Key Words
tinea, ringworm, candidiasis, Candida balanitis, vulvovaginitis, vaginosis, Pityrosporum folliculitis, moniliasis, pitted keratolysis, intertrigo, oral candidiasis
Dermatophyte Fungal Infections
These dermatophytes include a group of fungi (ringworm) that under most conditions have the ability to infect and survive only on dead keratin (that is, the top layer of the skin – the stratum corneum or keratin layer), the hair, and the nails. They cannot survive on mucosal surfaces such as the mouth or vagina where the keratin layer does not form. Very rarely, dermatophytes undergo deep local invasion and multivisceral dissemination in the immunosuppressed host. Dermatophytes are responsible for the vast majority of skin, nail, and hair fungal infections. Lesions vary in presentation and closely resemble other diseases; therefore laboratory confirmation is often required. There is evidence that genetic susceptibility may predispose a patient to dermatophyte infection. Studies show that although several blood-related members of a family may share similar manifestations of disease, spouses, despite prolonged exposure, do not become infected. Patients with chronic dermatophytosis have a relatively specific defect in delayed hypersensitivity to Trichophyton, but their cell-mediated responses to other antigens are somewhat depressed. There also is a greater frequency of atopy in chronically infected patients.
Classification.
Dermatophytes are classified in several ways. The ringworm fungi belong to three genera: Microsporum, Trichophyton, and Epidermophyton. There are several species of Microsporum and Trichophyton and one species of Epidermophyton .
Place of Origin.
The anthropophilic dermatophytes grow only on human skin, hair, or nails. The zoophilic varieties originate from animals but may infect humans. Geophilic dermatophytes live in soil but may infect humans.
Types of Inflammation.
The inflammatory response to dermatophytes varies. In general, zoophilic and geophilic dermatophytes elicit a brisk inflammatory response on skin and in hair follicles. The inflammatory response to anthropophilic fungi is usually mild.
Types of Hair Invasion.
Some species are able to infect the hair shaft. Microscopic examination of infected hairs shows fungal spores and hyphae either inside the hair shaft or both inside and on the surface. The endothrix pattern consists of fungal hyphae inside the hair shaft, whereas the ectothrix pattern consists of fungal hyphae inside and on the surface of the hair shaft.
Spores of fungi are either large or small. The type of hair invasion is further classified as large- or small-spored ectothrix or large-spored endothrix.
Clinical Classification.
Tinea means fungal infection. Clinically, dermatophyte infections are classified by body region. The dermatophytes, or ringworm fungi, produce a variety of disease patterns that vary with the location and species. Learning the numerous patterns of disease produced by each species is complicated and unnecessary because all dermatophytes respond to the same topical and oral agents. It is important to be familiar with the general patterns of inflammation in different body regions and to be able to interpret accurately a potassium hydroxide (KOH) wet mount preparation of scale, hair, or nails. Species identification by culture is necessary only for scalp infections, inflammatory skin infections, and some nail infections.
The Active Border.
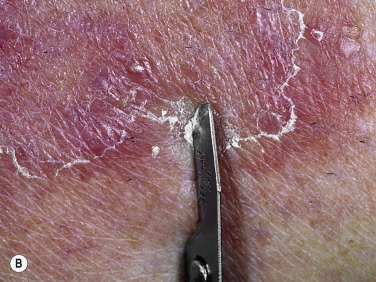
One very characteristic pattern of inflammation is the active border of infection. The highest numbers of hyphae are located in the active border, and this is the best area to obtain a sample for a KOH examination. Typically the active border is scaly, red, and slightly elevated ( Fig. 13.1 ). Vesicles appear at the active border when inflammation is intense ( Fig. 13.2 ). This pattern is present in all locations except the palms and soles.
Diagnosis
Potassium Hydroxide Wet Mount Preparation.
The single most important test for the diagnosis of dermatophyte infection is direct visualization under the microscope of the branching hyphae in keratinized material.
Sampling Scale.
Scale is obtained by holding a no. 15 surgical blade perpendicular to the skin surface and smoothly but firmly drawing the blade with several short strokes against the scale. If an active border is present, the blade is drawn along the border at right angles to the fringe of the scale. If the blade is drawn from the center of the lesion out and parallel to the active border, some normal scale may also be included.
Wet Mount Preparation.
The small fragments of scale are placed on a microscope slide and gently separated, and a coverslip is applied. Potassium hydroxide (5% to 20% solution) is applied with a toothpick or eye dropper to the edge of the coverslip and allowed to run under via capillary action. The preparation is gently heated under a low flame (heating should not be used with KOH solutions with dimethyl sulfoxide [DMSO]) and then pressed to facilitate separation of the epithelial cells and fungal hyphae. Potassium hydroxide dissolves material that fuses cells but does not distort the epithelial cells or fungi. Lowering the condenser of the microscope and dimming the light enhance contrast, making hyphae easier to identify.
Nail plate keratin is thick and difficult to digest. The nail plate can be adequately softened by leaving the fragments along with several drops of KOH in a watch glass covered with a Petri dish for 24 hours. Hair specimens require no special preparation or digestion and can be examined immediately.
Microscopy.
The preparation is studied carefully by scanning the entire area under the coverslip at low power. The presence of hyphae should be confirmed by examination with the ×40 objective. Slight back-and-forth rotation of the focusing knob aids visualization of the entire segment of the hyphae, which may be at different depths. It is not uncommon to find one small fragment of scale containing many hyphae and the rest of the preparation free of hyphae. The entire preparation should be studied carefully.
Interpretation.
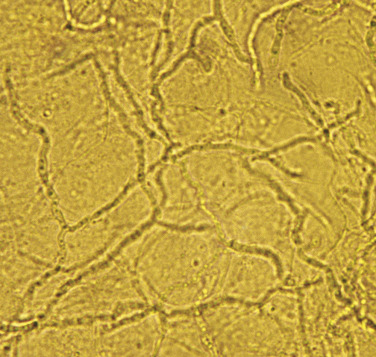
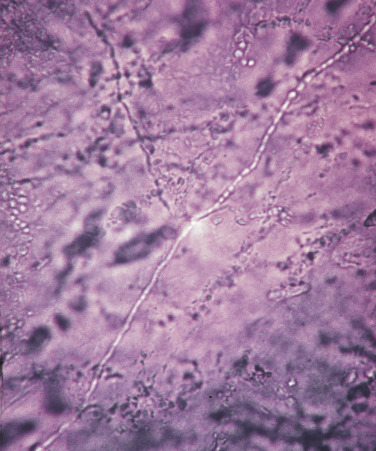
The interpretation of KOH wet mounts takes experience. Dermatophytes appear as translucent, branching, rod-shaped filaments (hyphae) of uniform width, with lines of separation (septa) spanning the width and appearing at irregular intervals ( Figs. 13.3 and 13.4 ). The uniform width and characteristic bending and branching distinguish hyphae from hair and other debris. Hair tapers at the tip. Lines that intersect across cell walls at different planes of the scale are viewed using the fine adjustment knob of the microscope. Some hyphae contain a single-file line of bubbles in their cytoplasm. Hyphae may break into round or polygonal fragments that look like spores. Hyphae may be seen in combination with scale or floating free in the KOH.
Artifact.
Confusion may arise with the so-called mosaic artifact produced by lipid droplets appearing in a single-file line between cells, especially from specimens taken from the palms and soles ( Fig. 13.5 ). These lipid droplets disappear when the cells are separated further by additional heating (KOH without DMSO) and pressure. Although spores and branching hyphae as well as short, nonbranching hyphae are seen in superficial Candida infections and tinea versicolor, only branching hyphae are seen in dermatophyte infections. Longitudinal, rod-shaped KOH crystals that simulate hyphae may appear if the wet mount is heated excessively.
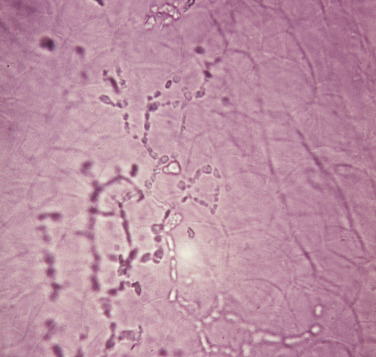
Special Stains.
Hyphae may be difficult to find in a KOH wet mount because of artifacts. Chlorazol black E (an acidic triazo dye, available in a DMSO 10% and 5% KOH solution) may be added to stain the chitin in the hyphae. Other stains such as Swartz-Lamkins fungal stain, or Parker blue ink clearly stain hyphae, rendering them visible under low power. The specialized stains are available from Delasco ( www.delasco.com ).
Culture.
In most cases it is not necessary to know the species of dermatophyte infecting skin because the same oral and topical agents are active against all of them. Fungal culture is necessary for hair and nail fungal infections. Scalp hair infections in children may originate from an animal that carries a typical species of dermatophyte. The animal can then be traced and treated or destroyed to prevent further infection of other humans. Nail plate, especially of the toenails, may be infected with nondermatophytes, such as the saprophytic mold Scopulariopsis, which do not respond to treatment. Identification of the genus of fungus responsible for nail plate infection is therefore necessary before embarking on a long course of treatment.
Cotton Swab Technique for Culture.
A sterile cotton swab that is moistened with sterile water or agar from an agar plate and rubbed vigorously over the lesion produces results comparable to those obtained by scraping with a scalpel blade. A light sweep over the lesion does not collect sufficient material; therefore the swab must be rubbed vigorously over the active part of the lesion and then over the surface of the agar. The swab is useful in areas that are difficult to scrape, such as the scalp, eyelids, ears, nose, and between the toes. The sterile swab is less threatening than a blade, and it is safer in situations in which the sudden movement of a young patient could lead to a painful stab or cut.
Culture Media for Tinea.
Dermatophytes are aerobic and grow on the surface of media. The three types of culture media used most often for isolation and identification are dermatophyte test medium (DTM), mycobiotic agar, and Sabouraud’s dextrose agar. Many hospital laboratories lack the experience to interpret fungal cultures and instead send them to outside laboratories for analysis. Material to be cultured can be sent directly to a laboratory because, unlike many bacteria, fungi remain viable for days in scale and hair without being inoculated onto media. Alternatively, many hospitals and individual practitioners now rely on DTM for faster but slightly less accurate results.
DTM is a commercially available medium supplied in vials that are ready for direct inoculation. The yellow medium, which contains the indicator phenol red, turns pink in the presence of the alkaline metabolic products of dermatophytes in approximately 6 or 7 days but remains yellow in the presence of the acid metabolic products of nonpathogenic fungi. It must be discarded after 2 weeks because saprophytes can induce a similar color change after the 2-week period. Species identification is possible with DTM but is more accurately determined with mycobiotic agar and Sabouraud’s agar because the dye in DTM may interfere with interpretation.
DTM and Onychomycosis.
DTM can be used to confirm dermatophyte infections in patients with presumed onychomycosis. Diagnosis of onychomycosis requires confirmation of dermatophyte infection only, not identification of genus and species. DTM fulfills this requirement and has a diagnostic yield comparable to that of a central laboratory culture. This allows diagnosis in the office and should fulfill the requirement when health care insurers demand confirmation of toenail infection before approving the use of oral antifungals.
Mycobiotic agar is a modification of Sabouraud’s medium that contains cycloheximide and chloramphenicol to prevent the growth of bacteria and saprophytic fungi; the dextrose content of Mycosel agar has been lowered and the pH has been raised to allow for better growth of dermatophytes.
Sabouraud’s agar, which does not contain antibiotics, allows the growth of most fungi, including nondermatophytes. This may be useful for nail infections because the detection of nondermatophytes is desirable in nail infections; but the more selective mycobiotic agar is best for evaluation of hair tinea because only dermatophytes cause hair tinea. Cultures usually become positive in 1 to 2 weeks.
Culture Media for Yeast.
Yeast may be isolated on plates obtained from the hospital laboratory. Acu-Nickerson is a commercially available medium in a slant for use in the isolation and identification of Candida species.
Wood’s Light Examination.
Light rays with a wavelength greater than 365 nm are produced when ultraviolet light is projected through a Wood’s filter. Hair, but not the skin of the scalp, fluoresces with a blue-green color if infected with Microsporum canis or Microsporum audouinii . The rarer Trichophyton schoenleinii produces a paler green fluorescence of infected hair; no other dermatophytes that infect hair produce fluorescence. Fungal infections of the skin do not fluoresce, except for tinea versicolor, which produces a pale white-yellow fluorescence. Erythrasma – a noninflammatory, pale brown, scaly eruption of the toe webs, groin, and axillae caused by the bacteria Corynebacterium minutissimum – shows a brilliant coral-red fluorescence with the Wood’s light. Wood’s light examination should be performed in a dark room with a high-intensity instrument. The fluorescence of hair may be caused by tryptophan metabolites.
Tinea
Clinically, dermatophyte infections have traditionally been classified by body region. Tinea means fungus infection. The term tinea capitis, for example, indicates “dermatophyte infection of the scalp.”
Tinea of the Foot
The feet are the most common area infected by dermatophytes (tinea pedis, or “athlete’s foot”). Shoes promote warmth and sweating, which encourage fungal growth. Fungal infections of the feet are common in men and uncommon in women; although uncommon, tinea pedis does occur in prepubertal children. Tinea should be considered in the differential diagnosis of children with foot dermatitis. The occurrence of tinea pedis seems to be inevitable in immunologically predisposed individuals regardless of elaborate precautions taken to avoid the infecting organism. Locker-room floors contain fungal elements, and the use of communal baths may create an ideal condition for repeated exposure to infected material. White socks do nothing to prevent tinea pedis. Once established, the individual becomes a carrier and is more susceptible to recurrences. There are many different clinical presentations of tinea pedis. The clinical diagnosis of tinea pedis can be misleading, since it features lesions that can also be present in some other skin diseases, and direct microscopy may be insufficient to confirm the diagnosis. Culture may be necessary for a definitive diagnosis.
Clinical Presentations.
Tinea of the feet may present with the classic “ringworm” pattern ( Fig. 13.6 ), but most infections are found in the toe webs or on the soles.
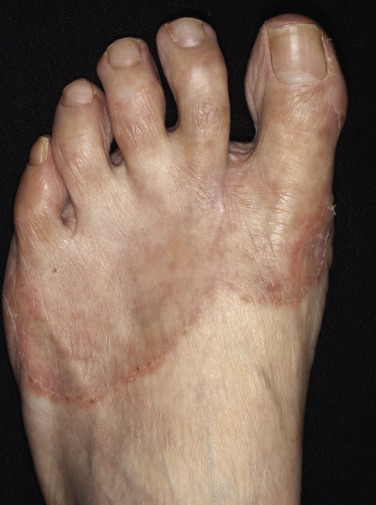
Interdigital Tinea Pedis (Toe-Web Infection).
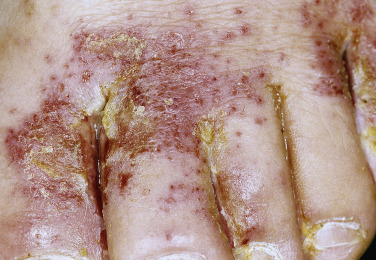
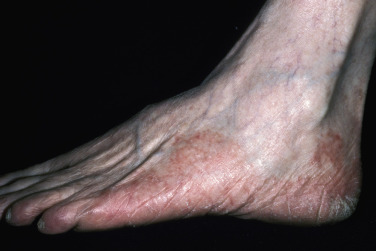
Tight-fitting shoes compress the toes, creating a warm, moist environment in the toe webs; this environment is suited to fungal growth. The web between the fourth and fifth toes is most commonly involved, but all webs may be infected. The area between the fourth and fifth toes is subject to friction between the bones of the metatarsal and phalanx, resulting in callous formation (soft corn). After adequate treatment for fungus, the callus may still remain and should be mechanically removed to prevent further infection. The web can become dry, scaly, and fissured or white, macerated, and soggy ( Fig. 13.7 ). Itching is most intense when the shoes and socks are removed. The bacterial flora is unchanged when the tinea-infected webs demonstrate scale and peeling without maceration. Overgrowth of the resident bacterial population determines the severity of interdigital toe web infection. The macerated pattern of infection occurs from an interaction of bacteria and fungus. Dermatophytes initiate the damage to the stratum corneum and, by the production of antibiotics, influence the selection of a more antibiotic-resistant bacterial population. The prevalence of Staphylococcus aureus, Gram-negative bacteria, Corynebacterium minutissimum, Staphylococcus epidermidis, and Micrococcus sedentarius increases. Extension out of the web space onto the plantar surface or the dorsum of the foot is common and occurs with the typical, chronic, ringworm type of scaly, advancing border or with an acute, vesicular eruption ( Fig. 13.8 ). Identification of fungal hyphae in the macerated skin of the toe webs may be difficult.
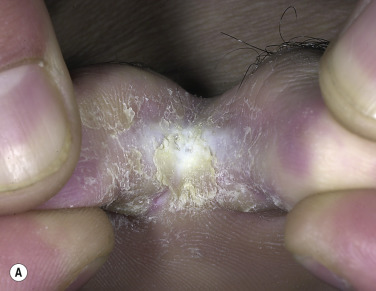
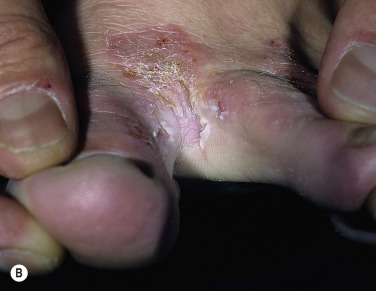
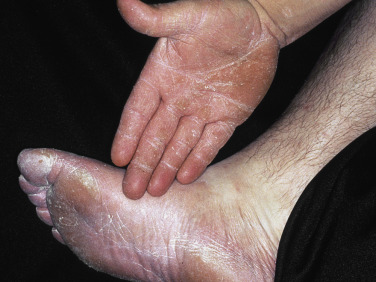
Two Feet–One Hand Syndrome.
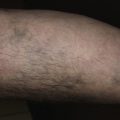
The two feet–one hand syndrome involves dermatophyte infection of both feet and tinea infection of the right or left palm ( Fig. 13.9 ). Nail infection of the hands and feet may also be present. Most cases occur in men. The same organism infects the feet, hand, and nails. Trichophyton rubrum is the causative organism in most cases. The development of tinea pedis/onychomycosis generally precedes the development of tinea of the hand. Tinea manuum usually develops in the hand used to excoriate the feet or pick toenails. Patients whose occupation involves a high intensity of use of the hands are more likely to develop the disease at an earlier age.
Treatment.
The newest class of antifungal agents produces higher cure rates and more rapid responses in dermatophyte infections than do older agents such as clotrimazole. They produce a higher cure rate and lower relapse rate than the antifungal/corticosteroid combination (e.g., clotrimazole/betamethasone).
Terbinafine 1% cream applied twice daily for 1 week results in a high cure rate in interdigital tinea pedis. In one series, terbinafine gave progressive mycologic improvement; at 5 weeks after treatment, 88% of the patients were clear of infection. Effective short-course therapy with potent fungicidal drugs such as terbinafine may avoid treatment failure caused by noncompliance with fungistatic agents, such as clotrimazole, that require 4 weeks of treatment. Butenafine applied twice daily for 1 week is also highly effective in treating interdigital tinea pedis. Econazole nitrate has activity against several bacterial species associated with severely macerated interdigital interspaces. Recurrence is prevented by wearing wider shoes and expanding the web space with a small strand of lamb’s wool. Powders, not necessarily medicated, absorb moisture. The powders should be applied to the feet rather than to the shoes. Wet socks should be changed.
Hyperkeratotic, moccasin-type tinea of the plantar surface responds slowly to conventional therapy. Oral terbinafine 125 mg daily for 4 weeks produced sustained cure rates of 95%. Griseofulvin 250 to 500 mg twice a day for 6 weeks resulted in a 27% to 35% cure rate.
Acute vesicular tinea pedis responds to wet Burow’s solution compresses applied for 30 minutes several times each day. Oral antifungal drugs control the acute infection. Secondary bacterial infection is treated with oral antibiotics. A vesicular id reaction sometimes occurs at distant sites during an inflammatory foot infection. Wet dressings, group V topical steroids, and, occasionally, prednisone 20 mg twice a day for 8 to 10 days are required for control of id reactions.
Tinea pedis has been effectively treated with pulse doses of fluconazole 150 mg orally once weekly, with itraconazole 200 mg daily for 2 weeks or 200 mg twice a day for 1 week, and with terbinafine 250 mg daily for 2 weeks.
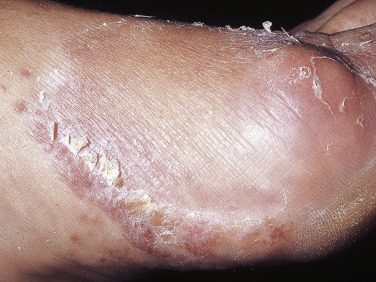
Chronic Scaly Infection of the Plantar Surface.
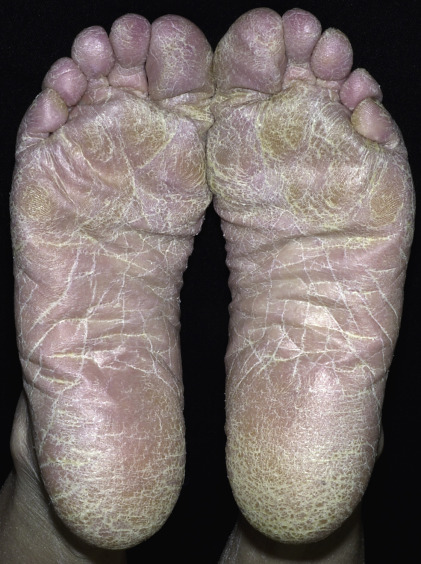
Plantar hyperkeratotic or moccasin-type tinea pedis is a particularly chronic form of tinea that is resistant to treatment. The entire sole is usually infected and covered with a fine, silvery white scale ( Figs. 13.10 to 13.12 ). The skin may be pink, tender, and/or pruritic. The hands may be similarly infected. It is rare to see both palms and soles infected simultaneously; rather, the pattern is infection of two feet and one hand or of two hands and one foot. Trichophyton rubrum is the usual pathogen. This pattern of infection is difficult to eradicate. T. rubrum produces substances that diminish the immune response and inhibit stratum corneum turnover.
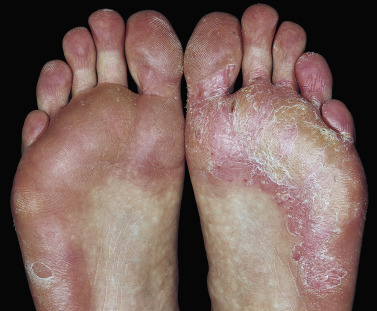
Acute Vesicular Tinea Pedis.
A highly inflammatory fungal infection may occur, particularly in people who wear occlusive shoes. This acute form of infection often originates from a more chronic web infection. A few or many vesicles evolve rapidly on the sole or on the dorsum of the foot. The vesicles may fuse into bullae or remain as collections of fluid under the thick scale of the sole and never rupture through the surface. Secondary bacterial infection occurs commonly in eroded areas after bullae rupture. Fungal hyphae are difficult to identify in severely inflamed skin. Specimens for KOH examination should be taken from the roof of the vesicle. A second wave of vesicles may follow shortly in the same areas or at distant sites such as the arms, chest, and along the sides of the fingers. These itchy sterile vesicles represent an allergic response to the fungus and are termed a dermatophytid, or id, reaction. They subside when the infection is controlled. At times the id reaction is the only clinical manifestation of a fungus infection. Careful examination of these patients may show an asymptomatic fissure or area of maceration in the toe webs.
Pitted Keratolysis
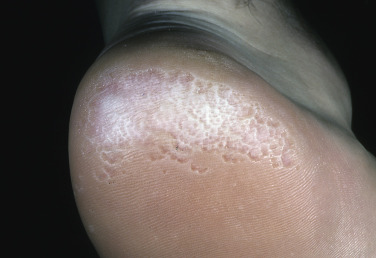
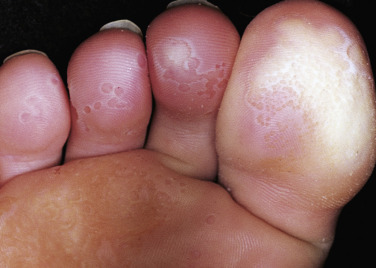
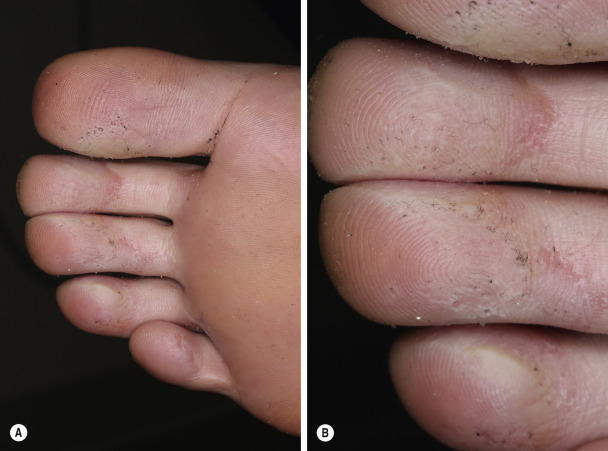
Although bacterial in origin, pitted keratolysis is often misinterpreted as a fungal infection. Mimicking tinea pedis, it is an eruption of the weight-bearing surfaces of the soles. The most common sites of onset are the pressure-bearing areas, such as the ventral aspect of the toe, the ball of the foot, and the heel. Lesions are rarely seen on the nonpressure-bearing locations. Hyperhidrosis is the most frequently observed symptom. Malodor and sliminess of the skin are also distinctive features.
Pitted keratolysis is characterized by many circular or longitudinal, punched-out depressions in the skin surface ( Figs. 13.13 to 13.15 ). Most cases are asymptomatic, but painful, plaquelike lesions may occur in both adults and children. The eruption is limited to the stratum corneum and causes little or no inflammation. Hyperhidrosis, moist socks, or immersion of the feet favors its development. There may be a few circular pits that remain unnoticed, or the entire weight-bearing surface may be covered with annular furrows. Several bacteria have been implicated, including Dermatophilus congolensis , Corynebacterium species, Actinomyces keratolytica , and Streptomyces and Kytococcus (formerly Micrococcus) sedentarius. These bacteria produce and excrete exoenzymes (keratinase) that are able to degrade keratin and produce pitting in the stratum corneum when the skin is hydrated and the pH rises above neutrality. These organisms are not easily cultured, but the filamentous and coccoid microorganisms can be demonstrated by hematoxylin and eosin staining of a formalin-fixed section of shaved stratum corneum prepared for histopathologic examination. The clinical presentation is so characteristic that laboratory confirmation is usually not necessary.
Treatment.
Wash feet daily with soap or antibacterial cleanser. Treatment consists of promoting dryness. Socks should be changed frequently. Rapid clearing occurs with application of 20% aluminum chloride twice a day. Lazerformalyde solution (10% formaldehyde), which is a potent antiperspirant, is useful. Treatment can then be applied periodically when necessary. Antibiotics are effective even without aluminum chloride or formaldehyde. The application twice a day of alcohol-based benzoyl peroxide may also be useful. Treatment with acne medications such as topical erythromycin solution or clindamycin solution is also curative. Mupirocin (Bactroban) ointment or cream and fusidic acid cream may also be effective. Oral erythromycin is an alternative.
Tinea of the Groin
Tinea of the groin (tinea cruris, “jock itch”) occurs often in the summer months after sweating or wearing wet clothing and in the winter months after wearing several layers of clothing. The predisposing factor, as with many other types of superficial infection, is the presence of a warm, moist environment. Men are affected much more frequently than are women; children rarely develop tinea of the groin. Itching worsens as moisture accumulates and macerates this intertriginous area.
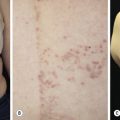
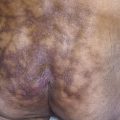
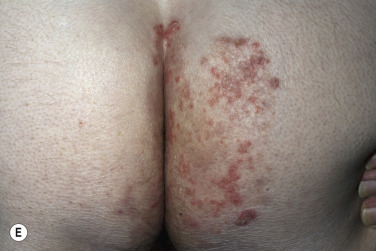
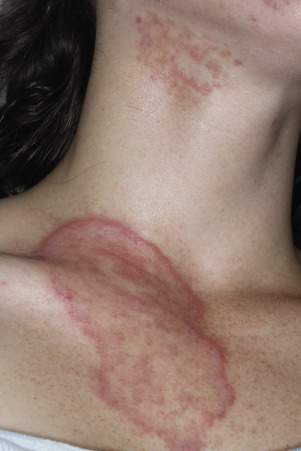
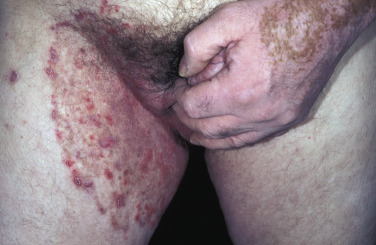
The lesions are most often unilateral and begin in the crural fold. A half moon–shaped plaque forms as a well-defined scaling, and sometimes a vesicular border advances out of the crural fold onto the thigh ( Fig. 13.16 ). The skin within the border turns red-brown, is less scaly, and may develop red papules ( Fig. 13.17 ). Acute inflammation may appear after a person has worn occlusive clothing for an extended period. The infection occasionally migrates to the buttock and gluteal cleft area. Involvement of the scrotum is unusual – unlike Candida reactions, in which it is common ( Fig. 13.18 ). Specimens for KOH examination should be taken from the advancing scaling border.
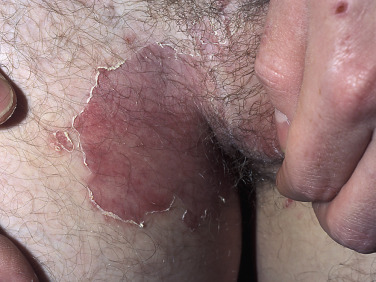
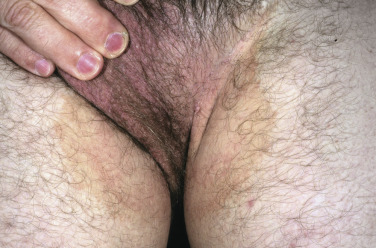
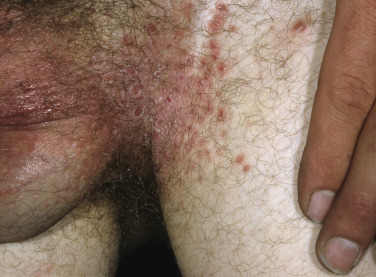
Topical steroid creams are frequently prescribed for inflammatory skin disease of the groin, and they modify the typical clinical presentation of tinea. The eruption may be much more extensive, and the advancing, scaly border may not be present ( Fig. 13.19 ). Red papules sometimes appear at the edges and center of the lesion. This modified form (tinea incognito) may not be immediately recognized as tinea; the only clue is the history of a typical, half moon–shaped plaque treated with cortisone cream. Scale, if present, contains numerous hyphae.
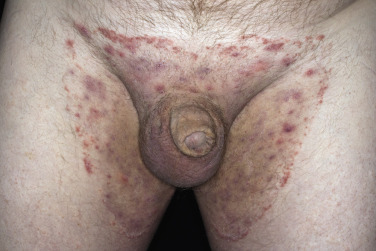
Differential Diagnosis
Intertrigo.
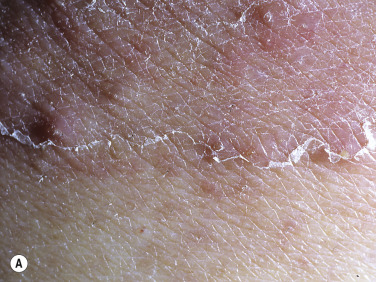
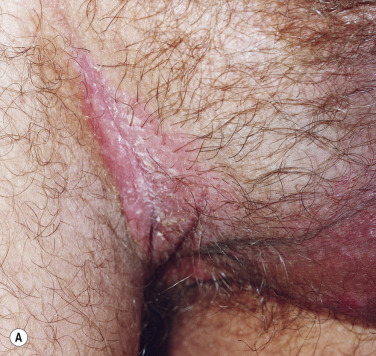
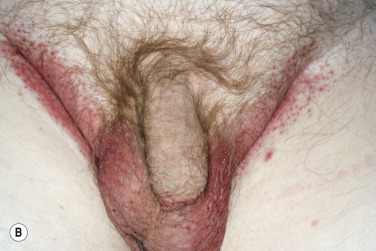
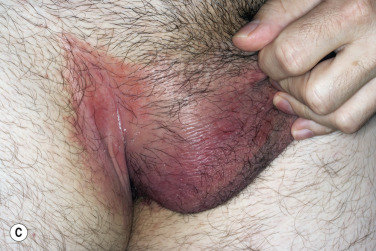
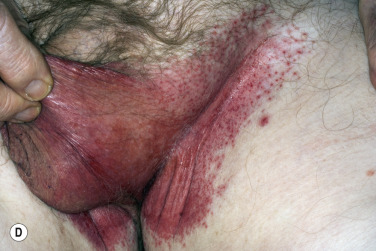
A red, macerated, half moon–shaped plaque, resembling tinea of the groin and extending to an equal extent onto the groin and down the thigh, forms after moisture accumulates in the crural fold ( Fig. 13.20A–B ). The sharp borders touch where the apposed surfaces of the skin folds of the groin and thigh meet. Obesity contributes to this inflammatory process, which may be infected with a mixed flora of bacteria, fungi, and yeast. Painful, longitudinal fissures occur in the crease of the crural fold ( Fig. 13.20C–D ). Groin intertrigo recurs after treatment unless weight and moisture are controlled. Psoriasis and seborrheic dermatitis of the groin may mimic intertrigo (see the section Candidiasis of Large Skin Folds, p. 516 ).
Erythrasma.
This bacterial infection (C. minutissimum) may be confused with tinea cruris because of the similar, half moon–shaped plaque ( Fig. 13.21 ). Erythrasma differs in that it is noninflammatory, it is uniformly brown and scaly, and it has no advancing border. The organism produces porphyrins, which fluoresce coral-red with the Wood’s light; tinea of the groin does not fluoresce. Erythrasma of the vulva may be misinterpreted as a candidal infection, especially if the Wood’s light examination is negative. The most common site of erythrasma is in the fourth interdigital toe space, but infection is also seen in the inframammary fold and the axillae. Gram stain of the scale shows Gram-positive, rod-like organisms in long filaments. However, the scale is difficult to fix to a slide for Gram stain. One technique is to strip the scale with clear tape and then carefully stain the taped-scale preparation. A biopsy demonstrates rods and filamentous organisms in the keratotic layer. Erythrasma responds to erythromycin (250 mg four times a day for 5 days) or clarithromycin (single 1-g dose) or topically to miconazole, clotrimazole, and econazole creams (but not ketoconazole). Topical acne medication such as clindamycin or erythromycin applied two times a day for 2 weeks is effective. Some topical antibiotics contain alcohol and may be irritating when applied to the groin.

Treatment for Tinea of the Groin.
Tinea of the groin responds to any of the topical antifungal creams listed in the Formulary. Lesions may appear to respond quickly, but creams should be applied twice a day for at least 10 days. The fungicidal allylamines (naftifine and terbinafine) and butenafine (allylamine derivative) allow for a shorter duration of treatment compared with fungistatic azoles (clotrimazole, econazole, ketoconazole, oxiconazole, miconazole, and sulconazole).
Moist intertriginous lesions may be contaminated with dermatophytes, other fungi, or bacteria. Antifungal creams with activity against Candida and dermatophytes (e.g., miconazole) are applied and covered with a cool, wet Burow’s solution, water, or saline compress for 20 to 30 minutes two to six times daily until macerated, wet skin has been dried. The wet dressings are discontinued when the skin is dry, but the cream is continued for at least 14 days or until all evidence of the fungal infection has disappeared. Any residual inflammation from the intertrigo is treated with a group V through VII topical steroid twice a day for a specified length of time (e.g., 5 to 10 days). A limited amount of topical steroid cream is prescribed to discourage long-term use. Absorbent powders, not necessarily medicated (e.g., Z-Sorb), help to control moisture but should not be applied until the inflammation is gone. Resistant infections respond to any of the oral agents listed in Table 13.1 .
| Griseofulvin (Ultramicrosize) | Fluconazole (Diflucan) | Itraconazole (Sporanox) | Terbinafine (Lamisil) | |
|---|---|---|---|---|
| Tinea corporis and cruris | Adult: 500 mg/day for 2–4 weeks Child: 5–7 mg/kg/day for 2–6 weeks | 150 mg once a week for 2–4 weeks | 100 mg daily for 1–2 weeks 200 mg daily for 1 week | 250 mg daily for 1–2 weeks |
| Tinea capitis | 15–25 mg/kg/day (microsize) or 15 mg/kg/day (ultramicrosize) for 6–8 weeks | 5 mg/kg/day for 4–6 weeks 6 mg/kg/day for 20 days 8 mg/kg once weekly for 4–16 weeks | 5 mg/kg/day for 4–6 weeks 5 mg/kg/day for 1 week plus 1–3 pulses 3 weeks apart 3 mg/kg/day (oral suspension) for 1 week plus 1–3 pulses 3 weeks apart Capsule-simplified dosing: 10–20 kg: 100 mg every other day 21–30 kg: 100 mg daily 31–40 kg: 100 and 200 mg on alternate days 41–50 kg: 200 mg daily >50 kg: 200–300 mg daily | 20–40 kg: 125 mg daily for 2–4 weeks >40 kg: 250 mg daily for 2–4 weeks |
| Onychomycosis | NR | 150 mg once a week for 9 months | 200 mg daily Fingernails: 6 weeks Toenails: 12 weeks Pulse dosing: 200 mg bid 1 week on, 3 weeks off Toenails: 3–4 months Fingernails: 2–3 months | 250 mg daily Fingernails: 6 weeks Toenails: 12 weeks |
| Tinea pedis | Adult: 500 mg daily for 6–12 weeks Child: 5–7 mg/kg/day for 6–12 weeks | 50 mg once a week for 3–4 weeks | Moccasin tinea pedis: 200 mg bid for 1 week | Moccasin tinea pedis: 250 mg daily for 2 weeks 200 mg daily for 3 weeks |
| Tinea versicolor | Not effective | 300- or 400-mg single dose; repeat in 2 weeks if needed | 200 mg daily for 1 week Prophylaxis: 200 mg bid 1 day per month for 6 months for recurrent disease | Oral not effective Topical is effective |
| Vaginal candidiasis | Not effective | 150-mg single dose 100 mg every 5–7 days | 200 mg for 3–5 days | Not effective |
Betamethasone dipropionate/clotrimazole cream or solution may be used for initial treatment if lesions are red, inflamed, and itchy. A pure antifungal cream should be used once symptoms are controlled. Prolonged use of this steroid/antifungal preparation may not cure the infection and may cause striae in this intertriginous area.
Systemic therapy is sometimes necessary. Tinea cruris is effectively treated by 50 to 100 mg of fluconazole daily or 150 mg once weekly for 2 to 3 weeks. Itraconazole 100 mg twice daily immediately after meals on days 1 and 8 or on days 1 and 2 may be effective. The standard treatments are itraconazole 100 mg daily for 2 weeks or 200 mg daily for 7 days, or 250 mg terbinafine daily for 1 to 2 weeks. Griseofulvin 500 mg daily for 4 to 6 weeks is also effective.
Tinea of the Body and Face
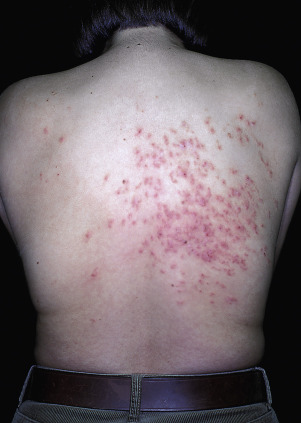
Tinea of the face (excluding the beard area in men), trunk, and limbs is called tinea corporis (“ringworm of the body”). The disease can occur at any age and is more common in warm climates. There is a broad range of manifestations, with lesions varying in size, degree of inflammation, and depth of involvement. This variability is explained by differences in host immunity and the species of fungus. An epidemic of tinea corporis caused by Trichophyton tonsurans was reported in student wrestlers.
Round Annular Lesions.
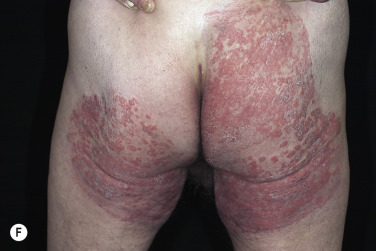
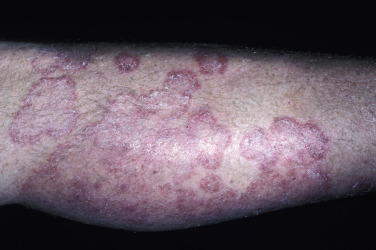
In classic ringworm, lesions begin as flat, scaly spots that then develop a raised border that expands at varying rates in all directions. The advancing, scaly border may have red, raised papules or vesicles. The central area becomes brown or hypopigmented and less scaly as the active border progresses outward ( Fig. 13.22 ). However, it is not uncommon to see several red papules in the central area ( Fig. 13.22D–F ). There may be just one ring that grows to a few centimeters in diameter and then resolves or several annular lesions that enlarge to cover large areas of the body surface ( Figs. 13.23 to 13.25 ). These larger lesions tend to be mildly itchy or asymptomatic. They may reach a certain size and remain for years with no tendency to resolve. Clear, central areas of the larger lesions are yellow-brown and usually contain several red papules. The borders are serpiginous or annular and very irregular.
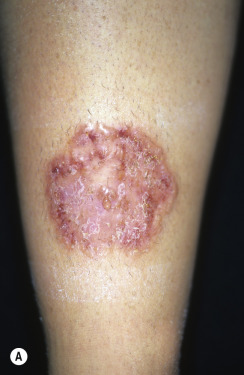
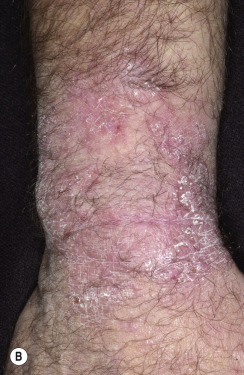
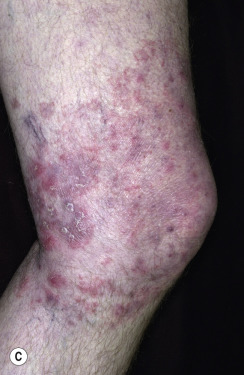
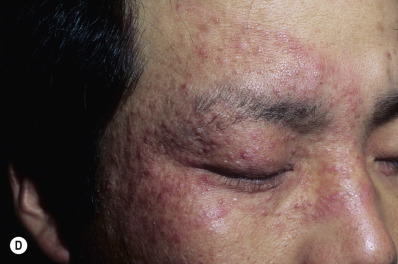
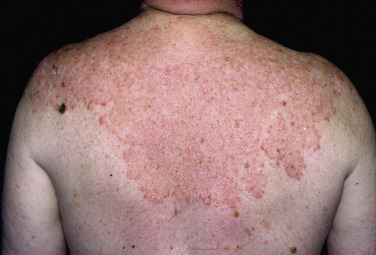
Pityriasis rosea and multiple small annular lesions of ringworm may appear to be similar. However, the scaly ring of pityriasis rosea does not reach the edge of the red border as it does in tinea. Other distinguishing features of pityriasis rosea include rapid onset of lesions and localization of the trunk. Tinea from cats may appear suddenly as multiple round to oval plaques on the trunk and extremities.
Tinea Corporis (Tinea Gladiatorum)
Tinea corporis has become common in competitive wrestling. Most reported cases are caused by T. tonsurans . Person-to-person contact is probably the main source of transmission. The role of potential asymptomatic carriers of dermatophytes is unknown.
Deep Inflammatory Lesions.
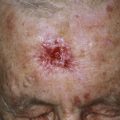
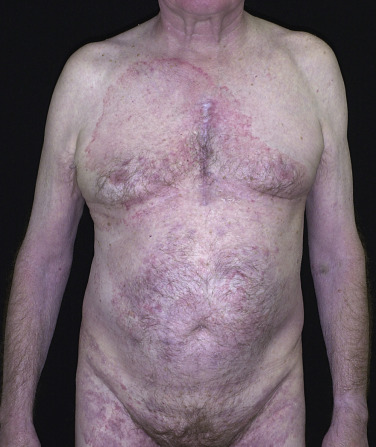
Zoophilic fungi such as Trichophyton verrucosum from cattle may produce a very inflammatory skin infection ( Figs. 13.26 to 13.29 ). The infection is more common in northern regions, where cattle are confined in close quarters during the winter. The round, intensely inflamed lesion has a uniformly elevated, red, boggy, pustular surface. The pustules are follicular and represent deep penetration of the fungus into the hair follicle (see Figs. 13.26 and 13.28 ). Secondary bacterial infection can occur. The process ends with brown hyperpigmentation and scarring (see Fig. 13.27 ). A fungal culture helps to identify the animal source of the infection.
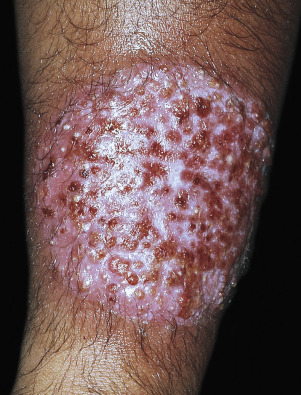
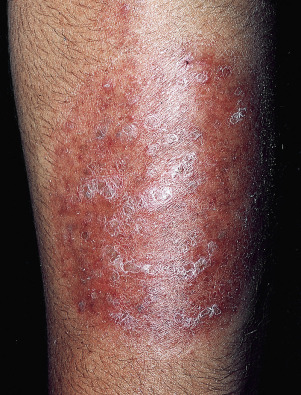
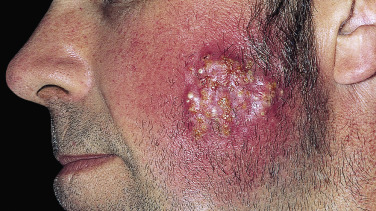
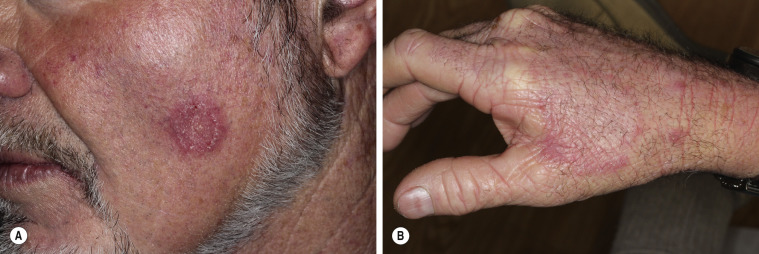
A distinctive form of inflammatory tinea called Majocchi granuloma, caused by T. rubrum and other species, was originally described as occurring on the lower legs of women who shave, but it is also seen at other sites on men and children. The primary lesion is a follicular papulopustule or inflammatory nodule. Intracutaneous and subcutaneous granulomatous nodules arise from these initial inflammatory tinea infections. Lesions have necrotic areas containing fungal elements; they are surrounded by epithelioid cells, giant cells, lymphocytes, and polymorphonuclear leukocytes, and they are believed to result from the rupturing of infected follicles into the dermis and subcutis, thus the term “granuloma.” There is marked variation from the usual hyphal forms. These include yeast forms, bizarre hyphae, and mucinous coatings. These variations may be a factor in allowing the dermatophytes to persist and grow in an abnormal manner. Lesions are single or multiple and discrete or confluent. The area involved covers a few to 10 cm and may be red and scaly, but it is not as intensely inflamed as the T. verrucosum infection described earlier. The border may not be well-defined. Skin biopsy with special stains for fungi is required for diagnosis if hyphae cannot be demonstrated in scale or hair.
Treatment.
The superficial lesions of tinea corporis respond to the antifungal creams described in the Formulary. Lesions usually respond after 2 weeks of twice-a-day application, but treatment should be continued for at least 1 week after resolution of the infection. Extensive superficial lesions or those with red papules respond more predictably to oral therapy (see Table 13.1 ). Tinea corporis is treated by 50 to 100 mg of fluconazole daily or 150 mg once weekly for 2 to 4 weeks, or by 100 mg of itraconazole daily for 2 weeks or 200 mg daily for 7 days. Itraconazole 100 mg twice daily immediately after meals on days 1 and 8 or on days 1 and 2 may also be effective. Terbinafine 250 mg daily for 1 to 2 weeks is also effective. The recurrence rate is high for those with extensive superficial infections. Deep inflammatory lesions require 1 to 3 or more months of oral therapy. Inflammation can be reduced with wet Burow’s solution compresses, and bacterial infection is treated with the appropriate oral antibiotics. Some authors believe that oral or topical antifungal agents do not alter the course of highly inflammatory tinea (e.g., tinea verrucosum), because the intense inflammatory response destroys the organisms. However, oral antifungals are safe, and few physicians would withhold such therapy. As with tinea capitis kerion infections, a short course of prednisone may be considered for patients who have highly inflamed kerions, such as the patient depicted in Fig. 13.26 .
Invasive Dermatophyte Infection.
Dermatophytes are typically confined within the keratinized, epithelial layer of the skin. The pathogenic potential is dependent, however, on a variety of local and systemic factors affecting the natural host resistance to dermatophytic infection. Underlying systemic conditions that cause depressed cellular immunity, such as malignant lymphomas and Cushing disease, as well as the administration of exogenous steroids or immunosuppressive agents, can lead to atypical, generalized, or invasive dermatophyte infection. Invasive dermatophyte infection should be included in the differential diagnosis of nodular, firm, or fluctuant masses (particularly on the extremities). Several dermatophyte species have caused a deep, generalized infection in which the organism invaded various visceral organs.
Tinea of the Hand
Tinea of the dorsal aspect of the hand (tinea manuum; Figs. 13.30 to 13.34 ) has all of the features of tinea corporis; tinea of the palm ( Fig. 13.35 ) has the same appearance as the dry, diffuse, keratotic form of tinea on the soles. The dry keratotic form may be asymptomatic and the patient may be unaware of the infection, attributing the dry, thick, scaly surface to hard physical labor. Tinea of the palms is frequently seen in association with tinea pedis. The usual pattern of infection is involvement of one foot and two hands or of two feet and one hand. Fingernail infection often accompanies infection of the dorsum of the hand or palm. Treatment is the same as that for tinea pedis and, as with the soles, a high recurrence rate can be expected for palm infection.
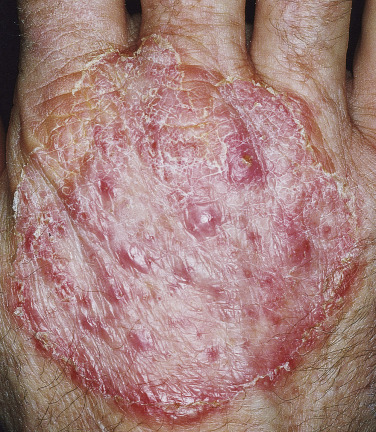
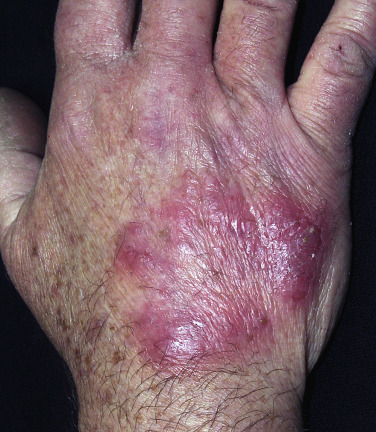
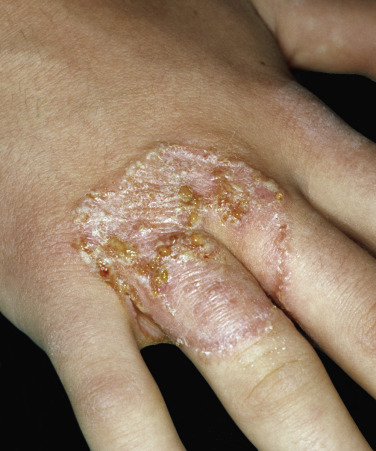
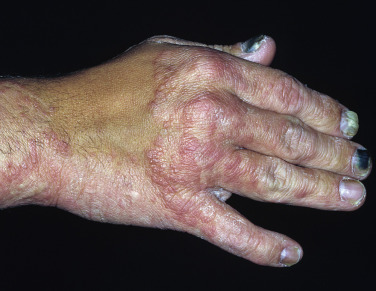
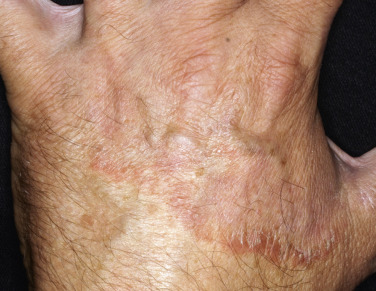
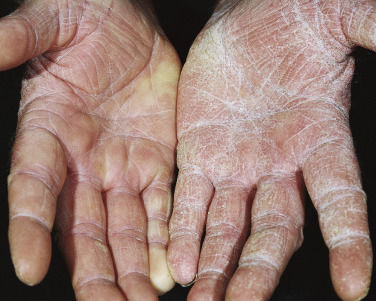
Tinea Incognito
Fungal infections treated with topical steroids often lose some of their characteristic features. Topical steroids decrease inflammation and give the false impression that the rash is improving while the fungus flourishes secondary to cortisone-induced immunologic changes. Treatment is stopped, the rash returns, and memory of the good initial response prompts reuse of the steroid cream, but by this time the rash has changed. Scaling at the margins may be absent. Diffuse erythema, diffuse scale, scattered pustules ( Figs. 13.36 to 13.39 ) or papules, and brown hyperpigmentation may all result. A well-defined border may not be present and a once-localized process may have expanded greatly. The intensity of itching is variable. Tinea incognito is most often seen on the groin, on the face, and on the dorsal aspect of the hand. Tinea infections of the hands are often misdiagnosed as eczema and treated with topical steroids. Hyphae are easily demonstrated, especially a few days after discontinuing use of the steroid cream when scaling reappears.
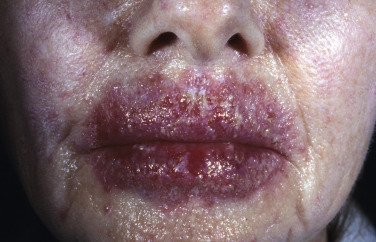
Tinea of the Scalp
Tinea of the scalp (tinea capitis) occurs most frequently in prepubertal children between 3 and 7 years of age. The infection has several different presentations. The species of dermatophyte likely to cause tinea capitis varies among different countries, but anthropophilic species (found in humans) predominate in most areas. Tinea capitis is most common in areas of poverty and crowded living conditions. The infection originates from contact with a pet or an infected person. Each animal is associated with a limited number of fungal species; therefore an attempt should be made to identify the fungus by culture to help locate and treat a possible animal source. Spores are shed in the air in the vicinity of the patient. Therefore direct contact is not necessary to spread infection. Unlike other fungal infections, tinea of the scalp may be contagious by direct contact or from contaminated clothing; this provides some justification for briefly isolating those with proven infection.
Organism and Transmission.
Large family size, crowding, and low socioeconomic status increase the chance of infection. Infectious fungal particles that have fallen from the infected person may be viable for months. Tinea capitis can be transmitted by infected persons, fallen hairs, animals, fomites (e.g., clothing, bedding, hairbrushes, combs, hats), and furniture. Zoophilic dermatophytes are acquired from contact with pets or wild animals. Microsporum canis is the most common cause of tinea capitis in central Europe. Sources of M. canis infection are cats, dogs, and guinea pigs. The animals may harbor the pathogen in their fur (colonization) although clinical symptoms may not be visible. Of the 3775 cases of tinea capitis reported to the European Confederation of Medical Mycology, 37.9% were M. canis, 23.1% T. tonsurans, 10.8% M. audouinii, 10.4% Trichophyton soudanense, and 9.7% Trichophyton violaceum. The pathogen spectrum shows clear geographic differences. A dramatic rise in T. tonsurans infections has been reported in the United States, which has not been the case in Europe. Farmers acquire T. verrucosum from touching the hide of infected cattle. Microsporum gypseum infection comes from contaminated soil.
Asymptomatic scalp carriage of dermatophytes by classmates and adults is probably an important factor contributing to disease transmission and reinfection. The asymptomatic carriage persists for an indefinite period.
Hair Shaft Infection.
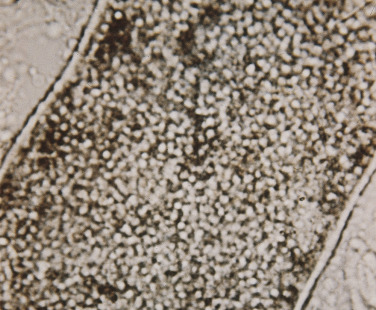
Hair shaft infection is preceded by invasion of the stratum corneum of the scalp (see Chapter 24 ). The fungus grows down through this dead protein layer into the hair follicle and gains entry into the hair in the lower intrafollicular zone, just below the point where the cuticle of the hair shaft is formed. Because of the cuticle, the fungi cannot cross over from the perifollicular stratum corneum into the hair but must penetrate deep into the hair follicle to circumvent the cuticle. This may explain why topical antifungal agents are ineffective for treating tinea capitis. The fungi then invade the keratinized outer root sheath, enter the inner cortex, and digest the keratin contained inside the hair shaft. The growth of hyphae occurs within the hair above the zone of keratinization of the hair shaft and keeps pace with the growth of hair. Distal to this zone of active growth, arthrospores are formed within or on the surface of the hair, depending on the species of dermatophytes. Hyphae grow inside and fragment into short segments called arthrospores. The arthrospores remain inside the hair shaft in the endothrix pattern ( Fig. 13.40 ). In the ectothrix type, they dislodge ( Fig. 13.41 ), obscure and penetrate the surface cuticle on the hair shaft surface, and form a sheath of closely packed spheres. The arthrospores are either large (6 to 10 mm) or small (2 to 3 mm). Large spores can be seen as separate structures with the low-power microscope objective. Higher power is needed to see the small spores.