Introduction
Tissue engineering represents an interdisciplinary field, a forum where the principles of biomaterial engineering, the molecular biology of cells and genes, and the clinical sciences interact intensively through the combined efforts of scientists, engineers, and clinicians. Tissue engineering attempts to exploit the cells’ reproductive potential and to harness the body’s intrinsic capacity for healing and regeneration. These cells produce growth factors and cytokines, which function as architects of the repair processes. As defined by Robert Langer in 1993, tissue engineering comprises :
- •
The isolation and manipulation of individual cells or cell substitutes, used for therapeutic infusion
- •
The identification of tissue-inducing substances, such as growth factors, and their appropriate delivery to their target
- •
Cells placed on or within matrices that permit the delivery of nutrients but protect cells from immunological destruction
The possibilities of cell engineering, matrix development, and growth-factor therapies are extensive. Imagine how application of proangiogenic proteins may reverse hypoxia in flaps, or stimulate tissue integration; how peptides may attack stubborn infections in wounds; how autologous cells from biopsies may generate an autologous dermal and epidermal cover on extensive burn wounds. Based on CT-imaging, rapid prototyping may generate a pre-shaped 3-D scaffold in which these cells can be seeded ( Fig. 6.1 ). These scaffolds may come in the shape of an ear, a nose, a mandible, or a breast. The paucity of optimal donor tissues may be treated with off-the-shelf tissues using tissue-engineering strategies.

However, the speed of translation of tissue-engineering science into clinical reality is slow due to the intrinsic complexity of tissues.
In this chapter, we seek to provide a perspective on the current strategies used in translational tissue engineering. Additionally, describe five clinical surgical domains, in which the integration of tissue-engineering protocols is a fact.
Mechanisms of Action
Current Strategies in Tissue Engineering
Three major approaches have been established toward engineering implants for regeneration: (1) implantation of engineered scaffolds and matrices; (2) implantation of neo-tissue derived from cells; (3) implantation of cells seeded within matrices.
Complex Scaffolds and Matrices
The optimal bioengineered substitute must have a structure similar to the authentic tissues it must replace. It should be autologous to optimally integrate without rejection, 3-dimensional to bridge deep defects, porous to allow cell migration, bio-inductive for cells to proliferate and to topically produce extracellular matrix (ECM) components and growth factors, bio-inductive for vascular sprouts to develop within the construct and chemotactic for cells from the wound surroundings to infiltrate, stimulate vasculogenesis and optimize tissue integration. When selective absorption of the matrix occurs, its composition and structure should be maintained. The absorption process should not result in deposition of toxic side products. The accompanying inflammatory response should be limited not to hamper cellular migration and repopulation stimulated by site-specific cues for cellular adhesion. This extensive list of requirements is self-explicatory as to why the ideal off-the-shelf scaffold has not been developed yet, despite the detailed knowledge of many biocompatible materials. It is the collaboration between material scientists, chemists, clinicians, and molecular biologists that forms the multidisciplinary environment required to design a veritable biomimetic smart scaffold.
Fundamental Role of Vascularization
The absence of a vascular network capable of distributing oxygen and nutrients within the matrix is the key limiting factor in the overall success of tissue engineering. Nutrient supply and waste removal of limited-thickness engineered tissues such as skin, cartilage, or bone can initially be overcome by diffusion, until neovascularization takes over. In 1973, Folkman demonstrated that cells only survive within a 3-mm distance from a nutrient source. From clinical evidence using artificial dermis in burn patients, we estimate the critical thickness to be even less, between 1 and 2 mm. The ultimate purpose of tissue engineering is to incorporate an inherent vascular bed, ready to be connected to the host vascular system. A viable vascular network within the tissue construct represents the missing link between the host and the engineered implant.
The adult vascular network remodels itself by the enlargement of existing collaterals induced by shear forces, as well as by angiogenesis; the coordinated migration and proliferation of endothelial progenitor cells and pericytes from the existing vascular bed and their subsequent maturation into endothelial cells, stabilized by these enveloping smooth muscle cells. The primary stimulus causative for capillary sprouting is hypoxia. The extracellular matrix functions as a reservoir of guidance molecules, i.e. growth factors that induce incoming blood vessels (angio-induction) and as a scaffold to guide outgrowth of blood vessels. To further enhance vascularization, proangiogenic endothelial progenitor cells can be seeded into this framework. These phenomena can be induced by an in-situ or ex-vivo approach.
With in-situ cultivation of cells and tissues, the patient’s own body is used as incubator. Such a strategy sounds logical and efficient. However, infiltration of undesired tissues and the obligatory use of shaped biocompatible implants as scaffold make this approach far from evident.
On the other hand, an ex-vivo approach to overcome the invasion of undesired tissues requires an immediate reperfusion once the regenerated tissue is “transplanted” in situ. Moreover, the ex-vivo revascularization step should occur in an incubator that mimics the real-time wound environment. Such strategy is also not evident.
Both approaches are intensively investigated.
Proangiogenic Strategies
The most obvious strategy focuses on adding proangiogenic molecules to the recipient tissues or the scaffold: growth factors such as VEGF, bFGF, PDGF, IGF-1 function as coordinators of the wound healing process, turning a temporary scaffold into a vascularized scaffold by attracting vascular progenitor cells, which will interact with local cells to grow a vascular network in situ. These proangiogenic proteins are secreted by the cells in a specific sequence. In an in vivo model, these processes occur spontaneously once the cells are added to the wound. In a hostile milieu, missing cells and growth factors could be added to induce healing and integration. However, the half-life of externally added proteins to the healing wound is very short. In-vivo and ex-vivo gene transfer protocols may overcome the short-acting effect of proteins by turning local cells into production units of those proangiogenic growth factors. Nevertheless, to avoid tumor growth, it is essential to control this proangiogenic stimulus.
In a clinical setting, prefabrication and prelamination techniques can be used to induce the formation of a macroscopical, transplantable vascular supply to tissues or tissue constructs. Similarly, hypoxia and inflammation in response to wounding are triggers to induce angiogenesis.
Recent proof-of-principle strategies combine engineering with biomolecular approaches. Miller and colleagues described 3-dimensional printed networks of carbohydrate glass-molds coated with PLGA-copolymer. Constructs were encapsulated in a range of hydrogels along with living cells. Following ECM cross-linking, glass particles were dissolved to reveal patent fluidic PLGA-channels.
An alternative approach is the use of photo-cross-linkable gelatin methacrylate using projection stereolithography based on computer-aided design. The advantage of CAD/CAM-design is the large-scale reproducibility and the patient-specific scaffold production through the combination with CT-imaging.
Another option is to use stacking strategies, coined “cell-sheet engineering.” A temperature-responsive surface is used to culture cells. Upon reaching confluency, the surface can be cooled to 20°C to reduce its hydrophobicity, and the intact cell sheet can be removed, preserving cell-to-cell junctions, ECM and cell-surface proteins. These cell sheets can then be stacked to generate multi-layered, cell-dense tissues. Stacked layers of cardiomyocytes that beat simultaneously without the use of a formal scaffold are a powerful example of this method.
A controlled induction of vascular networks by ex-vivo or in-situ strategies will determine when tissue engineering will be incorporated successfully and substantially in clinical practice.
Decellularization of Tissues to Obtain the Ideal Scaffold
Decellularization is an approach that removes resident cells from a tissue or even an organ. This cellular removal can be achieved by physical, chemical and enzymatic methods. The aim is to preserve the native structure and function of the ECM, including its perfusable vasculature. These decellularized matrices can be re-seeded and repopulated with autologous cells to restore morphology and function. The strength of this approach may be the preservation of biomolecular environmental cues that are likely to direct cellular phenotype.
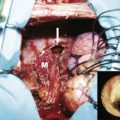
Song and colleagues decellularized cadaveric rat kidneys via detergent-perfusion through the renal artery. Acellular scaffolds were repopulated with human umbilical venous endothelial cells and rat neonatal kidney cells. After seeding, the organs were transferred to a perfusion bioreactor to provide whole-organ culture conditions. Upon transplantation in vivo, urine was produced.
The primary aim of such decellularization strategies is to deplete the tissues and organs of their immunogenic elements and to repopulate them with autologous cells. Similar whole-organ decellularization methodologies are tailored for heart, liver, lung and trachea.
Cells and Stem Cells: the Substrate
Scaffolds deliver the structural guidance and support for proliferating and migrating cells through their honeycomb structure. They transmit tissue-specific mechanical forces that cue the behavior of cells within it. The appropriate parenchymal-cell populations, are the living building units of new tissues. They are elementary in the generation of 3D-tissues that can be incorporated into the defect.
Cell cultivation allows expansion of the number of cells procured from a small tissue biopsy. However, adult cells may be limited in growth potential, may differentiate too easily, or may rapidly deteriorate by senescence. Stem cells could overcome these problems. Stem cells have the unique ability to self-renew, proliferate indefinitely, and create offspring that differentiates into specialized, mature tissues by asymmetric replication. Nevertheless, stem cells, represent a miscellaneous group of cells, with different levels of differentiation potential. From a clinical reconstructive perspective, it is desirable that they are derived from autologous tissues to avoid immune-mediated reactions.
Pluripotent Stem Cells for Use in Autologous Conditions
Induced Pluripotent Stem Cells
In 2006, Takahashi and Yamanaka reported how adult human dermal fibroblasts or other human somatic cells could be directly reprogrammed by the introduction of transcription factors. Induced pluripotent stem cells (iPSCs) possess a differentiation potential equal to human embryonic stem cells. In other words, they are capable of differentiating into tissues of all three germ-cell layers given the correct culture conditions, growth factors, and genetic milieu.
As these cells are derived from adult tissues, there are no ethical issues. Another advantage is the possible autologous source of the stem cells, unlike embryonic stem cells. Nevertheless, iPSCs also carry the risk of teratoma formation and genetic instability. At the moment, these cells cannot be efficiently used in clinical, reconstructive settings.
Multipotent Stem Cells for Use in Autologous Conditions
Multipotent stem cells are capable of differentiating into multiple cell types. Adult stem cells or organ, specific stem cells can be isolated from several tissue sources, including bone marrow, peripheral blood, skeletal muscle, central nervous system, retina, adipose tissue, and epithelia of skin and digestive tract. Multipotent stem cells from lipoaspirates are extensively investigated and used in a clinical reconstructive context.
These cells preferentially generate differentiated cells of the same lineage as their tissue of origin. However, experiments suggest that adult stem cells from various organs could contribute to the regeneration of dissimilar organs with stem cells crossing germ layers. This process is called transdifferentiation . This has led to the suggestion of a “stem-cell highway”, in which stem cells can transit through the circulation with access to all organs. Homing signals (the “niche” or microenvironment) might influence stem-cell migration to specific sites. Secreted growth and differentiation factors are important extracellular signals that control stem-cell fate. Still, vigilance is essential when using adult multipotent stem cells, since ex vivo culture of mesenchymal stem cells promotes chromosomal instability.
Unipotent Progenitor Cells
Blood Outgrowth Endothelial Cells
Lin et al and Yoder et al characterized late outgrowing endothelial-like colonies of collagen-adherent cells with much higher proliferation capacity than early-outgrowth endothelial progenitor cells (EPCs). These cells are named blood-outgrowth endothelial cells (BOECs). BOECs stimulate angiogenesis actively by incorporation into the host vessel, and passively by secretion of proangiogenic growth factors. BOECs can be isolated from umbilical cord blood or adult blood, with the former showing higher proliferation potential but also susceptibility to karyotypic aberrations.
In the future, these stem cells, derived from autologous tissues, may play a growing role in cell-based tissue-engineering techniques. Meticulous identification and purification will be essential to ensure predictability and safety in clinical protocols.
Tissue Engineering-Based Clinical Applications
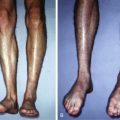
In 1997, Cao and colleagues published a report of an athymic mouse with a human ear grown on its back. This ear consisted of an ear-shaped cartilage structure, implanted subcutaneously. The group seeded the synthetic, biodegradable mold with bovine chondrocytes. This report was viewed worldwide and ignited the fascination for tissue engineering.
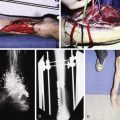
Walles performed a Medline search using the term “tissue engineering”, which provided 37,000 hits. The majority were proof-of-principle studies, only a few resulted in clinical protocols. For instance, in the context of auricular cartilaginous reconstruction, more than 20 studies have been published in the past decade using cell-based techniques and matrices for ear reconstruction. Clinical engineering however, has only been reported in singular cases. Major shortcomings are resorption of the biological scaffold and collapse of the construct in an in-vivo clinical setting.
The progression from bench to bed in other tissue-engineering protocols has also been hampered by many difficulties, mostly related to insufficient vascularization and loss of tissue strength and coherence.
In this section, clinical translational tissue-engineering applications for skin, urethra and bladder, bone, trachea and the cardiovascular system will be discussed in more detail.
Skin
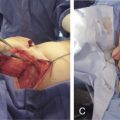
Reconstitution of skin wounds deeper than the basement membrane requires a restoration of all components to restore thickness, texture, and elasticity, while providing biomechanical resistance to aggressors. Skin substitutes were developed for clinical use to treat extensive wounds such as third-degree burns.
Distinct features categorize skin substitutes or skin equivalents:
Natural polymers used in skin tissue-engineering include chitosan, fibrin, gelatin, and hyaluronic acid. Chitosan, a polysaccharide acting as an analog to glycosaminoglycan, is biodegradable and biocompatible. It is used as a hemostatic agent and possesses antibacterial properties. Hyaluronic acid shows excellent biocompatibility. Disadvantages of natural polymers include low mechanical strength, shrinkage and contraction, rapid biodegradability and risk of immunological rejection.
Synthetic materials include nylon, polyglycolic acid and polylactic acid. Due to the ease of fabrication, synthetic polymers such as nylon (Biobrane ® , TransCyte ® ) and polyglycolic acid/polylactic acid (Dermagraft ® ) are less expensive. However, synthetic materials possess limited tissue compatibility, cellular recognition, and incorporation. These substitutes show their importance mainly in combination with natural polymers.
Skin substitutes can also be described according to cell content; cell-free ( acellular ) versus cell-containing ( cellular ) substitutes:
- •
Acellular products contain matrix only, based on natural or synthetic materials. These products serve as a template for dermal reconstitution, allowing migration of host cells during wound healing.
- •
Cellular products contain living cells such as keratinocytes and/or fibroblasts with or without matrix.
These scaffolds could be applied for short periods to stimulate autologous healing, serving as biological dressings . Cell-free biodegradable scaffolds may stimulate colonization by autologous cells in the wound environment. Cell-containing skin substitutes may provide immediate functional skin replacement. However, in a clinical context, allogenic cells will be rejected.
Skin substitutes can also be divided according to the skin layer they represent: epidermal, dermal or combined ( Table 6.1 ).
| Materials | Composition | Thickness | Brand | Indication |
|---|---|---|---|---|
| Biological | ||||
| Alloderm | Acellular human dermis | 0.79–3.3 mm | Lifecell Corporation, NJ, USA | Burns, soft tissue defects |
| Allomax | Acellular human dermis | 0.8–1.8 mm | Bard Davol, RI, USA | Soft tissue defects |
| DermaMatrix | Acellular human dermis | 0.2–1.7 mm | Synthes, PA, USA | Soft tissue defects |
| Glyaderm | Acellular human dermis | 0.2–0.6 mm | Beverwijck, The Netherlands | Full thickness wounds |
| Graftjacket | Acellular human dermis | 1; 1.4; 2 mm | Wright Medical Technology, TN, USA | Soft tissue defects |
| Oasis | Porcine small intestine submucosa acellular collagen | 0.15–0.3 mm | Healthpoint Ltd, TX, USA | Burns, chronic wounds |
| Permacol | Acellular porcine dermis | 0.4 or 1.5 mm | Covedien, OH, USA | Full thickness wounds |
| Strattice | Acellular porcine dermis | 1.5–2 mm | LifeCell, NJ, USA | Soft tissue reconstruction |
| SurgiMend | Acellular bovine dermis | 0.4–1.54 mm | TEI Biosciences, MA, USA | Soft tissue reconstruction |
| Tiscover | Acellular human dermis autologous FB | 1–2 mm | A-SKIN, BV, The Netherlands | Chronic wounds |
| Xenoderm | Acellular porcine dermis | 0.3 mm | MBP Neustadt, Germany | Full thickness wounds |
| Composite | ||||
| Apligraf | Allogenic neonatal FB | 0.4 mm | Organogenesis, MA, USA | Donor sites, EB |
| Allogenic neonatal KC | 0.75 mm | |||
| Dermagraft | Mesh + allogenic FB | 0.19 mm | BioHealing, CA, USA | Wounds, diabetic ulcers |
| Hyalomatrix | Hyaluron-based scaffolds with autologous FB | 1.2 mm | Fidia | Burns, chronic wounds |
| Integra | Human collagen I with GAG and silicone top | 1.3 mm | Integra Life Sciences, NJ, USA | Burns, chronic wounds |
| Matriderm | Bovine collagen I, elastin | 1 and 2 mm | Care AG, Germany | Burns, chronic wounds |
| OrCel previously CCS | Collagen I sponge gel allogenic FB and KC | 1 mm | Ortec International, NY, USA | Chronic wounds, donor sites |
| Renoskin | Bovine collagen I and GAG | 1.5–2.5 mm | Perouse Plastie, France | Burns, defects |
| Terudermis | Calf collagen polyester mesh ± silicone top | 4 types | Olympus Terumo Biomaterials, Japan | Burns, mucosal defects |
| TransCyte | Collagen with neonatal FB nylon mesh + silicone top | 1.2 mm | Sciences, Inc, CA, USA | Burns |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree