11 Replantation and revascularization
Synopsis
 Replantation should be considered for all amputated parts; the only absolute contraindication to replantation is preserving life over limb.
Replantation should be considered for all amputated parts; the only absolute contraindication to replantation is preserving life over limb.
 A string sign or ribbon sign in the digital vessels suggests the digit is not suitable for replantation.
A string sign or ribbon sign in the digital vessels suggests the digit is not suitable for replantation.
 Skeletal shortening should be considered. Bone shortening facilitates a primary nerve and vessel repair.
Skeletal shortening should be considered. Bone shortening facilitates a primary nerve and vessel repair.
 Rigid bone fixation is desired for early mobilization. Interosseous wires offer an excellent option for rigid fixation.
Rigid bone fixation is desired for early mobilization. Interosseous wires offer an excellent option for rigid fixation.
 Revascularization of wrist and more proximal amputations should be performed before 12 hours of cold ischemia or a maximum 6 hours of warm ischemia. If ischemia time is over 4 hours, consideration should be given to temporary arterial shunting prior to bone fixation.
Revascularization of wrist and more proximal amputations should be performed before 12 hours of cold ischemia or a maximum 6 hours of warm ischemia. If ischemia time is over 4 hours, consideration should be given to temporary arterial shunting prior to bone fixation.
 Pharmacological management of replantation and revascularization remains controversial. Our current protocol includes heparin irrigation at a dilution of 100 units/ml during surgery and a heparin bolus of 50–100 units/kg before release of microvascular clamps.
Pharmacological management of replantation and revascularization remains controversial. Our current protocol includes heparin irrigation at a dilution of 100 units/ml during surgery and a heparin bolus of 50–100 units/kg before release of microvascular clamps.
 The psychological implications of amputation and replantation must be considered. The hand plays a significant role in a patient’s identity and psyche.
The psychological implications of amputation and replantation must be considered. The hand plays a significant role in a patient’s identity and psyche.
Historical perspective
Malt performed the first replantation on May 23, 1962 at the Massachusetts General Hospital on a 12-year-old boy who had his arm amputated in a train accident.1 The first digital replantation was performed in Japan on January 7, 1965 by Komatsu and Tamai.2 Subsequent to these historical events, replantation surgery took off with a flourish. China was the epicenter of early replantation surgery. The American Replantation Mission to China report in 1974 highlighted the replantation efforts in China to an astonished American surgical audience.3 The boundaries of replantation surgery continue to expand, now reaching into the realm of extremity transplantation.2
Basic science/disease process
Pathophysiology of ischemia and reperfusion
Tissue tolerance to ischemia varies significantly. In normal ambient temperature, irreversible changes can occur in muscle with only 2 hours of ischemia; skin properly cooled and stored in a nutrient media can be viable for over a month.4 Ischemia causes tissue hypoxia and the conversion from aerobic to anaerobic metabolism. Adenosine triphosphate (ATP) is produced by glycolysis which leads to the buildup of lactic acid in the tissue and the resultant reduction of intercellular pH.5 As the supply of ATP is diminished, intracellular Na+ and Ca2+ concentrations are increased. Chemical mediators and enzymes are triggered by ischemia, resulting in the production of phospholipase A2 and lysozymes. If continued beyond the critical point these processes lead to eventual cell necrosis.
Revascularization is essential before these processes become irreversible, but revascularization can lead to its own problems. Reperfusion injury can cause problems as severe as the initial ischemic insult. When ischemic tissue is suddenly reperfused, reactive oxygen species (ROS) are produced. These include superoxide (O–2), hydrogen peroxide (H2O2), and hydroxyl radical (OH–).6 The ROS react with the cell membranes, especially in the endothelial cells. They cause direct cell damage and produce inflammatory mediators, complement activation, and leukocyte adhesion. This cascade increases vascular permeability and can cause cell death despite revascularization. Reperfusion-generated mediators may have systemic effects when vessels are unclamped and the inflammatory mediators are released to the circulation. This can lead to a decreased level of consciousness, jaundice, cardiac arrhythmia, metabolic acidosis, myoglobinuria, and multifailure organ system.7 Methods to prevent or reduce ischemia–reperfusion injury include hypothermia, interarterial flushing, ischemic preconditioning, antithrombotic agents, free-radical scavengers, and leukocyte inhibitors.4,5 While many of these techniques show promise experimentally or in elective free flap surgery, pretreatment is not an option in traumatic replantation and revascularization. Aside from expedient revascularization the only current useful techniques are cooling of the amputated part, interarterial flushing, and antithrombotic agents. In major limb replantation, once arterial flow is established, the vein should be allowed to bleed to eliminate ROS, prior to establishing flow to the systemic circulation.
Diagnosis/patient presentation
Transportation
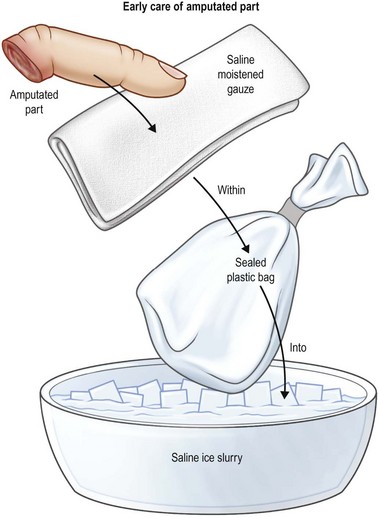
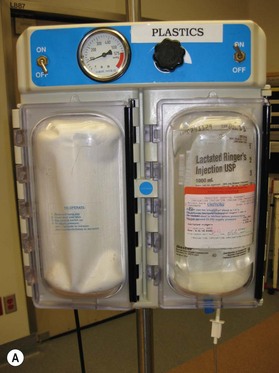
The most critical factors in replantation and revascularization surgery is management of the patient and the amputated part. Proper handling of the amputation is essential to revascularization and replantation success. The first responders must be educated in the proper transportation of the injured part. This includes proper cooling of the amputated part and stabilization of the patient. The part is wrapped in moist saline gauze and placed in a waterproof plastic bag. The plastic bag is placed in a container of ice (Fig. 11.1). The part should not come into direct contact with the ice. Freezing the severed extremity can cause direct irreversible damage to the microvascular system. Timely, efficient transportation of the patient and the amputated part to the replantation center is critical for survival and function. Because of the rapid irreversible changes that occur in muscle, this is essential in major limb amputations. Muscle undergoes irreversible damage with ischemia times greater than 6–9 hours. Digits tolerate longer periods of ischemia. Successful replantation can be achieved even after prolonged ischemia. VanderWilde et al. reported a successful hand replantation after 54 hours of cold ischemia.8 Wei et al. reported a series of three successful digital replants with 84, 86, and 94 hours of cold ischemia.9 All patients had satisfactory functional results and no vascular compromise. Lin et al. reviewed their experience with 31 replantations (two thumb, 21 finger, and two hand amputations), all with over 24 hours of either cold or warm ischemia. The overall success rate was 64%. Only one of the hands was successfully replanted; 15 of the 23 digits were salvaged.10
Replantation center
Digital and major limb replantation can be performed in any hospital with a microscope or even a pair of high-powered surgical loupes. A replantation center, usually a trauma center, offers a dedicated team to serve the patient better. The replantation center should include an emergency room and paramedic system familiar with the transportation of the injured patient and the amputated part (Table 11.1). This often includes a medical air transportation system to facilitate rapid transportation from the accident scene. At the replantation center, the operating room should be large enough to allow two independent surgical teams to operate, one on the patient and the second on the amputated part (Fig. 11.2). The replantation center should have a minimum of two sets of microsurgical instruments and two microscopes if possible. Chung et al. reviewed the database from the Agency for Healthcare Policy and Research, evaluating 304 cases of finger replantation. Over 906 hospitals were represented in the database but only 15% performed a finger replantation in the year reviewed; 60% of these hospitals performed only one replant. Only 2% of the hospitals in the database performed 10 or more cases. He postulated that replantations should be best performed at high-volume centers.11 Chen and Narayan reviewed the economic trends of upper extremity replantations over a 10-year period and found an increasing tendency for these surgeries to be performed at large teaching institutions.12 A survey of members of the American Society of Surgery of the Hand found that only 56% of its members perform replantation surgery, despite 94% of the responders describing their microsurgical training as good or excellent.13
Table 11.1 Replantation center criteria
Patient selection
Indications and contraindications
Needless to say, the critical patient selection criterion for replantation is a patient with an amputation. The amputated part must be both available and in satisfactory condition for the replantation (Table 11.2). Digits recovered from the stomach of an attacking animal or extracted from industrial grinders generally do not meet this criterion. Outside these obvious examples, replantation should at least be considered for all amputated parts. This includes not only amputation of the upper extremity but the lower extremity or any other body part. Accepted indications for replantations in the upper extremities are thumb amputations, multidigit amputations and amputations in children.14 Replantation of parts otherwise not generally thought to be replantable are often indicated in the child. Often adult patients with single-digit amputations, especially the index finger, are generally not considered candidates for replantation.15 Replantation of a single digit is generally indicated in children. This is partly due to improved healing in children but also because of the future potential of the child. A replanted digit may allow a child to follow her dream as a great musician or athlete, whereas an amputation revision may preclude this possibility. Lower-extremity and upper arm replantations may also be indicated in a child. The child possesses improved nerve regeneration that may allow improved function for amputations at these levels.
Table 11.2 Indications for replantation
| Strong indications |
| Relative indications |
Patients who have lost multiple digits also deserve an extra consideration in replantation. Where a single digit loss may be well tolerated, patients who have lost multiple digits have a significantly greater disability. Salvaging one or two fingers in a mutilated hand may preserve essential function of the hand. In multiple-digit amputations consideration should also be made for “spare-parts surgery.” If an amputation is through the metacarpophalangeal (MCP) joint of the long finger and more proximal on the index finger, replantation of the index finger to the long finger ray, preserving the joint, may increase the patient’s overall function. Replantation above the elbow is advocated for preservation of elbow function but generally distal function in the hand is poor.16 Fingertip replantations, once considered nonindicated, are now seen as an excellent level of replantation. Replantation of a digit distal to the flexor digitorum superficialis (FDS) insertion generally has excellent function because only one flexor tendon requires repair, and should be strongly considered.
The only absolute contraindication to replantation is preserving life over limb. A patient with life-threatening injuries or multiple medical problems who cannot withstand prolonged surgery should not undergo replantation. An uncooperative patient is also a contraindication for replantation.14 In a patient with acute major life-threatening injuries, an amputated hand or digit may be kept in cold storage and the patient reassessed in 24 hours. If the patient is stable enough to undergo surgery, delayed replantation may be considered.
There are several relative contraindications to digital replantation17 (Table 11.3). These contraindications are related more to microvascular success and hand function as compared to an amputation revision. All patients must be evaluated individually to determine if they are a candidate for digital replantation. While age is not an absolute contraindication for replantation, older patients carry a greater risk of arteriosclerosis of the vessel, joint stiffness, and generally decreased function secondary to less recovery of sensation. Okada et al. reviewed eight replantations in patients over 65 years old. Function of these digits was generally classified as poor, although all patients were satisfied with their results. Expectations in the elderly patient may be less than those of a younger patient.18
Table 11.3 Relative contraindications to replantation
Treatment/surgical technique
Operative sequence
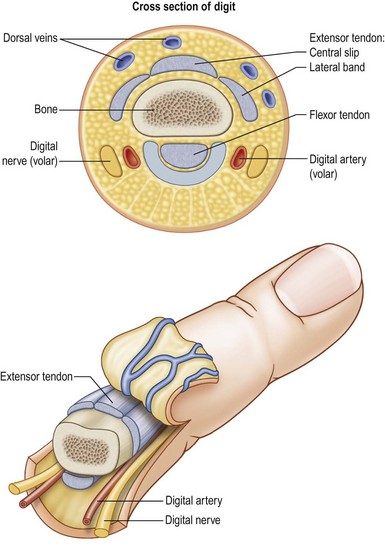
If two surgical teams are available (at least for a short time during this surgery) both the amputated part and the patient can undergo simultaneous surgery. If there is any delay in getting the patient to the operating room, the amputated part is brought to the operating room first. Using loupe magnification, all structures are examined. The blood vessels and nerves are usually examined at high magnification with the operating microscope before committing to perform a replantation. Under the operating microscope, the part is examined for suitability of replantation. Tissue loss is evaluated, along with the level of amputation. Multiple levels of injury may be found which were not apparent during the emergency room examination. The neurovascular structures should be evaluated for evidence of avulsion injuries. A string sign or ribbon sign means the digit is not suitable for replantation. These signs are indicated by a red streaking of blood along the path of the artery, usually implying an avulsion injury. The artery and nerves should be identified and tagged with a 7-0 polypropylene suture or microclips. If the digital vessels cannot be readily identified through the traumatic wound, the digital vessels can be exposed through a midaxial incision (Fig. 11.3). Dorsal veins are identified and tagged in a similar fashion. The location of the veins is more variable, and they may not be readily visible. Two or three veins should be identified. Preliminary bone fixation and methods of tendon repair are considered at this time. The digital core tendon suture may be placed while the part is on the back table. The distal aspect of the bony fixation can also be secured, whether this is with K-wires, interosseous wires, or a mini fixation plate.
The sequence of the repair in digital replantation varies among surgeons, with some advocating arterial repair after bony fixation and flexor tendon repair while others advocate repair of the dorsal structures, including the extensor tendon, dorsal veins, and skin prior to repairing any volar structures.17 The author favors repairing the volar structures first, and then proceeding to the dorsal structures. Repair of the extensor tendon prior to releasing the tourniquet is advantageous in those cases where visualizing the vein is difficult and better seen when the digit is revascularized.
Bone fixation
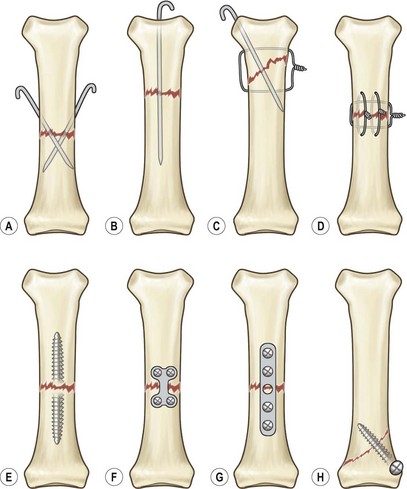
The time spent in achieving secure digital fixation is well worth the additional effort required in this fixation (Fig. 11.4). Adequate fixation of the replanted digit is essential not only for short-term healing but also for long-term function. Rigid fixation of the bony fracture can reduce pain, allow early mobilization of the hand, and improve long-term function. Conventional thinking suggests that time is of the essence and bony fixation should be rapid in order to get to the ultimate goal of the microvascular repair. This logic ignores the fact that the final outcome of the replantation lies in the ultimate function of the hand. Function is linked to motion, and motion is optimized by early mobilization. Rigid bone fixation is desired for early mobilization. Skeletal shortening should be considered in all replantations; this can allow more secure bone fixation and lessen the need for vessel and nerve grafting.14 In a thumb, shortening of the digit should be performed judiciously.19
Kirschner wires offer control of rotational deformity but greater care must be taken with early digital motion. Cross K-wires give an acceptable result although early rigid fixation is not achieved. Late digital stiffness and fracture malunion may occur. Kirschner wires should be reserved for cases where rapid fixation is necessary and in children.19
Interosseous (I-O) wires offer an excellent option for rigid fixation in amputations. I-O wires can be technically demanding to perform, but offer the advantage of rigid fixation and the ability to correct for any rotational deformity prior to final tightening of the wires. Three sets of wires, two longitudinal and one radial ulnar, give as much rigidity as a fixation plate. The exposure granted by the amputation allows easier access to the bone for I-O wire placement. The ends of the wires should be turned in to the fracture site to add additional strength to the configuration and to avoid impingement on the tendons.20 Ninety-ninety-wire bone fixation has a low nonunion rate and provides secure compression of the fractures.17,19,21–23
Plate fixation offers secure fixation for fractures. Standard miniplates, while offering secure fixation and compression, have the disadvantage of requiring significant periosteal stripping and more extensive soft-tissue exposure. Small “H” plates offer similar fixation strength with less exposure and periosteal stripping. One disadvantage of plates is the lack of adjustability. If distinct registration points are not present on the bone, improper fixation can lead to rotational deformities. The increased dissection and periosteal stripping may lead to increased scarring and tendon adhesions. Generally plate fixation is more time-consuming than other methods of fixation. A low-profile H-plate may allow less dissection; however, this application is technically demanding and angulation and malunions can occur.19
Tendon repair
Controversy exists among surgeons as to whether both and FDS and flexor digitorum profundus (FDP) tendons should be repaired in zone 2 replantations.24 Waikakul et al., in their series of 1018 replantations, found patients who had a sharp guillotine zone 2 amputation had good to acceptable results in terms of range of motion with repair of both tendons. In crush, degloving, or avulsion injuries, repairing only the FDP tendon or both tendons gave poor results. Repairing the FDP stump to the proximal superficialis tendon gave the best result, which was statistically significant.25 Ross et al. prospected and evaluated tendon function after replantation using a modified Kessler repair with 4-0 polyester suture. The average total active motion was 129°. Patients with zone 1 and 5 injuries had a significantly better active range of motion. Replantation after avulsion injury resulted in lower range of motion compared to sharp injuries. When they evaluated patients who had one tendon repaired versus those with a repair of both tendons, the average motion for fingers with both tendons repaired was 136° compared to 111° for fingers with only the FDP tendon repaired. This was statistically significant. Of all factors in the surgeon’s control, the most important was initiating early therapy. Early therapy increased both active and passive range of motion and did not significantly increase the incidence of tendon rupture.24
Artery repair
A patent arterial repair is essential to successful replantation. The red line sign, or ribbon sign, is an indication of torsion or stretch of the vessel and suggests a relative contraindication to replantation.26 Arteries should be debrided to beyond the zone of injury. The lumen of the artery should be explored to make certain there are no intimal flaps which can cause thrombosis. The arteries should be examined and cut back, as necessary, prior to inflation of the tourniquet. The vessel is flushed with a dilute heparin solution at a concentration of 100 units/mL. The heparinized saline solution can be placed in a pressure bag to assure a constant, accurate irrigation pressure (Fig. 11.5). Manual irrigation with a standard 3-cc syringe may produce excessive irrigation pressure. Yan et al. found that irrigation pressures under 80 mmHg are as effective as higher pressures at removing clots and did not damage the vessel endothelium.27 After irrigation, the vascular clamp is released to visualize the arterial flow. If the artery does not have a strong pulsatile flow, the vessel is cut back and irrigated until pulsatile flow is obtained. It is preferable to repair both digital arteries, if possible. Zumiotti et al. found that when two arteries and more than one vein were anastomosed, the success rate was highest.28 In the index finger, long finger, and thumb, the ulnar digital artery is usually larger and should be preferentially repaired. In the small finger, the radial digital artery is usually of larger diameter. There is usually no difference in size in the ring finger vessels. Adequate exposure to the blood vessels is essential. Skin hooks attached to rubber bands may facilitate artery exposure. If this is not adequate, additional incisions should be made to aid exposure. In the proximal and middle phalanx, zigzag incisions are preferable, while in distal replantations a mid-axial incision is preferred.14
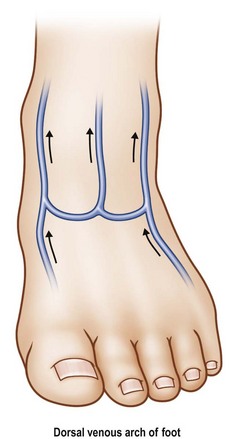
If after proper debridement the gap precludes primary repair, a vein graft should be used. Theoretically an increased potential of thrombosis exists when vein grafts are used since there are two microvascular anastomoses. However, clinical studies do not support an increased incidence of thrombosis. When vein grafts are utilized, venous discrepancies may be managed by a variety of techniques, including fish mouth incisions, sleeve anastomosis, or end-to-side coupling or step-down vein grafts.29 Vein grafts are usually harvested from the palmar side of the thenar eminence of the hand or from the distal volar forearm or the dorsal foot (Fig. 11.6).30 For arterial reconstruction in the finger, the author prefers the dorsal foot veins because they are thicker-walled and better match the digital artery. For more proximal amputations the greater or lesser saphenous veins can be harvested.
Lee et al. prospectively followed 75 finger replantations with Doppler examinations for 60 days postoperatively. In 37% of successful replantations Doppler signals, which were initially present, were not heard on postoperative day 15. This rate was five times greater for crush injuries than for guillotine amputation. Their replantation center now routinely administers anticoagulants for 2 weeks in all replantations, regardless of the degree of tissue damage. They postulate that by continuing anticoagulation, reduced morbidity such as cold intolerance and sensory abnormalities may be achieved.31
Vein repair
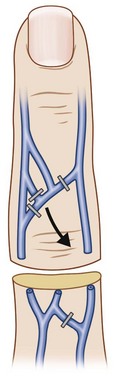
Repair of the veins is one of the most crucial steps in replantation. The number of veins repaired correlates well with success in digital replantation.32,33 In a devascularized extremity or digit, a small skin bridge of only a few millimeters can allow adequate venous outflow. In a complete amputation, the vein repair is critical. Dorsal veins can be found by identifying bruising or small hematomas just under the skin (Fig. 11.7). The veins are usually repaired after repair of one or both of the digital arteries. If ischemia time is prolonged, the vein can be repaired after the first digital artery and then the hand supinated to repair a second digital artery. Veins grafts to bridge segments of venous loss are used as required. Additional length on dorsal veins may be achieved by dividing the interconnecting branches between the veins. At least one vein should be repaired prior to release of the arterial clamp. If a vein could not be identified on the initial exploration of the amputated part, release of the arterial clamp will allow visualization of the dorsal veins as they fill with blood; but the resulting bleeding may obscure the clean microvascular field.
A functional venous anastomoses is important to overall success rate in replantation; however, the more distal the amputation, the less this factor comes into play. Distal amputations can be successfully replanted even without a venous anastomosis as long as some method of venous egress is assured.34–38 In a series of 120 replantations distal to the FDS insertion a 91.7% success rate was achieved when two veins were repaired. This success rate dropped to 84.4% when only one vein was repaired, but with no vein anastomosis, a 40% success rate was still achieved.39 Various methods of venous outflow other than direct vein coaptation have been described. These include use of leeches (Hirudo medicinal), arterial venous anastomoses, nail plate removal, heparin rubs, subcutaneous heparin instillation, nail ablation, and dermal implantation.34,35,39–41
Nerve repair
Sensibility in the replanted finger is an important factor in final functional outcome. Sensibility is even more important than range of motion.14 Primary repair of the nerve can be performed in most clean guillotine-type injuries. Bone shortening facilitates a primary nerve repair.42 Patient factors influencing nerve recovery are age, level of injury, and mechanism of injury. Surgeon-controllable factors include digital blood flow and postoperative sensory re-education. Repair of more than one artery improves nerve recovery.43 If there is any question of the quality of the nerve it should be debrided to identify a good fascicular pattern. Nerve grafts or conduits can be used for small nerve defects. Conduits may be constructed utilizing a variety of materials, including segments of vein, collagen, silicone, Gore-Tex®, polyester, and polyglycolide.44 Conduits are best limited to defects of less than 2 cm in length.45 Some conduits are extremely rigid and may impede motion if placed over a joint or are at risk for erosion of overlying skin. Conduits are best used for sensory nerve defects; for motor nerves standard nerve grafts are preferred. Nerve grafts can be harvested from a variety of donor sites. In the upper extremity these include the posterior interosseous or median antebrachial cutaneous nerves. The sural nerve is the standard donor graft in the lower extremity. The fascicles of the sural nerve match the digital nerve topology well. After successful nerve repair, replanted fingers typically achieve protective sensation with a two-point discrimination under 15 mm. The majority will achieve static two-point discrimination less than 12 mm.46
Skin closure
Skin closure should be performed with special precautions not to constrict arterial inflow or, more importantly, venous outflow. Tight closures may compromise success in replantation. If there is any concern a skin graft should be utilized. A small graft over the mid-lateral incision covering the vascular pedicle is far safer than a tight closure. If there is a deficit of skin and an arterial defect requiring a vein graft, consideration should be given to using a venous flowthrough flap (vascularized by the digital artery).47 The flap is harvested from the distal ipsilateral volar forearm. A vein of sufficient size is identified and a skin flap slightly larger than the skin defect is designed, centered over the vein. The flap should not extend more than 1.5 cm from the vein. The vein is reversed and used to bridge the arterial defect and the skin island covers the skin defect.