Keywords
Ustekinumab, Stelara, Psoriasis, Interleukin-12, Interleukin-23, PASI
Key points
- •
Ustekinumab is a human immunoglobulin G κ monoclonal antibody that binds to the p40 protein subunit shared by interleukin-12 (IL-12) and IL-23 cytokines.
- •
Ustekinumab is safe and efficacious for the treatment of moderate-to-severe plaque psoriasis and psoriatic arthritis.
- •
Efficacy, tolerability, ease of use, and long dosing intervals contribute to patient satisfaction and adherence.
Introduction
It can be argued that psoriasis is the most successfully treated immune-mediated disease and a promising example of the impact of translational research. Improved understanding of the pathophysiology of psoriasis in the late twentieth and early twenty-first century facilitated the evolution of treatments from nonspecific, immunosuppressive medications to novel targeted therapies. Several biologic agents have been developed for the treatment of psoriasis over the past decade. Biologics act with greater target specificity and generally have not demonstrated end-organ toxicities more commonly observed with other effective systemic agents. Although concern over adverse events (AEs; infections and malignancy) exists with some biological therapies, studies have shown that the risks are minimal and benefits are well documented. Targeted therapies, such as tumor necrosis factor (TNF)-α inhibitors, have considerably enhanced the treatment of moderate-to-severe plaque psoriasis and improved overall quality of life in psoriasis patients.
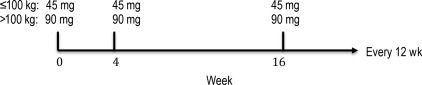
Ustekinumab is a human, monoclonal antibody that binds to the shared p40 subunit of interleukin (IL)-12 and IL-23. It was US Food and Drug Administration approved for the treatment of moderate-to-severe plaque psoriasis in 2009 and has proven to be a safe and efficacious treatment. Recommended dosing occurs at weeks 0, 4, and every 12 weeks thereafter ( Fig. 11.1 ). Infrequent dosing, high efficacy, and tolerability could positively affect adherence in psoriasis patients, who, in the past, have not treated their disease adequately.
Mechanism of action
Although monoclonal antibodies directed against TNFs proved to be effective in treating psoriasis, more specific targets were sought. Ustekinumab is a human immunoglobulin G κ (IgG κ ) monoclonal antibody that binds to the p40 protein subunit shared by IL-12 and IL-23 cytokines, preventing their interactions with the heterodimeric IL-12 receptor subunit. IL-12 and IL-23 are naturally occurring cytokines involved in inflammatory and immune responses. In transgenic mice, the overexpression of IL-12 has been linked to the development of inflammatory skin lesions; not surprisingly, lesional skin in psoriasis patients has demonstrated increased expression of IL-12 compared with nonlesional skin.
IL-12 and IL-23 are produced primarily by antigenic stimulation of dendritic cells and macrophages. IL-12 is composed of a p35 and a p40 subunit, the latter being largely expressed in psoriatic lesional skin; its receptor is made up of IL-12RB1 and IL-12R2 subunits. Binding of IL-12 to its receptor causes activation of a JAK-STAT signaling pathway, causing T cells to be assigned to the Th1 pathway and downstream secretion of its effector interferon-γ (IFN-γ).
IL-23 is composed of p19 and p40 subunits (both largely expressed in lesional skin). It binds to a receptor made of the IL-12RB1 and IL-23R subunits. Similar to IL-12, binding of the p40 subunit with IL-12RB1 and the p19 subunit with IL-23R signals through JAK-STAT signaling, activating STAT3 and causing a Th17-driven response.
Ustekinumab blocks IL-12 and IL-23 from binding to the IL-12Rβ1 receptor chain of IL-12 (IL-12Rβ1/β2) and IL-23 (IL-12Rβ1/23R) receptor complexes on the surface of natural killer and T cells. In vitro models showed ustekinumab equally disrupted the action of IL-12 and IL-23, blocking STAT 3,4 phosphorylation and ultimately IFN-γ, IL-22, and IL-17 production.
Pharmacokinetics
Similar to endogenous IgG, ustekinumab has an approximate half-life of 3 weeks. Peak serum concentration of ustekinumab occurred at approximately 13.5 days after a 45-mg dose and 7 days after a 90-mg dose. Steady-state drug concentrations were achieved by week 28. Ustekinumab is metabolized much the same as human IgG. An Fc receptor binds to IgG and carries it across the cell membrane to be degraded within the cell.
Efficacy
Compared with conventional therapies, many biologics have exhibited increased efficacy, and ustekinumab is not an exception. Efficacy in psoriasis patients is measured by improvement in Psoriasis Area Severity Index (PASI), body surface area (BSA), Physician Global Assessment (PGA), and dermatology-related quality of life. Ustekinumab has demonstrated short- and long-term efficacy in the treatment of moderate-to-severe psoriasis and remains among the best evaluated biologic in the treatment of psoriasis. Two landmark, phase III, prospective, long-term extension studies, PHOENIX 1 and PHOENIX 2, observed efficacy, measured by clinical response, and safety of ustekinumab in psoriasis patients for up to 5 years.
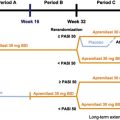
PHOENIX 1 was a multicenter, randomized, double-blinded, placebo-controlled trial that studied 766 patients receiving ustekinumab 45 mg, ustekinumab 90 mg, or placebo in a 1:1:1 ratio. Dosing occurred at weeks 0, 4 and every 12 weeks thereafter, with limited flexibility. Patients initially treated with placebo were rerandomized to receive ustekinumab 45 mg or 90 mg at week 12. The primary efficacy endpoint was the proportion of patients experiencing a 75% improvement from baseline PASI (PASI-75) score at week 12. Patients initially assigned to ustekinumab, who had achieved PASI-75 at weeks 12 and 28, were rerandomized at week 40 to continue on ustekinumab every 12 weeks, or switched to placebo. Patients randomized to placebo at week 40 were placed back on treatment when they lost 50% of PASI improvement ( Fig. 11.2 ).
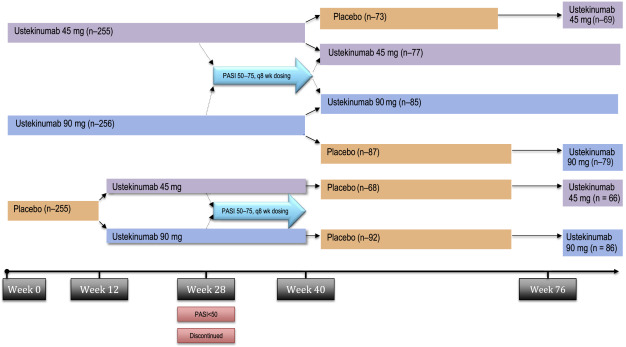
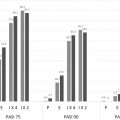
For many, improvement began within 2 weeks of their first dose. Approximately 67% of patients receiving ustekinumab 45 mg ( P <.0001) and 66% of patients receiving ustekinumab 90 mg ( P <.0001) achieved PASI-75 at week 12. Three percent of placebo-treated patients achieved PASI-75 at week 12. Furthermore, roughly 60% of patients receiving ustekinumab 45 mg and ustekinumab 90 mg were deemed clear or almost clear by the physician at week 12. By week 28, 50% of patients achieved 90% reduction from their baseline PASI score (PASI-90), a new clinical standard for efficacy.
Patients initially assigned to ustekinumab, who had achieved PASI-75 at weeks 12 and 28, were rerandomized at week 40 to continue on ustekinumab every 12 weeks, or switched to placebo. Patients randomized to placebo at week 40 were placed back on treatment when they lost 50% of PASI improvement. Importantly, rebound psoriasis was not reported in patients withdrawn from treatment. The median time to loss of PASI-75 was approximately 15 weeks, and 85% of patients reinitiated on treatment saw a PASI-75 within 12 weeks of restarting treatment. Efficacy of therapeutics in psoriasis was also measured by patient-reported outcomes. At week 12, 50% of patients reported that psoriasis had little to no effect on patient’s quality of life. These results were sustained for patients continued on therapy and worsened for patients when treatment was withheld.
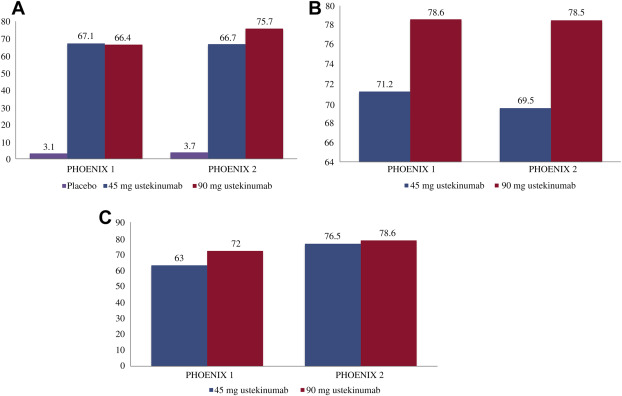
The PHOENIX 2 study enrolled 1230 patients and had the same study design as PHOENIX I through the first 28 weeks; however, it sought to answer whether dosing intensification would improve response in partial responders. Partial responders, defined as patients obtaining greater than 50% PASI improvement and less than 75% PASI improvement, were rerandomized at week 28 to receive every-8-week dosing or continue on every-12-week dosing. Week 12 data were similar to that in PHOENIX 1, with ustekinumab performing superior to placebo, with a PASI-75 of 63.1% and 72% in the 45-mg and 90-mg groups, respectively.
In the portion of patients treated with ustekinumab 45 mg, 22% were partial responders compared with 15.8% receiving 90 mg. Data through 1 year revealed increasing dosing frequency to every 8 weeks in patients receiving 45 mg of ustekinumab did not result in greater efficacy compared with continuing treatment every 12 weeks. However, dosing ustekinumab 90 mg every 8 weeks resulted in a high PASI-75 rate. This finding prompted investigators to allow more dosing flexibility in the long-term extension phase. Patients receiving 45 mg every 12 weeks were able to increase dose 45 mg every 8 weeks and then to 90 mg every 8 weeks. Similarly, patients receiving 45 mg every 8 weeks could increase frequency to 90 mg every 8 weeks; and patients who were receiving 90 mg every 12 weeks could increase their dose to every 8 weeks.
Most patients receiving ustekinumab maintained their clinical response over time. The long-term extension phase of PHOENIX 1 demonstrated in the overall population that 63.4% of patients receiving 45 mg ustekinumab and 72% of patients receiving 90 mg ustekinumab maintained a PASI-75 after 5 years. Comparably, high levels of clinical response were maintained over 244 weeks in patients studied in the PHOENIX 2 study; 76.5% (ustekinumab 45 mg) and 78.6% (ustekinumab 90 mg) of patients achieved a PASI-75, and 50.0% (ustekinumab 45 mg) and 55.5% (ustekinumab 90) attained PASI-90. Notably, improved response was observed following dosing adjustments, allowing clinicians to customize treatment for their patients. Ustekinumab was also noted to improve nail psoriasis until up to 1 year of treatment in those receiving maintenance treatment ( Fig. 11.3 ).
Several randomized, controlled trials have compared efficacy of ustekinumab against other systemic and biologic agents. The Active Comparator (CNTO1275/Enbrel) Psoriasis Trial (ACCEPT) study was a direct comparator study establishing greater efficacy of ustekinumab over etanercept. The Psoriasis Longitudinal Assessment and Registry (PSOLAR) is a prospective observational study (sponsored by Janssen Biotech) comparing the safety and efficacy of 3 TNF inhibitors (adalimumab, infliximab, etanercept) with ustekinumab. At 6 and 12 months, comparisons of percentage decrease in BSA, PGA, and patient-reported outcomes showed ustekinumab to be more effective than the TNF inhibitors. At 12 months, 59.2% of patients receiving ustekinumab achieved a PGA of clear or almost clear, compared with 56.5%, 42.0%, and 57.6% for adalimumab, infliximab, and etanercept, respectively. The mean improvement of BSA from baseline for patients treated with ustekinumab after 1 year was 16.3%. Patients treated with infliximab saw a decrease of 17.6% BSA after 1 year, whereas patients on etanercept and adalimumab saw less significant decreases of 13.8% and 12.3%, respectively. The mean improvement in dermatology quality-of-life index from baseline was 7.5 for ustekinumab, compared with 6.9, 5.4, and 4.9 for infliximab, etanercept, and adalimumab, respectively.
Factors effecting decreased efficacy
Although ustekinumab has demonstrated efficacy for psoriasis, there remains a portion of patients who have shown insufficient response. It is unclear what is responsible for the lack of response; however, heavier patients are notoriously difficult to treat, and there are well-documented effects of body mass index on disease incidence, severity, and response to biologic treatment. PHOENIX 2 investigators noted patients who needed increased dosing in ustekinumab were heavier, had more severe psoriasis, and were more likely to have comorbidities. The highest incidence of dosing adjustments was observed among patients weighing greater than 100 kg who were originally randomized to 45 mg. Clinical and pharmacokinetic data support a 90-mg dose in patients (>100 kg); lower serum concentrations were noted in heavier patients at each dose. Per the package insert, the recommended administration for patients weighing less than or equal to 100 kg (220 lbs) is 45 mg initially and 4 weeks later, followed by 45 mg every 12 weeks. For patients weighing greater than 100 kg, the recommended dose is 90 mg initially and 4 weeks later, followed by 90 mg every 12 weeks.
In the PHOENIX 2 study, partial responders were more likely to have failed treatment with at least 1 conventional systemic or biological agent compared with nonresponders. In some clinical trials, patients with prior TNF inhibitor use had worse PGA and percentage BSA responses compared with biologic-naïve patients.
PSOLAR reinforced the effect of weight on response to biologic therapy; patients with lower weight experienced superior results compared with heavier patients. Disease severity correlated with larger decreases in BSA but a lower PGA response. PSOLAR demonstrated patients with prior TNF inhibitor use had less of a response compared with bionaïve patients, whereas prior ustekinumab use had no effect on response.
Efficacy in palmoplantar psoriasis
Palmar plantar psoriasis (PPP) is a disabling and disfiguring form of psoriasis characterized by the presence of plaques with and without sterile pustules and fissures on the palms and soles in conjunction with psoriatic plaques in the typical distribution. PPP is associated with a higher degree of pain and an increased morbidity compared with psoriasis patients without palm and sole involvement. It is also notoriously difficult to treat using previous conventional therapies.
In an open-label, 24-week study to evaluate the safety and efficacy of ustekinumab in patients with moderate-to-severe PPP, 20 subjects received either 45 mg (patients weighing ≤100 kg), or 90 mg (patients weighing >100 kg), of ustekinumab subcutaneously at weeks 0, 4, and 16. After 16 weeks of treatment with ustekinumab, 7 of 20 subjects achieved clinical clearance (defined as a Palm-Sole PGA of clear or almost clear). After 16 weeks of treatment, 6 of 9 subjects who received 90 mg of ustekinumab achieved clinical clearance, compared with only 1 of 11 subjects who received 45 mg.
Efficacy in pediatric psoriasis
Ustekinumab has been shown to be safe and effective in treatment of pediatric psoriasis. The CADMUS trial is a phase 3, multicenter, double-blinded, placebo-controlled study of 110 male and female patients aged 12 to 17 years old. Patients were randomly assigned to receive a standard dose adjusted by weight (0.75 mg/kg [<60 kg], 45 mg [60–100 kg], 90 mg [>100 kg]), a half-standard dose (0.375 mg/kg [<60 kg], 22.5 mg [60–100 kg], 45 mg [<100 kg]) at weeks 0, 4, followed by every 12 weeks, or placebo at week 0 and 4 with crossover to standard or half-standard dosing at week 12.
The results of this study were apparent very quickly. By week 4, approximately one-third of patients in each ustekinumab group were clear or almost clear. At week 12, 67.6% of patients receiving the half-standard dose and 69.4% of patients receiving the standard dose achieved a PGA of 0 or 1, and 54.1% of patients receiving the half-standard dose and 61.1% of those receiving the full dose achieved PASI-90. In time points beyond week 12, clinical response was more favorable in the standard dose compared with the half-standard dose. Pharmacokinetic and efficacy date for the dose adjustment of 0.75 mg/kg was appropriate for patients weighing less than 60 kg. Adverse events in this study were similar across treatment groups, with no observed dose effect.
Efficacy in psoriatic arthritis
IL-12, IL-23, and IL-17 play a pivotal role in the synovial changes seen in psoriatic arthritis. Treatment modalities involved nonsteroidal anti-inflammatory drugs, disease modifying antirheumatic drugs, and TNF inhibitors. Several studies assessed the efficacy and safety of ustekinumab in patients with psoriatic arthritis. Clinical response in psoriatic arthritis is measured by a 20% improvement in baseline from the American College of Rheumatology (ACR20). In 1 phase 3, international, placebo-controlled trial to assess the efficacy and safety of ustekinumab in patients with psoriatic arthritis, patients were randomized to receive ustekinumab 45 mg, 90 mg, or placebo in a 1:1:1 ratio, and 50% of patients receiving ustekinumab also received methotrexate. ACR20 responses were significantly higher in patients treated with ustekinumab as were improvements in skin disease, dactylitis, enthesitis, and disease activity as measured by C-reactive peptide. Ustekinumab proved to be effective independent of methotrexate use.
Another phase 3 randomized, controlled study assessed patients previously treated with a TNF inhibitor. Investigators reinforced that ustekinumab was effective in treating psoriatic arthritis in patients already exposed to TNF inhibitor, although not as effective as those who were bionaïve. These 2 studies continued for 2 years and showed clinical and radiographic evidence that ustekinumab is safe and effective in psoriatic arthritis.
Safety
Ustekinumab has been the foremost biologic evaluated for safety in patients with psoriasis. The PHOENIX 1, PHOENIX 2, and ACCEPT trials are the preeminent sources of long-term safety data collected for patients treated with ustekinumab. Investigators found no statistically significant difference in overall AEs between ustekinumab- and placebo-treated patients. Most reported AEs were nasopharyngitis, upper respiratory infections, headaches, and arthralgias. More than 5 years in the PHOENIX 1 and 2 trials, there was no evidence of cumulative end-organ toxicity. There was a low incidence of autoantibodies to ustekinumab. One ustekinumab-treated patient developed reversible posterior encephalopathy syndrome; however, there were no reported cases of demyelinating disease.
Theoretically, a blockade of the actions of IL-12 and IL-23 could predispose a person to viral, bacterial, and fungal infections. Rates of infections and serious infections were comparable during the placebo-controlled portions of randomized, controlled trials, and rates of serious infections in ustekinumab-treated patients were comparable to those expected in patients with psoriasis treated with conventional systemic drugs. Although patients on TNF inhibitors are at increased risk for reactivation of latent tuberculosis (TB) reactivation, ustekinumab does not appear to increase this risk.
In placebo-controlled periods, rates of infections, serious infections, and malignancies were comparable with those of patients receiving placebo. Rates of malignancies other than nonmelanoma skin cancer in ustekinumab-treated patients were consistent with rates expected in the general population, suggesting that ustekinumab does not increase rate of malignancy. Of more than 12,000 patients (40,388 patient-years) observed in PSOLAR, overall incidence rates for malignancy (0.68/100 patient-years), serious infection (1.6/100 patient years), and mortality (0.93/100 patient years) were significantly lower when compared with other biologics. Most common reported types of serious infections were pneumonia and cellulitis. PSOLAR demonstrated a higher risk of serious infections with adalimumab and infliximab compared with nonbiologic treatment, while also observing no increased risk of serious infections with ustekinumab or etanercept.
Early studies exhibited a higher risk of major adverse cardiovascular events (MACEs) in patients receiving anti-IL-12/23 antibodies compared with those in placebo groups. Concern over a possible link between ustekinumab and MACEs prompted an in-depth investigation. PSOLAR found ustekinumab to have a significantly lower rate of MACEs (0.33/100 patient-years) compared with other biologics. The risk of MACEs in patients treated with anti-IL-12/23 biological was evaluated in 2 large meta-analysis studies that came to conflicting conclusions. Reich and colleagues analyzed 3000 patients on ustekinumab and found no increase in MACEs compared with patients in the general or psoriasis populations.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree