Introduction
The skull base is a complex anatomic region through which the major blood vessels of the head and neck, cranial nerves, and spinal cord pass. A variety of congenital malformations and neoplasms can involve the skull base. As surgical techniques, instrumentation, and imaging technology have improved over recent years, many skull base lesions can now safely be resected using an endonasal approach, thus minimizing morbidity and obviating the need for extensive reconstruction. However, many lesions still require an open resection in order to achieve clear margins. Reconstructive plastic surgeons thus play a major role in reconstructing the functional, anatomic, and cosmetic defects that such skull base resections often create.
The goals of skull base reconstruction include sealing dural leaks, dead space obliteration, separating the aerodigestive tract from the intracranial space, protecting the brain, and restoring cranial nerve function and appearance. Historically, reconstructive efforts were limited to skin grafts, pedicled flaps, and staged tubed flaps; such reconstructions were plagued by a variety of wound healing complications and significant cosmetic deformity. As such, skull base resections were associated with poor outcomes.
As is true for many aspects of reconstructive surgery, the advent of microsurgery brought about development of the tools necessary to reliably improve the cosmetic and functional results associated with skull base resections. Presently, microvascular techniques are considered the gold standard for most skull base reconstructions, providing the means to transfer well-vascularized tissue wherever needed, thereby allowing for a watertight, durable partition between the central nervous system and external environment. In many cases, advances in reconstruction now permit treatment of lesions that were deemed unresectable in the past due to the morbidity associated with their removal.
Anatomy
The skull base separates the extracranial and intracranial anatomy. Located on the extracranial side of the skull base are the sphenoid sinuses, ethmoid sinuses, pterygopalatine fossae, infratemporal fossae, and orbits. On the intracranial side, the skull base forms the anterior, middle, and posterior fossae. Multiple classification systems have been introduced based on anatomic boundaries, pathology, and reconstructive requirements. The Irish classification system has emerged as the most popular, due to its simplicity and clinical utility. Irish and colleagues reviewed 77 cases and divided the skull base into three regions, based on the relationship of the tumor to the anterior, middle, and posterior cranial fossae ( Fig. 27.1 ). In this classification, resections can involve removal of skull base bone with or without removal of the underlying dura.
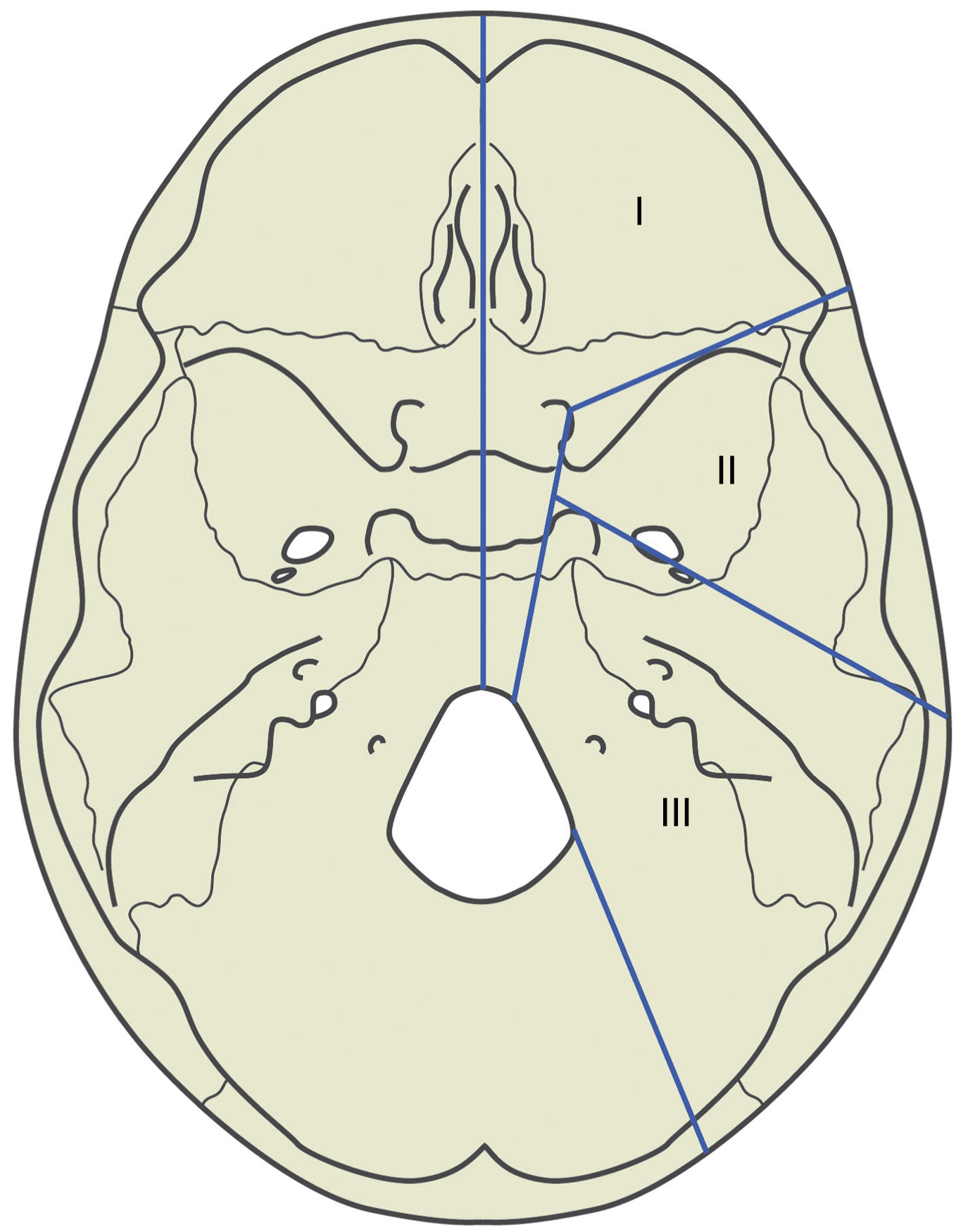
Region I defects typically involve or arise from structures that abut the anterior cranial fossa, including the orbits and orbital contents, the nose, the paranasal sinuses, and the cribriform plate. The posterior boundary of Region I includes the clivus and foramen magnum, as tumors in this region are also approached anteriorly. We have further subdivided Region I into midline and lateral or paramedian regions. Midline Region I tumors often originate from the ethmoid and sphenoid sinuses or the cribriform plate. These include esthesioneuroblastomas, for example. Lateral Region I tumors include those that arise from the orbit or periorbital structures, such as lacrimal gland tumors.
Region II extends from the posterior orbital wall to the petrous portion of the temporal bone. Region II defects result from tumors originating from the lateral skull base, primarily the infratemporal and pterygopalatine fossae, which can extend into the middle cranial fossa. Tumors that originate in this region include nasopharyngeal carcinomas, glomus jugulare tumors, sphenoid wing meningiomas, and clival chordomas, as well as parotid tumors that become deeply infiltrative and maxillary or cheek neoplasms with posterior spread. The latter usually occurs via the maxillary division of the trigeminal nerve (V 2 ).
Region III defects are usually associated with tumors arising from the ear, parotid, or temporal bone, and may extend intracranially into the middle or posterior cranial fossae. Region III tumors include cutaneous tumors involving the external auditory canal, such as squamous and basal cell carcinomas, as well as parotid malignancies and, occasionally, sarcomas that arise directly from the temporal bone.
Reconstructive Approach
The location of the tumor predicts the reconstructive needs. In the following sections, we will discuss flap selection and specifics of flap design and inset for each skull base region. Occasionally, defects will encompass more than one region and it will be necessary to modify flap design to address the reconstructive needs of two or more regions, or potentially perform a double free flap reconstruction. In addition to selection of the reconstructive flap, the possibility of needing grafts or implants should also be considered. For example, Region I defects of the orbital walls and frontal calvarium may require skeletal reconstruction in order to prevent change in eye position, pulsatile exophthalmos, and cosmetic deformity. Autologous bone grafts or manmade implants, such as titanium mesh, porous polyethylene, or polyetheretherketone (PEEK), can be considered for these applications.
Cranial nerve deficits should be anticipated based on the location of the tumor, as well as by a full preoperative physical exam of cranial nerve function, which should be documented in detail in the preoperative note. Various procedures to address facial nerve paralysis should be discussed with the patient preoperatively in any case where resection of the facial nerve is a possibility. , Such procedures include direct nerve repair, cable nerve grafting, dynamic reanimation with vascularized muscle transfer, anastomosis to other cranial nerves (e.g., masseteric nerve to facial nerve or hypoglossal nerve to facial nerve transfers), and static reanimation with a fascial graft and periocular procedures, such as gold eyelid weight placement, canthoplasty, lateral tarsorrhaphy, and browlift. ,
Other functional deficits might include those arising from loss of glossopharyngeal (IX), vagus (X), spinal accessory (XI), and hypoglossal (XII) nerve function. Resection of the glossopharyngeal nerve can result in uncoordinated swallowing and aspiration, which may be addressed with temporary or permanent gastrostomy tube placement as well as postoperative speech and swallowing therapy. Unilateral hypoglossal nerve deficits are generally well tolerated, but may also require speech and swallowing therapy. Vocal cord paralysis following transection of the vagus nerve can be addressed by an otolaryngologist postoperatively with vocal cord injection of various filler materials and/or medialization thyroplasty. Transection of the spinal accessory nerve results in shoulder drop that is best addressed with physical therapy.
A discussion of the patient’s desire for an orbital or ear prosthesis, if loss of the eye or auricle is a possibility, should take place preoperatively since this may influence flap choice, particularly for an orbital prosthesis. Both orbital and auricular prostheses can be secured either with adhesive or through the use of osseointegrated implants. If an osseointegrated implant is preferred, and postoperative radiation is a possibility, consideration should be given to placing the implant at the time of tumor resection; many have reported decreased implant reliability when placed after radiation is given. Similarly, if the skull base resection includes removal of the middle ear components, an otologist should be involved preoperatively for placement of a bone-anchored-hearing aid (BAHA), which also requires an osseointegrated implant. In many cases, placement of an osseointegrated implant is challenging after substantial resection of bone, since fairly thick bone stock must surround the implant in order for it to be stable. In such cases, an implant-retained prosthesis may not be possible.
Region I
Midline Region I tumors requiring an open extirpation are usually resected with an anterior craniofacial approach, resulting in communication of the nasal cavity with the intracranial space. Such a resection involves endonasal surgery or a lateral rhinotomy combined with a bicoronal incision followed by a frontal craniotomy. If the defect is small and limited to the upper nasal cavity, the intracranial space can be separated from the sinuses with a pedicled pericranial flap or galeafrontalis flap, both of which derive their blood supply from the supratrochlear and supraorbital vessels. In situations where these blood vessels have already been transected, or in larger defects involving the external nose, forehead, and/or orbit, a free flap may be necessary. If there is no skin defect and the flap is needed as a watertight partition between the aerodigestive tract and intracranial space, then it may be buried and monitored with an implantable Doppler probe. Moderate size muscle free flaps, such as the rectus abdominis, vastus lateralis, and serratus anterior muscle free flaps, are good options in this situation.
Lateral Region I defects frequently involve orbital exenteration ( Fig. 27.2 ). If the resection spares the medial and inferior walls of the orbit, it is well suited to the eventual use of a prosthesis. In order to accommodate a prosthesis, the orbit must be resurfaced with thin, pliable, well-vascularized tissue that will maintain the concave shape of the cavity. This can be achieved with a free radial or ulnar forearm fasciocutaneous free flap. A pedicled temporalis muscle or temporoparietal fascia flap with a skin graft may also be an option here, although removal of some of the lateral orbital bone is usually necessary to allow these flaps to reach the medial orbit. If the orbital exenteration results in communication with the sinuses or nasal cavity, then a bulkier free flap is preferred. The anterolateral thigh (ALT) and rectus abdominis myocutaneous (RAM) free flaps are highly customizable and allow for the design of flap with a variable amount of muscle, tailored to the defect’s requirements. The muscle conforms to the complex geometry of the defect, sealing exposed dura and/or sinonasal cavities. This feature helps to minimize the occurrence of a sinonasocutaneous fistula; such fistulae are exceedingly difficult to heal owing to persistent pressure from the nasal airway.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree