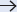15 Vascular Anatomy of the Skin and Muscles Abstract The evolution of plastic surgery has been intimately related to the knowledge of the arterial and venous anatomy of skin, soft tissues, and bone because their transfer and complete survival depend on an adequate blood supply. The first anatomical works, though elegant and precise, were purely descriptive without incorporating a great deal of clinical and surgical correlations in their findings. However, as flap surgery began to rise, particularly during the World Wars, the intrinsic relationship between flap survival and blood supply became evident, prompting surgeons to reappraise the anatomical works of previous anatomists and later on to return to the dissection lab in the search of vascular sources that would provide a reliable supply to a block of tissue. The description of the blood supply to the fascia, the classification of muscles according to their blood supply, the angiosome concept, and more recently the perforasome concept, are all part of a fascinating evolving period that has led us to the current status of flap surgery and tissue transfer that we know today. Nevertheless, despite the great advances that have contributed to increase the safety and reliability of tissue transfer, we still see portions of flaps becoming congested or ischemic, which makes us wonder whether we should have taken that extra perforator or designed our flap differently. Ultimately, the exact territory supplied by a single (perforator) vessel remains a mystery. This chapter describes the different sources of blood supply to the skin and muscles together with an anatomical outline of the vascular sources most commonly used in flap surgery. Keywords: angiosome, blood supply, direct cutaneous vessel, musculocutaneous perforator, perforasome, septocutaneous perforator, skin Plastic surgery is a constant battle between blood supply and beauty. — Sir Harold Gillies, 1957 If a flap is defined as a block of tissue that is transferred from one area to another while maintaining its own blood supply, then it becomes clear that a thorough knowledge of the anatomy of the vascular network is one of plastic surgery’s essential pillars. The knowledge and understanding of the blood supply to the skin and muscles has evolved gradually over the past 135 years. The initial works provided a detailed and exquisite anatomical description. Subsequent studies, inspired by the preceding ones, began to incorporate important clinical interpretations with the aim of offering a surgical solution to a clinical problem. However, the evolution from anatomical descriptions to (plastic) surgical applications was not as straightforward as one would imagine. Belatedness in scientific communication, language barriers, historical conflicts, egos, and personalities were likely responsible for the delay in the application of anatomical findings to reconstructive surgery. For example, because the works of Manchot and Salmon were written in French, those of Pieri in Italian, and those of Spalteholz and Esser in German, they all remained unknown to most of the (English speaking) surgical community until they were translated to English decades later. Another example is that, while Sir Harold Gillies reconstructed wounded British soldiers with his randomly based tubed flaps, Esser in Germany used “arterial” or “biological” flaps, which incorporated a known vascular axis on their base, hence ensuring a more reliable blood supply. Once it became evident that a block of tissue could be safely transferred based on a known vascular pedicle and that such pedicle had a relatively constant anatomy, surgeons were urged to return to the lab to perform anatomical dissections to reappraise the findings of their pioneers and put them in a clinical context. Thus, thanks to the invaluable and exhaustive work of many landmark plastic surgeons, anatomical research steadily met flap surgery until their separate histories converged into the completely interdependent and inseparable marriage we know today. In addition, the discovery of radiology and the development of sophisticated methods of vascular imaging have greatly enhanced the relationship between vascular anatomy and flap surgery as well as significantly aided in the preoperative planning and execution of a large number of flaps. The skin is the body’s largest organ, accounting for 8% of the total body mass, with a surface area of approximately 2 square meters. From a vascular point of view, the skin is supplied by a rich and fully interconnected network, hierarchically organized into layers with distinct anatomical features and physiological functions. The cutaneous circulation delivers nutrients and oxygen and removes waste metabolites, and it also orchestrates thermoregulation, one of the skin’s main tasks. Even though skin has relatively low metabolic demands, it is endowed with a remarkable ability to increase or decrease its capacity by as much as 20-fold as part of its thermoregulatory function, dissipating or conserving heat as required. Moreover, in normal conditions, the blood supply to the skin far exceeds its nutritional requirements, a feature to consider when designing, executing, and monitoring skin flaps. The skin is irrigated by cutaneous branches originating from underlying larger vessels. After piercing the deep fascia, cutaneous vessels begin to ramify to form five interconnected vascular plexuses, which are ultimately responsible for supplying the integument. These plexuses can be found in close association with the different layers of the skin. Three of them are found within the skin, irrigating its adnexal structures, whereas the other two are located deeper in the subcutaneous tissue and deep fascia. From superficial to deep, the cutaneous vascular plexuses are subpapillary, dermal, subdermal, subcutaneous, and fascial, which includes subfascial, intrafascial, and suprafascial (prefascial) components ( Subpapillary plexus: The subpapillary plexus, also called the subepidermal plexus, is located just below the dermal papillae, at the junction between the papillar and reticular dermis. Capillary loops emanate from this plexus into each dermal papilla to supply the overlying epidermis. Due to their terminal configuration, the vessels contained within the subpapillary plexus have no muscular layer and serve a primarily nutritional function. Dermal plexus: The dermal plexus is located within the reticular dermis. It contains a rich venous network as well as arterioles with a discontinuous muscular wall. The main function of this plexus is thermoregulation. This particular function is possible due to the presence of glomera (plural for glomus), which consist of arteriovenous anastomoses surrounded by connective tissue and muscle fibers. Glomera control the flow of blood toward or away from the superficial plexuses in response to autonomic vasomotor control. Subdermal plexus: Located between the dermis and the underlying subcutaneous fat, the subdermal plexus has long been considered the main vascular supply to the skin. The arteries running at this level have a continuous muscular layer, with their function being mainly the distribution of blood flow. From these arteries, branches are given off to both the overlying dermis and the underlying adipose tissue. Within the dermis, these branches join each other to form vascular arcades from which vessels forming more superficial plexuses originate. Subcutaneous plexus: The subcutaneous plexus runs parallel to the skin and is located at the level of the superficial fascia (e.g., Scarpa’s fascia in the trunk). This plexus is nourished by branches coming from direct and indirect vessels (described below), and in turn it sends off branches to supply the overlying network. Fig. 15.1 Blood supply to the skin, including the subpapillary (subepidermal), mid-dermal (dermal), subdermal, subcutaneous, and fascial plexuses. (Reproduced from Zenn, Jones, Reconstructive Surgery Anatomy, Technique, and Clinical Applications, ©2012, Thieme Publishers, New York.) Fascial plexus: As early as 1936, Michel Salmon noted that “the fascia is a very poorly vascularized structure and it is rare to encounter an arteriole within its substance.” Later on Lang identified a fine intrafascial plexus running within the fascia, which was subsequently confirmed by Schafer and Batchelor et al, who concluded that the intrafascial plexus is supplied by the suprafascial and subfascial plexuses. Thus we know now that fascia contains three plexuses—suprafascial, intrafascial, and subfascial. The blood supply to these plexuses comes from branches of perforating vessels (described later in this chapter) and from fascial feeders, which are vessels that do not penetrate the fascia but take part in the subfascial plexus and have no accompanying vena comitantes ( The anatomical configuration of the cutaneous vascular supply has been a matter of debate, particularly regarding denomination and classification of vessels. In fact, systems classifying vessels into 2, 3, and up to 10 different types have been proposed, hampering communication and teaching. Moreover, because the classification of the skin’s vasculature has direct implications in flap terminology and classification, several definitions and systems have also been proposed for perforator flaps. Fig. 15.2 Diagram showing the vessels nourishing the fascia and its associated vascular plexuses (subfascial, intrafascial, and suprafascial). Mc, musculocutaneous perforator; Sc, septocutaneous perforator; F, fascia; Ff, fascial feeder. Although there are some specific regional exceptions, the overall configuration of the skin’s vascular supply is based on source vessels from which cutaneous vessels (perforators) stem to reach the skin. While source vessels are located deep to the deep fascia, perforators travel through different pathways to pierce the deep fascia and reach the skin. Cutaneous vessels can be broadly classified as direct or indirect, according to the route they make on their way to the skin ( Fig. 15.3 Basic configuration of vessels supplying the skin. (Reproduced from Zenn, Jones, Reconstructive Surgery Anatomy, Technique, and Clinical Applications, ©2012, Thieme Publishers, New York.) Direct vessels: These vessels travel between tissues to reach their main destination, which is the skin. In addition, direct vessels can be subclassified into direct cutaneous and septocutaneous. Direct cutaneous vessels, also known as axial vessels, pierce the deep fascia directly without previously passing through muscle or an intermuscular septum. Once above the fascia, these vessels travel parallel to the skin in the subcutaneous plane, giving off branches that supply the overlying plexuses. Direct cutaneous vessels form the vascular basis of axial flaps; the dorsalis pedis, supratrochlear, and superficial circumflex iliac arteries are good examples of direct cutaneous vessels supplying the well-known dorsalis pedis, forehead, and groin axial flaps, respectively. Direct septocutaneous vessels (also called septocutaneous perforators) travel within an intermuscular septum before piercing the deep fascia and reaching the skin without experiencing a significant change in caliber. Examples of septocutaneous vessels include perforators from the descending branch of the lateral circumflex femoral artery running between the rectus femoris and the vastus lateralis to supply the anterolateral thigh flap or perforators from the peroneal artery traveling in the posterior intermuscular septum to supply the skin paddle of a fibular osteocutaneous flap. It is important from a semantic and clinical point of view not to confuse or interchangeably use the terms septocutaneous and fasciocutaneous. As we have illustrated here, septocutaneous describes the anatomical course of a blood vessel on its way to the skin, whereas fasciocutaneous refers to a particular type of skin flap that includes the deep fascia for a specific reconstructive purpose or to enhance vascularization. Indirect vessels: These vessels originate from source vessels and reach the skin secondarily after traveling through muscle, which they supply through numerous side branches. A good example of indirect vessels includes the deep inferior epigastric artery perforator (DIEP) flap supplying the abdominal wall. From a surgical point of view, the importance in differentiating indirect vessels with direct ones is that, in general, flaps based on the former are more technically demanding because the perforator needs to be dissected off its whole intramuscular course all the way down to its source vessel. It can be seen that different terms have been used to name the same structure. This is not an unusual situation when describing the vessels supplying the integument. The simplest way to understand how cutaneous vessels are classified is to appreciate that they are named based on one main concept—the pathway they take to get to the skin upon branching from their source vessel. As already described, direct cutaneous, direct septocutaneous, and indirect vessels supply the integument. Notwithstanding the anatomical accuracy of such a description, from a surgical standpoint it may be considered too broad and simplistic because, as noted by Nakajima et al, cutaneous vessels may actually follow six different pathways on their way to the skin ( Table 15.1 Terms’ equivalence of cutaneous blood vessels
15.1 Introduction
15.2 Basic Science
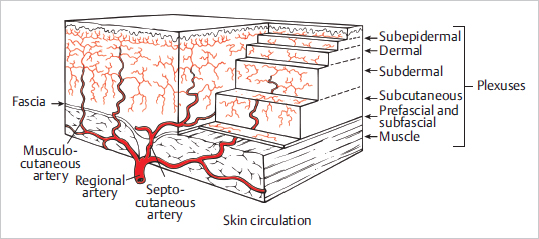
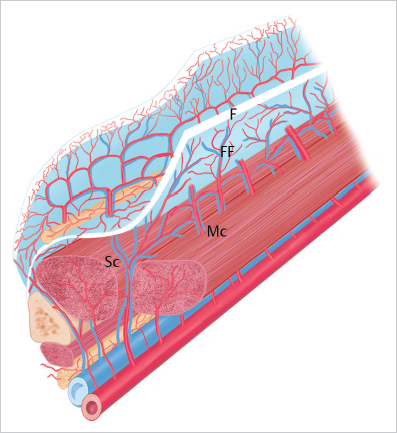
15.2.1 Blood Supply to the Skin
Cutaneous Vascular Plexuses
 Fig. 15.1).
Fig. 15.1).
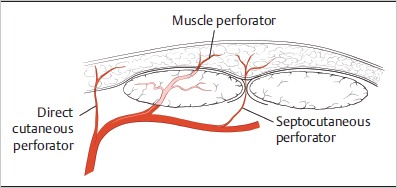
 Fig. 15.2). It is important to note that, because the deep fascia varies in different areas of the body, so may its vascular supply.
Fig. 15.2). It is important to note that, because the deep fascia varies in different areas of the body, so may its vascular supply.
Cutaneous Vessels: Direct and Indirect
 Fig. 15.3).
Fig. 15.3).
 Table 15.1 illustrates the equivalence of these terms.
Table 15.1 illustrates the equivalence of these terms.
Six Types of Vascular Input to the Skin
 Fig. 15.4). The first type is the direct cutaneous vessel, which, after branching off the source artery, bypasses all structures en route to its final destination to the skin. These vessels are typically found around the joints and areas of loose skin because these areas lack muscle and provide space for vessels to directly contact the skin. Direct septocutaneous vessels provide direct blood supply to the skin, but their pathway to get there is slightly different because it traverses through the intermuscular septum of the extremity muscles before reaching the skin. Importantly, even though these vessels travel between muscles, they do not supply them. The third type corresponds to direct cutaneous branches of muscular vessels. As their name implies, the main destination of these vessels is the skin; however, before reaching it they travel through intermuscular spaces. Additionally, in the strict sense, unlike the previous two, direct cutaneous branches of muscular vessels do not stem from the source vessel directly but from one of its muscular branches. Despite their anatomical differences, the three types of vessels discussed so far share in common being direct suppliers to the skin; that is, they do not branch to any other structure along their course, in contrast to the following three, which do supply other tissues (mainly muscles) apart from skin. Perforating cutaneous branches of a muscular vessel branch from the source artery and travel through and supply a muscular structure before reaching the skin. Septocutaneous perforators course in the intermuscular septum; however, in doing so, they give off branches to supply the fascial plexus as well muscle in some cases. Lastly, musculocutaneous perforators are branches from a muscular artery located within the muscle that emerge above the deep fascia and arborize to form the fascial plexus.
Fig. 15.4). The first type is the direct cutaneous vessel, which, after branching off the source artery, bypasses all structures en route to its final destination to the skin. These vessels are typically found around the joints and areas of loose skin because these areas lack muscle and provide space for vessels to directly contact the skin. Direct septocutaneous vessels provide direct blood supply to the skin, but their pathway to get there is slightly different because it traverses through the intermuscular septum of the extremity muscles before reaching the skin. Importantly, even though these vessels travel between muscles, they do not supply them. The third type corresponds to direct cutaneous branches of muscular vessels. As their name implies, the main destination of these vessels is the skin; however, before reaching it they travel through intermuscular spaces. Additionally, in the strict sense, unlike the previous two, direct cutaneous branches of muscular vessels do not stem from the source vessel directly but from one of its muscular branches. Despite their anatomical differences, the three types of vessels discussed so far share in common being direct suppliers to the skin; that is, they do not branch to any other structure along their course, in contrast to the following three, which do supply other tissues (mainly muscles) apart from skin. Perforating cutaneous branches of a muscular vessel branch from the source artery and travel through and supply a muscular structure before reaching the skin. Septocutaneous perforators course in the intermuscular septum; however, in doing so, they give off branches to supply the fascial plexus as well muscle in some cases. Lastly, musculocutaneous perforators are branches from a muscular artery located within the muscle that emerge above the deep fascia and arborize to form the fascial plexus.
Direct cutaneous |
| Axial vessel |
Direct septocutaneous |
| Septocutaneous perforator |
Indirect vessel |
| Musculocutaneous perforator |
Fig. 15.4 The classification of cutaneous vessels according to Nakajima. Type A, direct cutaneous branch of muscular vessel; Type B, septocutaneous perforator; Type C, direct cutaneous; Type D, musculocutaneous perforator; Type E, direct septocutaneous; Type F, perforating cutaneous branch of a muscular vessel. (Reproduced from Zenn, Jones, Reconstructive Surgery Anatomy, Technique, and Clinical Applications, ©2012, Thieme Publishers, New York.)
15.2.2 Angiosome Concept
Over the last 35 years, Ian Taylor and his large number of contributors and colleagues have provided some remarkable foundations to the current knowledge and understanding of the vascular supply to the skin. Inspired mainly by the works of Manchot and Salmon ( Fig. 15.5 and
Fig. 15.5 and  Fig. 15.6), Taylor and his colleagues have undertaken an impressive amount of anatomical research with a strong clinical perspective providing several key concepts for flap surgery. A detailed description of these works is beyond the scope of this chapter—for a full review on the topic the reader is referred to Taylor’s numerous articles and seminal work The Angiosome Concept and Tissue Transfer.
Fig. 15.6), Taylor and his colleagues have undertaken an impressive amount of anatomical research with a strong clinical perspective providing several key concepts for flap surgery. A detailed description of these works is beyond the scope of this chapter—for a full review on the topic the reader is referred to Taylor’s numerous articles and seminal work The Angiosome Concept and Tissue Transfer.
An angiosome (from the Greek angiome [vessel] and somite [segment or sector of the body]) is a three-dimensional block of tissue supplied by one source artery and its accompanying veins. An angiosome usually spans all the way from skin to bone, though in some cases it can be restricted to the deeper layers. Similarly, the representation of an angiosome is not always the same in the deep tissues and the skin—in some areas an angiosome may include a wide portion of deep tissues but a comparatively small skin territory, and vice versa. In their initial work, Taylor and Palmer described 40 different vascular territories (angiosomes), each of them based on a different source artery ( Fig. 15.7). These authors also observed that contiguous angiosomes were interconnected by vessels without a change in caliber (true anastomoses) or, more commonly, by vessels of reduced caliber, which they named choke vessels. These choke vessels have the capacity to dilate in response to metabolic and oxygen requirements. This increase in diameter (and hence improvement of blood supply) is maximal at 48–72 hours after surgery and once established is permanent and irreversible. One of the most important clinical applications derived from the angiosome concept was the fact that, when harvesting a flap, it is possible to safely include tissues from an immediately adjacent angiosome; however, capturing the one beyond may lead to necrosis of the distal portion unless the different angiosomes are linked by “true” anastomoses or a delay procedure is performed prior to flap transfer.
Fig. 15.7). These authors also observed that contiguous angiosomes were interconnected by vessels without a change in caliber (true anastomoses) or, more commonly, by vessels of reduced caliber, which they named choke vessels. These choke vessels have the capacity to dilate in response to metabolic and oxygen requirements. This increase in diameter (and hence improvement of blood supply) is maximal at 48–72 hours after surgery and once established is permanent and irreversible. One of the most important clinical applications derived from the angiosome concept was the fact that, when harvesting a flap, it is possible to safely include tissues from an immediately adjacent angiosome; however, capturing the one beyond may lead to necrosis of the distal portion unless the different angiosomes are linked by “true” anastomoses or a delay procedure is performed prior to flap transfer.
Fig. 15.5 Illustration of the territories supplied by cutaneous arteries according to Carl Manchot, 1889. (Reproduced from Zenn, Jones, Reconstructive Surgery Anatomy, Technique, and Clinical Applications, ©2012, Thieme Publishers, New York.)
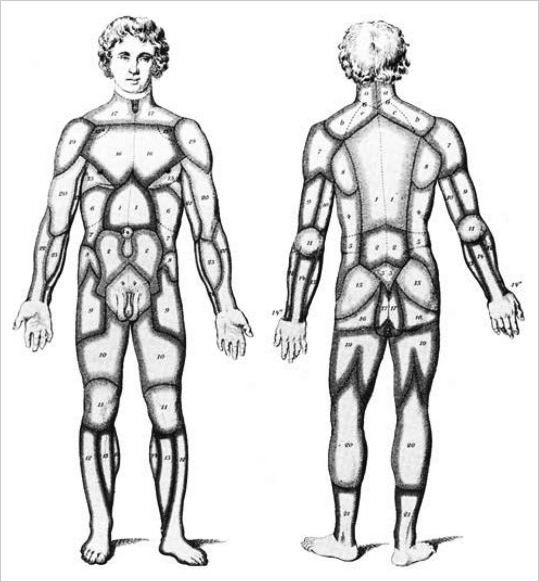
Fig. 15.6 (a,b) The vascular territories of the human body according to Michel Salmon, Artères de la Peau, 1936. (Reproduced from Zenn, Jones, Reconstructive Surgery Anatomy, Technique, and Clinical Applications, ©2012, Thieme Publishers, New York.)
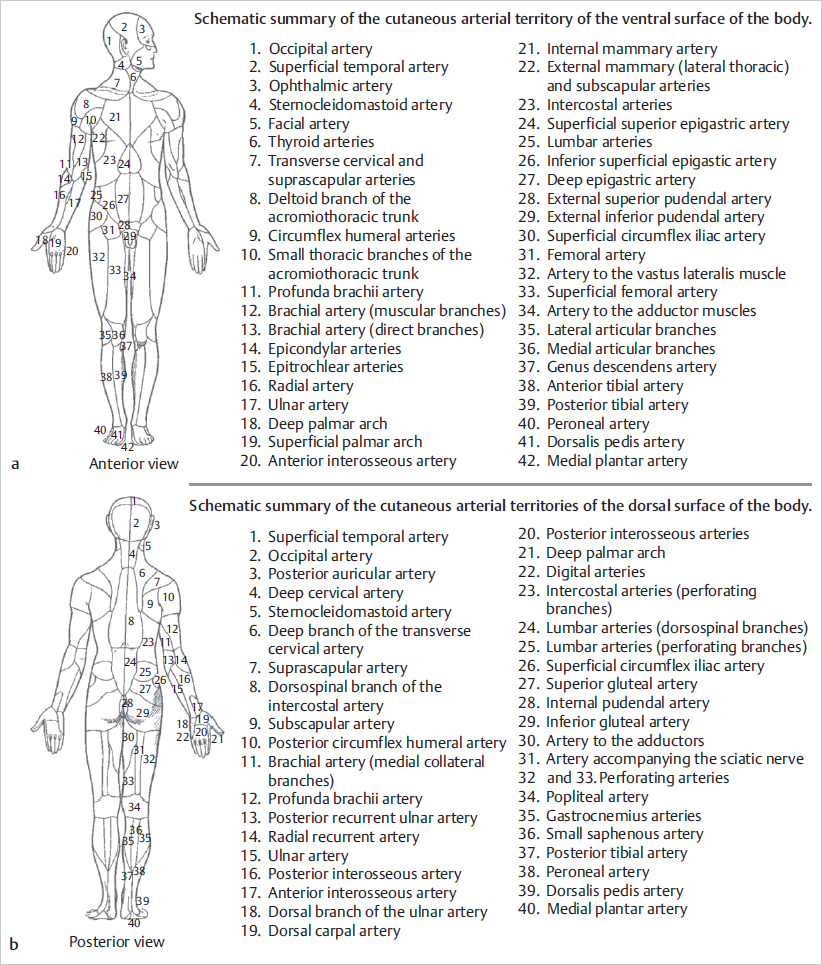
Fig. 15.7 The vascular territories (angiosomes) described by Taylor. Vascular territories of the integument of the skin are delineated according to the source vessel of the perforator. 1, thyroid; 2, facial; 3, buccal internal maxillary; 4, ophthalmic; 5, superficial temporal; 6, occipital; 7, deep cervical; 8, transverse cervical; 9, acromiothoracic; 10, suprascapular; 11, posterior circumflex humeral; 12, circumflex scapular; 13, profunda brachii; 14, brachial; 15, ulnar; 16, radial; 17, posterior intercostals; 18, lumbar; 19, superior gluteal; 20, inferior gluteal; 21, profunda femoris; 22, popliteal; 22A, descending geniculate saphenous; 23, sural; 24, peroneal; 25, lateral plantar; 26, anterior tibial; 27, lateral femoral circumflex; 28, adductor profunda; 29, medial plantar; 30, posterior tibial; 31, superficial femoral; 32, common femoral; 33, deep circumflex iliac; 34, deep inferior epigastric; 35, internal thoracic; 36, lateral thoracic; 37, thoracodorsal; 38, posterior interosseous; 39, anterior interosseous; 40, internal pudendal. (Reproduced from Zenn, Jones, Reconstructive Surgery Anatomy, Technique, and Clinical Applications, ©2012, Thieme Publishers, New York.)
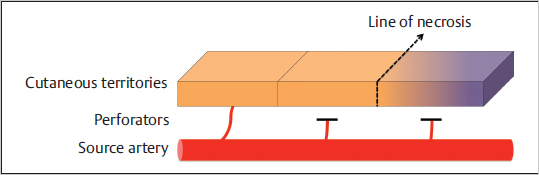
The perforators originating from an angiosome’s source artery supply the cutaneous territory of that angiosome. According to Taylor, each perforator has its own territory, which connects radially with its neighbors via true anastomoses or choke vessels, so that all perforators together make up for the angiosome’s cutaneous territory. The same author has noted that the territory irrigated by a single perforator is influenced by its size and the density of perforators in the same area. Thus, in regions like the head and neck, torso, and proximal limbs, where perforators are large and low in number, one can anticipate that a single perforator supplies a large area; conversely, in the presence of small and numerous perforators, the territory of each is likely small, as is usually the case in distal limbs, hands, and feet. Again, as before, the territory beyond the one adjacent to the selected perforator is at higher risk of necrosis ( Fig. 15.8). These findings and others have provided remarkable insights not only to the design and harvest of flaps but also to the understanding of ischemic complications seen in flap surgery.
Fig. 15.8). These findings and others have provided remarkable insights not only to the design and harvest of flaps but also to the understanding of ischemic complications seen in flap surgery.
Remember 
An angiosome is a three-dimensional block of tissue supplied by one source artery and its accompanying veins. Adjacent angiosomes are linked by true anastomoses or choke vessels.
15.2.3 Anatomic, Dynamic, and Potential Territories and the Delay Procedure
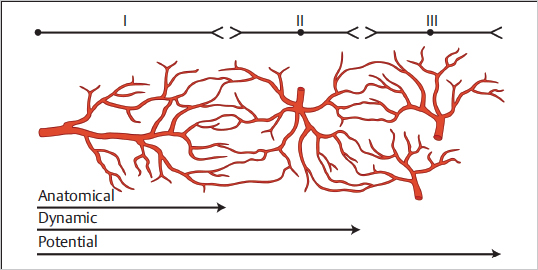
Cormack and Lamberty in their work The Arterial Anatomy of Skin Flaps describe three different types of cutaneous vascular territories, namely, anatomical, dynamic, and potential ( Fig. 15.9).
Fig. 15.9).
The anatomical territory of a vessel refers to the area irrigated by that vessel before anastomosing with any of its neighbors. This is analogous to Taylor’s cutaneous territory of a single perforator. The observation that whenever a cutaneous vessel is occluded the adjacent one supplies its territory, led to the denomination of the dynamic territory. Dynamic territories are therefore developed following flap elevation where a cutaneous vessel is divided and nevertheless the area it used to irrigate remains viable thanks to a rearrangement of the local blood flow that allows an adjacent vessel to “invade” that territory. This concept correlates with Taylor’s observation that the safe area of a single cutaneous perforator is formed by its own territory and those immediately adjacent to it. The potential territory of a cutaneous vessel includes areas beyond its dynamic territories and can only be captured following a delay procedure.
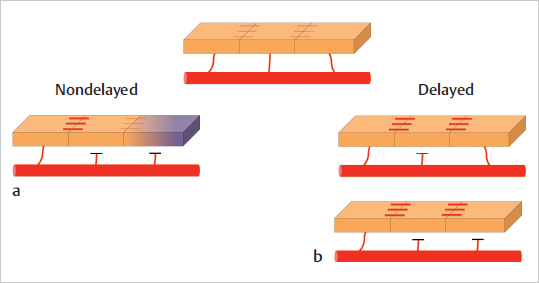
Bearing the concepts of angiosome and cutaneous vascular territories in mind, the understanding of the delay procedure becomes easier. Flap delay consists in the segmental interruption of blood supply to a piece of tissue (most commonly skin, but can be muscle as well) in a staged manner so that the vascular inflow from the main pedicle gradually extends to the distal portion of the flap, which would otherwise not survive if raised in one stage. The opening of choke vessels and the subsequent capture of potential territories form the basic anatomical and physiologic basis of the delay phenomenon ( Fig. 15.10). An important aspect in flap delay is the fact that after partial elevation, the flap is sutured back at the donor site so that it can adapt to the new vascular situation without the “stress” elicited by complete elevation and transfer. Chapter 16 provides a more detailed explanation of the surgical steps and the physiologic changes of flap delay.
Fig. 15.10). An important aspect in flap delay is the fact that after partial elevation, the flap is sutured back at the donor site so that it can adapt to the new vascular situation without the “stress” elicited by complete elevation and transfer. Chapter 16 provides a more detailed explanation of the surgical steps and the physiologic changes of flap delay.
Fig. 15.8 The vessel supplying one territory is able to supply the adjacent one. However, capturing one territory beyond usually leads to necrosis of the distal portion.
Fig. 15.9 The anatomical, dynamic, and potential territories of cutaneous vessels. Although anatomical and dynamic territories can be safely harvested based on a single vessel, potential territories will not be adequately perfused unless a delay procedure is performed. (Reproduced from Zenn, Jones, Reconstructive Surgery Anatomy, Technique, and Clinical Applications, ©2012, Thieme Publishers, New York.)
15.2.4 Perforasome Concept
Earlier concepts introduced by Taylor and Nakajima as already discussed helped to formulate the work of Saint-Cyr, who described the perforasome theory in 2009. In this work, the authors used dye injections followed by tomographic angiography to assess vascular anatomy and flow characteristics for the unique vascular arterial territory of a given perforator—termed a perforasome. The theory blends classic concepts of vascular anatomy and introduces four principles that define the perforasome.
First principle: Each perforasome is linked with neighboring perforasomes via linking vessels that can vary in size. The larger vessels directly link one perforator to the next and as such are termed direct vessels. Conversely, indirect vessels are smaller in size, located in the subdermal plexus, and represent recurrent flow to adjacent perforator branches. The principle in this theory further postulates that these two distinct and different flow patterns are protective mechanisms that help to ensure blood flow throughout the perforasome in the event of vascular injury.
Second principle: Flap design should be based on the direction of linking vessels. The reason for this is because the orientation of linking vessels corresponds to maximal blood flow. In the extremities, this blood flow follows the axial direction of the involved limb, whereas the direction of flow in the trunk is perpendicular to the midline. Larger perforasomes typically contain more linking vessels and have more variability. Therefore, perforators with a very large perforasome will have a higher degree of freedom in flap design and orientation.
Third principle: The filling of vessels from the source artery begins with perforators of that same source artery and then continues to include perforators of adjacent source arteries. Only after the source artery fully perfuses the vessels within its own perforasome do the linking vessels perfuse adjacent perforasomes.
Fourth principle: Source vessels found near joints and articulations have branching vessels that preferentially lead away from the articulation. In contrast, perforators located at a midpoint between two articulations have flow in many directions.
The perforasome theory helps to explain the mechanism by which large perforator flaps can be harvested based on a single perforator. Linking vessels of different sizes enable shared perfusion of adjacent perforasomes and act as a protective feature to ensure collateral blood flow in the event of disruption.
15.3 Blood Supply to the Muscles
The use of muscle (flaps) to reconstruct defects of varying nature brought forward a whole new perspective as to the possibilities of reconstructive surgery, which until then were “limited” to employing skin flaps only. Their bulk, reliable blood supply, and, for most, consistent anatomy, were seen as major advantages, as was also the fact that they were based on sizeable vascular pedicles. Later on, when it became evident that a muscle was able to support an overlying skin paddle or that they could be used to provide functional restoration of a paralyzed segment, muscle flaps became a fundamental tool in the plastic surgeon’s armamentarium.