21 Repair and grafting of bone
Synopsis
 This chapter will concentrate on the principles and concepts of bone repair, grafting, and reconstruction.
This chapter will concentrate on the principles and concepts of bone repair, grafting, and reconstruction.
 The embryology, physiology, microanatomy, and histochemistry of bone will be reviewed.
The embryology, physiology, microanatomy, and histochemistry of bone will be reviewed.
 Principles of mechanotransduction and cellular mechanisms of bone turnover will also be discussed.
Principles of mechanotransduction and cellular mechanisms of bone turnover will also be discussed.
 Pathophysiology of traumatic injury to bone (fractures, segmental loss, defects) will be categorized and reviewed.
Pathophysiology of traumatic injury to bone (fractures, segmental loss, defects) will be categorized and reviewed.
 Bone-remodeling mechanisms (osteoconduction, osteoinduction, osteointegration) will be described.
Bone-remodeling mechanisms (osteoconduction, osteoinduction, osteointegration) will be described.
 After a brief history of autogenous bone grafting, the clinical application of bone transfer and transplantation will be captured by a brief atlas of harvest of each subtype.
After a brief history of autogenous bone grafting, the clinical application of bone transfer and transplantation will be captured by a brief atlas of harvest of each subtype.
 The reader will also be provided with a brief overview of bone substitutes.
The reader will also be provided with a brief overview of bone substitutes.
Microanatomy and histochemistry
The formation of bone occurs by two distinct mechanisms: intramembranous ossification and endochondral ossification. Direct condensations of mesenchymal stem cells (MSCs) initiate the process of intramembranous ossification to create the flat bones of the craniofacial skeleton. During intramembranous ossification, osteoblasts that line the surfaces of the skeleton deposit new bone upon previously laid bone in a process called apposition. Alternatively, endochondral ossification occurs when MSCs form a hyaline cartilage template that is subsequently replaced by bone. Endochondral ossification is responsible for the development for the tubular long bones of the appendicular skeleton as well as the bones of the vertebral column and pelvis.1–4
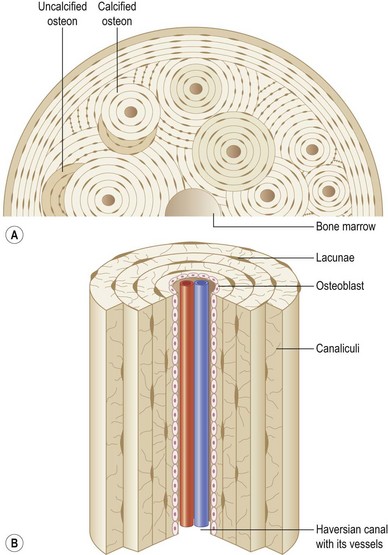
Bone is characterized by the presence of osteocytes located within open cavities within bone, called lacunae. Osteocytes project radiating processes that travel through microscopic canals called canaliculi that adjoin neighboring lacunae. Osteocytic cytoplasmic processes are linked to one another via gap junctions,5 through which osteocytes are thought to communicate.6
Cortical versus cancellous bone
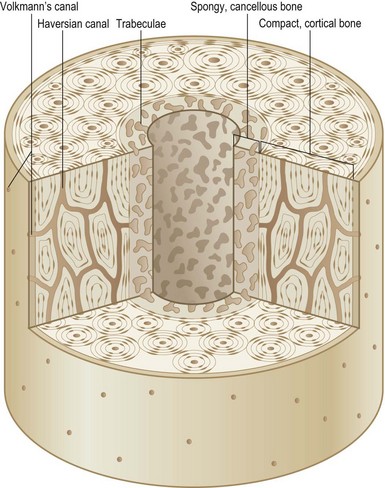
The entire skeleton is comprised of two types of bone: cortical bone and cancellous bone (Fig. 21.1). Cortical or compact bone comprises the outer layer of the skeleton and accounts for 80% of all bony tissue. It is characteristically dense, strong, and stiff, and serves to support the body and protect the internal organs. A two-layered cellular membrane known as the periosteum covers cortical bone. The outer layer is composed of a dense fibrous membrane that contains flat fibroblast-like cells and serves as an attachment for muscles and tendons. Large plump cells line the inner periosteal layer (cambium layer) and can differentiate into osteoblasts when induced. In addition, bone contains a thin layer of vascular connective tissue that lines the medullary cavity, known as the endosteum.
The primary functional unit of cortical bone is the osteon, or haversian system (Fig. 21.2). Each osteon consists of concentric layers of cortical bone tissue (lamellae) that surround a central canal (haversian canal). The haversian canal contains nervous tissue and blood vessels that innervate and supply bone. Nutrient vessels within haversian canals anastomose to blood vessels within the bone marrow and periosteum. Volkmann’s canals run perpendicular to haversian canals and are responsible for the aforementioned interconnection between the haversian efferent vessels and those in the bone marrow and periosteum.
The chemical composition of bone
Inorganic phase
Bone is a calcified tissue composed of 60% inorganic matter, 30% organic matter, and 10% water.7–9 The inorganic component of bone is responsible for approximately 40% of bone volume, while the organic component and water comprise 35% and 25%, respectively.7–10 The inorganic phase primarily consists of calcium phosphate (hydroxyapatite), which has the following chemical formula: Ca10(PO4)6(OH)2. Hydroxyapatite is a mineral crystal that measures 20–25 nm in length, 15 nm in width, and 2–5 nm in thickness.7 It is the hydroxyl endmember of the apatite group; however, impurities arise when hydroxyl groups are replaced by potassium, magnesium, or sodium, or phosphate groups are replaced by carbonate.7,10–14 By storing a mixture of minerals in the form of impure hydroxyapatite, bone serves as an important reservoir of such minerals within the human body.
Organic phase
The organic component of bone, know as demineralized organic bone matrix or osteoid, is deposited by osteoblasts during bone formation. Osteoid primarily consists of type I collagen, but also includes nearly 30 noncollagenous proteins.9 Collagen is a trilaminar protein composed of two α1 chains and one α2 chain that interact to form a unique right-handed helical structure. Within the rough endoplasmic reticulum of osteoblasts, procollagen undergoes hydroxylation and glycosylation before subsequent transfer to the Golgi apparatus, where it is packaged and secreted. Outside the cellular membrane, procollagen peptides are cleaved into tropocollagen. Lysyl oxidase, an important copper-dependent enzyme, mediates covalent cross-linking within and between tropocollagen molecules to create mature collagen fibers. The strength of bone is primarily derived from interactions between collagen fibers and inorganic minerals.
The cellular composition of bone
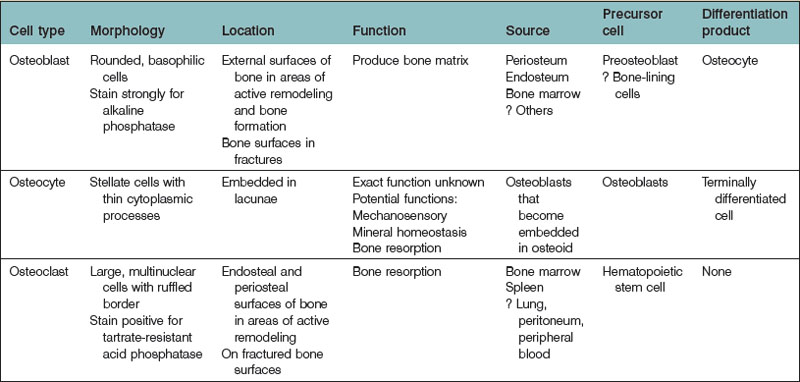
Three cell types are predominantly found in bone: osteoblasts, osteocytes, and osteoclasts (Table 21.1). Osteoblasts are responsible for the formation of bone matrix. Osteocytes are the terminal differentiation product of osteoblasts. Both cell types are derived from a common MSC precursor. Osteoclasts, which are responsible for bone resorption, are derived from hematopoietic stem cells.
Osteoblasts
Histology and function
Osteoblasts are derived from pluripotential MSCs that have the capacity to differentiate into several connective tissue cell types.15 Within the appropriate osteogenic environment, MSCs first differentiate into osteoprogenitor cells and become committed to an osteoblastic lineage. Though they may continue to proliferate, osteoprogenitor cells eventually become preosteoblasts, prior to their maturation into osteoblasts. Osteoprogenitor cells are seemingly ubiquitous in bone, located within Volkmann’s and haversian canals, the cambium layer of periosteum, the endosteum, and within the perivascular tissue adjacent to bone.16
Osteoblasts are plump, basophilic cuboidal or columnar cells located on the external surfaces of bone at sites of active bone formation during bone development and fracture repair. Ultrastructurally, osteoblasts have a vast endoplasmic reticulum capable of abundant collagen production. Osteoblasts also contain many mitochondria and a large Golgi apparatus, both necessary to package and secrete large amounts of procollagen. Osteoblasts are chiefly responsible for the production of the organic component bone matrix. They also play a role in the mineralization of osteoid by aiding the process of hydroxyapatite deposition and also by the liberation of matrix vesicles.16–18 Osteoblasts may eventually surround themselves with bony matrix, at which time that may terminally differentiate into osteocytes.
The phenotypic characteristics of an osteoblast depend upon its stage in maturation. As an osteoprogenitor cell matures into a preosteoblast, it will begin to express enzymes and markers characteristic of the osteoblastic lineage. For example, preosteoblasts, which are typically located near but not at bone-forming surfaces, express low levels of alkaline phosphatase (ALP), a well-described marker of early osteogenesis.4,19,20 Preosteoblasts also produce bone matrix proteins, though not to the extent of mature osteoblasts.
As osteoblasts mature, they begin to take their characteristic basophilic appearance, stain strongly for ALP, and lose the ability to proliferate. In addition to osteoid, osteoblasts secrete numerous noncollagenous proteins, including bone sialoprotein (BSP), osteonectin, osteopontin, and osteocalcin. Osteoblasts also secrete colony-stimulating factors, which play an important role in myelopoiesis.16,21
Mature osteoblasts express receptors for hormones that have important roles in both bone metabolism and mineral homeostasis, including parathyroid hormone receptor (PTH-R) and 1,25-dihydroxyvitamin D receptor (1,25-(OH)2D3-R). Mature osteoblasts also secrete a number of cytokines with many pleiotropic functions, including transforming growth factor-beta (TGF-β), bone morphogenetic proteins (BMPs), insulin-like growth factors (IGFs), and platelet-derived growth factors (PDGFs). Eventually, most osteoblasts undergo apoptosis or become bone-lining cells; nearly 20%, however, differentiate to osteocytes (see below).22
Regulation of osteoblast differentiation
Major signaling pathways
Under the control of multiple signaling pathways, preosteoblasts differentiate into mature osteoblasts. The Wnt pathway,23–26 the TGF-β/BMP superfamily19,27–29 (see below), notch signaling,30–32 hedgehog proteins,33–36 and fibroblast growth factors37–40 (FGFs: see below) have all been implicated in the molecular signaling of osteogenesis. Although the exact mechanisms of osteogenesis are complex and only partially elucidated, advances have been made regarding the initiation and molecular control of this process. Wnt/β-catenin signaling has been shown to control the differentiation of both osteoblasts and osteoclasts, which may be important in postnatal bone acquisition.41 Wnt/β-catenin signaling is also important during skeletogenesis in the fetus, and is considered to be partially responsible for both osteoblast and chondrocyte differentiation.42,43 Notch signaling is a highly conserved signaling system involving cell–cell communication. In addition to cell fate division and homeostatic maintenance, the notch pathway is believed to be important in osteogenesis because of notch1–BMP-2 interactions that promote osteogenic differentiation.44 Additionally, Engin et al.45 showed with osteoblast-specific gain of Notch function studies that Notch not only stimulates terminal osteoblast differentiation, but that it also plays a role in the proliferation of immature osteoblasts. In addition, loss of notch signaling is associated with the expression of an osteoporotic phenotype.45
Hedgehog signaling plays a role in many embryonic processes and also is involved with the maintenance of stem cells in adults. There are three mammalian Hedgehog orthologs: Desert, Sonic, and Indian. Sonic Hedgehog and Indian Hedgehog both have important roles in osteogenesis.33,34,46–49 Specifically, Indian Hedgehog is critical for endochondral bone formation,50 whereas Sonic Hedgehog appears to be important for skeletal patterning.51
Transcriptional regulation
In addition to numerous signaling pathways, many transcription factors play a significant role in osteogenesis. Runt-related transcription factor 2/core-binding factor alpha 1 (Runx2/cbfα-1) and osterix have many important effects on osteoblast differentiation.52–57 Runx2 is the mammalian homologue of the Drosophila transcription factor Runt and is believed to be evolutionarily conserved in humans to serve a critical role throughout the many steps of osteoblastogenesis, including osteoblast induction, proliferation, and maturation. Homozygous deletions of the Runx2 gene are uniformly lethal in mice due to a complete lack of mineralized bone matrix.53 Runx2 haploinsufficiency causes cleidocranial dysplasia, an autosomal-dominant disease in humans characterized by hypoplastic clavicles, dental deformities, shortened stature, brachycephaly, hypertelorism, and other skeletal defects.58 While the exact mechanisms that control the regulation of Runx2 have not been fully decoded, studies have shown that various histone acetyltransferases are important Runx2 cofactors.59,60 Additionally, microRNAs appear to regulate Runx2 protein expression.61 MicroRNAs play critical roles in stem cell function,62,63 and thus may have important clinical implications for diseases due to dysfunctional MSC/Runx2 interactions. Many other regulators also play an important role in Runx2 function64–70; however, their exact roles are currently under study.
The zinc finger-containing protein osterix (Osx) is also a key transcription factor to bone formation.55,71 Osx-null mice form neither cortical nor cancellous bone. Unlike Runx2-null mice, however, osx-null mice still produce normal cartilage.72 As a result of the findings that Osx expression is absent in Runx2 knock out mice, whereas Runx2 expression is normal in Osx-null mice, the Osx gene is thought to be downstream to Runx2.55 Nishio et al.73 confirmed this using overexpression experiments to show that Runx2 transactivates the Osx gene promoter significantly, indicating that there is a Runx2-binding element within the promoter region.
The signaling pathways and transcription factors responsible for osteoblastogenesis are exquisitely detailed and only a brief review has been described. For the reader interested in further study, excellent review articles have been published.74,75
Osteocytes
Histology and function
Osteocytes are terminally differentiated cells of the osteoblast lineage and comprise 90–95% of all bone cells. Despite being the most abundant cell in bone, osteocytes are the least well characterized. Osteocytes are located within lacunae. Multiple cytoplasmic processes radiate from osteocyte somas and travel through canaliculi between lacunae. Gap junction connections between adjacent cytoplasmic processes enable osteocytes to communicate with one another. Osteocytes communicate with osteoblasts by way of this canalicular network as well. Processes also must extend to the haversian canals so that osteocytes can rid themselves of waste products and receive nutrients necessary for survival. Compared with osteoblasts, osteocytes have qualitatively similar cellular organelles, but the organelles are smaller in both size and number.76
Osteocyte functions
Bone is a dynamic substance that maintains its viability by constantly adapting to the environment in which it lives. Victim to mechanical loading and various traumas, bone must have a way to recognize such stressors. It is believed that osteocytes are mechanosensory cells that translate (via mechanotransduction) physical stress into chemical and/or electrical signals that stimulate bone remodeling.77–79 This hypothesis has been derived from various lines of evidence. Anatomically, osteocytes form a syncytium with surrounding cells via their cellular processes. These connections may provide communication between mechanical stimuli and effector cells (osteoblasts and osteoclasts). Recent evidence has shown that osteocyte processes also attach to ECM proteins and may mediate mechanotransduction by the amplification of shear stress experienced at the ECM.80 In addition, mechanical loading alters osteocyte gene expression. Osteocytes rapidly produce increased levels of c-fos, IGF-1, prostaglandin (PG), and nitrous oxide (NO) when mechanically stimulated.81–83 These and many other molecules (see below) have numerous effects on bone turnover. Molecular studies have implicated the activation of the Wnt/β-catenin pathway in response to loading.84,85 The osteocytic Wnt/β-catenin pathway appears to be triggered by crosstalk with various signaling pathways when the osteocyte senses a load strain, thereby decreasing negative regulators of the Wnt/β-catenin pathway,82 an important regulator of bone mass. Finally, Tatsumi et al.86 have shown that targeted ablation of osteocytes induces osteoporosis with defective mechanotransduction.
One potential function of the osteocyte, termed periosteocytic osteolysis, has remained elusive. Osteocytes may fulfill an osteoclast-like activity by resorbing varying amounts of calcified bone matrix surrounding their lacunae. Evidence for periosteocytic osteolysis comes from studies that have shown a larger than expected size of lacunae in conditions characterized by increased bone resorption.87–89 However, criticisms for these studies were raised regarding the methodology used to determine the size of lacunae. More recently, the administration of parathyroid and thyroid hormone has been shown to induce microradiographic enlargement of lacunae, thought to be the result of the hormones’ actions on osteocytes.90,91
Osteoclasts
Histology and function
Bone resorption is a complex process by which crystalline hydroxyapatite is dissolved and organic bone matrix is degraded. In vitro bone resorption models using primary osteoclast isolates provided the basis for the resorption cycle.92,93 The cycle is a complex series of actions which includes osteoclast migration to the resorption site and attachment to bone, membrane polarization, dissolution of hydroxyapatite followed by degradation of organic bone matrix, removal of degradation products, and osteoclast inactivation or apoptosis.94 Clinically, bone resorption is an extremely important process, as evidenced by pathologic conditions characterized by dysfunctional resorption. For example, increased osteoclastic function leads to excess bone resorption, thereby causing osteoporosis. In contrast, osteoclast underactivity decreases bone resorption, which causes osteopetrosis.
Bone resorption is key for both bone remodeling at sites of injury and also as a mechanism of calcium homeostasis. Osteoclasts are stimulated to resorb bone by growth factors and cytokines secreted by local osteoblasts.95 Once osteoclasts localize to the area in need of resorption they form a tight seal that separates the area of bone to be resorbed from the extracellular environment. Evidence has pointed to integrins (in particular integrin αvβ3) and cadherins as playing a prominent role in the attachment of this sealing zone.96–98 Myosin and actin-binding proteins are also important for such attachments.99,100 After sealing zone attachments are completed, osteocytes polarize such that the area adjacent to the resorbing bone surface becomes ruffled. The ruffled border is a specialized membrane domain formed by the fusion of intracellular acidic vesicles and functions as the osteocyte’s resorbing organelle.101,102 Finger-like projections from the ruffled border increase the effective surface area of resorption. Before proteolytic cleavage of the organic matrix occurs, osteoclasts dissolve hydroxyapatite crystals by targeted secretion of hydrochloric acid through the ruffled border toward the resorption pit (Howship’s lacuna).94 After the dissolution of inorganic matter, many proteolytic enzymes, including matrix metalloproteinase-9 and cathepsin K, are secreted into the resorption lacuna to degrade the organic components of bones.103,104 Both phases of bone are subsequently removed from the resorption lacuna via transcytosis through the ruffled border and sent to the secretory domain of the osteoclast, which then expels these degradation products into the extracellular space outside the osteoclast.105–107 The osteocytic marker tartrate-resistant acid protease (TRAP) has been localized to transcytotic vesicles of resorbing osteoclasts, and is believed to generate reactive oxygen species that degrade matrix degradation products within these vesicles.108
Osteoclast differentiation
The mechanisms surrounding osteoclast differentiation are complex and have been the subject of intense study. Depending on local stimuli, hematopoietic stem cells may differentiate into osteoclasts, macrophages, or dendritic cells. Macrophage colony-stimulating factor (M-CSF) and receptor activator for nuclear factor κ B (RANK) and RANK ligand (RANKL) are early mediators that signal osteoclastogenesis.109–114 In murine studies, M-CSF is functionally lacking in op/op mice, which express an osteopetrotic phenotype.115,116 In addition, in order for osteoclastogenesis to commence, RANKL (found on the cellular membrane of osteoblasts) must interact with RANK, a receptor found on osteoclast precursors.117–119 RANK–RANKL interaction must occur in order for osteoclast precursor cells to begin expressing phenotypic markers (e.g., TRAP) that characterize osteoclasts. Similar to op/op mice with functionally absent M-CSF, RANK and RANKL knockout mice also express an osteopetrotic phenotype.112,114,120
Osteoprotegerin (OPG), a TNF-related soluble protein secreted by osteoblasts, is also an important regulator of osteoclastogenesis. OPG has effects on bone density and bone mass117,121 by acting as a decoy receptor for RANK, thereby blocking RANK–RANKL interaction. Transgenic mice that overexpress OPG suffer from an osteopetrotic phenotype,121 while OPG knockout mice display an osteoporotic phenotype.122,123
The RANK molecule lacks enzymatic activity, and thus must recruit adaptor proteins when stimulated by RANKL to promote differentiation.124–126 TNF receptor-associated factor (TRAF) family members appear to be important adaptor proteins, with TRAF6 serving the most critical role.127–129 TRAF6 knockout mice develop severe osteopetrosis as a result of either the dysfunction or total absence of osteoclasts.130,131 Moreover, TRAF6 activates NF-κB, another essential modulator of osteoclast differentiation.132,133 NF-κB enters the nucleus of osteoclasts stimulated with RANKL, and regulates transcription of target genes critical for osteoclastogenesis.134
Extracellular matrix
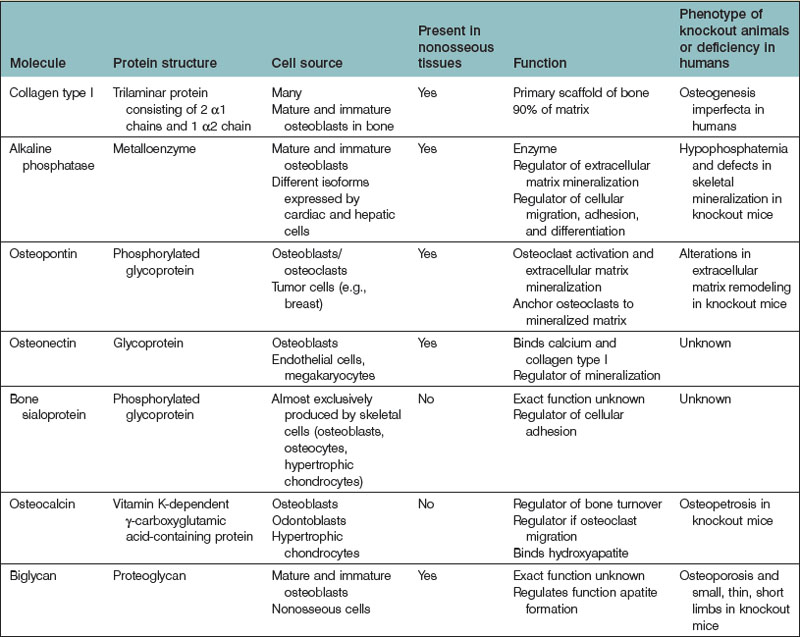
The extracellular matrix (ECM) is a complex scaffolding from which bone derives much of its strength and characteristic architecture. Most molecules found within the ECM are produced by osteoblasts. The ECM contains nearly 30 types of collagen, as well as noncollagenous phospho- and glycoproteins. Type I collagen is the predominant form found within the ECM, making up approximately 90% of bone matrix. The significance of type I collagen is highlighted by patients with osteogenesis imperfecta (OI), a disease characterized by bone fragility and repeated fracture. OI often occurs in patients with pro-α1 or pro-α2-chain gene mutations, or from a mutation in one of the enzymes responsible for posttranslational hydroxylation of collagen.135 In addition, bone mineralization takes place within the ECM. Bone mineralization is a well-orchestrated process during which hydroxyapatite crystals are laid down within the organic matrix of bone and calcify proteins that are secreted by osteoblasts.
In addition to type I collagen, many noncollagenous proteins are found within the ECM (Table 21.2). Osteopontin is a ubiquitous phosphoprotein that exists in multiple forms. It has been localized to most cells within bone, including osteoblasts, osteocytes, osteoclasts, and bone precursor cells.136–138 Osteopontin plays a role in the nucleation of hydroxyapatite crystals during matrix mineralization and also in osteoclast adhesion during bone remodeling.139–144
Osteonectin is a glycoprotein that binds to calcium and type I collagen and also initiates and regulates bone mineralization.145,146 BSP is another important noncollagenous protein that, in contrast to osteopontin and osteonectin (both of which are expressed by many tissues throughout the body), is mainly expressed by cells within the skeleton.147 BSP is an acidic phosphoprotein that binds to collagen and also promotes hydroxyapatite nucleation.148 BSP-null mice express a phenotype characterized by small and undermineralized bones.149
Osteocalcin (bone Gla protein) is a vitamin K-dependent γ-carboxyglutamic acid-containing protein, similar to factors II, VII, IX, and X of the coagulation cascade. The most abundant noncollagenous protein in bone, osteocalcin is secreted by osteoblasts and odontoblasts and is an important marker of increased bone turnover.150 Osteocalcin plays a role in bone turnover,151,152 osteoclast differentiation,153 and energy metabolism.154,155
Osteoblasts also produce proteoglycans, the most abundant of which is biglycan (BGN). BGN, a leucine-rich noncollagenous protein, is believed to play a role in the mineralization of bone given that bgn-deficient mice express an osteoporotic phenotype.156 However, BGN seems to play an important role outside the skeleton as well, as bgn-knockout mice manifest spontaneous aortic rupture and dissection,157 dental and muscular abnormalities, thinning of skin, and osteoarthritis.158
A variety of enzymes produced by bone cells also function within the ECM. The most prominent is ALP. Like osteocalcin, ALP is often used as a marker of bone turnover.159 While the exact function of ALP is unknown, it is believed to be important for bone mineralization, during which it regulates apatite formation.
Principles of bone homeostasis and turnover
Unique to bone is its ability to undergo regeneration rather than repair with scar formation, as is typified by most other tissues within the body. As described previously, bone is subject to constant remodeling, a process mediated by osteoclasts and osteoblasts. Physiologically, bone homeostasis is maintained by the counterbalancing of bone resorption with formation, and vice versa. When one of these two processes becomes dysfunctional, patients often manifest a distinct pathology. Excessive bone resorption resulting in an excessive loss of bone is the fundamental pathogenesis underlying osteoporosis.160 Conversely, decreased bone resorption due to decreased osteoclast activity leads to an osteopetrotic phenotype.161 Decreased bone resorption due to increased osteoblast activity, however, will produce an osteosclerotic phenotype.
The structure of bone is a function of its material composition as well as the genetic blueprint of an individual. In addition, various loads to which bone is subjected define its structure. This process of functional adaptation mediates repair of damaged bone and also facilitates prevention of damage before it may occur. Bone exhibiting suppressed remodeling and resorption suffers increased microdamage and fracture accumulation.162,163 It has been demonstrated that sites of microcracks are subsequently associated with new bone remodeling more often than expected by chance,164–166 which has led to the belief that bone remodeling and repair are often targeted to specific locations.167
Wolff’s law and mechanotransduction
Wolff (1892) derived a mathematical formula to describe how changes in the form and function of bone could lead to a change in its internal architecture and external structure.168 Wolff believed that, during the functional adaptation of bone to new loads that occur during one’s life (e.g., as a result of trauma), the trabeculae within bone would reorient to align with the new principal stress trajectories of these environmental loads. While various authors have criticized Wolff’s beliefs because he did not take into account that bones are subject to dynamic applied loads,169 the overarching importance of his contributions to our understanding of bone adaptation cannot be denied.
Since Wolff’s time, ongoing research has examined the initial events that stimulate bone adaptation. It is believed that the stimulus for bone remodeling is strain (the physical deformation of bone tissue).170 Osteocytes, the mediators of mechanotransduction,171 respond molecularly to mechanical loads. Osteocytes undergo direct deformation by bending and also are deformed by the electrical potential induced by the flow of surrounding interstitial fluid.172,173 In addition, osteocytes are subjected to shear stress by the physical flow of interstitial fluid around their cellular membranes.174–178
When strained, osteocytes convert the mechanical stimulus into chemical or electrical activity in a process thought to require various signaling molecules, including NO and PGs.171 Interestingly, NO synthase, a calcium/calmodulin-dependent enzyme, becomes activated under conditions of increased intracellular calcium; intracellular calcium concentrations appear to increase in response to fluid flow.179,180 Not surprisingly, Klein-Nulend and colleagues181 demonstrated that a pulsating fluid flow model led to the release of NO from osteocytes. Taken together, these studies indicate that NO may be important in osteocyte mechanotransduction.182 Additional studies have showed that pulsating fluid flow treatment also stimulates osteocytes to synthesize increased levels of PGE2 and PGI2,183,184 as well as increased levels of cyclooxygenase-2 (COX-2) mRNA.185 COX-2 production is also stimulated by mechanical loading.186,187 Thus, COX-2 induction and the production of PGs are thought to be important mediators in the conversion of mechanical shear forces into signals that trigger bone turnover.
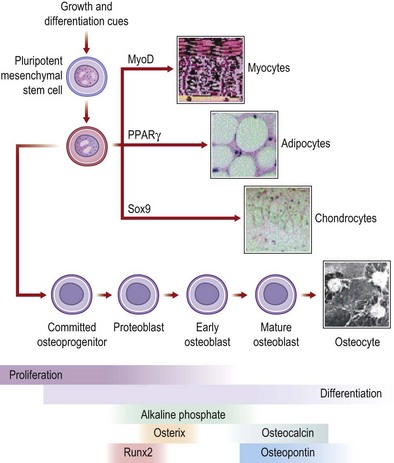
Bone regeneration: the role of the stem cell
In the last decade, the field of regenerative medicine has exploded with the discovery of new techniques to isolate cells capable of differentiating into many different tissues. In particular, the MSC (Fig. 21.3) has considerable therapeutic potential for fracture repair and other bone pathologies. For concordance, Dominici and colleagues188 have proposed that MSCs meet the following criteria: (1) isolated MSCs must adhere to plastic in culture; (2) MSCs should express the CD105, CD73, and CD90 surface antigens as well as not express the CD34, CD45, CD14 or CD11B, CD79A or CD19 and human leukocyte antigen-DR (HLA-DR) surface antigens; and (3) MSCs must have the capacity to differentiate into osteoblasts, adipocytes, and chondroblasts. MSCs and MSC-like are derived from bone marrow, but can also be expanded from skeletal muscle,189 adipose tissue,190 dental pulp,191 the circulatory system,192 synovium,193 amniotic fluid,194 the umbilical cord,195 and fetal tissues.196,197
The capability of MSCs to differentiate into and assist with the restoration and repair of bone has been extensively studied.198,199 MSCs are also used clinically in patients with various bone disorders.200 In a clinical trial200 that examined MSC administration to children with OI, bone mineral density, growth velocity, and ambulation all improved after MSC administration. However, the concentration of MSCs detected in bone, skin, and other tissues was less than 1%. These data and evidence from additional studies201,202 have led authors to postulate that MSCs may actually secrete soluble factors that alter tissue microenvironment, which may be an important mechanism by which MSCs repair tissue.203,204
Molecular mechanisms of bone regeneration
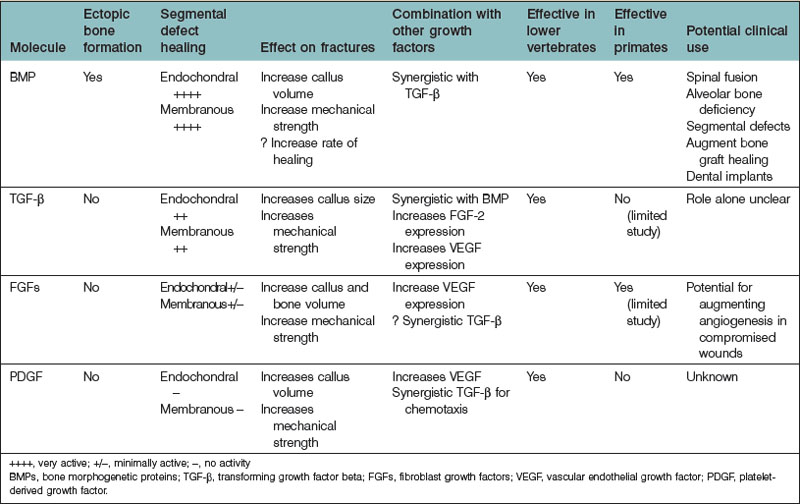
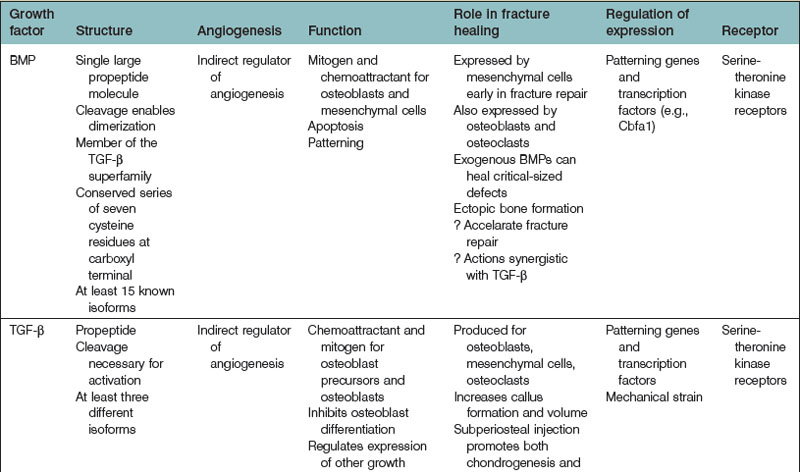
Bone regeneration consists of a complex interplay of molecular processes that promote MSC migration, proliferation, and differentiation. Recently, there has been a great deal of research aimed at the identification of these molecular processes, and advances in our understanding of the molecular mechanisms of bone regeneration have been made. Many have been able to identify important signaling molecules as well as transcriptional regulators of bone regeneration (Table 21.3).
Bone morphogenetic protein
In the 1960s, Urist205 discovered the function of BMP from their work with demineralized bone matrix (DBM). To date, approximately 20 BMP isoforms have been characterized. BMPs are pleiotropic members of the TGF-β superfamily. BMPs, while important for brain and bone formation in utero,206–208 have generated much of their recognition from their osteoinductive properties.
BMPs are synthesized as large precursor molecules that consist of characteristic subsections. A series of seven cysteine residues are located at the carboxyl termini of BMPs, and is important for proper protein folding. BMPs also contain a signal peptide, a prodomain, and a mature peptide.209 BMPs are thought to be secreted from cells in their active form.
BMPs are natural ligands for type I and type II serine/threonine kinase transmembrane cellular surface receptors. There are three type I and three type II receptors that are capable of binding BMPs.208 Upon ligand (BMP) binding, a heterotetrameric complex is formed, after which type II receptor kinase domains phosphorylate Gly-Ser domains in the type I receptor kinases. This process activates type I receptors to recruit and phosphorylate intracytoplasmic Smad proteins (Smads 1, 5, and 8). Following Smad phosphorylation, Smad 1, 5, or 8 will bind to Smad 4, and this Smad4-phosphorylated Smad 1, 5, or 8 complex will relocate to the cell nucleus. Transcription factor activation occurs, leading to the transcription of bone morphogenetic target genes.210
Bone morphogenetic protein function
BMP exerts pleiotropic effects throughout the lifespan of an organism. In the early stages of human development, BMP plays a key role in skeletogenesis.207 Also during this time period, BMP signals epidermal induction,211 directs the development of neural crest cells,212 and induces a sympathetic adrenergic phenotype.213 BMP-2-null mice typically die between 7 and 10 days of gestation due to cardiac defects, even before the formation of bone begins.
BMP is the only signaling molecule capable of singly inducing de novo bone formation. It has been hypothesized that BMP-2, -6, and -9, and to a lesser extent, -4, and -7, various BMP isoforms, have the greatest osteogenic capacity.19,209 Luu and colleagues19 infected HEK293, C2C12, and C3H10T1/2 cells with adenoviral vectors containing various BMP isoforms (AdBMP). The authors measured the expression of early (ALP) and late (osteocalcin) osteogenic markers in vitro. They also assessed in vivo induction of heterotopic ossification by implanting AdBMPs into nude mice and evaluating radiographic and histologic results at 3 and 5 weeks. BMP-2, -4, -6, and -7 were shown to induce increased ALP elevation compared to other BMPs. In addition, BMP-2, -4-, -6, -7, and -9 stimulated increased osteocalcin expression compared to others. The authors also found that BMP-2, -6, -7, and -9 induced the greatest degree of osteogenesis in vivo. The authors concluded that BMP-2, -6, and -9 have the greatest osteogenic potential, and that BMP-4, and -7 also have a significant degree of osteogenic potential.
BMP-2 promotes osteoblast differentiation by stimulating Runx2 in a process mediated by Smad proteins.214 In addition, BMP-2 affects osteoblast differentiation via activation of the β-catenin signaling pathway.215 Interestingly, endogenous activation of β-catenin induces ALP mRNA expression; however, BMP-2 must be present for β-catenin signaling to induce osteocalcin expression as well.216 Thus, β-catenin-dependent differentiation processes likely require BMP-2 to promote the later stages of osteoblast differentiation. Recently, it has been demonstrated that BMP-2-mediated osteoblast differentiation is also dependent upon Akt2 selective signaling.217 IGFs are important for osteoblast differentiation and normal bone growth,218–221 and Akt2 is a key molecule within the insulin signaling pathway. Mukherjee et al.217 showed that, although BMP-2 signaling is normal, osteoblast differentiation does not occur in Akt2-knockout mice. Delivery of Runx2 to Akt2-deficient mice restores osteoblast differentiation; thus, Akt likely serves a specific role in osteoblast differentiation through its regulation of Runx2 gene expression.
Furthermore, BMP-2 appears to improve bone healing in large osseous defects. Many studies have evaluated recombinant human BMP-2 (rhBMP-2) in in vivo animal models. In rabbits, rats, dogs, and nonhuman primates, rhBMP-2 has been shown to augment bone growth successfully and also close critical-sized osseous defects.222–226 Both retrospective and prospective studies have assessed the efficacy of rhBMP-2 in humans as well.227–236 Slosar et al.237 demonstrated that anterior lumbar allografts filled with rhBMP-2 experienced higher fusion rates than lumbar allografts without rhBMP-2 in patients who underwent lumbar fusion. The authors demonstrated that at 6 months and at 2 years, allografts with rhBMP-2 shortened the timeframe for expected improvement, and also improved patients’ Oswestry Disability Index scores. Thus, it has been concluded allografts with rhBMP-2 can offer reliable and clinically significant benefits.
At the present time, rhBMP-2 (INFUSE, Medtronic, Inc.) is one of only two rhBMPs that are approved by the Food and Drug Administration (FDA) for human application. The other is rhBMP-7 (rhOP-1). As described, rhBMP-2 is often used in spinal fusion, as well as orthopedic trauma and dental procedures. rhBMP-7 has shown clinical benefits in spinal fusion and in tibia repair.238,239 Interestingly, although BMP-2 may be more osteoinductive than BMP-7 based on in vitro analyses,29 there has been relatively little in vivo data comparing the two. Barr and colleagues240 have found that rhBMP-7 promotes similar bone quality but greater bone volume induction than rhBMP-2 in murine muscle pouch assays analyzed by microscopic computed tomography and histology. However, other studies have found different results.28,241 Conflicting results among these studies may be due to differences in experimental design.
Transforming growth factor-β
TGF-β also has protean effects on cellular processes within the human body. TGF-β has been implicated in cell cycle regulation, angiogenesis, wound healing, and skeletogenesis. TGF-β also appears to have significant roles in human disease.242 There are three distinct human TGF-β isoforms (TGF-β1, -β2, and-β3). Though they exhibit differences, each isoform shares similar functions with one another, including roles pertaining to the regulation osteogenesis and osseous repair.
TGF-β, like BMP, contains a cluster of conserved cysteine residues.243 In addition, each isoform is produced as a precursor molecule consisting of TGF-β and a propeptide region. In contrast to BMPs, however, TGF-β is secreted as an inactive molecule and stored in its latent form within the ECM. TGF-β becomes active after the noncovalent disulfide bond linking TGF-β and its propeptide region is broken.
TGF-β signal transduction has been the subject of much study.243–245 There are four TGF-β receptors identified to date: types I, II, and III, and endoglin. TGF-β-initiated signaling, like BMP-mediated signaling, uses Smad protein intermediates. Active TGF-β binds to either type III or type II receptors, both of which promote the phosphorylation of type I receptors. Phosphorylation of Smad2 or Smad3 follows, which in turn leads to the binding of Smad4 to the phosphorylated Smad2 or Smad3 proteins. This complex translocates to the cellular nucleus, where transcription factors activate specific target gene transcription, thereby mediating the effects of TGF-β at the cellular level.
Evidence that TGF-β may play a role in osteogenesis has come from experiments demonstrating that TGF-β stimulates the production of collagen and osteopontin, as well as studies demonstrating that TGF-β is produced by osteoblasts.246–250 In addition, many in vivo studies have demonstrated an osteoinductive effect of TGF-β.251–253 TGF-β1, in particular, has been the subject of a considerable body of literature with respect to its osteogenic potential, in part because it is the dominant TGF-β isoform expressed by bone cells.254 In vitro analyses have demonstrated that TGF-β1, by recruiting and stimulating the proliferation of osteoblast progenitors, increases bone formation.255 Interestingly, TGF-β1 seems to have a differential effect on osteoblast differentiation. Early phases of differentiation appear to be promoted by TGF-β1, while later phases are blocked.256
In addition, TGF-β1 modulates the processes of osteoclastogenesis and bone resorption. At high TGF-β1 concentrations (0.1–10 ng/mL), osteoclastogenesis appears to be downregulated.255 TGF-β1 binds to its receptor on osteoblasts and augments the expression of OPG while also decreasing RANKL expression. A lower RANKL : OPG ratio results in maximal RANKL occupation by OPG, thus decreasing maturation of osteoclasts. TGF-β1 appears to serve a bifunctional role, however, as low concentrations are associated with high levels of both RANKL and RANK, and subsequent osteoclastogenesis.
TGF-β1 may also be important for the coupling of bone resorption and formation, as is found at bone resorption sites. Along these lines, Tang and colleagues257 injected green fluorescent protein (GFP)-labeled osteogenic bone marrow stromal cells (BMSCs) into the femur cavity of Tgfb1+/+ and Tgfb1−/− immunodeficient mice. Using anti-GFP antibodies, injected BMSCs were detected at both 1 and 4 weeks. After 1 week, BMSCs were localized to the bone surfaces in Tgfb1+/+ mice; BMSCs were widely dispersed in the bone marrow (i.e., not at bone surfaces) of Tgfb1−/− mice. After 4 weeks, BMSCs were embedded in trabecular bone in Tgfb1+/+mice but not in Tgfb1−/− mice. The authors concluded that TGF-β1 plays an essential role in the coupling of bone resorption to bone formation by their induction of osteogenic BMSCs to sites of resorption.
In addition, TGF-β expression is elevated during fracture repair.258 Given its apparent role in many physiologic bone processes, TGF-β has been considered for exogenous use to augment bone growth and repair. Studies support that TGF-β isoforms, both alone and in conjunction with other cytokines, can significantly induce the formation of endochondral bone at extracranial sites in rapid fashion.259,260 However, studies observing the effects of TGF-β on calvarial defects revealed little osteoinductive potential.261,262
Fibroblast growth factor
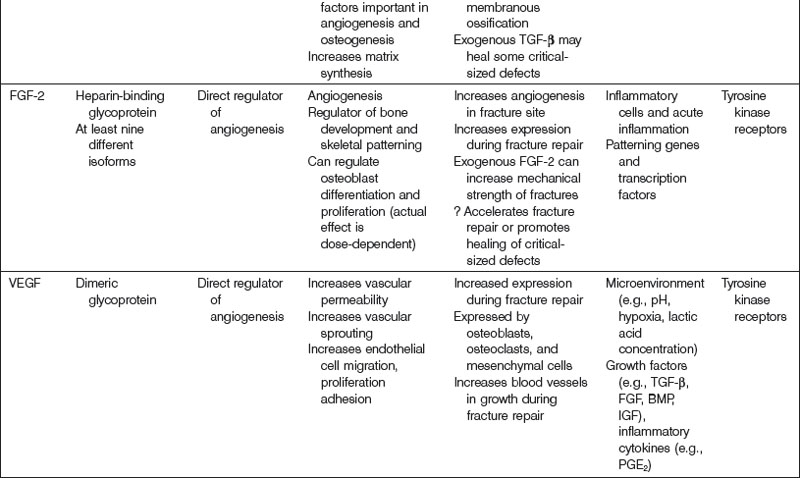
FGF consists of a family of structurally related cytokines that mediate processes such as cellular proliferation, migration, and differentiation, as well as mitogenesis, angiogenesis, embryonic development, and wound healing. There are at least 23 known members of the FGF family, all possessing a characteristically high affinity for heparin. Most FGF isoforms share a similar internal core region that has important FGF receptor (FGFR)-binding properties.263 Mutations in FGFs or FGFRs are thought to play an important role in the development of various skeletal dysplasias, including achondroplasia and craniosynostosis.38,264,265
There are four known FGFRs (FGFR-1, -2, -3, -4). Following FGF ligand binding, receptor dimerization occurs, which promotes the transphosphorylation of each receptor monomer by an intrinsic tyrosine kinase domain. Phosphorylated tyrosine residues on FGFRs become docking sites for adaptor proteins necessary for downstream signaling.266 Human gene studies have identified many associations between diseases of bone and the skeleton and FGFRs. Specifically, Pfeiffer syndrome has been linked to FGFR1 gene mutations,267 Crouzon syndrome to FGFR2 gene mutations,268 and achondroplasia to FGFR3 gene mutations.269
FGF has been implicated in a variety of osteogenic mechanisms. FGF-2, in particular, is thought to play an important role in endochondral bone growth. Coffin et al.270 have found that increased concentrations of FGF-2 are found at the epiphyseal growth plate, specifically within the proliferation and hypertrophic zones. FGF-2 also appears to accelerate fracture repair as well as closure of critical-sized defects in endochondral bone.271 In addition, FGF-2 appears to affect cell function during intramembranous bone growth.265 There appears to be a differential effect of FGF-2 treatment with respect to its method of delivery. Although continuous FGF-2 treatment stimulates osteoblast proliferation, it decreases levels of differentiation markers (e.g., ALP) and augments osteoclast formation, thus resulting in net bone resorption. Oppositely, intermittent FGF-2 treatment enhances both in vitro and in vivo bone formation.
The effects of FGF-2 in combination with other growth factors on osteoblast differentiation have also been studied. Maegawa and colleagues272 examined MSCs deposited in osteogenic media supplemented with BMP-2, FGF-2, or both. MSCs supplemented by both FGF-2 and BMP-2 concurrently expressed the highest levels of ALP, osteocalcin mRNA, and bone matrix formation. In addition, Sabbieti et al.273 investigated whether FGF-2 modulates the anabolic response of bone to PTH. Primary calvarial osteoblasts were isolated from FGF-2+/+ (wild type) and FGF-2−/− mice. By immunocytochemistry, FGF-2-null osteoblasts expressed decreased Runx2 protein synthesis as well as significantly less Runx2 expression within perinuclear and nuclear spaces than wild-type variants when exposed to PTH. Given the importance of Runx2 on osteogenesis (see earlier), it was concluded that FGF-2 expression is required for PTH to promote an anabolic response by bone.
Platelet-derived growth factor
PDGF plays an important role in a number of biological processes, including embryological development, inflammatory reactions, angiogenesis, organogenesis, and wound healing. Four unique isoforms of PDGF have been characterized (PDGF-A, -B, -C, -D), which are inactive in their monomeric forms, and thus must dimerize to become active. Five dimeric PDGF compounds have been indentified (PDGF-AA, -BB, -CC, -DD, and -AB).274 All PDGF molecules share a highly conserved PDGF/vascular endothelial growth factor (VEGF) homology domain, a motif that is also found in VEGF.275 PDGFs interact with two receptor tyrosine kinases, termed PDGF receptor-α (PDGFR-α) and PDGFR-β. PDGFRs as well must dimerize in order to activate.276 Receptor dimerization results in autophosphorylation at a specific tyrosine residue on a PDGFR, a necessary step for signal transduction to proceed. Although it has been shown in tissue culture-based systems that PDGF can promote similar responses by interacting with PDGFR-α or PDGFR-β, it is thought that these two receptor types in fact mediate distinct events during organism development because of their unique temporospatial expressions in vivo.277
PDGF is a potent mitogen and chemotactic agent for cells and tissues of mesenchymal origin and is crucial in bone homeostasis and repair. At sites of bone fracture, platelets congregate and release PDGF. PDGF attracts many other cell types important for tissue repair and the formation of granulation tissue. PDGF has also been shown specifically to attract osteogenic cells derived from calvaria, periosteum of long bones, trabecular bone, and BMSCs.278 In addition, angiogenesis is crucial for bony healing (to be discussed later) and PDGF, in addition to its direct mitogenic effect on osteoblasts, indirectly enhances bone regeneration by stimulating angiogenic factors such as VEGF.279,280
PDGF also appears to stimulate bone healing indirectly via interactions with other growth factors. Levi et al.281 studied the ability of IGF-1, PDGF-A, and a mixture of the two to induce osteogenic differentiation. IGF-1 promoted differentiation of human adipose-derived stromal cells down an osteogenic lineage, whereas PDGF-A did not. IGF-1 and PDGF-A in combination, however, enhanced osteogenesis more than IGF-1 alone. This finding may be the result of enhanced IGF-1 transcription in the presence of PDGF-A. In contrast, PDGF-BB may decrease IGF-2 mRNA expression by osteoblasts.282
Clinical studies of the effects of PDGF on osseous repair have been performed mainly within the disciplines of periodontology and orthopedics. For example, a randomized controlled trial was performed assessing the effectiveness and safety of the administration of 300 µg/mL of recombinant human PDGF-BB (rhPDGF-BB) with beta-tricalcium phosphate (β-TCP) for the treatment of advanced periodontal osseous defects.283 Compared to β-TCP alone, β-TCP plus rhPDGF-BB treatment promoted significantly greater linear bone gain as well as percent defect fill at 6 months. Patients who received rhPDGF-BB were also found to have a significant gain of clinical attachment level compared to patients who did not receive treatment with rhPDGF-BB at 3 months. Moreover, the efficacy of rhPDGF-BB in a β-TCP matrix as a bone graft material was evaluated in a pilot study of 20 adult patients with ankle or hindfoot fusion.284,285 Patients were randomly assigned to receive either a β-TCP matrix enhanced with rhPDGF-BB or an autologous bone graft. At 12 weeks postsurgery, computed tomography scan results showed that in 7 of 11 (64%) patients who received the β-TCP matrix with rhPDGF-BB, there was greater than 50% osseous bridging across the fusion site. Greater than 50% osseous bridging across the fusion site was found in only 3 of 6 (50%) patients who underwent bone graft with autologous tissue. These preliminary results have paved the way for a larger study of approximately 400 patients for which the results have not yet been published.278
Healing of fractures
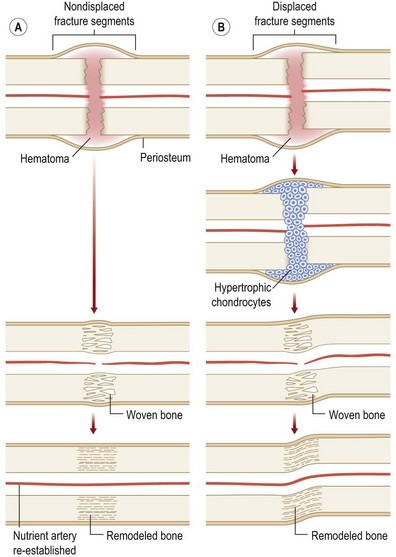
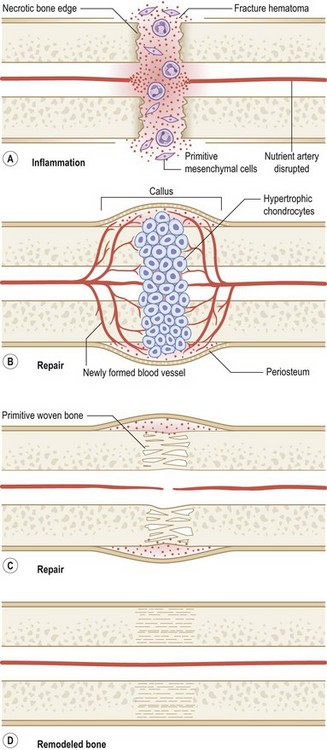
Originally distinguished based on their histology, two types of bone repair have been described (Fig. 21.4). Primary bone repair refers to direct cortical healing of two fracture ends without a cartilaginous intermediate. During secondary (callus) bone repair, a cartilaginous intermediate is formed by mechanisms that occur within the periosteum, soft tissues, and bone marrow. Most fracture repair is by secondary bone healing.
Primary bone repair
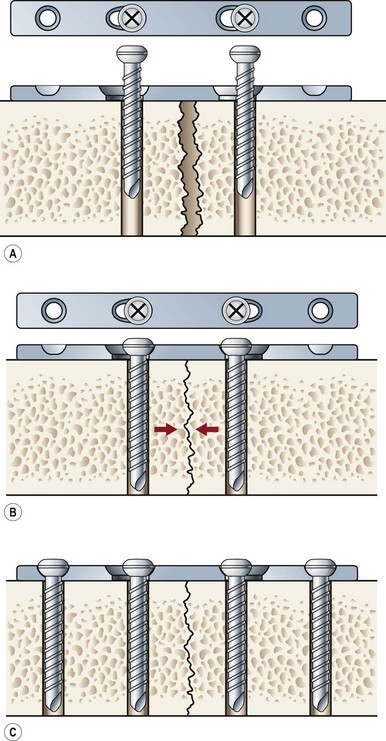
Primary bone repair involves the direct deposition of woven bone by osteoblasts to re-establish mechanical continuity between fracture fragments. Similar to intramembranous ossification, the periosteum provides osteoprogenitor cells and undifferentiated MSCs during primary bone repair. In order for primary bone repair to occur, the fracture fragments must be reduced to anatomic position.286 This is usually accomplished through rigid internal fixation. The interfragmentary strain must also be at a minimum for direct bone healing to occur.286 In addition, bone healing occurs under axial compression and fails under tension (Fig. 21.5).
During primary bone repair, new bone is synthesized in parallel to the long axis of the bone by osteoprogenitor cells. This process is initiated by the formation of cutting cones at the junction of live and necrotic skeletal segments. A cutting cone is a system of cells, mainly composed of active osteoclasts. It burrows its way through cortical bone across the fractured bone segments, thereby allowing the ingrowth of blood vessels and undifferentiated MSCs. MSCs then differentiate into bone-producing osteoblasts. New haversian systems are created and provide pathways for blood vessel penetration. Initially, bone is laid down in a woven-matrix configuration. Within a few weeks, the woven bone becomes more lamellar-like in its orientation. As the proportion of lamellar bone increases over the course of months, complete fracture repair occurs. The benefits of rigid plate osteosynthesis include increased stability, decreased incidence of non- and malunion, and decreased incidence of infection.287 Orthopedic surgeons, as well as cardiac surgeons, commonly perform rigid internal fixation.288
Secondary (callus) bone repair
Fractures left untreated or those that are treated with external or intramedullary fixation or by sling or cast immobilization heal by secondary bone repair without rigid fixation. During secondary or callus bone repair, undifferentiated MSCs proliferate and differentiate into cartilage prior to bone formation at the fracture site. This involves a combination of both intramembranous and endochondral ossification. There are the three overlapping phases of secondary bone repair: the inflammatory phase, reparative phase, and remodeling phase (Fig. 21.6). However, at least five distinct stages of healing have been described.289
When a bone fractures, the disruption of osseous tissue, surrounding soft tissue, and blood vessels initiate the inflammatory phase of secondary bone repair. This phase peaks at approximately 48 hours and typically lasts 7 days. The inflammatory phase supports immobilization at the fracture site by promoting pain and swelling.290 A hematoma forms at the site of fracture, which also serves to immobilize the area (via a fibrin network). In addition, the hematoma provides a source of signaling molecules that initiate cellular events necessary for fracture repair.291 Platelets within the hematoma release PDGF and TGF-β, which promote MSC proliferation and differentiation into an osteoblastic lineage. Moreover, neutrophils, lymphocytes, blood monocytes, and tissue macrophages are recruited to remove necrotic debris and stimulate angiogenesis at the fracture site.
The fracture callus undergoes both intramembranous and endochondral ossification.289 Endochondral ossification occurs during the second week of fracture repair, when abundant cartilage overlying the fracture site (formed by chondrogenesis of the callus) undergoes calcification. Blood vessels grow into this calcified cartilaginous callus, bringing with them osteoblast progenitor cells. As the calcified cartilage is resorbed by chondroclasts, osteoblasts begin to lay woven bone. Intramembranous ossification, on the other hand, occurs adjacent to the fracture site.
Variables influencing bone repair
Blood supply
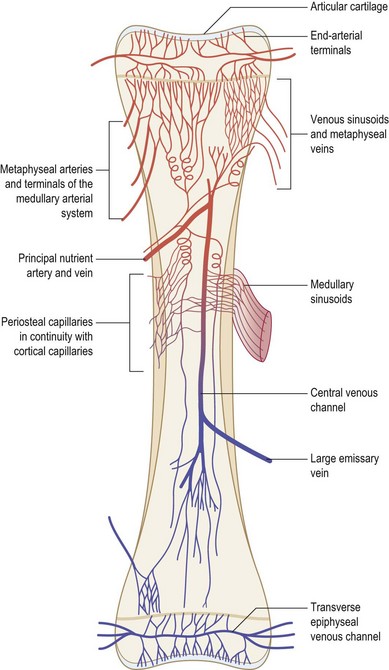
Blood flow to bone represents approximately 10–20% of total cardiac output. The pattern of vascularization to the majority of the appendicular skeleton is similar in organization to the vascular pattern of a typical long bone.292 A tubular long bone in the adult human receives blood by three distinct arterial systems that anastomose with one another extensively (Fig. 21.7). The primary supply of blood to the inner two-thirds of the cortex and the medullary cavity is derived from the diaphyseal nutrient artery. As the nutrient artery traverses the cortex, it passes through to the medullary cavity, where it divides into ascending and descending medullary branches. Metaphyseal and epiphyseal arteries represent the second significant source of blood flow. These vessels, which arise from arteries supplying adjacent joints, deliver blood to cancellous bone found at the end of long bones. The third principal blood flow source stems from periosteal arteries, which carry blood to the outer one-third of bone cortex.
As blood moves through bone, from endosteum to periosteum, it flows through sinusoids and arterioles within the haversian system. Exchange vessels (i.e., vessels connecting the arterial and venous systems) within osteons lie in parallel to the axis of the bone. Collecting sinuses and veins with similar names to their arterial counterparts receive deoxygenated blood from exchange vessels. A high intramedullary pressure is likely responsible for the centrifugal flow of blood through bone.293
Angiogenesis is critical for bone growth maturation, and repair.294 Although the mechanisms that regulate angiogenesis during fracture repair have not been fully elucidated, many important angiogenic cytokines have been described (Table 21.4). The prototypical molecule thought to play a pivotal role in angiogenesis within developing and healing bone is VEGF. Molecules described previously (BMP, TGF-β, FGF-2, and PDGF) have also been implicated in angiogenic processes necessary for osteogenesis. VEGF and FGF-2 are the only two directly acting regulators of angiogenesis; other growth factors serve indirect roles. In addition, the hypoxia-inducible factor-1α (HIF-1α) pathway is crucial to the coupling of angiogenesis with osteogenesis.295,296 Discoveries associated with this pathway have led some authors to hypothesize that skeletal angiogenesis and osteogenic–angiogenic interactions are rate-limiting processes in bone growth.294 Not surprisingly, the HIF-1α pathway is intimately linked to VEGF expression.297
A potent regulator of both vasculogenesis and angiogenesis, VEGF mediates many important functions during the embryologic stage of development, and throughout the lifetime of organisms. Deletion of even a single VEGF allele is associated with lethality in utero in a mouse model.298,299 VEGF, which is structurally related to PDGF, is expressed by many different of cell types, typically in especially high concentrations within highly vascularized tissues. VEGF mediates its effects on cells through its interactions with two receptor tyrosine kinases (VEGF-1 and VEGF-2).
The hypothesis that VEGF plays an essential role in skeletogenesis mainly comes from studies examining endochondral bone formation. At the epiphyseal growth plates of long bones, hypertrophic chondrocytes have been shown to express high levels of VEGF.300,301 In addition, injection of an anti-VEGF monoclonal antibody results in inhibition of vascular invasion of the nonhuman primate growth plate, thereby hampering longitudinal bone growth.302 Moreover, mice treated with a VEGF inhibitor manifest significantly decreased bone capillary invasion and trabecular bone formation.301 Cessation of anti-VEGF treatment is associated with capillary invasion and restoration of bone growth.
Fracture fixation
Critical to the success of fracture repair in long bones is the extent of movement at the fracture site after fixation.303 Delayed union and nonunion are both associated with excess motion; however, some movement, known as micromotion, can enhance bone healing. The mechanism by which absolute immobilization hampers fracture repair may be related to increased resorption in a stress-protected environment. In addition, the degree of mechanical strain endured at the site of a fixed fracture has differential effects on chondrogenesis and osteogenesis.304
Age
Increasing age causes characteristic changes in bone. Pediatric fractures tend to heal at an accelerated rate compared to adult fractures.305,306 The reasons for this are multifactorial. Age appears to affect angiogenesis,307 and angiogenesis affects fracture healing (see earlier). With this in mind, Lu and colleagues308 designed a study to examine the effect that age has on vascularization during fracture healing. Tibial fractures were created in 4-week-old, 6-month-old, and 18-month-old (juvenile, middle-aged, elderly, respectively) rats. At 7 days postfracture, there was a higher surface density of blood vessels in juvenile fracture calluses compared to those of middle-aged and elderly rats. In addition, juvenile rats expressed increased levels of HIF-1α and VEGF at 3 days postfracture versus older rats. Although these results do not provide definitive data that a younger age augments osteogenesis via vasculature-mediated mechanisms, they do offer insight into the fact that angiogenic potential during fracture healing does change throughout the lifespan of rats. Age may also influence fracture repair via its effects on periosteal structure and cell population309 and chondrogenic potential,310 stem cell function,311 and biologic signaling.312
Bone remodeling
Osteoinduction
Albrektsson and Johansson313 have written that osteoinduction is the process by which undifferentiated pluripotent cells are stimulated to become cells within the osteoblast lineage. This phenomenon occurs naturally during skeletogenesis and fracture repair.
Although osteoinduction is an intrinsic biologic process, it can also be manipulated exogenously. For example, purified and recombinant human isoforms of BMP are commonly used in experimental settings to induce bony formation. Various biomaterials have also been used for the same purpose. Bioglass and other osteoinductive materials have shown promise when used for craniomaxillofacial applications.314 After bioactive glass is mixed with saline or blood, an apatite surface layer is formed at the biomaterial–bone interface, which incorporates collagen and proteins from surrounding native bone. In addition to producing a chemical bone at the interface, the apatite layer stimulates the production of osteogenic cytokines from surrounding osteoprogenitor cells, and this results in new bone formation.315
The use of osteoinductive effector cells such as MSCs and osteoprogenitor cells is currently an area of large focus. This is because the use of exogenous or autologous stem cells capable of differentiating into bone, in addition to osteoinductive cytokines, appears to have clear benefits. Adipose tissue may provide an ideal source of MSCs capable of undergoing osteoinduction as they have been shown to have the ability to differentiate into osteogenic cells.316 In addition, Caballero et al.317 examined whether and to what extent MSCs from the umbilical cord of neonates and palatal periosteum of older infants could undergo osteoinduction. After isolation and subsequent MSC incubation within osteogenic media, it was found that umbilical cord- and palatal periosteum-derived MSCs both stained positive for calcium deposition and also expressed increased levels of BMP-2, ALP, and osteocalcin. Given that MSCs from both sources appear to be capable of undergoing osteoinduction, they may be of benefit for children with critical osseous defects (defects that cannot ossify on their own without intervention).
Osteoconduction
Osteoconduction refers to the ability of a material to serve as a scaffold on to which bone can attach and grow. During primary bone repair, opposing fragments at the fracture site mediate osteoconduction. In secondary bone repair, callus ECM acts as a scaffold on to which new bone can attach and grow. In the field of regenerative medicine, many implanted materials serve an osteoconductive role. It is believed that an osteoconductive substrate must be porous to allow for the ingrowth of bone and fibrovascular tissue during osteoinduction.318 Currently, there are many osteoconductive materials that are used in an effort to improve bone healing in clinical practice. Surgeons can choose from allografts (processed human bone), autogenous bone grafts, purified collagen, calcium phosphate (CaP) substitutes, and synthetic polymers. Different osteoconductive materials have differing innate osteoinductive potentials as well. Native bone, for example, will mediate both osteoconduction and osteoinduction, whereas CaP substrates are purely osteoconductive. CaP, however, can express osteoinductive properties if enhanced with osteoinductive substances such as BMPs.319