div class=”ChapterContextInformation”>
26. Surgical Reconstruction of Urethral Injury After Artificial Urinary Sphincter Infection-Erosion
Keywords
MaleUrethral injuryRadiationProstatectomyArtificial urinary sphincterUrinary incontinenceUrethroplastyReconstruction26.1 Introduction
Although the artificial urinary sphincter remains the gold standard for treating men with moderate to severe stress urinary incontinence (SUI), management of urethral cuff erosion is a feared complication that is often debated. Though contemporary descriptions of cuff erosion management report a role for urethroplasty at the setting of device removal, most of these series are limited by single institutional designs with small cohorts and short-term follow-up.
Approximately up to 50% of patients who undergo AUS placement will eventually require surgical revision after first implantation [1]. The predominant causes of AUS device failure are controversial, such as subcuff urethral atrophy, cuff leakage or pressure reservoir balloon (PRB) malfunction, which result in recurrent incontinence. Early cuff erosion most commonly results from unrecognized dorsal urethral injury during cuff placement and usually present as urinary leakage and periurethral catheter bleeding or hematuria following catheter removal. Most commonly, late urethral erosion, which occurs predominantely within the first 2 years after implantation, is associated with an established capsule around the cuff which does not usually cause urine leakage but hematuria and irritative voiding symptoms instead. Definitive risk factors for delayed cuff erosion have been identified and include initial erroneous selection of an undersized cuff, traumatic endourological manipulation and urethral catheterization with incomplete device deactivation. The diagnosis of cuff infection-erosion, an event characterized by a binomial relationship, is easily made by cystourethroscopy and surgical removal of the cuff is mandatory. In the presence of purulent infection, the entire AUS device must be explanted.
Because “selection is the silent partner of the surgeon”, a rigorous choice of candidates for AUS implantation is critical. As with all surgical complications in general, the most important aspect is avoiding – to the extent possible – the problem in the first place. As such, in this chapter we will review best practices for avoiding erosion and risk factors for erosion before turning our attention to how to manage erosions, many of which are unavoidable. We also need to better know the natural history and clinical course of men who underwent AUS cuff explantation for erosion in large, international cohort studies that could provide valuable insights into future management.
26.2 Radiation and Other High Risk Factors
Patient risk factors – specifically pelvic radiation , prior AUS erosion, history of urethral stent placement, neurogenic bladder dysfunction and previous urethroplasty – have been responsible for an increased risk of AUS complications, specifically infection and erosion [2]. Some studies have also suggested that patients with additional medical comorbidities, including hypertension, coronary artery disease, and low testosterone are more likely to develop erosion of their AUS [3, 4].
A prior AUS revision, especially for erosion or infection of the cuff, predisposes to another similar event. The reason for this occurrence is still unclear but it is likely that erosion reduces urethral blood supply due to scar tissue it induces. Hypertension and coronary artery disease may also add to the vascular insufficiency leading to tissue breakdown and urethral cuff erosion. This is the leading cause of early AUS revision, with sub-cuff urethral atrophy being the usual cause of late (>12 months) revision.
The role of prior radiation in subsequent cuff infection-erosion rate has been controversial in prior published literature. It is known that radiation causes long-term biological effects such as obliterative endarteritis of small vessels, which result in tissue ischemia, fibrosis, necrosis and abnormal tissue repair. These effects contribute to a higher likelihood of infection-erosion [2–9]. The erosion rate is higher in patients receiving radiotherapy than in patients without a prior history of irradiation. Additionally, in patients who do eventually suffer from erosion, a history of radiation decreases the median time to erosion , versus those without that history [10].
The management strategies for AUS revisions for infection-erosion should include placing the cuff at alternate cuff sites (more proximal or distal to the original one), transcorporal approach and downsizing the cuff size. The use of the UroLume® stent (no longer available in the United States and Europe) across post-erosion stricture site has been recommended by some authors [11]. However, other authors have reported dismal results [12].
Despite patient characteristics and risk factors , there are also surgeon-dependent factors. Not only patient selection is critical, but showering with a chlorhexidene soap starting several days prior to the procedure [13], a thorough scrub of the operating field, meticulous attention to sterile technique, scrupulous use of pre- and possibly peri-operative broad spectrum antibiotics, avoidance of hematomas or excessive use of electrocautery, and copious irrigation of the surgical field are good surgical principles to which to adhere. Delaying device activation for 4–6 weeks, and perhaps up to 8 weeks in patients with fragile urethras or following irradiation are generally followed practices. In high-risk patients, we may postpone reimplantation for up to 6 months to be sure that the urethra is completely healed. If a non-significant stricture develops, a direct vision internal urethrotomy may be attempted once; otherwise, the patient should undergo open urethroplasty. In our experience, one of the authors (FEM) always tries to perform synchronous urethral repair at the time of revisional surgery for cuff infection-erosion (see below). In most cases where the erosion is not circumferential, the urethra is not necrotic and the gap between both urethral ends is not too great, an anastomotic urethroplasty is feasible (data in preparation for publication). This approach, however, is controversial, and a multi-institutional retrospective study suggested that the type of repair at the time of erosion did not influence subsequent urethral stricture rates, although complete erosions were more likely to result in strictures compared to partial erosions [14]. In our opinion, a urethral stent should be avoided at all costs, as we have never had any successful patient after such stent placed at the bulbar urethra or bladder neck followed by AUS implantation later (unpublished data).
26.3 Surgical Approach for Initial Implantation
Positioning/Approach
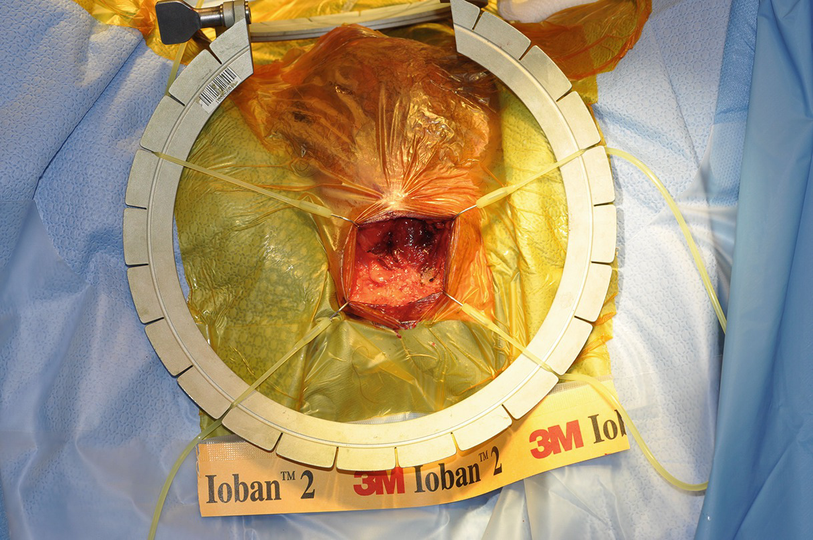
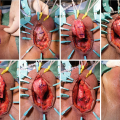
Incision through perineal fat
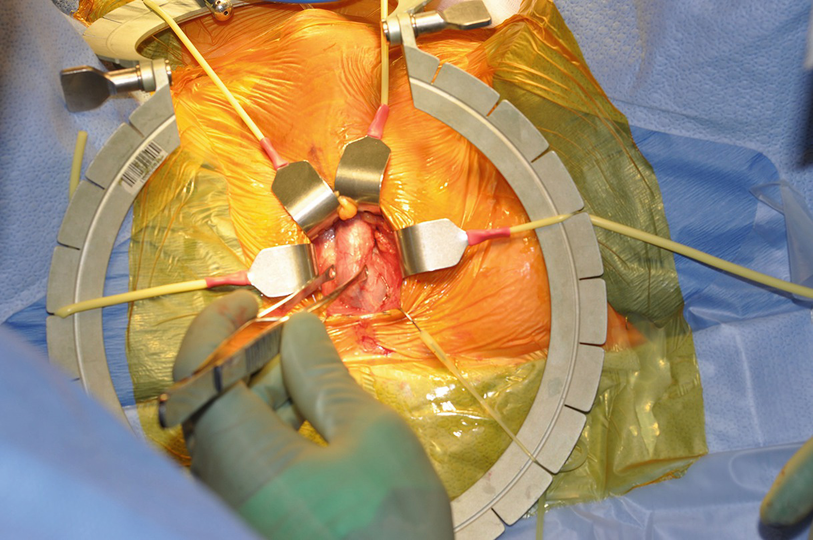
Demonstration of the bulbar urethra
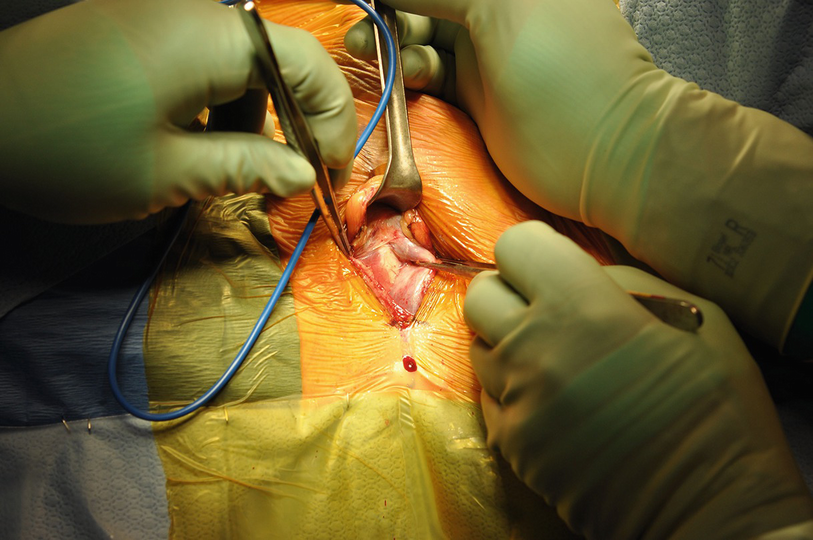
Initial dissection for a periurethral cuff placement
Cuff Location(s) and Considerations
Depending on age and comorbidities, it is likely that the AUS will require revision during the patient’s lifetime, often several times. For this reason, it is best to plan surgery with the option of later successful revisions. In general, we recommend placing the AUS for the first time at the area of the crus, with subsequent cuffs placed in either proximally (at the area of the perineal body or more proximal) or distally (especially if a transcorporal approach is used). In the past, double cuffs have been used more commonly, but the major disadvantage of double cuff placement is that the wide area of urethra that is used will take away some future flexibility of placement (see below).
Sharp Dissection vs Blunt
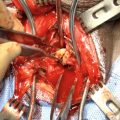
The urethra is eccentric within the bulb, with thin spongiosal tissue dorsally. The corpus spongiosum is densely adherent to the tunica albuginea in the area of the septum. If blunt dissection is performed, this may lead to urethral injury, and the dorsal urethra is the most common site of iatrogenic injury. If not recognized, this will lead to cuff erosion. For these reasons, we recommend sharp dissection, with care to visualize all aspects of the dissection until the urethra is completely encircled.
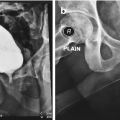
Urethral Irrigation with Proximal Occlusion to Demonstrate Lack of Urethral Injury (Fig. 26.4)
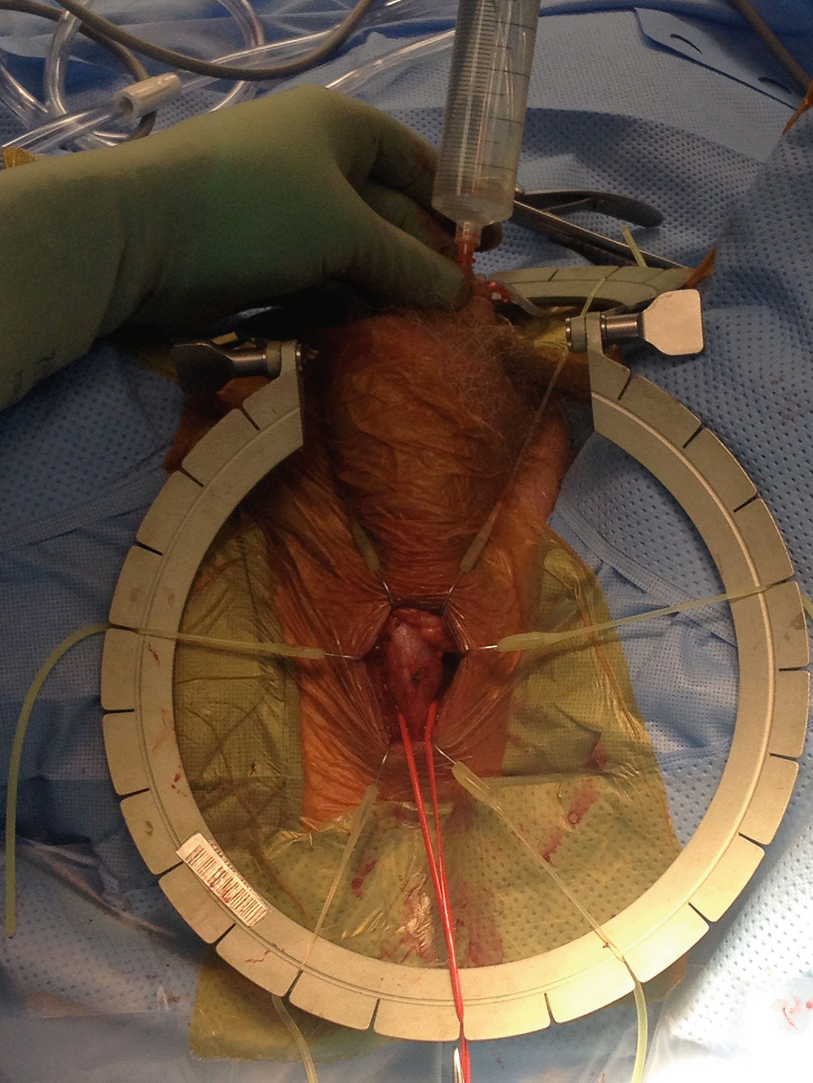
Urethral irrigation with proximal occlusion to demonstrate lack of urethral injury
Single Cuff vs Double vs Transcorporal (TC)
Although placement of a double cuff (either at one surgery or added later as a staged procedure) is tempting due to initial improvements in continence, we have found that, ultimately, incontinence rates are equal and there is a higher rate of revision and erosion, particularly of the distal cuff [16, 17]. Additionally, the two cuffs remove some location flexibility for placement of future cuffs. For these reasons, we recommend single cuff placement if at all possible.
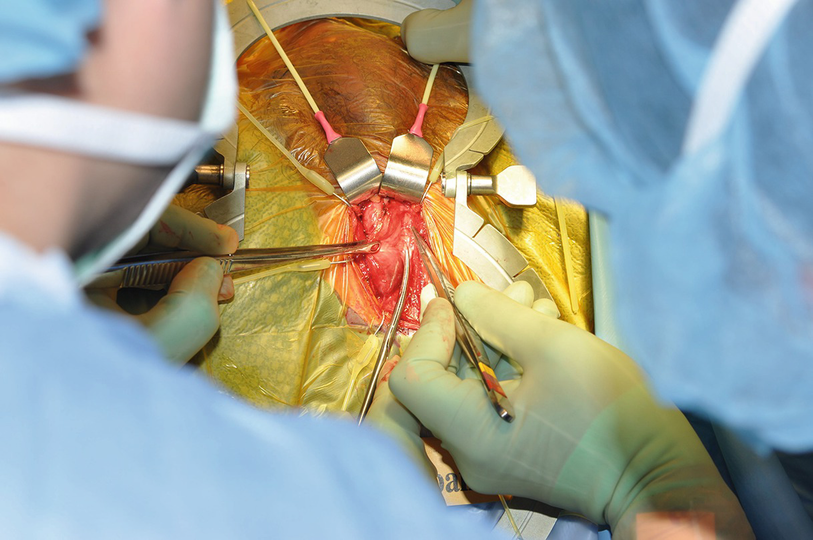
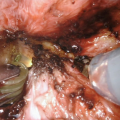
Clearing off the tunica albuginea (TA)
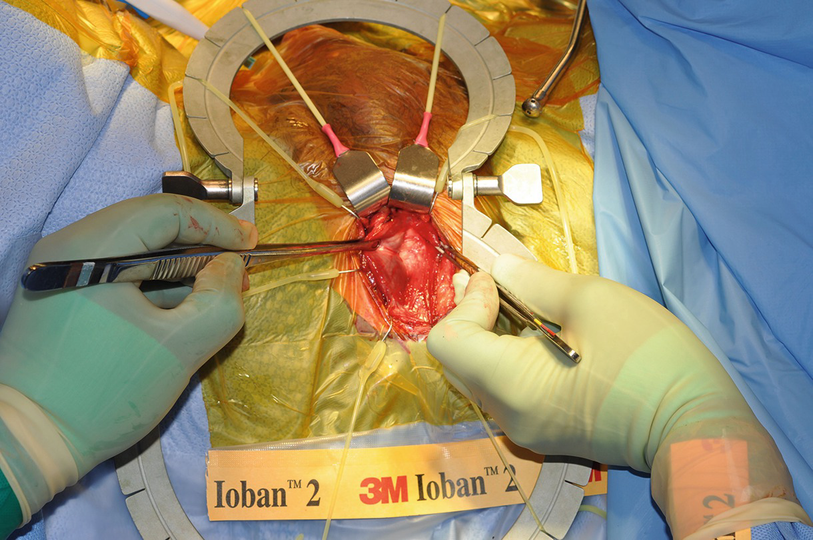
Exposure of the TA
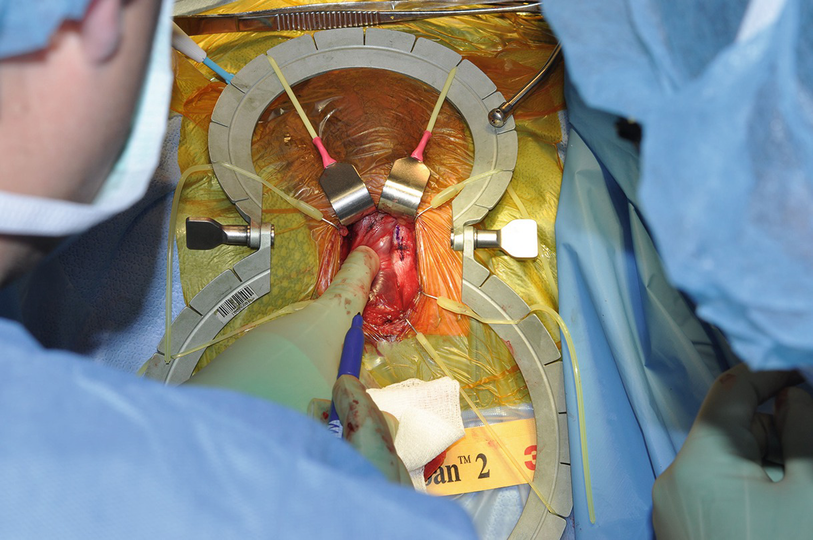
Marking the TA
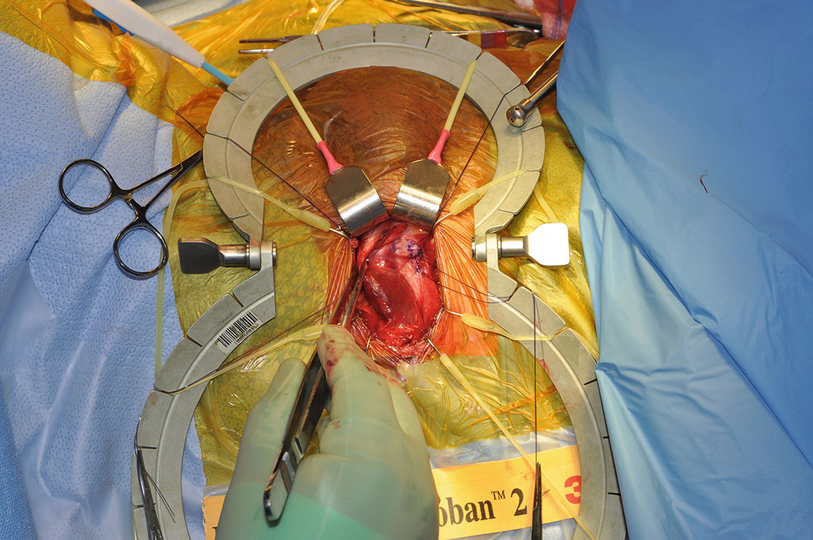
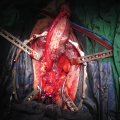
Placing sutures lateral to the proposed corporotomy
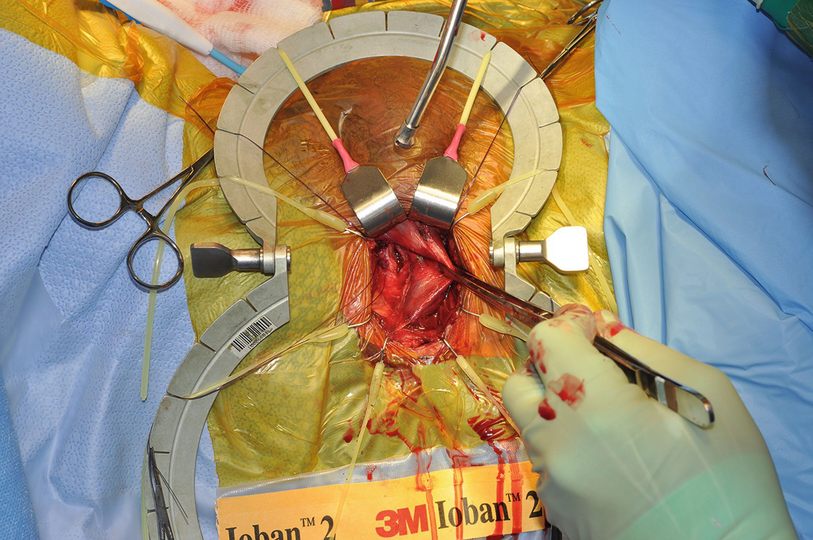
2 cm corporotomy
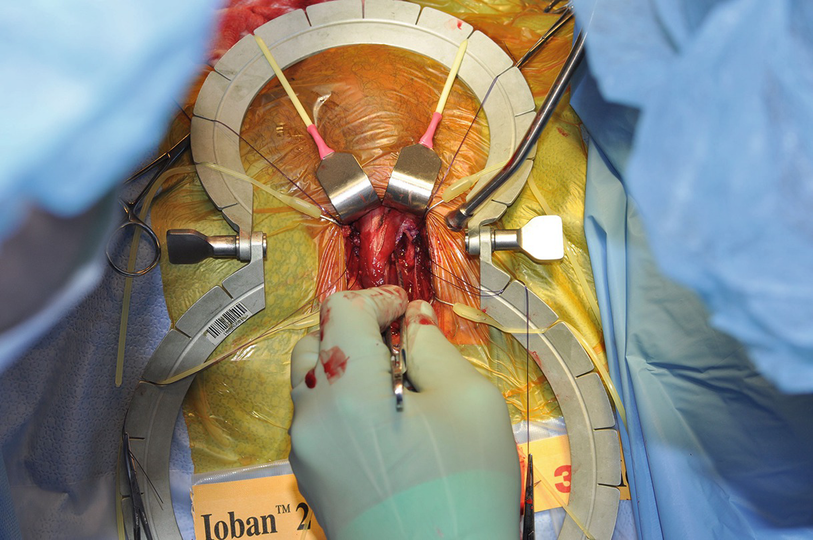
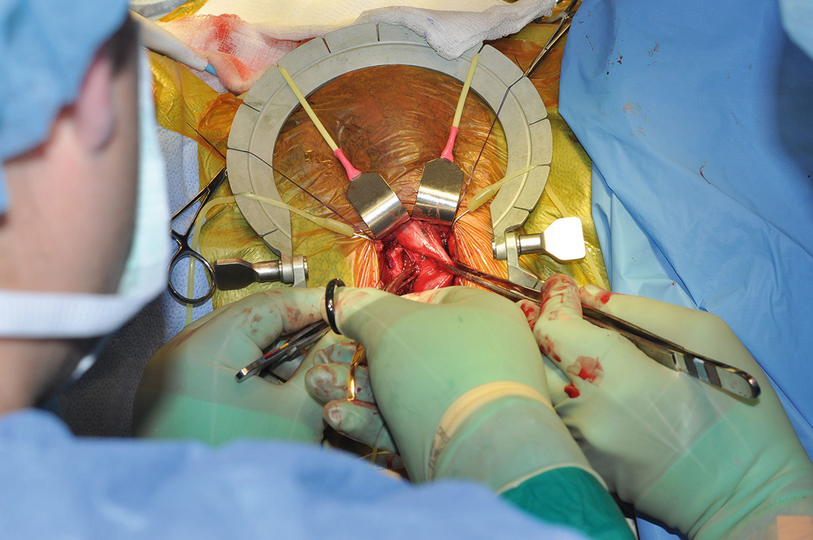
Contralateral corporotomy

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree