patients and is extremely contagious, given the much higher mite load compared with classic scabies infection. Skin findings can mimic eczema and psoriasis and can be hyperkeratotic.37 This form of scabies can be the source for epidemic involvement in hospitals.
CAPSULE SUMMARY
SCABIES
Scabies infestation is caused by the S. scabiei mite and presents as a very pruritic rash. This infection occurs in all ages, from infancy to adulthood, with the highest prevalence under age 2. Newborns and infants present shortly after infestation with very inflamed papulovesicles and nodules that can be crusted and serpiginous. Scabies nodules are primarily seen in infants. Lesions are reddish-brown and most commonly occur on the trunk, genital region, and axillae.
The pathologic findings and differential diagnosis are discussed in detail in Chapter 21 about parasitic infestations.
PAPULAR URTICARIA
Definition and Epidemiology
This is a common rash in childhood that presents with pruritic erythematous urticarial papules that are most commonly caused by a hypersensitivity reaction to arthropod bites. This can be seen in children and adults of all ages, but is most prevalent in children ages 2 to 10 years.1
Etiology
This can be caused by a number of different bites, including mosquitoes, fleas, bedbugs, and mites.
Clinical Presentation
Lesions are typically urticarial and clustered erythematous papules; they are often in a linear array and can vesiculate. They can occur anywhere on the body, although they frequently spare intertriginous areas.1 Excoriation and crusting can be seen, and a central punctum is often visible (Figure 6-9). Each individual lesion resolves over 1 to 2 weeks. Recurrence is common, especially if there is reexposure.
Histologic Findings
The histopathologic findings present in association with arthropod bite reactions/papular urticaria are variable (Figure 6-10): those depend on the arthropod subtype, the duration of the clinical lesion, the immunologic reaction, the presence or absence of arthropod parts, and the discharge of toxins. Changes caused by tick bites involve both the epidermis and the dermis. In the epidermis, there is variable acanthosis, which in some cases can be exuberant and have a pseudoepitheliomatous appearance. Epidermal spongiosis can also be seen, particularly in the acute phases. The spongiotic reaction can be localized to the site of the bite, or more extensive, with multiple intraepidermal vesicles and sometimes frank necrosis. Eosinophilic spongiosis can be a particular feature seen in arthropod bites. The dermal reaction is characterized by wedge-shaped dense foci of inflammatory cells consisting of lymphocytes, histiocytes, plasma cells, and eosinophils especially in the periappendageal region. Secondary lymphoid follicles with germinal centers are formed in some lesions. Mouth parts of the tick can be present sometimes, typically in the mid dermis and covered by a down-growth of the epidermis. The arthropod parts can be polarizable. Reactions to fleas, fire ants, mosquitoes, and spider envenomation show a predominance of neutrophils with vasculitic foci. Hemorrhage and edema can also be prominent.
Pseudolymphomatous changes can also be seen: the pleomorphism of the mononuclear infiltrate may simulate mycosis fungoides or other lymphomas of the skin including Hodgkin disease, especially when accompanied by eosinophils. Cells resembling those of Sternberg-Reed may sometimes be present. Usually, the clinical history and the
presence of a single lesion help exclude lymphoma, but, when lesions are numerous, differentiation may be difficult. The abundance of plasma cells in the insect bite may aid in the differentiation.
presence of a single lesion help exclude lymphoma, but, when lesions are numerous, differentiation may be difficult. The abundance of plasma cells in the insect bite may aid in the differentiation.
CAPSULE SUMMARY
PAPULAR URTICARIA
This is a common rash in childhood that presents with pruritic erythematous urticarial papules that are most commonly caused by a hypersensitivity reaction to arthropod bites. Lesions are typically urticarial and clustered erythematous papules; they are often in a linear array and can vesiculate. They can occur anywhere on the body, although they frequently spare intertriginous areas. The dermal reaction is characterized by wedge-shaped dense foci of inflammatory cells consisting of lymphocytes, histiocytes, plasma cells, and eosinophils, especially in the periappendageal region. Secondary lymphoid follicles with germinal centers are formed in some lesions.
ANNULAR (GYRATE) ERYTHEMAS
This reactive group of skin conditions is characterized by serpiginous and annular eruptions, including erythema annulare centrifugum (EAC), annular erythema of infancy, and erythema chronicum migrans (ECM). These are entities that need to be differentiated from other annular conditions such as erythema multiforme, tinea corporis, and SSLR.
ERYTHEMA ANNULARE CENTRIFUGUM
Epidemiology and Etiology
Clinical Presentation
Skin lesions are annular plaques with central clearing and often have a fine raised trailing scale and can extend peripherally rapidly. They are usually asymptomatic and tend to be on the trunk and buttocks. Lesions can last weeks to months, and some can have recurrent lesions over the course of years.40
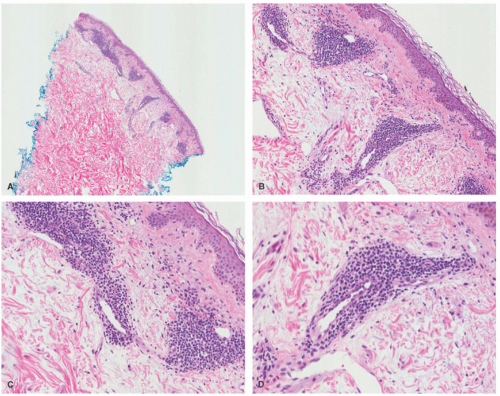
Histologic Findings
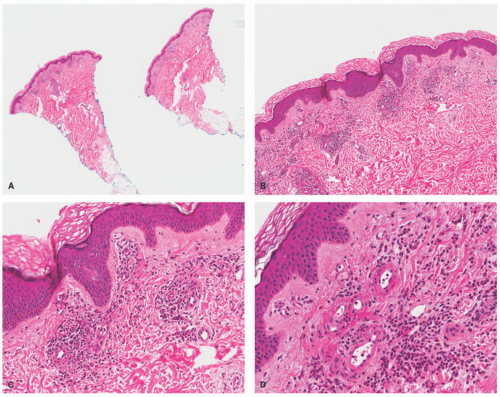
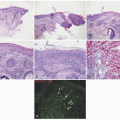
The biopsies of EAC show the prototypic findings of a gyrate erythema, a lymphocytic vasculitis: there is a superficial to mid dermal, very tight, perivascular mostly lymphocytic infiltrate (Figure 6-11). The epidermis is relatively normal. However, the superficial variant of EAC can have spongiosis, exocytosis of lymphocytes, and areas of parakeratosis. Papillary dermal edema can be sometimes present. The tight perivascular character of the lymphocytic infiltrate has also been described to as having a “coat sleeve” or “pipe stem” appearance. As opposed to other types of vasculitis, fibrinoid changes and extravasated red blood cells are not seen. In a small number of cases,
scattered dermal eosinophils are present. A “deep” form of gyrate erythema has been described, in which the inflammatory infiltrate extends to involve the deep vessels in the reticular dermis. The latter form lacks epidermal changes.41,42,43,44
scattered dermal eosinophils are present. A “deep” form of gyrate erythema has been described, in which the inflammatory infiltrate extends to involve the deep vessels in the reticular dermis. The latter form lacks epidermal changes.41,42,43,44
Differential Diagnosis
Other gyrate erythemas (described in the following paragraphs: annular erythema of infancy, erythema marginatum, and ECM) should always be included in the differential diagnosis of EAC. Arthropod bite reactions can also be considered in the differential diagnosis. As opposed to EAC, they typically have both a superficial and deep character of inflammation, a wedge-shaped appearance, sometimes a punctum (site of arthropod bite), and an abundance of dermal eosinophils. Tumid lupus, reticular erythematous mucinosis, and Jessner’s lymphocytic infiltrate can also have a tight perivascular lymphocytic inflammation. As opposed to EAC, there is a significant amount of interstitial dermal mucin. Polymorphous light eruptions also have a tight lymphocytic perivascular inflammation. However, the latter shows a significant amount of edema, and sometimes epidermal changes. The superficial form of EAC can have a similar differential diagnoses to subacute and chronic spongiotic dermatitis (eczema, contact dermatitis, id reactions, atopic dermatitis, spongiotic drug reactions).
CAPSULE SUMMARY
ERYTHEMA ANNULARE CENTRIFUGUM
Skin lesions are annular plaques with central clearing and often have a fine raised trailing scale and can extend peripherally rapidly. They are usually asymptomatic and tend to be on the trunk and buttocks. The biopsies of EAC show the prototypic findings of a gyrate erythema, a lymphocytic vasculitis: there is a superficial to mid dermal, very tight, perivascular mostly lymphocytic infiltrate.
ANNULAR ERYTHEMA OF INFANCY
Epidemiology and Etiology
This annular erythema usually starts shortly after birth and is thought to be a hypersensitivity reaction to an unknown antigen.45
Clinical Presentation
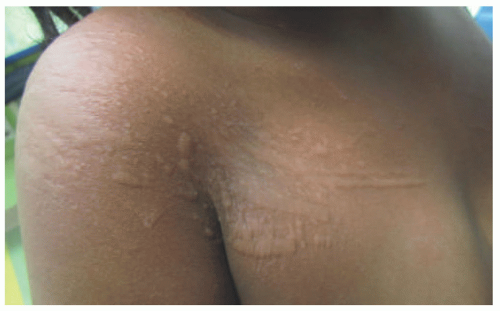
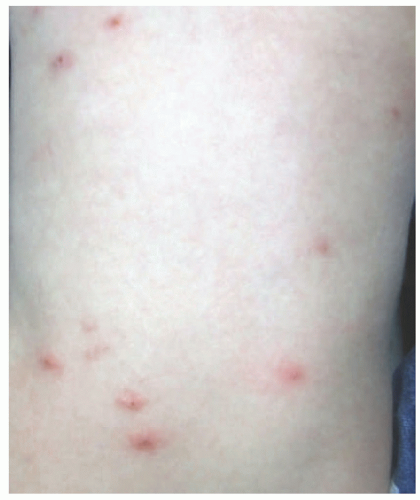
Infants develop an asymptomatic 2 to 3 cm arcuate and annular eruptions with raised borders that classically last a few days, but frequently can recur for months to rarely years (Figure 6-12). Lesions resolve without hyperpigmentation or scarring, and there are no systemic symptoms.46,47
Histologic Finding
It is important to emphasize that a variety of histopathologic patterns have been linked to this condition. It appears likely that more than one process is grouped into this particular disease category. The majority of the pathologic reports describe a dense perivascular inflammatory infiltrate composed of lymphocytes, with scattered eosinophils and plasma cells. Others have documented a more interstitial pattern of inflammation with a predominance of eosinophils. An additional case report describes a case with perivascular leukocytoclasis45,46,48,49
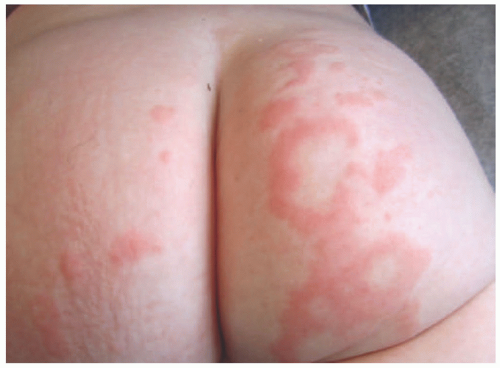 FIGURE 6-12. Recurrent erythematous annular plaques on the buttocks of an infant representing annular erythema of infancy. No definitive cause was identified. |
CAPSULE SUMMARY
ANNULAR ERYTHEMA OF INFANCY
This annular erythema usually starts shortly after birth and is thought to be a hypersensitivity reaction to an unknown antigen. Infants develop an asymptomatic 2 to 3 cm arcuate and annular eruptions with raised borders that classically last a few days, but frequently can recur for months to rarely years. A variety of histopathologic patterns have been linked to this condition. The majority of the pathologic reports describe a dense perivascular inflammatory infiltrate composed of lymphocytes, with scattered eosinophils and plasma cells.
ERYTHEMA CHRONICUM MIGRANS
Epidemiology and Etiology
This is the earliest sign of Lyme disease, which is caused by a tick bite from the spirochete Borrelia burgdorferi. Both children and adults can be affected. Cases have been reported in North America, Europe, and Asia. In the United States, the majority of cases are in the northeastern, north-central, and western states.50
Clinical Presentation
Localized erythema migrans presents as an annular erythematous lesion with central clearing that expands to be several centimeters over days; this is the typical bull’s eye rash that appears between 1 and 30 days after the tick bite.50 Sometimes, the center of the lesion can vesiculate or ulcerate. Without treatment, the lesion can last for 1 to 4 weeks. In early disseminated disease, multiple lesions are seen and can be accompanied by fever and joint pains.50
Histologic Findings
The pathologic findings are similar to those of other gyrate erythemas (Figure 6-13). This condition is also referred to as Borrelia-induced lymphocytoma cutis. There is a superficial and deep, mostly perivascular tight lymphocytic infiltrate with the presence of plasma cells. The biopsies typically lack epidermal changes. Scattered eosinophils can also be present. A particular perineural infiltration of lymphocytes and plasma cells has been described in some cases of ECM. An interstitial pattern of inflammation has also been documented in some cases. Special stains for microorganisms (Warthin-Starry stain) and polymerase chain reaction analysis for Lyme can help in establishing the diagnosis. Other
findings that can be seen in association with Lyme disease include acrodermatitis chronica atrophicans and cutaneous lymphoid hyperplasia.51,52,53,54
findings that can be seen in association with Lyme disease include acrodermatitis chronica atrophicans and cutaneous lymphoid hyperplasia.51,52,53,54
Differential Diagnosis
Other gyrate erythemas typically lack the plasma cell infiltrate typical of ECM. Tumid lupus/Jessner’s/reticular erythematous mucinosis can also share the presence of plasma cells. However, ECM lacks the significant amount of mucin present in those conditions.
CAPSULE SUMMARY
ERYTHEMA CHRONICUM MIGRANS
ECM is the earliest sign of Lyme disease, caused by a tick bite from the spirochete Borrelia burgdorferi. Both children and adults can be affected. Localized erythema migrans presents as an annular erythematous lesion with central clearing that expands to be several centimeters over days; this is the typical bull’s eye rash that appears between 1 and 30 days after the tick bite. Pathologically, there is a superficial and deep, mostly perivascular tight lymphocytic infiltrate with the presence of plasma cells.
ERYTHEMA MARGINATUM
Definition and Etiology
This is a form of annular erythema that occurs in approximately 2% to 4% of patients with rheumatic fever. It is seen more frequently in children than in adults. The pathogenesis remains unknown. A study documenting the accumulation of bradykinin in the stromal tissue and endothelial cells in the skin biopsy of erythema marginatum has been reported. Bradykinin appears to be important in the pathogenesis of erythema marginatum associated with hereditary angioedema.55,56,57,58
Clinical Presentation
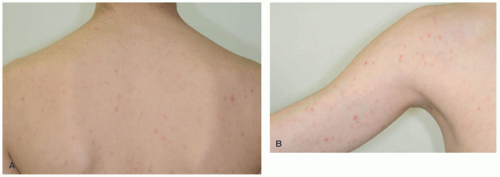
The lesions appear on the trunk (particularly the abdomen) and proximal extremities as pink macules and papules that often have an annular and polycyclic pattern.
Histologic Findings
The biopsies of this condition show a neutrophilic infiltrate within the dermal papillae and around the superficial vessels. There is no definitive evidence of vasculitis. Leukocytoclasis is present. Scattered apoptotic keratinocytes in the epidermis can be present. A rare case report of an urticarial
presentation of this form of gyrate erythema has been described (with similar pathologic changes to those seen in urticaria). Neutrophilic figurate erythema of infancy may have identical pathologic findings, but is not associated with rheumatic fever.11,49,56,57,58
presentation of this form of gyrate erythema has been described (with similar pathologic changes to those seen in urticaria). Neutrophilic figurate erythema of infancy may have identical pathologic findings, but is not associated with rheumatic fever.11,49,56,57,58
CAPSULE SUMMARY
ERYTHEMA MARGINATUM
This is a form of annular erythema that occurs in approximately 2% to 4% of patients with rheumatic fever. It is seen more frequently in children than in adults. The lesions appear on the trunk (particularly the abdomen) and proximal extremities as pink macules and papules that often have an annular and polycyclic pattern. The biopsies of this condition show a neutrophilic infiltrate within the dermal papillae and around the superficial vessels. There is no definitive evidence of vasculitis.
PERNIOSIS (CHILBLAINS)
Definition and Epidemiology
Pernio is an inflammatory process that occurs because of an abnormal exuberant response to cold temperatures in predisposed individuals. It is more common in colder climates such as the northern United States and northern Europe and is most common in young women aged 15 to 30.59 Within the pediatric population, it occurs most in teenagers, but can also occur in younger ages.60 In children, the presence of cryoproteins can be associated with pernio.61 In adolescents, it has been reported in the context of anorexia and may be related to impaired thermoregulation.62 Thin habitus was also noted in one series to be a common characteristic, and in one pediatric patient, weight loss secondary to celiac disease was speculated to contribute to the development of pernio lesions.59,63
Etiology
Some studies have suggested that pernio may be caused by vasospasm, which in turn is caused by cold temperatures, leading to inflammatory changes secondary to ischemia.64 Others postulate that it may be caused by endothelial cell injury leading to abnormally persistent vasoconstriction of the deep cutaneous arterioles with dilation of the smaller superficial vessels.60
Clinical Presentation
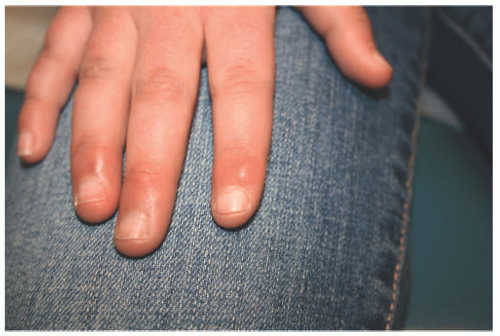
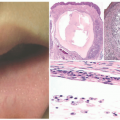
Typical acute lesions occur on the fingers, toes, and nose and appear several hours after exposure to cold and typically resolve within 1 to 3 weeks. They appear as blanching macules in its mild form and progress to firmer edematous nodules that can be pink to violaceous; lesions can be painful, pruritic, or burning (Figures 6-14 and 6-15).59 Lesions can blister or ulcerate. The condition tends to begin in the fall and winter and resolve by the spring and summer, although in chronic cases with repeated exposure to cold, the lesions can become persistent through the warmer months as well.59 Dorsal fingers and toes, heels, lower legs, nose, and ears can be affected as well. If lesions occur in unusual circumstances or are persistent, then evaluation for other possible causes is warranted. Possible underlying conditions include connective tissue disease and hematologic disorders.65 Bloodwork checked includes complete blood count, antinuclear antibody, cryoglobulin, cryofibrinogen, and cold agglutinin. In adults, serum protein electrophoresis is often checked for monoclonal gammopathy, but this is much less likely in children. Genetic disorders have also been associated with perniosis, including Aicardi-Goutieres syndrome (particularly those with dominant type TREX1 mutation) and familial chilblain lupus (heterozygous mutation in TREX1).66
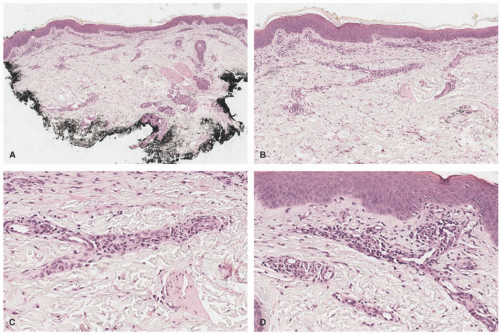
Histologic Findings
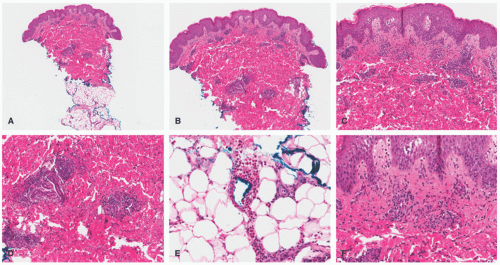
Biopsies from perniosis show a superficial and deep, perivascular and periadnexal lymphocytic infiltrate in an acral location (Figure 6-16). Marked papillary dermal edema is also seen. Sometimes, small vascular thrombi and fibrinoid changes of the vascular walls are present. Some cases can also have increased dermal mucin, and interface changes. In most cases, the epidermis is relatively unremarkable.59,61,67,68
Differential Diagnosis
Lupus erythematosus should always be considered in the differential diagnosis. The clinical, serologic findings, and the presence of increased dermal mucin can help in establishing a diagnosis of lupus. Other forms of gyrate erythemas can also be considered in the differential diagnosis. But, the acral location of perniosis can easily discern between such diagnostic possibilities. A small-vessel vasculitis (LCV) can also be considered in the differential diagnosis.
However, LCV shows a neutrophilic infiltrate with leukocytoclasis, features usually missing in cases of perniosis.
However, LCV shows a neutrophilic infiltrate with leukocytoclasis, features usually missing in cases of perniosis.
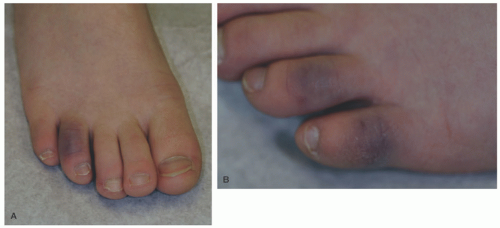 FIGURE 6-15. Firm, indurated nodules, with a dusky and hemorrhagic appearance are seen on the toes (A,B). |
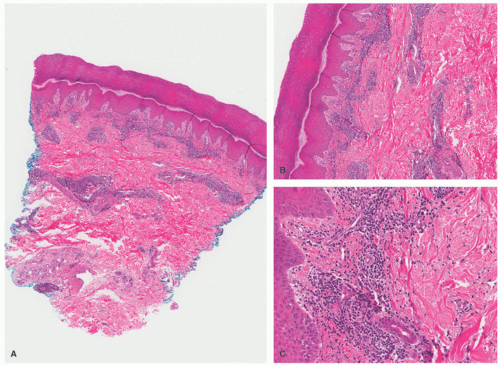 FIGURE 6-16. Perniosis. There is a superficial and deep, perivascular and periadnexal lymphocytic infiltrate in an acral location (A). Marked papillary dermal edema is also seen (B,C). |
CAPSULE SUMMARY
PERNIOSIS (CHILBLAINS)
Pernio is an inflammatory process that occurs because of an abnormal exuberant response to cold temperatures in predisposed individuals. Within the pediatric population, it occurs most in teenagers but can also occur in younger ages. Typical acute lesions occur on the fingers, toes, and nose and appear several hours after exposure to cold and typically resolve within 1 to 3 weeks. They appear as blanching macules in its mild form and progress to firmer edematous nodules that can be pink to violaceous; lesions can be painful, pruritic, or burning. Biopsies from perniosis show a superficial and deep, perivascular and periadnexal lymphocytic infiltrate in an acral location. Marked papillary dermal edema is also seen.
PITYRIASIS LICHENOIDES
Definition and Epidemiology
This a spectrum of skin diseases that is broken down into the acute and chronic forms, called PLEVA (pityriasis lichenoides et varioliformis acuta or Mucha-Habermann disease) and PLC (pityriasis lichenoides chronica). PLEVA occurs more acutely and presents with papulovesicles that can become necrotic and ulcerated; PLC presents with recurrent scaly lichenoid papules that tend to be recurrent.69
Pityriasis lichenoides is relatively frequent in the pediatric population. In one study, almost 20% of cases were seen in the pediatric age group.70 Most cases are reported in kids under the age of 10, with peaks in the preschool and early school years in one study of 124 children.69 It generally does not occur under age 2, although rare cases can occur in infancy.71,72 Winter and fall were the most common seasons for this disorder to occur.69
Etiology
Pityriasis lichenoides is considered an interface dermatitis with an associated lymphocytic vasculitis, that is likely triggered by an antigenic stimulus.73 There is frequently history of a preceding infection such as a viral illness, medication, or vaccination in children.74,75 Most PLEVA samples in one study were found to be monoclonal, which differed from the PLC cases that were mostly polyclonal. It is hypothesized that host immune response determines the clinical presentation and histology of this condition.76 Although its potential evolution to malignancy is unclear, there are rare reports of cutaneous T-cell lymphoma in patients with a history of PLEVA/PLC.77,78 In the author’s perspective, such reports are biased by a selection of cases, and in most circumstances, MOST patients with PLEVA or PLC DO NOT progress to a cutaneous T-cell lymphoma, such as mycosis fungoides or lymphomatoid papulosis.
Clinical Presentation
One study reported equal frequencies of occurrence between PLEVA and PLC, with many cases having an overlap presentation.74 In both forms, the distribution of lesions was mostly diffuse in the majority of patients (75%) and localized (in about 20%).69
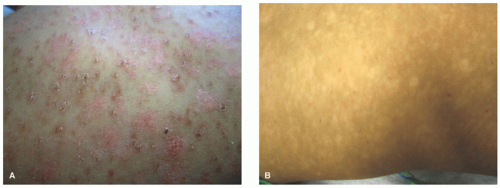
PLEVA begins suddenly as 2 to 3 mm reddish-brown macules and papules on the trunk and proximal extremities that appear in crops and quickly develop vesicles, purpura, and sometimes necrosis (Figure 6-17). They can be asymptomatic or pruritic. In this stage, lesions can appear similar to varicella, bug bites, and impetigo. Lesions then develop fine scaling and can resolve with a small scar as well as transient hyperpigmentation or hypopigmentation. The time to resolution ranges anywhere from weeks to many months.69
The febrile ulceronecrotic form is rare and presents with ulceronecrotic nodules and plaques with fever; half of all cases have been in children.79,80
PLC lesions are scaly lichenoid papules that resolve with hypopigmentation (Figure 6-18A). At times in
darker-skinned patients, hypopigmentation may be the most prominent finding (Figure 6-18B).
darker-skinned patients, hypopigmentation may be the most prominent finding (Figure 6-18B).
The course of PLC tends to be more prolonged than PLEVA and lasts several months, often with recurrence.81
Histologic Findings
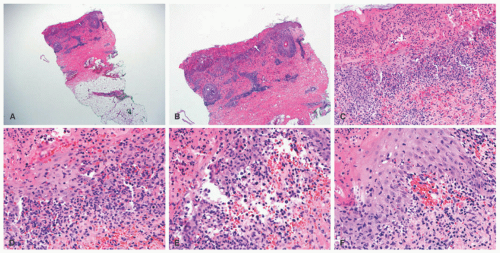
Biopsies of PLEVA and PLC show a range in severity, density, and pattern of lichenoid and interface changes.76,82,83 Biopsies of PLEVA classically show variable thickness of the epidermis, depending upon the developmental stage of the lesion, which is being biopsied. Lesions with chronic evidence of trauma may show psoriasiform changes. Thick parakeratosis is present. There is an interface tissue reaction with numerous dyskeratotic and apoptotic keratinocytes, in addition to vacuolar degeneration of the basal keratinocytes (Figures 6-19 and 6-20). In some cases, extensive and diffuse epidermal necrosis can be seen. A superficial to deep, lymphocytic infiltrate in the dermis is present, with a wedge-shaped appearance. The infiltrate shows extension into the surface epidermis. A lymphocytic vasculitis is also
seen, in the form of tight perivascular collections of lymphocytes with dermal edema and extravasated red blood cells. Eosinophils are not frequently seen.
seen, in the form of tight perivascular collections of lymphocytes with dermal edema and extravasated red blood cells. Eosinophils are not frequently seen.
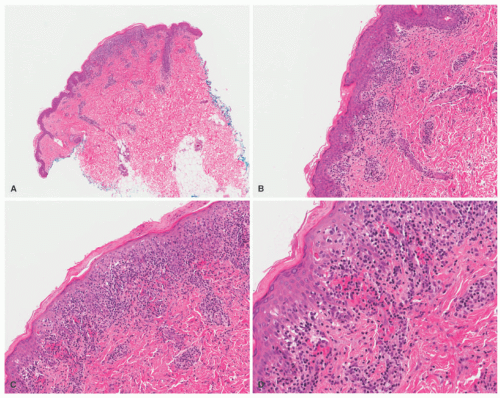
Under the microscope, PLC displays both interface lichenoid changes and mild spongiosis in the epidermis (Figure 6-21). Lymphocytes approximate the dermal-epidermal junction, accompanied by basovacuolar change and scattered dyskeratotic keratinocytes in the epidermis. Small intraepidermal lymphocytes with regular nuclear contours are seen. There is variable ortho-and parakeratosis. The dermis typically shows a superficial perivascular lymphocytic infiltrate. Eosinophils are rarely present. Fibrin deposition may be observed within vessel walls, but frank vasculitis is not seen. Other common histologic findings include the presence of extravasated red blood cells and/or melanophages in the upper dermis.
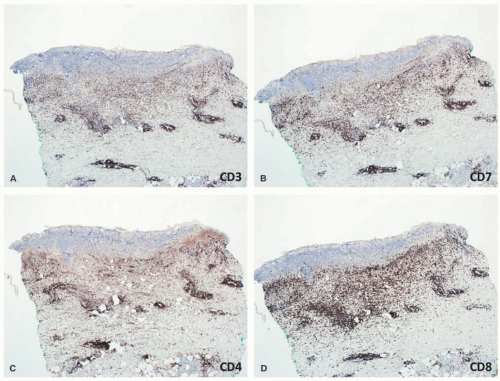
Although PLEVA and PLC are thought to be closely related conditions, or even different manifestations of a single disorder by some, they may manifest different immunophenotypes. The infiltrate of PLEVA has been found to more often display an elevated CD8:CD4 ratio (Figure 6-20), whereas the T lymphocytes in PLC show a predominance of CD4+ cells rather than CD8+ cells. Moreover, T-cell intracellular antigen-1 (TIA-1) is more likely to be expressed in the T lymphocytes of PLEVA, compared with PLC which shows increased expressed of FOXP3, a marker of T-regulatory cells. Interestingly, one recent study found that 4 out of 23 cases diagnosed as PLC with a benign clinical course showed a predominantly γδ T-cell phenotype, even though γδ T-cells have more traditionally been associated with aggressive lymphomas.
Clonal rearrangement of the T-cell receptor gene may be observed in PLC; on the basis of this finding PLC has been classified as a T-cell dyscrasia. A study by Magro and colleagues found evidence of T-cell clonality in 33/35 patients with pityriasis lichenoides. The patients studied were interpreted to have either PLEVA or PLC considering both clinical and histopathologic findings, with the majority of the patients classified as having PLC. Two other studies have found occasional clonal TCR-γ gene rearrangement in their
PLC patients, in 1/13 and 3/16 patients, respectively. By contrast, Kim et al. did not detect clonal TCR-γ gene rearrangement in any of the 13 PLC patients. The differences in these studies may reflect some of the difficulties in differentiating PLC from early lesions of mycosis fungoides.73,76,83,84,85,86
PLC patients, in 1/13 and 3/16 patients, respectively. By contrast, Kim et al. did not detect clonal TCR-γ gene rearrangement in any of the 13 PLC patients. The differences in these studies may reflect some of the difficulties in differentiating PLC from early lesions of mycosis fungoides.73,76,83,84,85,86
Differential Diagnosis
The histopathologic changes of PLC closely resemble those of PLEVA, because both show lymphocyte exocytosis, interface dermatitis with basovacuolar alteration, mild spongiosis, a superficial lymphocytic infiltrate, and papillary dermal hemorrhage. However, the microscopic findings in PLC are typically more subtle than in PLEVA; the lymphocytic infiltrate of the latter is more intense in both the epidermal and dermal compartments, with a wedge-shaped perivascular lymphocytic inflammation extending into the reticular dermis. In addition, ulceration is not a characteristic observation in PLC but is seen with some frequency in more severe cases of PLEVA, often in conjunction with a serum crust containing neutrophils.
Some variants of mycosis fungoides may be difficult to distinguish from PLC, both clinically and histologically. A recent study detailed four patients with eventual diagnoses of papular mycosis fungoides, all of whom had small reddish-brown papules present between 5 and 25 years.87 On the basis of initial clinical-pathologic correlation, three of the four patients were initially felt to have pityriasis lichenoides, but additional biopsies in each case demonstrated characteristic findings of mycosis fungoides on histology, including epidermotropism with lymphocyte atypia and elevated CD4:CD8 ratios. Hypopigmented mycosis fungoides often demonstrates overlapping histologic findings with PLC, and in these cases, clinical-pathologic correlation is essential.
Other diagnostic considerations include pityriasis rosea, guttate psoriasis, small plaque parapsoriasis (digitate dermatosis), arthropod bite reactions, and lymphomatoid papulosis. As opposed to the more confluent pattern of parakeratosis, pityriasis rosea only shows mounds of it In addition, the “tilting” phenomenon of the parakeratotic mounds is typical of pityriasis rosea (PR), but not of pityriasis lichenoides (PL). PR also differs on the spongiotic pattern, as opposed to the interface changes seen in both PLEVA and PLC. Guttate psoriasis has a very different histologic appearance, with only mild or very modest pathologic changes, which include vascular telangiectasia, mild acanthosis, and few scattered neutrophils in the epidermis or corneal layer. Digitate dermatosis lacks the interface changes of PLC/PLEVA and the red cell extravasation. Although arthropod reactions can share the wedge-shaped dense inflammation in the dermis, they have an abundance of dermal eosinophils, a feature not present in PLC/PLEVA. Lymphomatoid papulosis has different histologic patterns, all of which are based on the finding of atypical CD30+ large cells. The lesions can also have the scarring seen in PLC/PLEVA.
CAPSULE SUMMARY
PITYRIASIS LICHENOIDES
This is a spectrum of skin diseases that is broken down into the acute and chronic forms, called PLEVA and PLC. PLEVA occurs more acutely and presents with papulovesicles that can become necrotic and ulcerated; PLC presents with recurrent scaly lichenoid papules that tend to be recurrent. Biopsies of PLEVA and PLC show a range in severity, density, and pattern of lichenoid and interface changes.
SWEET SYNDROME (ACUTE FEBRILE NEUTROPHILIC DERMATOSIS)
Definition and Epidemiology
Sweet syndrome, also known as acute febrile neutrophilic dermatosis, presents with painful edematous lesions along with fever and leukocytosis. It is estimated that about 5% of cases are seen in the pediatric population.88,89 The mean age is reported to be just over 5 years old, and cases can present in infancy with the youngest presentation at 3 days old.90,91,92 There is a male predominance in patients under age 3 and an equal gender distribution in cases over age 3.90 Most patients who present before age 6 weeks tend to have a serious underlying condition such as immunodeficiency or malignancy.91,93
Etiology
Etiology is not known, and this condition is thought to represent a hypersensitivity response to triggers. The classic pediatric case presents with a history of an upper respiratory infection or GI illness about 1 to 3 weeks prior to the onset of skin lesions.90,91 Other associated diseases in children include autoimmune conditions such as systemic lupus erythematosus (SLE),94,95 immunodeficiencies,96 and other infectious processes such as otitis media40 and HIV.97 Medications including trimethoprim-sulfamethoxazole and granulocyte colony-stimulating factor have also been reported.98,99,100 In children, about one-quarter of cases are linked with an underlying malignancy, usually hematologic. In addition, cases of Sweet syndrome with malignancy tend to occur in older age.90,91,101
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree