Abstract
Skin grafts are autologous portions of skin completely divided from their blood supply and transplanted into a recipient wound.
Full-thickness skin grafts are composed of epidermis and the full thickness of dermis, and if used selectively can provide a very good match of color, thickness, and texture.
Composite grafts are grafts consisting of tissue from two germ layers. In dermatologic surgery, the term composite graft is nearly synonymous with chondrocutaneous graft, a full-thickness skin graft connected to its underlying cartilaginous base by perichondrium.
Split-thickness skin grafts consist of epidermis and only a portion of the dermis and provide the surgeon a means to cover massive and even avascular defects.
This chapter aims to guide the reader on successful graft site selection and surgical technique and prepare the surgeon for proper postoperative care, techniques for revision, and management of complications.
Keywords
skin grafts, full-thickness skin grafts, split-thickness skin grafts, composite grafts, chondrocutaneous grafts
Introduction
Skin grafts are autologous portions of skin completely divided from their blood supply and transplanted into a recipient wound. Skin grafts vary in their thickness, composition, and intended purpose, and can be categorized broadly as either full-thickness skin grafts (FTSGs) or split-thickness skin grafts (STSGs). Used for thousands of years in a variety of clinical settings, the popularity of skin grafts has waned with the advancement of local flap design. When used in the appropriate setting, however, skin grafting remains a superb choice rivaling the results attained by local flaps. This chapter will instruct the reader on successful graft site selection and surgical technique, and prepare the surgeon for the management of proper postoperative care, revision, and complications.
Full-Thickness Skin Graft
Skin Graft Physiology
FTSGs, if used selectively, can provide a very good match of color, thickness, and texture. FTSGs are composed of epidermis and the full thickness of dermis, and therefore are relatively resistant to contracture and are replete with adnexal structures. The inclusion of adnexae provides the matured graft a much more natural and camouflaged appearance relative to its split-thickness brethren, although final appearance is more variable than with a flap. The full-thickness nature of this graft is not without its detractions, however, as it has significant nutritional requirements, and requires a rich vascular supply both to promote neovascularization and to develop a new collagenous matrix by which it adheres to the recipient site. Grafts are also constrained by their limited thickness and can only cover relatively shallow defects without appearing depressed.
Through careful study, three stages of wound healing have been identified in skin grafting. Imbibition is the first stage, and is a period of relative ischemia that occurs during the first 2 days. As the name suggests, the graft survives by imbibing nutrients from the plasma exudate of the wound bed via passive diffusion. Beginning on the second to third day and lasting as long as 10 days, inosculation, the second stage, marks the beginnings of true revascularization. During this phase the dermal vessels of the graft anastomose with the vessels of the recipient bed. It is for this reason that unsuccessful inosculation and graft failure may follow full-thickness skin grafting of exposed bone or cartilage. The final step in graft survival, typically beginning concurrently with inosculation, is neovascularization. A new microvascular plexus grows into the graft from the recipient bed, supplementing the anastomosed native vessels.
Several host and iatrogenic factors contribute to the success of a graft. The vascular and nutritional demands of a graft may not be met in a patient with diabetes mellitus, nutritional deficiencies, heavy alcohol consumption, or tobacco dependence. Graft failure due to hematoma or prohibitive clotting may occur in those with inherited or acquired bleeding disorders, uncontrolled hypertension, or iatrogenic anticoagulation. Wound beds harboring high microbial loads and immunosuppression may also interfere with graft survival. Therefore these factors must be analyzed when considering an FTSG for the repair of a defect.
Graft Selection
The donor site selection is critical to the long-term aesthetic result of skin grafting, given the concept of donor dominance that describes how FTSGs retain those native characteristics like texture and hair growth attributable to preserved adnexal structures. The surgeon must understand both the epidermal and dermal qualities of a cadre of potential donor sites as one aims to provide optimal match. This analysis must extend further to the sebaceous qualities, relative dyschromia, telangiectases, and epidermal texture, as it compares with the recipient wound, of any potential donor site. The following section will serve a guide to the surgeon on possible donor sites based on anatomic location of the recipient wound.
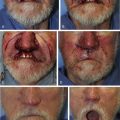
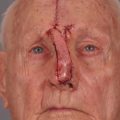
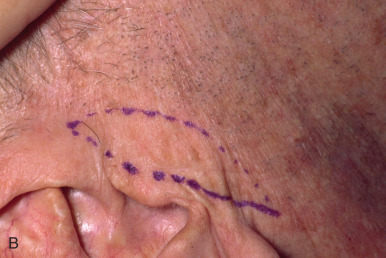
When reconstructing a wound on the nose with an FTSG, tremendous attention to the qualities of the skin and the individual cosmetic units of the nose is paramount. In general, the skin of the upper two-thirds of the nose (Zone 1) is thin and mobile relative to the thick, sebaceous lower third (Zone 2). This transition is highly variable among patients and must be closely studied prior to the design of a repair. Additionally, the nose is divided into cosmetic subunits based on convexities, concavities, natural borders, and the quality of skin. In certain instances, wounds may be extended so as to graft an entire subunit. The cephalad two-thirds of the nose lends itself quite well to skin grafting. The dermis in this region is of medium thickness, and the skin of the preauricular cheek lateral to the sideburn or the postauricular sulcus (if the skin of the recipient site is notably thin) are good matches ( Fig. 9.1 ). In males, an area of approximately 1.5 to 2 cm in the horizontal dimension can be harvested from the preauricular cheek without incorporating the terminal hairs of the sideburn. In women, the graft can be larger, but will likely include delicate vellus hairs that may detract from the cosmesis of the final appearance. In larger repairs, a defect template can be halved vertically, and two grafts can be harvested from the glabrous skin of a single preauricular donor site ( Fig. 9.2 ). The two grafts heal as one, especially if sutured together along the midline or along a cosmetic subunit. When sizable defects are involved, supraclavicular skin can be used, but caution must be taken to thin the dermis to an appropriate thickness to avoid a patchlike blob of tissue.
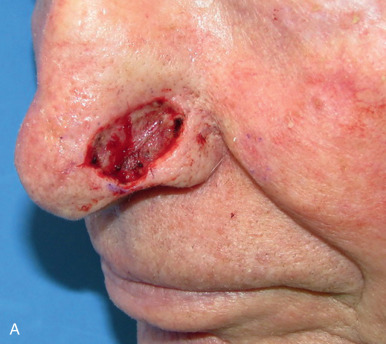
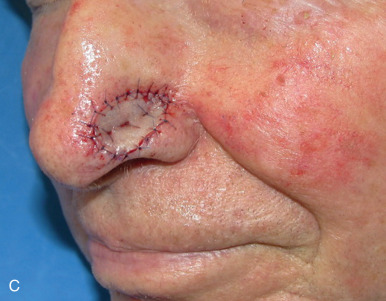
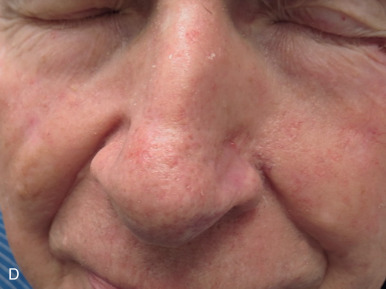
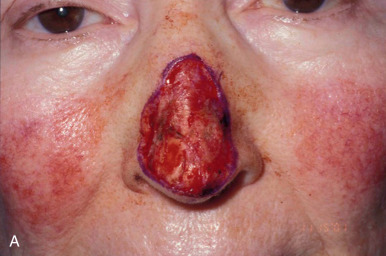
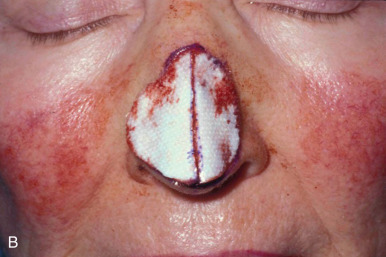
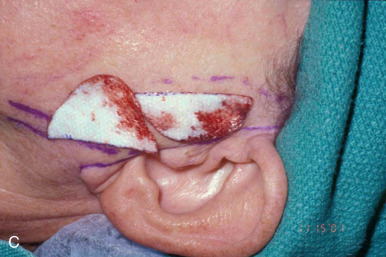
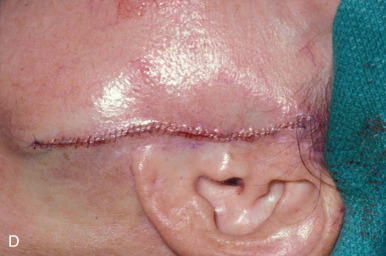
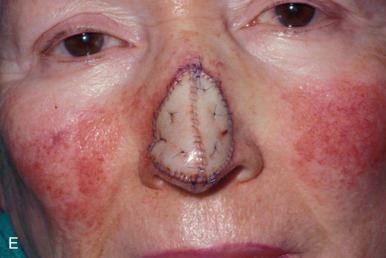
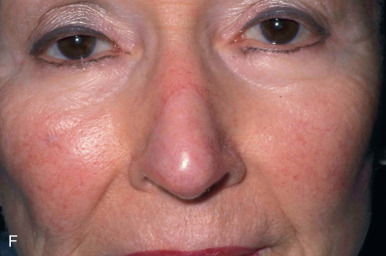
The distal third of the nose is significantly more sebaceous, and is better matched by a conchal bowl, forehead, or nasolabial skin. In women, the submental crease provides another excellent option. Grafts chosen for soft triangle and alar rim defects are often buttressed with cartilage to avoid notching. A Burow graft from a cephalad vertical triangle of skin can be harvested and transplanted into a more caudal defect. When designing a Burow graft, the triangle of skin should be of sufficient height so as to avoid significant tension in closing donor site when the graft is repositioned in its more caudal position. Failure to do so may lead to contralateral alar flare when using the Burow graft to repair nonmidline defects. A Burow graft can also be used to repair complex wounds spanning the medial cheek and nasal sidewall. First, a template of the defect confined to the nasal sidewall is created. The entirety of the medial cheek defect to its junction with the nasal sidewall is closed with a rotation or fusiform advancement flap, ensuring that the distal standing triangle is of sufficient size to serve as an FTSG for the nasal sidewall. The FTSG is then transplanted to the nasal sidewall defect and sutured into place. Defects of the columella are also well suited to repair with an FTSG.
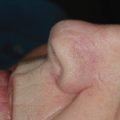
Wounds of the ear are also commonly repaired by FTSG. Many superficial wounds of the auricle, not involving the helical rim or sulcus, lobule, tragus, or antitragus, heal beautifully with second intention healing and require no reconstructive intervention. Wounds of the helical rim, however, often heal with perceptible notching. Similarly, defects involving the sulcus between the helix and antihelix can web and distort anatomic landmarks. Broad wounds of the auricle with exposed perichondrium can be resurfaced with a thin FTSG. In broad wounds with exposed perichondrium, strips of cartilage can be excised from the triangular fossa, antihelix, and concha prior to placing an FTSG over the entirety of the defect. This allows for imbibition and eventual anastomosis to arise from the skin and subcutaneous tissues of the medial ear. The auricle is best grafted with tissue from the postauricular sulcus, and the graft must be adequately thinned so as not to obscure the intricate topography of the ear. Thin freehand grafts harvested with double-edged razor blades from the mastoid process can be used to cover broad, thin wounds. The donor site is then allowed to heal secondarily.
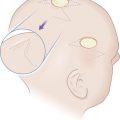
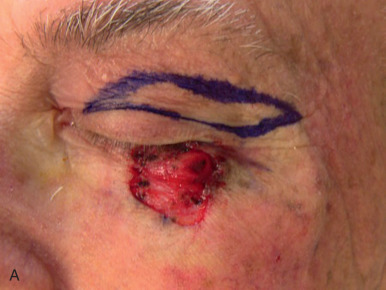
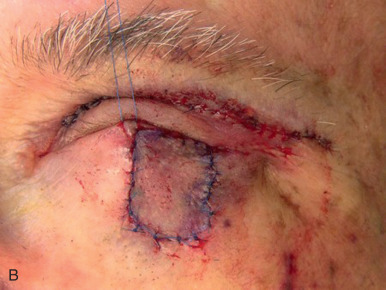
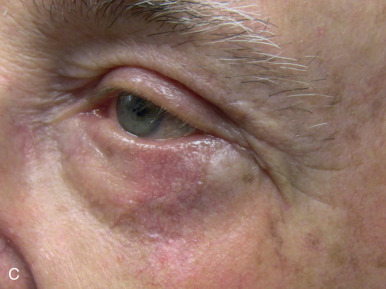
Eyelid defects involving exclusively the anterior lamella can be reproducibly reconstructed with FTSG. Defects of the eyelid are best repaired using FTSG of the upper eyelid or postauricular sulcus. Judicious use of Frost sutures when reconstructing periorbital defects is recommended to avoid ectropion ( Fig. 9.3 ). An option for large defects abutting the lower eyelid is to use guiding sutures to reduce defect size and optimize tension vectors in combination with FTSG, but leaving the guiding sutures intact during the healing phase. Careful postoperative observation is critical in the grafting of eyelids, and small aliquots of high-potency intralesional triamcinolone into early hypertrophic scarring will often stave off or reverse ectropion. This observation, coupled with the selective use of the Frost suture to suspend the lower lid by its tarsal plate, can significantly reduce the risk of eyelid malpositions. FTSG of the medial canthus is an excellent reconstructive choice because the graft can be appropriately thinned and made to fit into the contours of the defect with tacking sutures or a well-placed bolster. Local flaps, by contrast, tend to recruit excessive bulk. Grafts for the medial canthus are often obtained from the preauricular cheek or postauricular sulcus.
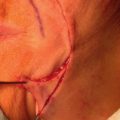
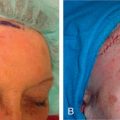
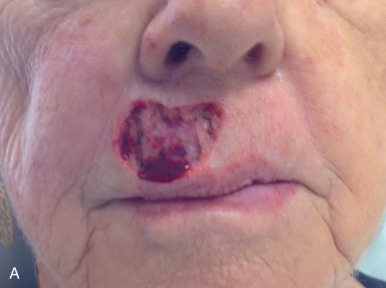
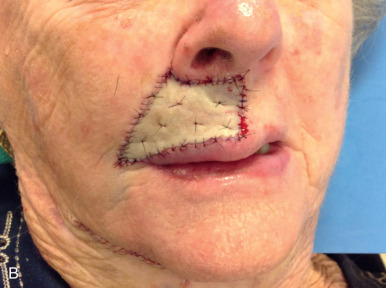
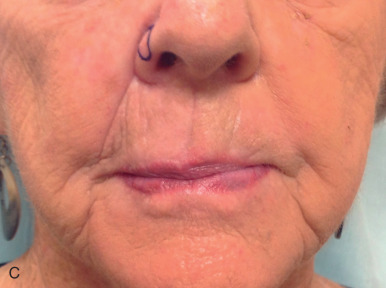
Grafts can be used throughout the face, although their use is less common than in ear, eyelid, and nose reconstruction. Their less frequent use is likely attributable to the availability of local tissue to closed primarily or with a local flap and the increased variability of the final cosmesis of grafts. Still, they are arguably less taxing on the patient and can be revised or even surgically extirpated at follow-up, and may represent the best choice for many patients. When encountered with massive defects of the forehead, temple, and cheek, the reconstructive surgeon is occasionally left no option but to repair all or part of a defect with an FTSG. In the cheek, FTSG, harvested either from the supraclavicular fossa or regionally (in the form of a standing triangle) can be paired with a rotation, rhombic, or V-to-Y advancement to repair sizable defects. The forehead, given its relatively inelastic nature, is less forgiving than the cheek. The forehead is most frequently repaired with an FTSG from the supraclavicular fossa or from a Burow graft of the temple or frontal scalp (in an alopecic male). A novel method of FTSG for the upper forehead and scalp, presented by Dr. David Brodland, is the marriage of a simple purse string closure with an FTSG. The defect is initially partially closed with purse string, and a template of the new, smaller defect is made. The FTSG, often harvested from the supraclavicular fossa, is sewn into place. The purse string is then released, drawing the graft over the defect like a drum head. This repair both reduces the size of the grafted area and provides a taut appearance to the FTSG that replicates the appearance of normal forehead or scalp. Grafting on the lip can prove invaluable in select scenarios. Very superficial defects involving delicate structures such as the philtral ridges can be grafted with thin FTSGs, often from the postauricular sulcus. If an entire cosmetic subunit has been extirpated, it can be redraped with a more sizable graft. As the lip is a very dynamic anatomic structure, the use of basting sutures and bolster dressings can aid in graft survival ( Fig. 9.4 ).
Full-Thickness Skin Graft Technique
There are multiple techniques for templating, harvesting, and insetting an FTSG. The following reflects the authors’ preferred techniques, with mention of other valuable alternatives.
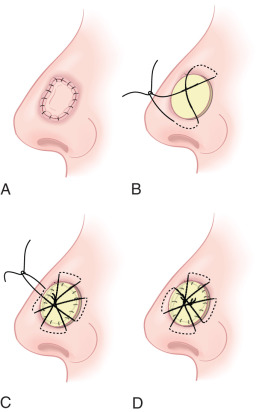
The first step of performing an FTSG is creating a template of the defect. The foil suture packaging, cut with scissors, allows the surgeon to sculpt his template and produce a three-dimensional representation of the defect. Alternatively, the wound edge can be inked with a waterproof pen, and a Telfa pad can be pressed to the defect and ink to create a template. Advocates of this method suggest oversizing the graft 5% to 10% to avoid an undersized FTSG; we prefer to precisely match the size while accounting for convexities and concavities that can cause sizing errors. Grafts should be placed under the same surface tension as the native skin elasticity to prevent pincushioning or distortion of free margins. Once completed, the template is then transferred to the donor site, and the skin underlying the template is excised to the subcutaneous fat. The graft is excised in as atraumatic a fashion as possible, with the authors’ preference being a single skin hook grasping the leading edge of subcutaneous fat and dermis. The harvested graft can be transferred to normal saline as the donor site is closed. With rare exceptions, most donor sites can be closed in a fusiform fashion. The graft can survive 1 to 2 hours in normal saline at room temperature without increased necrosis or failure. Similarly, the use of lidocaine with epinephrine at the donor site does not lead to poor outcomes.
Once the donor site has been closed, attention is redirected to inset of the graft into the recipient wound. The graft is stripped of subcutaneous fat, exposing the glistening white underbelly of the dermis. This is most commonly achieved by draping the graft over the digital pulp of the surgeon’s nondominant second digit, and tangentially snipping fat with sharp scissors. In deeper defects of the nose, some have achieved success leaving 1 to 5 mm of subcutaneous fat to add bulk and volume to match the defect. The authors recommend tie-over bolster dressing rather than bolster sutures for this type of less viable FTSG to improve adherence and take. This variation should be approached with caution in active or even former tobacco smokers. Conversely, the dermis can be thinned if necessary to match the depth of a thin defect, but this may damage adnexae and deter from the final cosmesis of the graft.
Once thinned, the graft is placed in the recipient wound. The wound must not be actively bleeding, as any seroma can cause graft necrosis and failure. The bevel of the recipient site is left as is, and the dermal edge of the graft can be slightly reverse-beveled by trimming with the scissors. Initially, one to four interrupted 5-0 or 6-0 sutures are placed in four quadrants, typically at 12, 3, 6, and 9 o’clock. Interrupted or running vertical epidermal sutures are then placed along the entire periphery of the graft. A vertically oriented bite through the epidermis and dermis of the graft, coupled with a slightly oversized bite through the wall of the recipient wound edge, ensures that the graft’s wound edge is closely apposed to its recipient wound bed.
Direct contact with the wound bed and strict immobilization are important to the survival of the graft. While there are several reports demonstrating bolster dressings may not be necessary, many surgeons use them. Numerous methods including basting sutures, bolsters, and thermoplastic casts have been used singly or in combination to achieve this goal. The authors’ technique is described herein. The graft is covered in a generous layer of petrolatum ointment. A tightly folded nonstick dressing or Vaseline impregnated gauze shaped to extend millimeters beyond the graft edge is held in place with dressing forceps by the surgical assistant. For bolsters in the periocular area, one should avoid the yellow Vaseline impregnated gauze that contains bismuth, because this is a known irritant to the conjunctival tissues. The dressing is then secured by as many bolster sutures as necessary (typically one or two) to securely position the bolster and prevent movement ( Fig. 9.5 ). A conventional pressure bandage is placed over the bolster. The patient removes the pressure dressing in 48 hours, and the bolster is left in place for 1 week. The patient must be instructed to avoid any strenuous activities that may risk trauma to the graft site, as the graft is at risk to be sheared from the wound bed for the first month.