Impairment
Measurement
Impairment
Measurement
Movement restriction
Goniometer
Impaired sensation/nerve injury
Monofilaments
Inclinometer Wire tracing
Two-point discrimination
Stereognosis
Tape measure
Moberg pickup test
Pollexograph
Ten test
Ninhydrin sweat test
Weakness
Dynamometer
Prehension
Box and blocks
Quantified muscle testing (QMT)
Nine hole peg test
Manual muscle testing (MMT)
Functional dexterity test
Edema
Volumetry
Test of in-hand manipulation
Tape measure
Pain
Visual analog scale
Wong-Baker faces
Face, legs, activity, cry, consolability scale
Table 5.2
Impairment ratings
Impairment | Tool | Normative data | ||||
|---|---|---|---|---|---|---|
First author and date | Citation | Measure | N | Age (years) | ||
Movement restriction | Goniometer | Soucie (2011) | [9] | Shoulder, elbow, and forearm PROM | 200 | 2–19 |
Goniometer | Barad (2013) | [10] | Elbow PROM | 1,361 | 1–16 | |
Pollexograph | de Kraker (2009) | [11] | Thumb abduction | 100 | 4–12 | |
Distance measurement | ||||||
Weakness | Grippit | Häger-Ross (2002) | [12] | Grip strength | 530 | 4–16 |
Jamar dynamometer | Bowman (1984) | [13] | Grip strength | 153 | 6–9 | |
Jamar dynamometer | Fullwood (1986) | [14] | Grip strength | 214 | 5–12 | |
Jamar dynamometer | De Smet (2001) | [15] | Grip strength | 487 | 5–15 | |
Jamar dynamometer | Holm (2008) | [16] | Grip strength | 376 | 7–12 | |
Jamar dynamometer | Ploegmakers (2013) | [17] | Grip strength | 2,241 | 4–15 | |
Jamar dynamometer | Mathiowetz (1986) | [18] | Grip strength | 571 | 6–19 | |
B & L pinch gauge | Pinch strength | |||||
Preston pinch gauge | Lee-Valkov (2003) | [19] | Pinch strength | 17 | 3–5 | |
Jamar dynamometer | Grip strength | |||||
Preston pinch gauge | Ager (1984) | [20] | Pinch strength | 474 | 2–13 | |
Jamar dynamometer | Grip strength | |||||
B & L pinch gauge | Surrey et al. (2001) | [21] | Pinch strength | 414 | 5–12 | |
Preston pinch gauge | De Smet (2006) | [22] | Pinch strength | 262 | 5–12 | |
Impaired sensation | Two-point discrimination | Cope (1992) | [23] | Discriminative touch | 112 | 2–13 |
Prehension | Box and block test of manual dexterity | Mathiowetz (1985) | [24] | Manual dexterity | 471 | 6–19 |
Functional dexterity test | Gogola (2013) | [25] | Manual dexterity | 175 | 3–17 | |
Nine hole Peg test | Smith (2000) | [26] | Manual dexterity | 826 | 5–10 | |
Poole (2005) | [27] | Manual dexterity | 409 | 4–19 | ||
Purdue Pegboard | Wilson (1982) | [28] | Manual dexterity | 206 | 2.6–5.1 | |
Skerik et al. [29] described a standardized process of assessment for all children with CAUE including analysis of available patterns of pinch and grip, observation of preferred patterns of usage, and measurement of ROM, pinch strength, and hand size. To measure outcomes following index finger pollicization, Percival et al. [30] developed a battery of seven tests for which a maximum score is 22. Included in this battery is a measure or observation of tip pinch and pulp pinch strength; opposition of the thumb to the middle, ring and small finger; grasping of two balls of different size, active movement of the thumb at three joints; two-point discrimination; and cosmesis (length and position of the thumb). Scores are characterized as excellent (>20) good (16–19), fair (12–15), or poor (<12). Ho and Clarke [31] conducted a systematic review of studies published between 1966 and 2003 aimed at evaluating outcomes following pollicization of the index finger or centralization for radial longitudinal deficiently. Of the ten studies reviewed, six attempted to measure ADL or functional use of the hand, but only one did so using a standardized instrument.
Since Ho and Clarke’s review [31], other outcome studies have been published in which standardized assessment tools were employed. Buffart et al. [32] set out to identify appropriate assessment tools for use with children with transverse or longitudinal reduction deficiency using as criteria inclusion of bimanual tasks, measures of quality of movement, and appealing tasks. These researchers recommended the AHA, Unilateral Below Elbow Test (UBET), ABILHAND-Kids, and PUFI. In a follow-up study [33] the AHA, UBET, ABILHAND-Kids, and PUFI were administered to 20 children with RLD, aged 4–12 years. The AHA and PUFI were deemed most valid for children with RLD, due to the relationships found with type of RLD (r = −0.82 and −0.64, respectively), functional hand grips (r = 0.58 and 0.46, respectively), and the therapist’s global assessment of hand function (r = 0.85 and 0.63, respectively). Kaplan and Jones [34] used the Pediatric Outcomes Data Collection Instrument (PODCI) to determine outcomes following microsurgical toe transfers for thumb reconstruction. Table 5.3 presents performance-based assessments specifically designed for children with CAUE, children with normal use of one hand only, or children with disability but no specific diagnostic population.
Table 5.3
Performance-based assessment
Assessment tool | Target populations | Target age (years) | Studies describing psychometrics | ||
|---|---|---|---|---|---|
Validity | Reliability | Responsiveness | |||
ABILHAND-Kids | Children with cerebral palsy | 6–15 | [33] | – | |
AHA | Children with typical function in one hand only | 1.6–12.8 | [36] | [37] | |
CHEQ | Children with typical function in one hand only | 6–18 | [39] | – | – |
PedsQL | Children with acute or chronic illness | 2–18 | [40] | [40] | [41] |
PEDI | Children with physical disability | 6 months–7.5 | [45] | – | |
PODCI | Children with orthopedic conditions | 0–18 | [46] | ||
PUFI | Children who use an upper extremity prosthesis | 3–18 | – | ||
UBET | Children with transverse reduction deficiency | 2–21 | [33] | – | |
Assessments that attempt to measure satisfaction with or perceptions of activity performance and participation should include the child with CAUE, but in some cases the caregiver may need to serve as proxy. Researchers have studied the extent to which parents and children agree on satisfaction with or perception of activity performance and participation. Netscher et al. [53] examined ability to participate in activities following index finger pollicization. In addition to measuring impairment level and ability to participate in simulated tasks reflecting participation in a larger activity, researchers administered a non-validated novel questionnaire to nine children and their parents to determine perceptions of appearance, social participation, and performance skills. The mean score for children was 22, with 12 being the best score and 60 the worst. Although parents tended to assess their children’s skills as slightly better than the children did of themselves, there was no statistically significant difference between parents’ and children’s scores, suggesting that parents may serve well as proxy. Ardon et al. [54] found similar results when parents and their children with CAUE separately completed the Pediatric Quality of Life Inventory (PedsQL). No statistically significant differences were observed for total score and the five domains (physical health, emotional functions, social functioning, school functioning, psychosocial health). The researchers noted analysis of individual scores showed children and parents tended to disagree and the variables that influenced disagreement included number of affected digits and bilateral involvement [54]. Similarly, in a large multi-center study significant differences were found between parents and their children with congenital below elbow deficiency (CBED) for upper extremity physical function (p < 0.001), pain/comfort (p < 0.05), and social functioning (p < 0.001) using the PODCI and PedsQL [55]. In summary, use of a parent as proxy should be limited; effort to elicit children’s participation is desirable.
Interventions to Address Impairments
Current estimates of the rate of congenital upper limb differences include 1 in 506 live births [56], 5.25 in 10,000 live births [57], and 21.5 per 10,000 live births [58]. In two reports of the incidence of all congenital limb reductions, 75–81 % involved the upper extremities [59, 60]. Within these estimates, not all children with CAUE will require surgical intervention and subsequent rehabilitation. When indicated, rehabilitation efforts may initially emphasize interventions to address impairment with simultaneous or subsequent attention to participation in activity. The studies presented in the following sections are not specific to children.
Edema
Edema management is often addressed via rest, ice, compression, and elevation. Postoperative dressings and casting provide rest and compression yet preclude icing. Chronic edema that persists after removal of postoperative immobilization may be treated with gentle compression. Younger children may not be amenable to elevation and the efficacy of elevation following hand surgery is unclear. In two prospective and randomized comparison trials, no statistically significant differences were noted in those who the limb and those who did not for adults undergoing Dupuytren’s release [61] or carpal tunnel decompression [62]. Gentle compression may be achieved with self-adhering wrap [63]; however, only one case report of an adult with burn injury could be located to support its use [64].
Scar
In addition to being cosmetically unappealing, postoperative scar may lead to motion restriction, pain, and puritis. These impairments may in turn reduce function and participation in activities. Intervention should first concentrate on prevention of hypertrophic scars, but when hypertrophic scars are present, efforts should be made to reduce the extent of the existing scar. Scars from surgical incisions may respond well to treatment including massage, pressure, topical application of a gel product, and reduction of tension on scar.
Massage
Shin and Bordeaux [65] conducted a systematic review of studies investigating the effectiveness of scar massage regimes for scars due to burn and trauma, and included four randomized controlled studies, three prospective controlled studies, one prospective study, and two case reports. Across ten reports, the total number of subjects was 220 with 144 receiving scar massage. The standardized outcome measures included the Observer Scar Assessment Scale and the Vancouver Scar Scale (VSS), as well as subjective assessments of scar thickness, perfusion, color, pain, and itching. For patients who had surgical scars and received massage, 90 % improved. Foo and Tristani-Firouzi [66] recommend that postsurgical scar massage commence during the proliferative phase, 2–3 times per day, for 3–5 min, for 3–4 months.
Pressure
Pressure application may be applied using self-adherent wraps, neoprene splints, tubular elastic, and custom fit pressure garments. Pressure inhibits fibroblastic activity [66], via ischemia and hypoxia resulting in degeneration of fibroblasts and slowed synthesis of collagen [67]. Despite a long history of inclusion of pressure in the treatment of scar, definitive evidence regarding its efficacy is lacking.
In a meta-analysis of six published randomized controlled trials and one unpublished trial examining the benefit of pressure therapy for burn scar, researchers found no difference between scars treated with pressure therapy and controls [68]. More recently, a randomized controlled study of treatment of burn scar demonstrated significant improvement on the VSS using pressure therapy alone (p < 0.001), but also found significant improvement with combined pressure therapy and application of silicone gel sheeting (p = 0.001), and combined pressure therapy and silicone spray (p < 0.001). Patients with two similar scars from split-thickness grafts were randomized into either a silicone gel sheeting group or silicone spray group, but all used pressure therapy. Differences between the groups were not significant [69]. Widgerow [83] suggests pressure garments are more appropriate for widespread scar seen in burn injury; however, in maintenance of tape or silicone gel sheeting on the hand of a young child can be challenging. Use of a pressure garment may discourage self-removal of treatment modalities held in place by the garment, including the garment itself. In a laboratory study of fibroblastic activity under pressure, researchers showed pressure application may be applied at higher levels over shorter periods of time or at lower levels for longer periods of time to reduce fibroblastic proliferation [66].
If using self-adherent wrap, care in wrapping and maintained supervision are indicated to avoid a tourniquet-like effect due to lifting, slippage, and rolling [72]. Use of neoprene patches or orthoses for at least 8 h per day was retrospectively studied in a small population of children and young adults with burn scar (n = 8 participants, 12 scars). Duration of treatment ranged from 1 to 11 months. Scars were evaluated pre and post treatment and differences for mean VSS was significantly lower after treatment (p = 0.0001). This study is useful to therapists working with children because neoprene splints are often used long term across several diagnostic groups for limb positioning and so could also serve to manage scar [73].
Silicone
Silicone gel may serve to prevent hypertrophic scars and improve characteristics of existing hypertrophic scar. In a narrative review of eight RCTs and an analysis of 27 trials, the International Advisory Panel on Scar Management concluded that use of silicone gel sheeting is “safe and effective”; however, the panel distinguished adhesive silicone gel sheeting from other adhesive gels, liquid silicone, and nonadhesive silicone gel sheeting [74]. O’Brien and Pandit [75] conducted a meta-analysis to determine the effectiveness of silicone dressings to prevent hypertrophic or keloid scarring in people with newly healed wounds and to treat established keloid or hypertrophic scars. The study included randomized or quasi-randomized controlled trials, and controlled clinical trials comparing silicone dressings to other nonsurgical treatment, no treatment or placebo. Included trials compared adhesive silicone dressings with control; non-silicone dressings; silicone gel plates with added vitamin E; laser therapy; triamcinolone acetonide injection, and nonadhesive silicone dressings. Scar quality was determined by blood flow, color change, hyperpigmentation, thickness, and shape. Studies that set out to determine effectiveness of silicone to treat existing scars measured change in scar size and did so using a ruler, taking an impression, or via ultrasound. Across 15 studies, 615 people between 2 and 81 years-of-age were included. Compared with no treatment silicone reduced the incidence of hypertrophic scar (RR 0.46, 95 % CI 0.21–0.98). For established keloid and hypertrophic scar SD significantly reduced scar thickness (RR −1.99, 95 % CI −2.13 to −1.85) and improved color (RR 3.05, 95 % CI 1.57–5.96). Silicone dressings produced superior results compared to controls in two trials, no difference was found in two trials, and the control group fared better in one trial. This study included clinical trials of varied rigor and most were subject to bias thus there is weak evidence for use of silicone dressing to prevent or improve scars. An update to this review was published in 2013; five new studies were included but the same conclusion was offered [76].
The proposed mechanism of action of silicone gel is thought to be hydration and occlusion [77], though non-silicone gels may be equally effective as silicone. In a prospective, randomized study patients (n = 24) with existing hypertrophic or keloid scars (n = 41) present for longer than 3 months, including incisional scars, were randomly assigned to one of three groups: treatment with silicone gel (n = 16 scars), treatment with non-silicone gel (n = 14 scars), or control (n = 11 scars). Treatment was applied 24 h per day for 4.5 months. No statistically significant differences were found between SD and NSD groups for color, size, induration, and symptoms, although significant differences were noted when SD and NSD were compared to controls for color, size, induration, and scar pliability [78].
Tape
Tension on scar is believed to stimulate collagen production due to mechanosensitive fibroblasts [71, 79, 80]. Tape applied to scars may reduce tension and prove effective in preventing hypertrophic scar [81, 82]. Porous tape should be applied longitudinal to and directly over the scar to adequately provide support and reduce tension [83]. When scars cross joints, use of an orthosis may help to reduce tension on scar.
Motion Restriction
Clinicians utilize AROM, active assisted ROM, passive range of motion (PROM), joint mobilization and orthoses to achieve greater range of movement. Michlovitz et al. [84] conducted a systematic review of interventions to promote joint motion in the upper extremity. The review included 26 studies that examined interventions in adults, but excluded children and congenital hand differences. In their summary, the researchers noted moderate evidence for the use of orthoses or casts and passive exercise to increase ROM after joint trauma or immobilization. Following this study, Glasgow et al. [85] published a narrative review to develop a set of recommendations for mobilizing the stiff hand. After a review of 29 studies of varying levels, these authors recommended active and active assisted exercise during all stages of tissue healing, passive exercise during the proliferative and remodeling phases, and joint mobilization during the remodeling phase. Orthoses for management of stiffness via mobilization were recommended during the proliferative and remodeling phases.
When the purpose of an orthosis is to increase motion, orthosis prescription must consider tissue compliance and the length of time the restriction has been present. Therapists must decide on orthosis type (including no orthosis), wear time (hours per day and duration), and the magnitude of force to apply. Flowers [86] offered a hierarchy for decision-making when treating stiff joints using a modified Week’s test [87]. After pre-conditioning, those whose PROM measures change by 20° may not need a splint; by 15° may require a static splint with no overpressure; by 10° may require a dynamic splint; and by 5° or less may required a static progressive splint with overpressure. This decision-making process may prove useful with older children; but may not be feasible with infants and toddlers due to required exposure to thermotherapy.
Consensus on wear time of an orthosis to resolve motion restrictions is lacking, although many studies provide guidance. Flowers and LaStayo [88] executed a study to determine if duration of orthosis use impacted outcomes for stiff joints. Patients (n = 15) with 20 PIP flexion contractures between 15° and 60° were randomly assigned to continuous casting for 6 days then 3 days or 3 days then 6 days. There was a statistically significant difference (p < 0.005) in gains made with 6 days of wear achieving a mean increase of 5.3° and 3 days of wear achieving 3°. Glascow et al. [89] prospectively investigated optimal hours of daily orthoses wear in 43 subjects with joint restrictions in the hand following trauma. Subjects with similar levels of stiffness—as determined via torque range of motion (TROM)—were randomly allocated to a <6 h or 6–12 h per day group. There was a statistically significant difference between the groups, with better TROM observed in the 6- to 12-h group. It is not clear if increasing time more than 12 h provides greater benefit. In a follow-up randomized study of 22 patients with PIP joint flexion contractures, no significant differences were found for PROM, AROM, or TROM between 6–12 h of wear and 12–16 h of orthosis wear after 8 weeks of treatment [90].
Interventions to Address Activity Performance and Participation
Assuming a child with CAUE is otherwise typically developing, interventions to improve activity performance or participation may occur immediately following surgery or intermittently—when the child encounters specific problems with activity performance or participation. Following surgery, impairment-based may be emphasized concurrently with interventions to promote activity performance and participation via activity modification or introduction to assistive devices [4]. In a qualitative study investigating perceptions of children 8–20 years of age with unilateral CBED, participants described their own activity performance and participation and generally reported no limitations. Further, these children reported similar levels of participation as peers without CBED. The researchers suggested, for children in this study, perceptions of activity participation might have been limited to actual chosen activities rather than potential chosen activities (activities that may have been chosen if participants had two hands) [5]. In a descriptive study of eight people with ulnar longitudinal deficiency (ULD), age 3–41, adult patients reported no difficulty with self-dressing, washing, toileting, eating, closing and opening dressing fasteners, managing the telephone, typing, or opening containers with screw on caps and parents of children with ULD reported no difficulty with bimanual self-care, play, or school related activity [91].
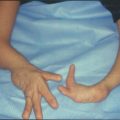
Health professionals should recognize there are multiple strategies to manage limitations in activity performance and participation that may be acceptable to the child with CAUE including using other body parts (Fig. 5.1), activity modification, choosing varying levels of participation, receiving assistance from another, using assistive devices, and prosthetic wear [5]. In the study by de Jong et al. [5] health professionals were less apt to recognize as many strategies as did children and their parents, and identified assistive devices and prosthetics more frequently as potential solutions for success in activity performance and increased participation.


Fig. 5.1
This child with thrompocytopenia absent radii has self-identified strategies for participation in activities
Diagnosis-Specific Intervention
Camptodactyly
Range of Motion Exercise
While orthotic management and surgery are intervention options for camptodactyly [92], ROM exercises may prove beneficial especially for children with an infantile onset of deformity. Rhee et al. [93] retrospectively evaluated the effectiveness of passive stretching to correct flexion deformities in children younger than 3 years with camptodactyly. Records of children with simple camptodactyly who had not received surgery or intervention with an orthosis were included, but those with flexion contractures of less than 10° were excluded. Parents were taught a PROM technique, to be implemented at home, requiring the PIP joint be extended with the wrist and metacarpophalangeal (MCP) joint in extension. Instructions were to complete gentle PROM, while the child was sleeping, 20 or more times per day with a hold time of 5 min. Exercise frequency was reduced to five or ten times per day when near full extension was achieved. Duration of intervention was individualized and poorly defined. The intervention could be realistically applied; however, the burden of applying PROM only when children are sleeping could compromise adherences rates. Pre- and post-intervention measurements, recorded by the same physician, were compared. Across groups, 13 males and 9 females with a mean age of 12 months (range 3–36 months) were included in the study. Digits were further classified into mild deformity (<30°, n = 12 digits), moderate deformity (30–60°, n = 36), and severe deformity (>60°, n = 13) as per goniometric measures. Groups were expected to be different with regard to extent of deformity but no analysis was performed to assure they were similar for age, sex, and dominance. Final PROM for PIP extension was compared to initial measures. Mean change in PROM were as follows: −20° to −1° for the mild group, −39° to −12° for the moderate group, and −75° to −28° for the severe group. Differences from pretest to posttest were significant for all groups: mild (p < 0.001), moderate (p < 0.001), and severe (p < 0.001). Mean time from start to end of intervention (either correction or cessation of change) for the mild group was 5 months, moderate group was 10 months, and severe group 13 months. Researchers found a relationship between degree of flexion contracture at the start of intervention and final measure. No relationship was found between initial flexion contracture, handedness, digit involvement, and number of digits or hands involved. Differences between pretest and posttest AROM values were statistically significant. No statistical analysis was performed to determine clinical significance, however all but two children (in the moderate group) improved and gains were maintained during a prolonged follow-up period (mean of 26 months, range of 12–47 months). The researchers concluded children under three who have camptodactyly should be treated with PROM only and orthoses are not necessary; however, this statement is unfounded since no comparison was made between PROM and use of an orthosis. The researchers recognized the weaknesses of the study including use of retrospective design and absence of a control group. The outcomes cannot be applied to all children with camptodactyly since only children under the age of three with simple syndactyly were studied, and children with syndromic or adolescent onset camptodactyly were not included [93].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree