20 Repair, grafting, and engineering of cartilage
Synopsis
 Cartilage is one of the most important tissue grafts in plastic surgery and is widely used in auricular reconstruction, rhinoplasty, and facial countoring.
Cartilage is one of the most important tissue grafts in plastic surgery and is widely used in auricular reconstruction, rhinoplasty, and facial countoring.
 This chapter introd uces the background knowledge and surgical skills required for harvesting cartialge graft and managing donor site tissues.
This chapter introd uces the background knowledge and surgical skills required for harvesting cartialge graft and managing donor site tissues.
 Additionally, recent development in engineered cartilage fabrication and their potential application in plastic surgery are described.
Additionally, recent development in engineered cartilage fabrication and their potential application in plastic surgery are described.
Introduction
Cartilage is a kind of connective tissue which is mainly composed of chondrocytes and their extracellular matrices (ECM) of type II collagen fibers, proteoglycans, and elastic fibers. According to its composition, cartilage can be classified into three types1: (1) hyaline cartilage; (2) fibrocartilage; and (3) elastic cartilage. Figure 20.1 shows the histology of the three types of cartilages.
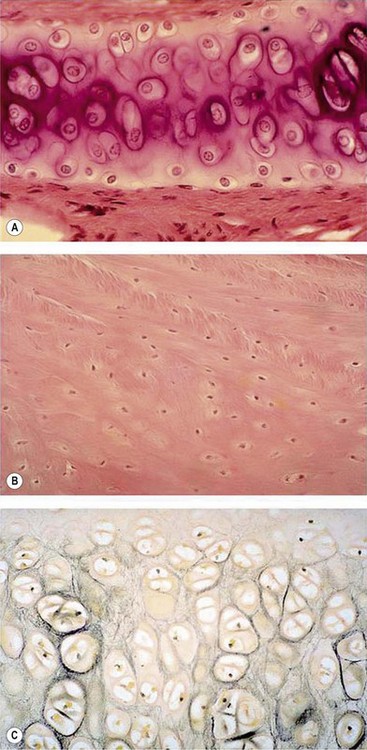
Fig. 20.1 Histology of (A) hyaline cartilage, (B) fibrocartilage, and (C) elastic cartilage (silver staining).
(Courtesy of Dr. Roger C. Wagner Prof. Emeritus of Biological Sciences at University of Delaware and Prof. Fred E. Hossler, East Tennessee State University.)
The perichondrium is a layer of dense irregular connective tissue that consists of two separate layers, an outer fibrous layer and an inner chondrogenic layer. Collagen fibers and fibroblasts constitute the fibrous layer. In contrast, the chondrogenic layer remains partially undifferentiated and is likely to contain mesenchymal stem cells and chondrogenic progenitor cells,2,3 which can play a role in cartilage repair and regeneration.
Cartilage is a unique tissue with low metabolic rate due to the sparsity of its cell population and its avascular structure. The glycolytic activity and oxygen consumption of cartilage approaches anaerobic condition and the tissue is nourished by tissue fluid diffusion. Because of this unique feature, cartilage graft is relatively easy to survive when being implanted. In term of cartilage grafting, Sushruta Samhita in India probably is the first person to perform cartilage grafting in the form of a composite graft.4,5 Cartilage graft is widely used for repairing nasal or auricular defects as well as for reconstructing other tissues either as a pure cartilage graft or as a composite graft. According to surgical procedures, cartilage can be transferred either as a free graft or as a microvascular composite graft. Generally, autologous cartilage graft will not have metaplastic changes even being transferred to a different locations.6 In addition, grafting of free perichondrial tissue is also a common procedure for cartilage reconstruction because it has been observed long time ago that perichondrium possesses the ability to regenerate cartilage.7
Autologous cartilage grafts and applications6
Auricular cartilage graft
As an elastic cartilage, auricular cartilage is an ideal graft for transplantation and perhaps is the most versatile of all cartilage grafts because it can be easily fashioned and contoured into different shapes for various uses. Auricular cartilage can be harvested easily under local anesthesia and a significant portion of the concha can be removed without causing donor site deformity.6
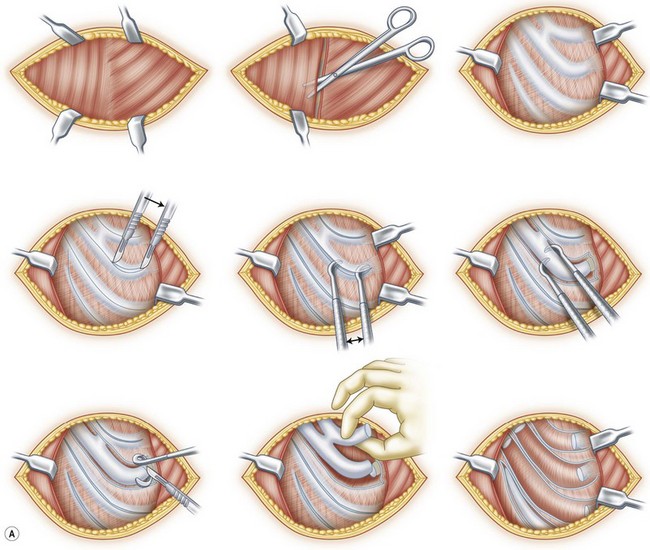
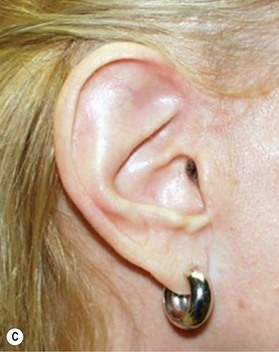
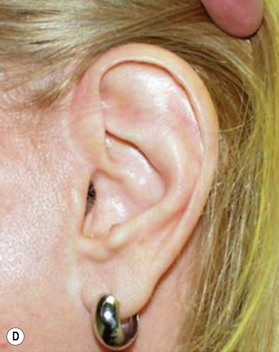
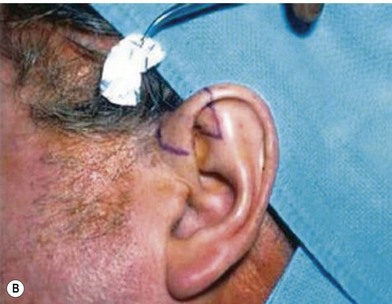
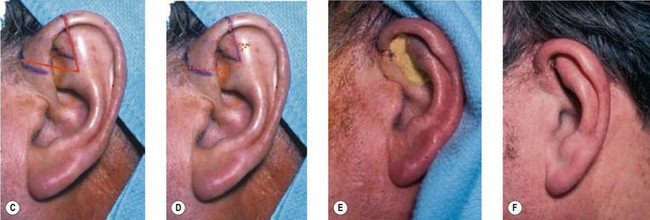
However, in practice, harvesting of the entire conchal cartilage is likely to cause collapsed conchal bowl, cymba concha, and a horizontally short ear. To overcome this problem, Han et al. proposed the surgical procedure that involves: (1) using a postauricular incision to minimize visible scars; (2) harvesting the entire cymba concha and cavum concha separately, with at least 5 mm of the helical crus, leaving a lateral extension as a strut between them, as well as a 2-mm outer rim along the conchal wall; and (3) by using a tie-over bolster dressing that can serve as a mold for the conchal bowl. Figure 20.2 presents the technique to avoid contour irregularity or deformity after harvesting the maximal amount of conchal cartilage graft.8
Auricular cartilage graft is often used as a framework for ear reconstruction or auricular deformity correction, as reported by Brent,9 Ono et al.,10 Firmin et al.,11 and others. In addition, conchal cartilage can be used as a single-layered graft for nasal, tarsal, and nipple reconstruction.6
The other important application of auricular cartilage is to transfer as a composite chondrocutaneous graft for nasal reconstruction. How to manage the donor site of auricular composite graft aesthetically is important for its successful application. Singh and Bartlett have summarized several techniques to deal with the donor site of free auricular composite grafts according to different applications12:
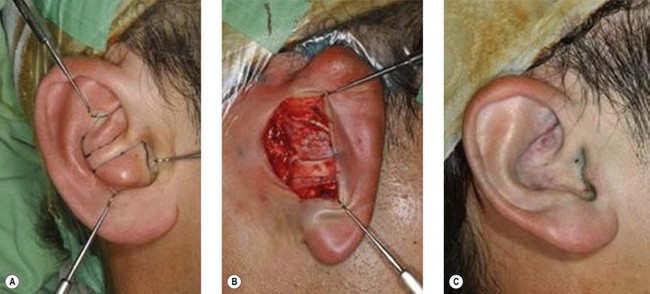
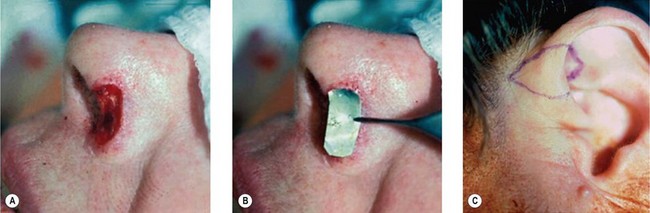
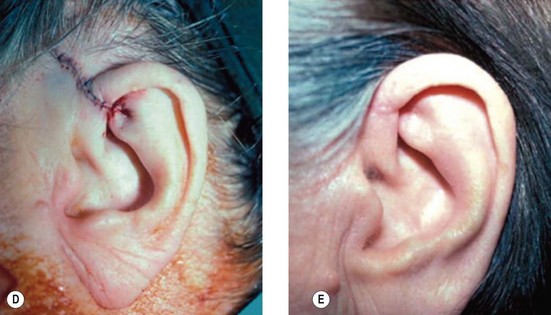
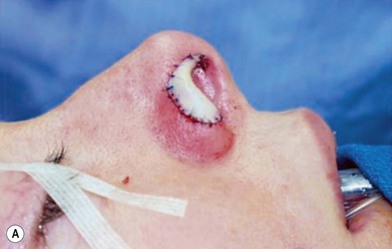
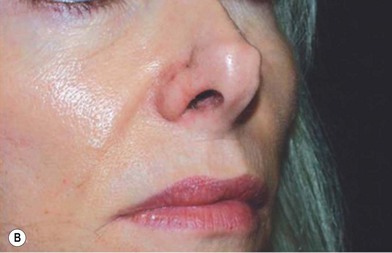
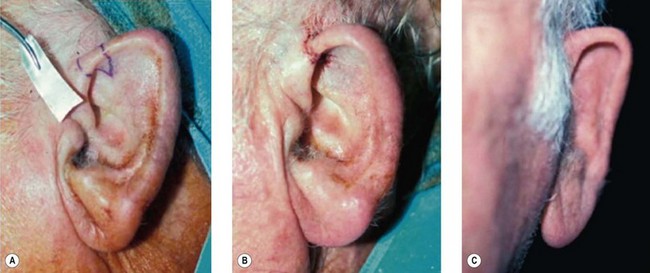
1. Technique 1: A composite graft that is less than 1 cm can be harvested from the root of the helix and the dog ear is run anteriorly toward the hairline with primary closure of the defect (Fig. 20.3).
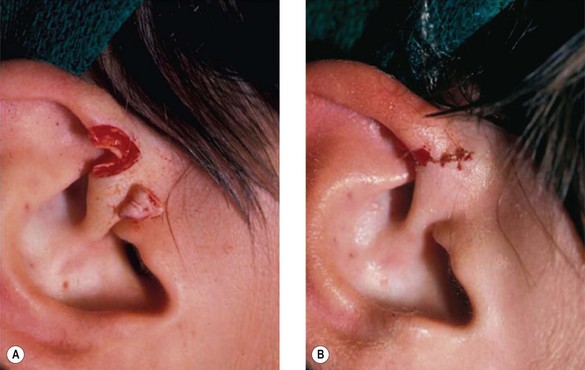
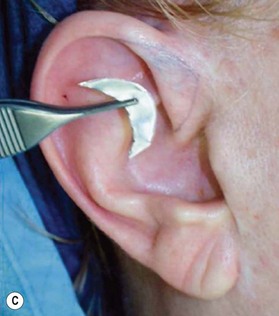
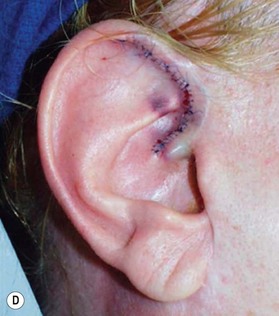
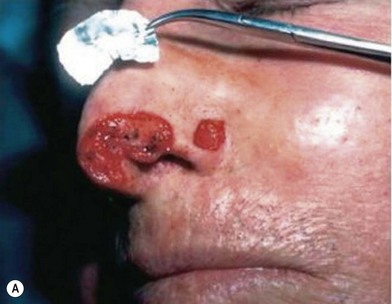
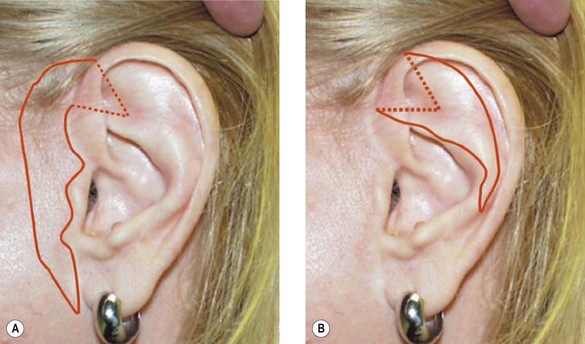
2. Technique 2: This is designed to close the donor site wound of the composite graft with cartilaginous base of 1–1.5 cm, which is usually needed to repair a wider defect of the alar rim (Fig. 20.4).
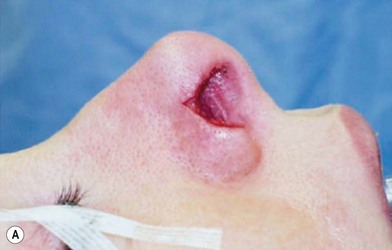
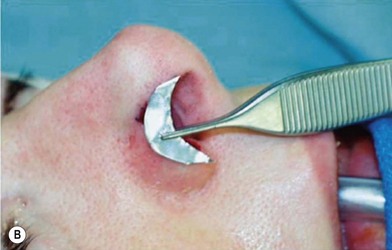
3. Technique 3: To repair a nasal defect with short vertical dimension (height), the composite graft can be harvested from the anterior aspect of the helical root and the dog ear must be run superiorly and inferiorly. As shown in Figure 20.5, this technique can provide adequate tissue for nasal reconstruction and an aesthetic closure without disturbing the size of the ear.
4. Technique 4: For harvesting grafts with width of 1–1.2 cm from the base of the helix, the defect may be closed primarily by advancing the helical rim forward. The possible resulting overprojection of the ear might be addressed with a postauricular incision and a scaphomastoid suture to match the prominence with the contralateral ear (Fig. 20.6).
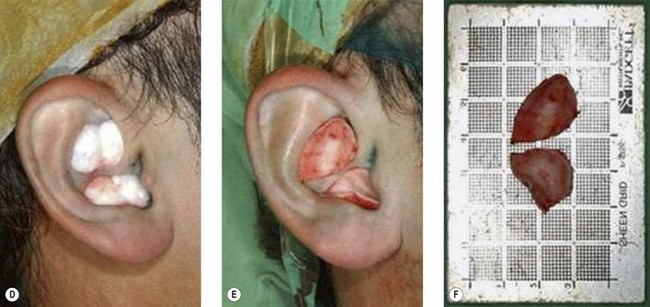
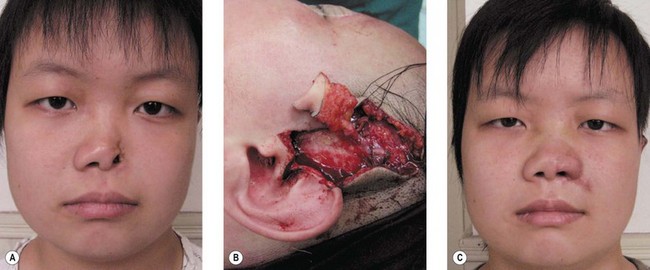
5. Technique 5: After harvesting composite graft wider than 1.5 cm from the base of helix, the closure of such a wound became more intricate. A simple helical advancement would result in significant distortion and overprojection (Fig. 20.7). To overcome this, a V-shaped wedge of skin is harvested, but a “half-star” pattern of cartilage at the apex of the V is taken (Fig. 20.8B–E). In addition, a postauricular incision with a scaphomastoid suture is needed. With these procedures, this technique can prevent cupping of the ear with closure (Fig. 20.8F).
6. When an extended skin graft component is needed with the composite graft, the composite tissues can be taken either preauricularly or postauricularly, as demonstrated by the markings on Figure 20.9.
The techniques described above provide simple methods to close the wounds of auricular chondrocutaneous composite grafts primarily with appreciable aesthetic outcome.12 Other methods of rotational flap or skin graft have also been described in the literature.13,14 Importantly, the functional and aesthetic aspects should be well balanced for both the repair site and donor site when a physician decides to harvest an auricular composite chondrocutaneous graft of a particular size and shape.
One concern for the clinical application of free auricular composite grafts is their limited size due to the lack of immediate blood supply after transplantation. In contrast, microvascularly transferred auricular composite grafts can overcome this limit and are particularly useful when repairing large nasal defects. Zhang et al. have recently reviewed their experiences of surgical treatment of large nasal defects with vascularized preauricular and helical rim flaps based on the superficial temporal vessels in 63 clinical cases. The repaired deformities include unilateral alar defect, alar and side wall defects, tip and columellar defects, entire lower third of the nose missing, and composite defects involving cheek and maxilla. The total flap survival rate reached 97%. The results demonstrate that such an approach, using vascularized preauricular and helical rim flaps, is a reliable method for reconstructing nasal defects.15 Figure 20.10 demonstrates the surgical technique for repairing an alar lobule defect with vascularized helical rim composite tissue and preauricular skin.15
Nasal cartilage graft
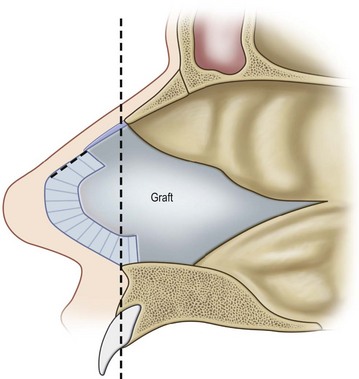
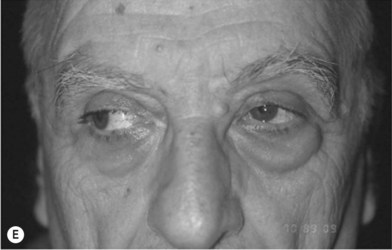
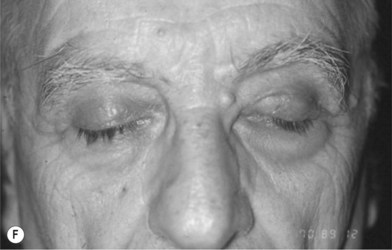
Although limited in its available amount, nasal cartilage has been employed as a composite chondromucosal graft for eyelid reconstruction. Septal cartilage is an important source of nasal cartilage graft. As reported by Murrell, the septal cartilage can be accessed via a hemitransfixion incision with dissection around the caudal margin of the quadrangular cartilage. After both sides of mucoperichondrium are raised, the septal cartilage can be harvested.16 Importantly, as shown in Figure 20.11, an L-shaped septal strut should be preserved to give nasal support and avoid nasal collapse.6,16 Nevertheless, the amount of L-shaped strut cartilage necessary to provide good support can vary greatly depending on the strength, thickness, and dimensions of the nasal septum and other nasal tissues (i.e., upper lateral cartilages, lower lateral cartilages, nasal bones).16 As early as 1962, Millard published his work of repairing eyelid defect with a chondromucosal graft harvested from nasal septum. Later, Mustardé also described the technique in the book Repair and Reconstruction in the Orbital Region.6
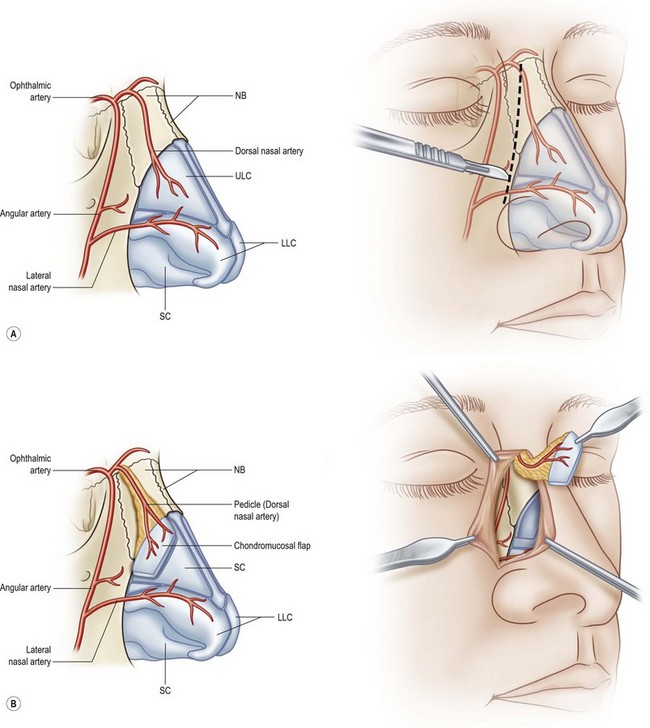
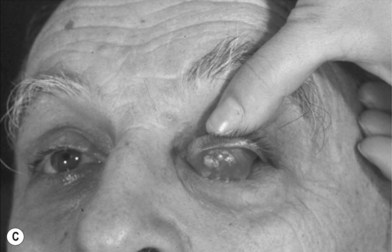
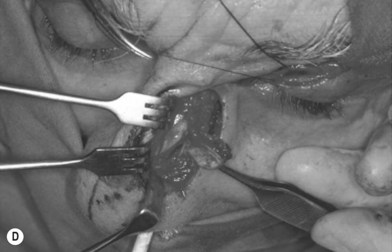
The other region available for harvesting nasal chondromucosal graft is the upper lateral nasal cartilage, as reported by Tessier in 1979.6 In clinical practice, repairing a large defect of the upper eyelid is always a challenge. Scuderi and colleagues have developed a surgical technique to address such a concern. In their report, a pedicled nasal chondromucosal flap was designed along the lateral nasal wall based on the terminal branch of the dorsal nasal artery. The flap includes the subcutaneous tissues down to the periosteum and the cranial portion of the upper lateral cartilage. It can be harvested either unilaterally or contralaterally and a skin graft is applied for cutaneous coverage. In their reported 15 patients, the flap was viable in every patient and satisfactory functional recover was achieved, indicating that this one-stage procedure can reconstruct a thin and mobile eyelid.17 Figure 20.12 demonstrates its design and surgical procedure as well as its clinical outcome.17 In addition to eyelid repair, septal cartilage graft has been used for dorsal augmentation,16 tracheal repair,18 and extended septal graft for controlling the projection shape of nose tip.19
Rib cartilage graft
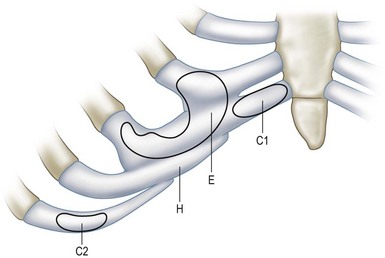
Costal cartilage may serve as the best donor site for cartilage graft in terms of available tissue amount and mechanical strength. The autologous rib cartilage can be virtually contoured into any desired shape and it can retain form and bulk after implantation if basic surgical principles are followed.6 The costal cartilage graft is often used as a cartilage framework for total ear reconstruction. Tanzer,20 Thomson et al.,21 and Brent22 have respectively described the technique to harvest costal cartilage to construct auricular framework. In the procedure, the synchondrosis of the sixth and seventh cartilages as well as the eighth costal cartilage is harvested, usually along with the perichondrium.20–23 In contrast, Nagata’s method for total auricular reconstruction requires the harvest of four costal cartilages in the first-stage operation and one or two costal cartilages for the second-stage operation.24–28 Figure 20.13 is a schematic illustration of the harvest of rib cartilage designed for ear reconstruction or chin contouring.29
In clinical practice, harvest of costal cartilage can be associated with donor site morbidity. Uppal et al. have reported their investigation on morbidity associated with the harvest of costal cartilage in 42 patients who underwent ear reconstruction. The commonest complaints included pain and clicking of the chest wall, which usually peaked in the first week postsurgery and gradually diminished over 3 months. The other problems were scar and chest wall deformity.30 Among them, the most challenging problems are pneumothorax during the surgical procedure and resulting abnormal chest wall contour. This is particularly true for Nagata’s method, which requires more and extra cartilage grafts for auricular reconstruction.24–28
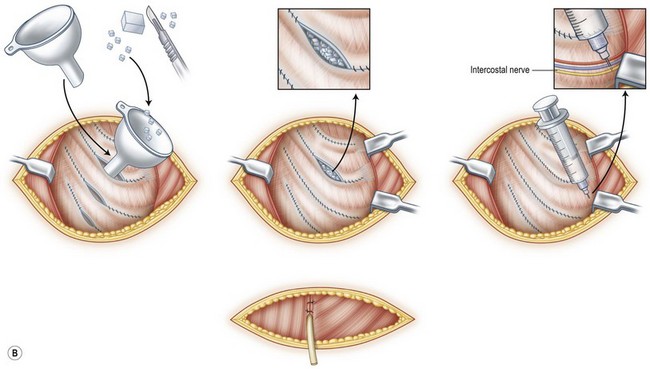
To address this concern, Kawanabe and Nagata developed a new method for harvesting rib cartilage to avoid intraoperative and postoperative complications and problems. In this modified procedure, the costal cartilages were harvested en bloc with the perichondrium left completely intact at the donor site. In addition, after the fabrication of the auricular cartilage frame, the remaining costal cartilage was cut into small blocks that acted as spacers to fill the dead space formed in the perichondrial pocket. The retained perichondrium not only helps to avoid the injury of pleura, but may also promote cartilage regeneration because of the presence of chondrogenic stem cells.2,3 In an investigation of 270 cases of total auricular reconstruction performed using Nagata’s method and the new method of rib cartilage harvesting, the incidence of infection and pneumothorax was reduced to less than 1%. More importantly, there were no postoperative chest wall deformities when costal cartilage was harvested with this new technique.31 Interestingly, a follow-up study revealed that returned hyaline cartilages were mixed with fibrocartilage with visible margins at 6 months postsurgery. Twelve months after the first-stage operation, homogeneous hyaline cartilage was observed histologically, and the regenerated cartilages were similar to native costal cartilages in their hardness, which enabled the second harvest.32 Figure 20.14 gives a schematic illustration of harvesting costal cartilage with the new method of retaining perichondrium and returning the remaining costal cartilage to the donor site.31
(Reproduced from Kawanabe Y, Nagata S. A new method of costal cartilage harvest for total auricular reconstruction: part I. Avoidance and prevention of intraoperative and postoperative complications and problems. Plast Reconstr Surg 2006;117:2011–2018.)