16 The bilateral pedicled TRAM flap
Synopsis
 The pedicled TRAM flap is still the most popular way of transferring abdominal tissue to the breast.
The pedicled TRAM flap is still the most popular way of transferring abdominal tissue to the breast.
 This technique, introduced by Dr Carl Hartrampf, is consistently successful if it is performed properly.
This technique, introduced by Dr Carl Hartrampf, is consistently successful if it is performed properly.
 The muscle and fascial sparing technique ensures abundant vascularity to the TRAM flap, while allowing secure and complication free closure of the abdominal wall.
The muscle and fascial sparing technique ensures abundant vascularity to the TRAM flap, while allowing secure and complication free closure of the abdominal wall.
 The shaping of the breast using the pedicled TRAM flap is enormously simplified in the immediate reconstructive setting.
The shaping of the breast using the pedicled TRAM flap is enormously simplified in the immediate reconstructive setting.
 We recommend utilizing the pedicled TRAM flap for bilateral reconstruction and the free TRAM flap for unilateral reconstruction.
We recommend utilizing the pedicled TRAM flap for bilateral reconstruction and the free TRAM flap for unilateral reconstruction.
History
In 1980, Dr Carl Hartrampf performed the first TRAM flap for breast reconstruction. In his landmark paper, Hartrampf described a procedure in which tissues that would normally be discarded during an abdominal lipectomy were instead transferred to a mastectomy defect based on the proximal rectus abdominis muscle and the superior epigastric vessels contained within it. The skin and subcutaneous tissue of the abdomen were left in continuity with the anterior rectus sheath and its perforators from the deep epigastric vascular system, on which the flap relied.1 The introduction of this procedure provided an attractive alternative to implant reconstruction and to the other options for autogenous breast reconstruction that either did not provide enough soft tissue (e.g., the latissimus dorsi myocutaneous flap) or involved multiple stages to be completed (e.g., the tubed abdominal flaps).
Basic science/anatomy
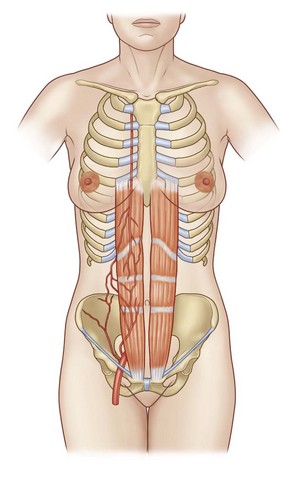
The TRAM flap is based on the blood supply from the perforating vessels through the rectus abdominis muscle and the deep epigastric system (Fig. 16.1). These perforating vessels are supplied primarily as branches from the deep inferior epigastric artery but can also be supplied by the adjacent territory of the superior epigastric artery. The anatomy of the perforating vessels is somewhat variable and may be predominantly along the lateral third of the muscle, in the central third of the muscle, or even in the medial third. The concept of the epigastric artery as the pedicle source was originally described by Mathes.2 The vascular anatomy of the epigastric arteries as they relate to the TRAM flap was described by Moon and Taylor.3 They described three basic arterial schemata. In a type I pattern, a single intramuscular vessel extends from the superior epigastric artery to the deep inferior epigastric artery. The majority of patients in their study exhibited a type II system, in which the inferior epigastric artery branches into two vessels at the arcuate line and communicates with the superior epigastric through anastomosing choke vessels. Patients with a type III pattern exhibited three branches of the inferior epigastric at the arcuate line. These patients, on average, showed an even greater number of anastomoses with the superior epigastric artery. Only 2% of patients showed symmetry between their rectus muscles; most showed different vascular patterns on each side. The epigastric vessels send perforators through the rectus muscle that cross the anterior rectus sheath generally in two rows, one medial and one lateral. The rectus abdominis muscle is a type III muscle, as described by Mathes2 and has, in addition to the superior and inferior epigastric vessels, circulation from branches of the 8th, 9th, 10th, 11th, and 12th intercostals vessels. These vessels enter the rectus muscle after anastomosis with the superior and deep inferior epigastric vessels posterior to the muscle. They penetrate the posterior rectus sheath just medial to the linea semilunaris. The rectus abdominis muscles also derive their motor innervations from the intercostals nerves T8 through T12.
The primary blood supply to the lower rectus muscle is from the deep inferior epigastric artery. Hence, the blood supply with the use of the inferior epigastric artery for the free TRAM flap or perforator flap is substantially richer than the blood supply available from the superior epigastric artery.4
The circulation from either rectus muscle across the lower abdomen is divided into four zones (Fig. 16.2) Zone I is the zone immediately overlying the rectus muscle. Zone II is described as a zone immediately across the midline. Zone III is the ipsilateral zone just lateral to the rectus muscle border, and zone IV is the contralateral zone lateral to the contralateral rectus border. The circulation is best in zone I, somewhat variable in zones II and III, and usually poor in zone IV, particularly with a pedicled TRAM flap. In each of these zones, the blood supply is improved to some degree by use of the perforator flap, free flap, or pedicled flap after a delay procedure.
Patient selection
The aims of breast reconstruction are to form a breast with natural appearance and feel, comfortable and in harmony with the contralateral breast. Additional aims include leaving the abdominal donor site undamaged and, if possible, improved in appearance. In order to accomplish these goals, several factors should be considered. These include the size and shape of the opposite breast, the plans for altering the opposite breast, the nature of the mastectomy defect, the pathologic stage of the breast cancer, the likelihood of postmastectomy irradiation, the general health of the patient, the availability of donor tissue, the lifestyle of the patient, and finally the patient’s goals and expectations. Once the factors are all taken into consideration, the reconstructive surgeon has a wide variety of options from which to choose, including prosthetic techniques, autologous techniques, and combined autologous tissue and implant techniques. In addition, a number of matching procedures are available for the contralateral breast to help with symmetry.5
If a TRAM flap is to be chosen, there are a number of options available. Surgeons generally choose these options based on the presence or absence of risk factors which significantly increase the risk of complications to both the TRAM flap and the donor site. These risk factors are obesity, smoking, diabetes, hypertension, collagen vascular disease, and other significant systemic illnesses (such as pulmonary, renal, or cardiac disease). In terms of circulation, the single-pedicled TRAM flap has its best application in patients who do not have any of these risk factors and who have enough tissue in the lower abdomen for reconstruction of the breast to the desired shape and size. Generally speaking, with the unilateral pedicled TRAM, approximately 50% of the lower abdominal tissue typically removed in an abdominoplasty procedure will be available in most patients without risk factors to provide tissue for breast reconstruction. Patients who have risk factors or who need more tissue than 50% of the lower abdomen need a TRAM flap with a more robust circulation. This moves them into the category of patients who would benefit from a free TRAM flap, a TRAM with a preliminary delay procedure of the deep inferior epigastric vessels, or a double pedicled TRAM flap.6,7 There is also the option of performing a single-pedicled TRAM flap with the addition of a prosthesis in patients who have no risk factors but who need more volume than the TRAM flap alone will provide. The deep inferior epigastric artery perforator (DIEP) flap is a free flap based on one, two, or more perforating vessels, but it does not carry the whole, most, or, in some circumstances, any of the lower rectus abdominis muscle. This procedure provides an enhanced circulation compared with a single-pedicled TRAM flap, but it is most likely not as hardy or as simple to harvest as a free TRAM flap, which carries most of the perforators and a small corresponding segment of rectus abdominis muscle.8
Treatment/surgical technique
Operative procedure
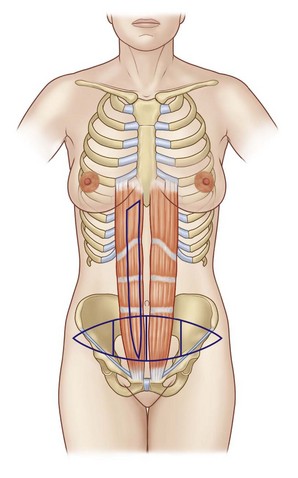
The abdominal flap is designed at this time. The lower transverse incision is located two fingerbreadths above the pubic symphysis. The upper incision is usually just at the upper border of the umbilicus. The lower incision extends out laterally and crosses over the anterior superior iliac spine and angles cephalad. The upper incision is essentially level extending laterally until it meets the rising lower incision. The suture technique is useful for ensuring that each line is equal (Fig. 16.3).
Hints and tips
• If the upper incision is straight across, the resulting scar will curve upward. If the upper incision is curved down laterally the resulting scar will be straight across.
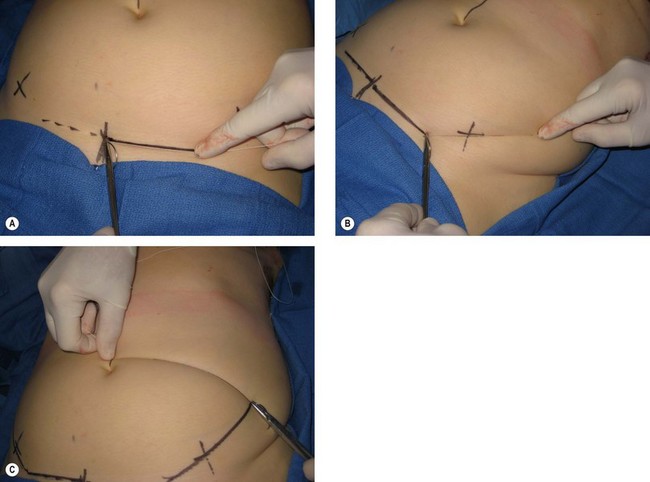
• The inframammary lines drawn preoperatively are reinforced and the tunnel positions on the right and the left are also drawn in place.
These tunnel positions are at the 5- and 7-o’clock positions on the right and the left respectively (Fig. 16.4)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree