Key Words
psoriasis, pityriasis rubra pilaris, seborrheic dermatitis, pityriasis rosea, lichen planus, lichen sclerosus, Grover disease, Reiter syndrome, topical steroid, pityriasis lichenoides
Papulosquamous diseases are a group of disorders characterized by scaly papules and plaques. These entities have little in common except the clinical characteristics of their primary lesion. A complete list of diseases characterized by scaly plaques appears in the section on primary lesions in Chapter 1 . The major papulosquamous diseases are described here.
Psoriasis
Psoriasis occurs in 2% to 3% of the general adult population, with two main peaks of onset, 20 to 30 and 50 to 60 years of age. Thirty percent of psoriasis begins in childhood, and the median age of onset of childhood psoriasis is between the ages of 7 and 10 years. The disease is transmitted genetically, with at least 10% of people inheriting one or more genes that may lead to psoriasis. A family history of psoriasis predicts an early age of onset. The disease is lifelong and characterized by chronic, recurrent exacerbations and remissions that are emotionally and physically debilitating.
There may be many millions of people who have the potential to develop psoriasis, with only the correct combination of environmental factors needed to precipitate the disease. Stress, for example, may precipitate an episode. Environmental influences may modify the course and severity of disease. Extent and severity of the disease vary widely. Psoriasis frequently begins in childhood, when the first episode may be stimulated by streptococcal pharyngitis (as in guttate psoriasis).
Pathogenesis
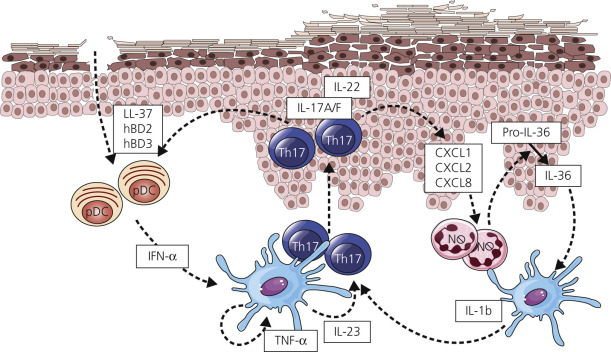
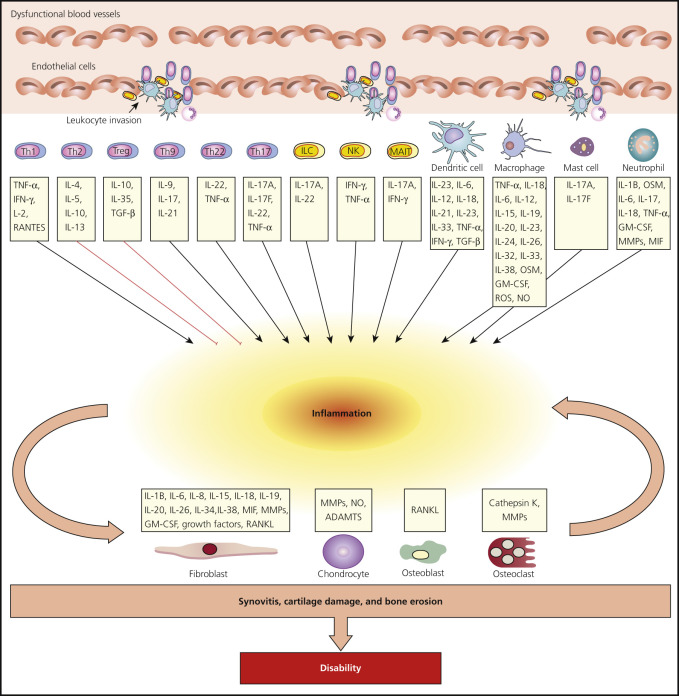
Understanding of the immune mechanism of disease has led to a burgeoning of target therapies. Psoriasis is an autoimmune disorder driven in large part by T cells (see Fig. 8.1 ). Various inflammatory pathways are important in the pathogenesis of various forms of psoriasis and form the basis for molecular targeted therapies ( Fig. 8.1 and Tables 8.1 and 8.2 ).
| Name | Drug Class | Mechanism of Action | Expected % PASI 75 | Dosage | Monitoring | Clinical Considerations |
|---|---|---|---|---|---|---|
| Methotrexate | Antimetabolites | Inhibitor of folate biosynthesis, antiinflammatory | 40 | 7.5–25 mg/week in a single dose | CMP, CBC, pregnancy | Hepatotoxicity. Consider supplementing with folate 1–5 mg/day or folinic acid 15 mg/week in 3 divided doses every 12 h, starting >6 h after methotrexate dose. Avoid in cases of alcohol abuse, hepatitis, and pregnancy. |
| Cyclosporine | Immunosuppressant | Calcineurin inhibitor | 60–70 | 3–5 mg/kg/day in 2 divided doses | CMP, CBC, magnesium, K, blood pressure | Associated with cumulative renal toxic effects; immunosuppressive; expensive. Fast acting. Safe to use in pregnancy. |
| Acitretin | Retinoid | Exact mechanism unknown. Believed to target RXR and RAR retinoid receptors | 20–40 | 10–50 mg/day increased as needed Maximum dose is 75 mg/day as a single dose to be taken with fatty meal | CMP, lipids, CBC, pregnancy | More effective when used in combination with psoralen–UVA or UVB phototherapy. Can be used safely in combination with other oral agents, topicals, and biologics. Avoid in cases of alcohol abuse, hepatitis, dyslipidemia, and pregnancy. Long half-life. Slow onset of action. Plaques may thin, but be persistent. |
| Apremilast (Otezla) | Phosphodiesterase 4 inhibitor | Reduces inflammation by decreasing PDE4 activity | 30 | 30 mg bid | Weight | Can cause GI upset and weight loss. Pregnancy C, registry for exposure reporting. |
| Tofacitinib | Janus kinase inhibitors | Modulates cytokines involved with the immune and inflammatory responses | 40–64 | 5–10 mg bid | CBC, LFT, lipids | FDA approved for rheumatoid arthritis at 5 mg dose; safety concerns in rheumatoid arthritis at 10 mg. Pregnancy C, registry for exposure reporting. Avoid simultaneous use of immunosuppressive medications. |
| Fumaric acid esters | Immunomodulators | Not fully understood | 40 | Titrated up to maximum of 720 mcg/day in 3 divided doses | CBC, LFT | Three times daily administration for maximum dose. Limited data. Appears safe in pediatric populations. |
| Mycophenolate mofetil | Immunosuppressant | Inosine monophosphate dehydrogenase inhibitor | 20 | 1–5 g/day in 2 divided doses | CMP, CBC, pregnancy | Significant number of partial responders and nonresponders. Lack of long-term toxicity. Pregnancy D. |
| Hydroxyurea | Antimetabolite | Reduces DNA replication within the basal layer of the epidermis | 40–50 yet poorly substantiated | 500–1500 mg daily as tolerated; alternatively 3–4.5 g/week | CBC, pregnancy | Poor quality data. Pregnancy D. |
| 6-Thioguanine | Antimetabolite | Inhibits purine synthesis | 50–80 yet poorly substantiated | 120 mg twice per week, up to 160 mg three times per week | CMP, CBC, pregnancy | Poor quality data. Pregnancy D. |
| Sulfasalazine | Sulfa drugs, antiinflammatory | Not fully understood. Acts as an antiinflammatory, immunomodulator, and TNF inhibitor | <40 | 1.5–4 g daily, as tolerated | CBC, LFT, pregnancy | Poor quality data. Pregnancy B. |
| Leflunomide | Antirheumatic | Pyrimidine synthesis inhibitor | 17 | 100 mg/day × 3 days loading dose, 20 mg/day | LFT, phosphate, CBC, pregnancy | May benefit patient with significant PsA. |
* These therapies can be used in conjunction with UVB and narrowband ultraviolet B light (NB-UVB). NB-UVB works by the rapid depletion of dermal and epidermal lymphocytes, macrophages, and dendritic cells. It is safe and effective, but requires many office visits.
| Name | Drug Class | Mechanism of Action | Indications | Dosage | Contraindications | Clinical Considerations |
|---|---|---|---|---|---|---|
| Ustekinumab (Stelara) | Interleukin-12 and -23 antagonist | Binds with high affinity and specificity to the p40 protein subunit used by both IL-12 and IL-23 cytokines | Patients with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy Patients with active psoriatic arthritis (alone or in combination with methotrexate). | Injection: 45 mg/0.5 mL or 90 mg/mL in a single-dose prefilled syringe. Recommended dosage 45 mg administered subcutaneously initially and 4 weeks later, followed by 45 mg every 12 weeks. Patients weighing more than 100 kg, recommended dosage is 90 mg administered subcutaneously initially and 4 weeks later, then 90 mg administered subcutaneously every 12 weeks Intravenous infusion injection: 130 mg/26 mL (5 mg/mL) solution in a single-dose vial | Serious hypersensitivity to ustekinumab or to any of its excipients | Increased risk of infections. Evaluate for tuberculosis (TB) prior to initiating medication. Reversible posterior leukoencephalopathy syndrome (RPLS). Serious infections from mycobacteria, salmonella and Bacillus Calmette–Guérin (BCG) vaccinations have been reported in patients genetically deficient in IL-12/IL-23. Allergic reactions can occur. Increased risk for malignancies. |
| Guselkumab (Tremfya) | Interleukin-23 blocker | IgG1 monoclonal antibody that selectively binds to the p19 subunit of IL-23 and inhibits its interaction with the IL-23 receptor | Patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy | 100 mg/mL in a single-dose prefilled syringe administered by subcutaneous injection at week 0, 4, and then every 8 weeks | Serious hypersensitivity to guselkumab or to any of its excipients | Increased risk of infections. Evaluate for TB prior to initiating medication. Allergic reactions can occur. |
| Secukinumab (Cosentyx) | Interleukin-17A antagonist | IgG1 monoclonal antibody that selectively binds to the IL-17A cytokine and inhibits its interaction with the IL-17 receptor | Moderate to severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy Adults with active psoriatic arthritis | Plaque psoriasis: Recommended dosage is 300 mg by subcutaneous injection at weeks 0, 1, 2, 3, and 4, followed by 300 mg every 4 weeks. A dose of 150 mg can be considered for some patients. Psoriatic arthritis: The recommended dosage with a loading dosage is 150 mg at weeks 0, 1, 2, 3, and 4 and every 4 weeks. Without a loading dosage is 150 mg every 4 weeks. Consider a dosage of 300 mg in patients who continue to have psoriatic arthritis. | Serious hypersensitivity reaction to secukinumab or to any of its excipients | Increased risk of infections. Evaluate for TB prior to initiating medication. Induced or exacerbated inflammatory bowel disease. Allergic reactions can occur. |
| Ixekizumab (Taltz) | Interleukin-17A antagonist | IgG4 monoclonal antibody that selectively binds with the IL-17A cytokine and inhibits its interaction with the IL-17 receptor | Adults with moderate to severe plaque psoriasis and who are candidates for systemic therapy or phototherapy | Recommended dose is 160 mg (two 80 mg injections) at week 0, followed by 80 mg at weeks 2, 4, 6, 8, 10, and 12, then 80 mg every 4 weeks | Serious hypersensitivity reaction to ixekizumab or to any of its excipients | Increased risk of infections. Evaluate for TB prior to initiating medication. Allergic reactions can occur. May induce or exacerbate Crohn disease or ulcerative colitis. |
| Etanercept (Enbrel) | TNF blocker | Inhibits TNF binding to TNF receptors | Rheumatoid arthritis Polyarticular juvenile idiopathic arthritis (JIA) for patients 2 years or older Psoriatic arthritis Ankylosing spondylitis Plaque psoriasis | Adult rheumatoid arthritis and psoriatic arthritis: 50 mg once weekly with or without methotrexate Ankylosing spondylitis: 50 mg once weekly Adult plaque psoriasis: 50 mg twice weekly for 3 months, followed by 50 mg once weekly JIA: 0.8 mg/kg weekly, with a maximum of 50 mg per week | Allergy to etanercept or to any of its excipients Use of live vaccines Same time use with abatacept or anakinra can increase risk of serious infection Same time use with cyclophosphamide | Avoid taking etanercept with an active infection. Consider empiric antifungal therapy for patients who develop a severe systemic illness and travel or reside in regions where mycoses are endemic. New or worsening demyelinating disease. Possible development of lymphoma can occur. New or worsening congestive heart disease. Pancytopenia or aplastic anemia. Monitor hepatitis B virus carriers for reactivation both during and several months after therapy. Allergic reactions can occur. Discontinue etanercept if lupus-like syndrome or autoimmune hepatitis develops. |
| Adalimumab (Humira) | TNF blocker | Binds to TNF-α, which blocks its interaction with the p55 and p75 cell surface TNF receptor | Rheumatoid arthritis Psoriatic arthritis Ankylosing spondylitis Crohn disease Patients with moderate to severe chronic plaque psoriasis and who are candidates for systemic therapy or phototherapy Hidradenitis suppurativa | Rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis: 40 mg every other week. Plaque psoriasis: 80 mg initial dose, followed by 40 mg every other week starting one week after initial dose Hidradenitis suppurativa
| Allergy to etanercept or to any of its excipients Same time use with anakinra can increase risk of serious infection Use of live vaccines | Avoid taking adalimumab with an active infection. Possible malignancies, including lymphoma. Monitor hepatitis B virus carriers for reactivation both during and several months after therapy. New or worsening heart failure or demyelinating disease. Allergic reactions can occur. Possible development of lupus-like syndrome, cytopenias, pancytopenia. |
| Infliximab (Remicade) | TNF blocker | Binds to TNF-α, blocking its action, resulting in a reduction of inflammation | Patients with moderate to severe chronic plaque psoriasis and who are candidates for systemic therapy or phototherapy Psoriatic arthritis Crohn disease Ulcerative colitis Rheumatoid arthritis (used with methotrexate) Ankylosing spondylitis | Psoriatic arthritis and plaque psoriasis: 5 mg/kg at 0, 2 and 6 weeks, then every 8 weeks | Same time use with abatacept or anakinra can increase risk of serious infection Use of live vaccines Doses >5 mg/kg in moderate to severe heart failure Allergy to infliximab or to any of its excipients | Increased risk of malignancies, including lymphoma. Allergic reactions can occur. Increased risk of HSTCL in male patients with Crohn disease or ulcerative colitis who are receiving azathioprine or 6-mercaptopurine treatment. Monitor hepatitis B virus carriers for reactivation both during and several months after therapy. Hepatotoxicity. New or worsening heart failure or demyelinating disease. Possible development of lupus-like syndrome, cytopenias. |
| Golimumab (Simponi) | TNF blocker | Binds to TNF-α, blocking its action, resulting in a reduction of inflammation | Moderate to severe rheumatoid arthritis (RA) in adults (in combination with methotrexate) Active psoriatic arthritis (PsA) in adults (alone or in combination with methotrexate) Active ankylosing spondylitis in adults | 50 mg administered by subcutaneous injection once a month | Same time use with abatacept or anakinra can increase risk of serious infection Use of live vaccines Allergy to golimumab or to any of its excipients | Avoid taking golimumab during an active infection. Consider empiric antifungal therapy for patients who develop a severe systemic illness and travel or reside in regions where mycoses are endemic. Monitor hepatitis B virus carriers for reactivation both during and several months after therapy. Increased risk of malignancies, including lymphoma. New or worsening heart failure and demyelinating disease may occur. Allergic reactions can occur. |
| Brodalumab (Siliq) | Interleukin-17 receptor A antagonist | IgG4k monoclonal antibody that selectively binds with IL-17A receptor | Plaque psoriasis | 210 mg subcutaneous injection at weeks 0, 1, and 2. Then, 210 mg every 2 weeks | Existing Crohn disease Use of live vaccines Allergy to brodalumab or to any of its excipients | Evaluate for TB before initiating treatment. Serious infection can occur; avoid use of brodalumab with an active infection. Allergic reactions can occur. Development of Crohn disease has been reported. |
| Certolizumab pegol (Cimzia) | TNF blocker | Binds to TNF-α, blocking its action, resulting in a reduction of inflammation | Severe plaque psoriasis Crohn disease Active ankylosing spondylitis Psoriatic arthritis Moderate to severe rheumatoid arthritis | Plaque psoriasis (weight ≤90 kg): Two 200 mg/mL injections on weeks 0, 2, and 4 loading dose. Maintenance: 200 mg/mL injection every two weeks | Allergy to certolizumab pegol or to any of the excipients | Evaluate for TB before treatment. Serious infection can occur; avoid use of certolizumab pegol with an active infection. Anaphylaxis or serious allergic reactions can occur. Cases of lymphoma and other malignancies have been observed. New or worsening heart failure or demyelinating disease. Monitor hepatitis B virus carriers for reactivation both during and several months after therapy. Possible development of lupus-like syndrome, cytopenias, pancytopenia. |
| Tildrakizumab-asmn (Ilumya) | Interleukin-23 antagonist | IgG1/k inhibits interaction with the IL-23 receptor by selectively binding to the p19 subunit of IL-23 | Adults with moderate to severe plaque psoriasis and who are candidates for systemic therapy or phototherapy | 100 mg delivered by subcutaneous injection at weeks 0, 4, and then every 12 weeks | Allergy to tildrakizumab or to any of the excipients | Increased risk of infection. Evaluate for tuberculosis before beginning treatment. |
| Risankizumab-rzaa (Skyrizi) | Interleukin-23 antagonist | IgG1 inhibits interaction with IL-23 receptor by selectively binding to the p19 subunit of IL-23 | Adults with moderate to severe plaque psoriasis who are candidates for systemic or phototherapy | 150 mg delivered by subcutaneous injection at weeks 0, 4 and then every 12 weeks | Allergy to risankizumab-rzaa or any of the excipients | Increased risk of infection. Evaluation for tuberculosis before beginning treatment. |
* These therapies can be used in conjunction with UVB and narrowband ultraviolet B light (NB-UVB). NB-UVB works by the rapid depletion of dermal and epidermal lymphocytes, macrophages, and dendritic cells. It is safe and effective, but requires many office visits.
“The Heartbreak of Psoriasis.”
Psoriasis for most patients is more emotionally than physically disabling. Psoriasis erodes the self-image and forces the victim into a life of concealment and self-consciousness. Patients may avoid activities, including sunbathing (which is the very activity that can clear the disease), for fear of being discovered. Therefore even when a patient has only a few asymptomatic, chronic plaques, the disease is more serious than it appears.
Clinical Manifestations
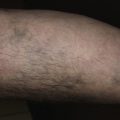
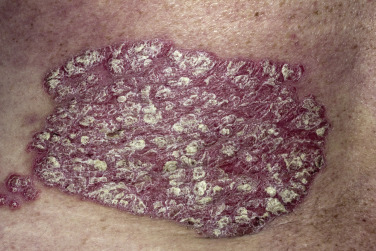
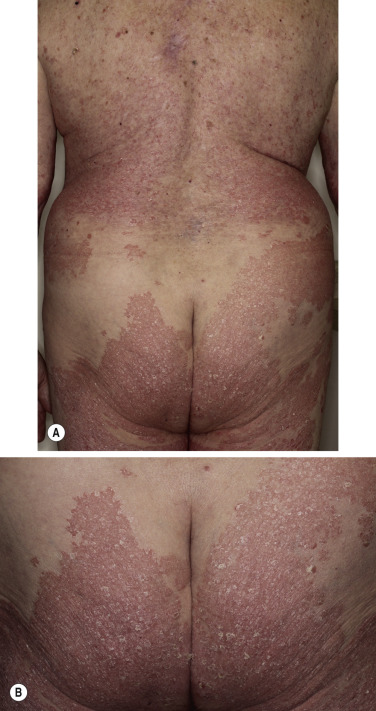
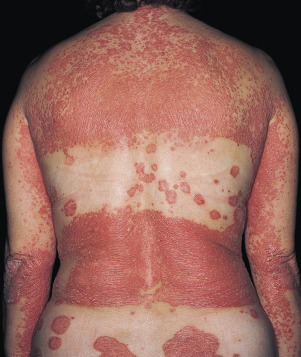
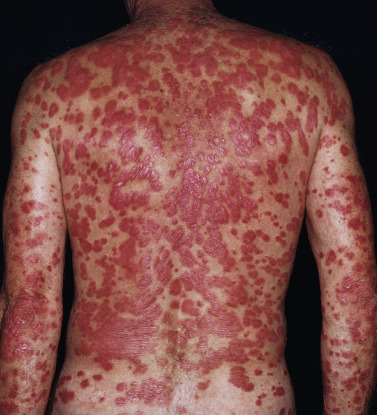
The lesions of psoriasis are distinctive. They begin as red, scaling papules that coalesce to form round to oval plaques, which can easily be distinguished from the surrounding normal skin ( Fig. 8.2 ). The scale is adherent and silvery white, and reveals bleeding points when removed (Auspitz sign). Scale may become extremely dense, especially on the scalp. Scale forms but is macerated and dispersed in intertriginous areas; therefore the psoriatic plaques of skin folds appear as smooth, red plaques with a macerated surface. The most common site for an intertriginous plaque is the intergluteal fold; this is referred to as gluteal pinking ( Figs. 8.3 and 8.4 ). The deep, rich red color is another characteristic feature and remains constant in all areas.
Psoriasis can develop at the site of physical trauma (scratching, sunburn, or surgery), the so-called isomorphic or Koebner phenomenon ( Fig. 8.5 ; see also Figs. 8.12 and 8.14 ). Pruritus is highly variable. Although psoriasis can affect any cutaneous surface, certain areas are favored and should be examined in all patients in whom the diagnosis of psoriasis is suspected. Those areas are the elbows, knees, scalp, gluteal cleft, fingernails, and toenails.
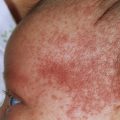
The disease affects the extensor more than the flexor surfaces and usually spares the palms, soles, and face. Most patients have chronic localized disease, but there are several other presentations. Localized plaques may be confused with eczema or seborrheic dermatitis (SD), and the guttate form with many small lesions can resemble secondary syphilis or pityriasis rosea (PR).
Drugs That Precipitate or Exacerbate Psoriasis ( Tables 8.3 and 8.4 )
Patients with new onset psoriasis, flares of existing psoriasis, and psoriasis recalcitrant to therapy should be evaluated for a possible underlying medication that is driving the psoriasis. Psoriasis usually remits with discontinuing the suspected medication.
| Drug Class | Drug | Mechanism | Psoriasis Type | Clinical Considerations |
|---|---|---|---|---|
| Beta-blockers | Propranolol Oxprenolol Practolol Metoprolol Atenolol Cetamolol Timolol Nadolol Pindolol | Bind to receptors in keratinocytes, leading to cAMP decrease. This increases intracellular calcium, causing increased cell proliferation but decreased differentiation | Psoriasiform dermatitis Eczematous eruption Lichenoid changes Psoriasiform pustulosis | Latency period between exposure and eruption can be up to 48 weeks Antipsoriatic medications are generally not effective for eruptions unless beta-blocker is discontinued |
| Lithium | Lithium carbonate Lithium citrate | Alters neuronal sodium transport Alteration to intracellular calcium homeostasis via cAMP levels and the inositol triphosphate pathway | Plaque psoriasis Palmoplantar psoriasis Pustular psoriasis Scalp psoriasis Erythroderma Nail psoriasis Psoriatic arthritis | Latency period up to 48 weeks, but average eruption at 20 weeks Cutaneous side effects more common in male patients |
| Antimalarial (AM) | Chloroquine Hydroxychloroquine | Inhibition of transglutaminase in the skin, causing decreased epidermal differentiation | Pustular psoriasis Onychoperiostitis | Psoriatic lesions resolve in one month on average after AM discontinuation 31% of patients with existing psoriatic lesions had aggravation of their psoriasis on AMs |
| Nonsteroidal Antiinflammatories | Naproxen Indomethacin Ibuprofen Meclofenamate Phenylbutazone | Inhibit the metabolism of arachidonic acid by the cyclo-oxygenase (COX) pathway leading to accumulation of leukotrienes, which are thought to flare psoriasis | Generalized pustular psoriasis | NSAIDS have a short latency period, averaging 1.6 weeks Some psoriatic lesions demonstrate resistance to LTB4, while others have no response to LTB4 |
| ACE inhibitors and ARBs | Captopril Enalapril Ramipril Candesartan | Current theories suggest ACE inhibitors cause a rise in bradykinin, which can result in psoriatic lesions | Guttate psoriasis Plaque psoriasis Palmoplantar psoriasis Pustular psoriasis Psoriasiform drug eruptions | ACE inhibitors are the cause of cutaneous side effects in 58.3% of patients Latency period of 4.1 to 8.4 weeks |
| Interferons | IFN-α IFN-β IFN-γ | IFNs can alter cells of the immune system as well as keratinocytes | Psoriatic plaques Punctiform psoriatic foci at injection sites (IFN-γ) | All forms of IFN can cause de novo psoriasis Latency period of 1 week to 6 months |
| Corticosteroids | Prednisone | Mechanism unknown | Generalized pustular psoriasis Erythrodermic exacerbations | |
| Antibiotics | Tetracycline | Decrease intracellular cAMP, exert effects on the arachidonic acid system, and photosensitize the skin | Koebner psoriasis | Tetracyclines have been shown to localize in psoriatic lesions |
| TNF-α inhibitors | Adalimumab Etanercept Infliximab Certolizumab | Proinflammatory cytokines produced by plasmacytoid dendritic cells are likely to be involved with inducing psoriasis | Plaque type psoriasis Pustular psoriasis Guttate psoriasis Scalp psoriasis Palmoplantar psoriasis | TNF-α induced psoriasis more frequently associated with females and smoking Anti-TNF-α-induced psoriasis can be managed with corticosteroids, but utilization of another biologic or TNF-α should be considered Psoriasis will recur after switching treatment in ~50% of cases |
| Immunomodulators | Imiquimod | Stimulates Toll-like receptor 7 | Plaque psoriasis Psoriatic erythroderma | |
| Fibrates | Gemfibrozil | No known mechanism | Psoriatic eruption | No other lipid-modulating agents are shown to induce psoriatic lesions |
| Allylamines | Terbinafine | Interferes with fungal cell wall synthesis (ergosterol biosynthesis) by inhibiting the enzyme squalene epoxidase | Plaque psoriasis Pustular psoriasis Inverse psoriasis | Latency period of 1.8 to 3 weeks |
| Aminoketone | Bupropion | No known mechanism | ||
| Angiotensin receptor blockers | Losartan Valsartan | Possible cross-reaction with ACE inhibitors, stimulating keratinocyte proliferation by increasing angiotensin II | Plaque psoriasis Possible ungual involvement |
| Latency | Drugs |
|---|---|
| <4 weeks = short | Nonsteroidal antiinflammatory drugs |
| 4–12 weeks = intermediate | Synthetic antimalarials |
| >12 weeks | Lithium |
| Variable | Beta-blockers |
Comorbidities Associated With Psoriasis
The concurrence of multiple diseases or disorders in association with a given disease (i.e., comorbidity) has been observed in psoriasis. Compared to the general population, patients with psoriasis are at a higher risk for comorbidities such as arthritis, heart disease, diabetes, cancer, and hypertension ( Table 8.5 ).
| Classification | Synonyms |
|---|---|
| Autoimmune diseases | Crohn disease and ulcerative colitis are 3.8 to 7.5 times more likely to occur in patients with psoriasis. |
| Cardiovascular disease | There is an increased risk of cardiovascular disease in patients with psoriasis. Patients with psoriasis are more frequently overweight, have an increased incidence of diabetes, have an increased incidence of hypertension, and have an atherogenic lipoprotein profile at the onset of psoriasis with significantly higher very-low-density lipoprotein cholesterol levels and high-density lipoprotein levels. |
| Metabolic syndrome | The combination of obesity, impaired glucose regulation, hypertriglyceridemia, reduced high-density lipoprotein, and hypertension is known as metabolic syndrome. Psoriasis patients have higher prevalence of metabolic syndrome. |
| Deficiency of serum concentration of 25-hydroxyvitamin D (OHD) | The 25-OHD values are significantly lower in psoriatic patients than in control subjects. Psoriatic patients with a body mass index of 27 or higher are likely to have vitamin D insufficiency. |
| Depression/suicide | The prevalence of depression in patients with psoriasis may be as high as 60%. |
| Psychologic and emotional burden of psoriasis | There are elevated rates of poor self-esteem, sexual dysfunction, and anxiety. |
| Alcohol | The prevalence of psoriasis is increased among patients who abuse alcohol. |
| Obesity | Patients with psoriasis have higher body mass index (BMI) than control subjects. |
| Quality of life | Psoriasis causes psychosocial morbidity and decrement in occupational function. |
Comorbidities tend to increase with age. Nearly half of psoriasis patients aged over 65 years have at least three comorbidities and two thirds have two or more comorbidities.
Clinical Presentations
Variations in the morphology of psoriasis
- •
Chronic plaque psoriasis
- •
Guttate psoriasis (acute eruptive psoriasis)
- •
Pustular psoriasis
- •
Erythrodermic psoriasis
- •
Light-sensitive psoriasis
- •
HIV-induced psoriasis
- •
Keratoderma blennorrhagicum (Reiter syndrome)
Variations in the location of psoriasis
- •
Scalp psoriasis
- •
Psoriasis of the palms and soles
- •
Pustular psoriasis of the palms and soles
- •
Pustular psoriasis of the digits
- •
Psoriasis inversus (psoriasis of flexural areas)
- •
Psoriasis of the penis and Reiter syndrome
- •
Nail psoriasis
- •
Psoriatic arthritis
Chronic Plaque Psoriasis
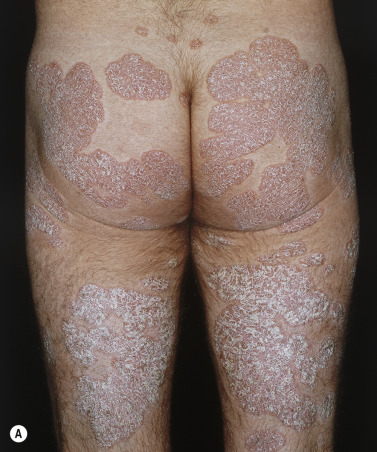
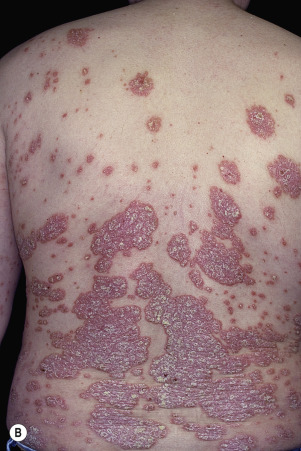
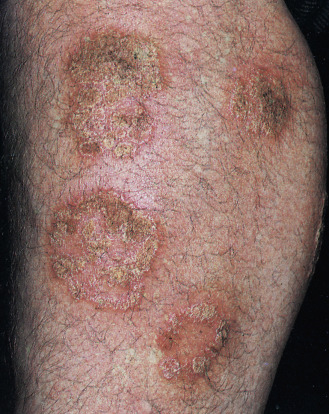
Chronic, noninflammatory, well-defined plaques are the most common presentation of psoriasis. The plaques are irregular, round to oval, with a predilection for extensor surfaces such as the elbows and knees. Plaques have a silvery surface scale and tend to be symmetrically distributed. They can appear anywhere on the skin surface. Plaques enlarge and then tend to remain stable for months or years ( Figs. 8.6 and 8.7 ). Smaller plaques or papules may coalesce into larger lesions. Numerous lesions may cover almost the entire body surface. A temporary brown, white, or red macule remains when the plaque subsides.
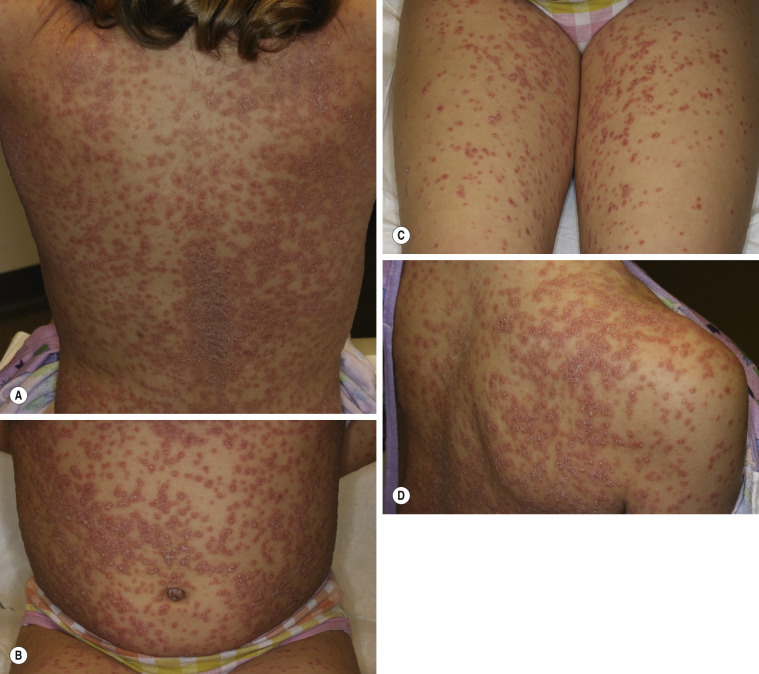
Guttate Psoriasis
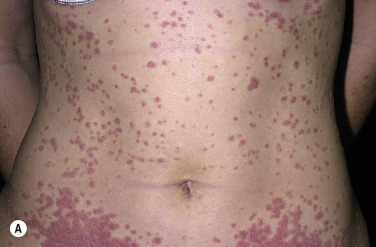
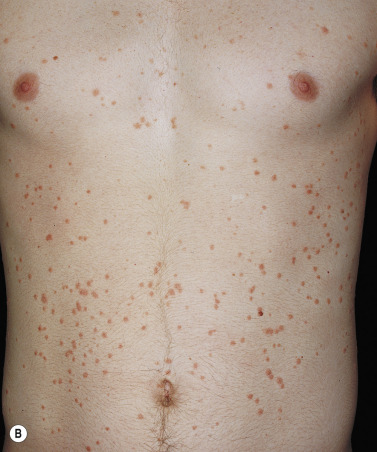
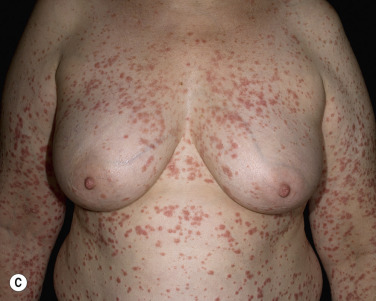
More than 30% of psoriatic patients have their first episode before age 20; in many instances, an episode of guttate psoriasis is the first indication of the patient’s propensity for the disease. Streptococcal pharyngitis or a viral upper respiratory tract infection may precede the eruption by 1 or 2 weeks. Scaling papules suddenly appear on the trunk and extremities, not including the palms and soles ( Figs. 8.8 and 8.9 ). Their number ranges from a few to many, and their size may be that of a pinpoint up to 1 cm. Lesions increase in diameter with time. The scalp and face may also be involved. Pruritus is variable. Guttate psoriasis may resolve spontaneously in weeks or months; it responds more readily to treatment than does chronic plaque psoriasis. Throat cultures should be taken to rule out streptococcal infection. Infected patients are treated for 10 days with penicillin or amoxicillin. Penicillin-allergic individuals are treated with a first-generation cephalosporin for 10 days, clindamycin or clarithromycin for 10 days, or azithromycin for 5 days.
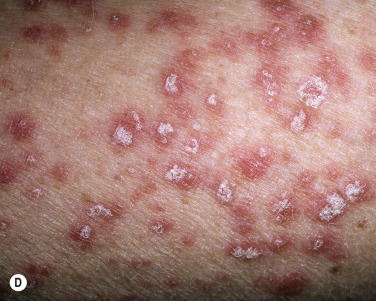
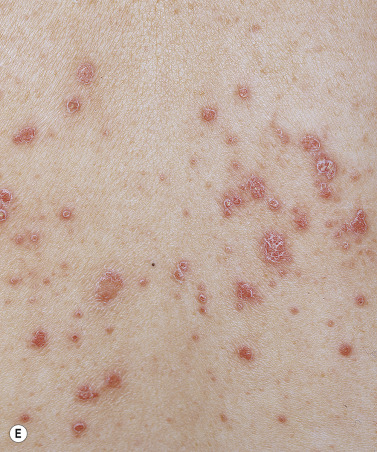
Generalized Pustular Psoriasis
This rare form of psoriasis is a serious and sometimes fatal multisystem disorder. Erythema suddenly appears in the flexural areas and migrates to other body surfaces. Numerous tiny, sterile pustules evolve from an erythematous base and coalesce into lakes of pus ( Fig. 8.10 ). The superficial, upper epidermal pustules are easily ruptured. Many patients are weak, febrile (temperatures up to 40° C) with arthralgia and myalgia. Laboratory abnormalities include elevated white blood cell count, with neutrophilia, C-reactive protein, and liver enzymes. Patients exhibit osteoarthritis, uveitis, neutrophilic cholangitis, acute respiratory distress syndrome, and cardiovascular shock. Rarely, familial cases of pustular psoriasis have been identified and homozygous and composite heterozygote loss of function mutations in the IL36R gene have been found. This genetic mutation is not seen in patients with plaque psoriasis without pustules, but may be seen in patients with palmar–plantar psoriasis (see below), acrodermatitis continua of Hallopeau (rare relapsing pustular psoriasis variant, involving distal hands and feet, may have bony changes), and acute generalized exanthematous pustular eruption. Patients with generalized pustular psoriasis and palmar plantar psoriasis have also been found to have CARD14 (caspase-activating recruitment domain, member 14) mutations. Pustular flares occur with infection, emotional stress, withdrawal from topical and systemic steroids, and pregnancy. Topical medications such as tar and anthralin may precipitate episodes in patients with unstable or labile plaque psoriasis. Wet dressings and group V topical steroids provide initial control. Acitretin, cyclosporine (CS), methotrexate (MTX), and infliximab are first-line therapies for those with generalized pustular psoriasis. They provide rapid relief in just a few days. Adalimumab, etanercept, and psoralen plus ultraviolet (UV) A light are second-line modalities.
Erythrodermic Psoriasis
Generalized erythrodermic psoriasis, like generalized pustular psoriasis, is a severe, unstable, highly labile disease that may appear as the initial manifestation of psoriasis but usually occurs in patients with previous chronic disease ( Fig. 8.11 ). Precipitating factors include the administration of systemic corticosteroids; the excessive use of topical steroids; the overzealous application of irritating topical therapy; and the consequences of phototherapy complications, severe emotional stress, and preceding illness, such as an infection. Discontinuation of CS, MTX, and ixekizumab have also resulted in erythrodermic psoriasis. Treatment includes bed rest, initial avoidance of all UV light, utilization of Burow’s solution compresses or colloidal oatmeal baths, liberal use of emollients, increased intake of protein and fluids, administration of antihistamines for pruritus, avoidance of potent topical steroids, and, in severe cases, hospitalization.
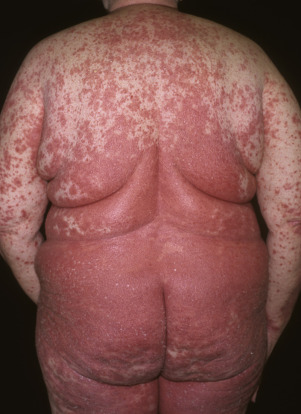
Cyclosporine and infliximab are the most rapidly acting agents. Acitretin and MTX are also appropriate first-line choices, although they usually work more slowly. Second-line agents include etanercept or combination therapy. Tar and anthralin may exacerbate the disease and should be avoided.
Light-Sensitive Psoriasis
Psoriatic patients wait for sunny summer months when, in most cases, the disease responds predictably to ultraviolet light. However, too much of a good thing can be dangerous, especially for the patient who gets sunburned in an anxious attempt to clear the disease rapidly. As a result of the Koebner phenomenon, guttate lesions or a painful, diffusely inflamed plaque forms in the burned areas ( Fig. 8.12 ). Plaques subsequently converge onto the clear, previously protected sites. Some patients do not tolerate ultraviolet light of any intensity.
Psoriasis of the Scalp
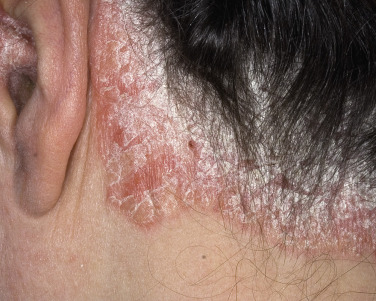
The scalp is a favored site for psoriasis and may be the only site affected. Plaques are similar to those of the skin except that the scale is more readily retained; it is anchored by hair. Extension of the plaques onto the forehead is relatively common ( Fig. 8.13 ). A dense, tight-feeling scale can cover the entire scalp. Even in the most severe cases, the hair is not permanently lost. Permanent hair loss may occur if scale is chronically removed along with the hair. Scalp psoriasis can be persistent and recalcitrant to therapy. A distinct scaling eruption of the scalp observed in children is described in this chapter in the section concerning SD.
Psoriasis of the Palms and Soles
The palms and soles may be involved as part of a generalized eruption, or they may be the only locations involved in the manifestation of the disease. There are several presentations. Superficial red plaques with thick brown scale may be indistinguishable from chronic eczema ( Fig. 8.14 ). Smooth, deep red plaques are similar to those found in the flexural area ( Fig. 8.15 ).
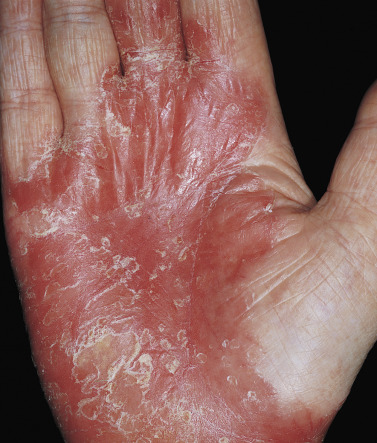
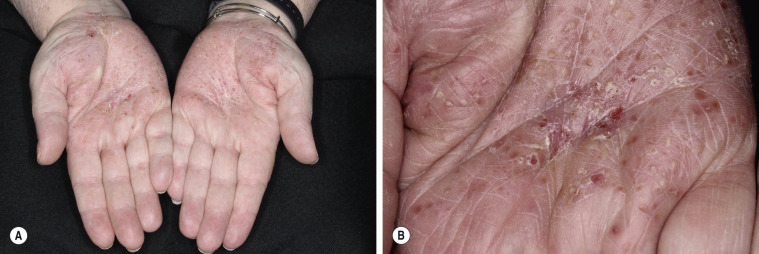
Pustular Psoriasis of the Palms and Soles
Deep pustules first appear on the middle portion of the palms and insteps of the soles; they may either remain localized or spread ( Figs. 8.16 and 8.17 ). The pustules do not rupture but turn dark brown and scaly as they reach the surface. The surrounding skin becomes pink, smooth, and tender. A thick crust may later cover the affected area. The course is chronic, lasting for years while the patient endures periods of partial remission followed by exacerbations so painful that mobility is affected. There is a considerably higher prevalence of smoking in these patients. Acitretin, MTX, psoralen plus ultraviolet light A (PUVA), narrowband ultraviolet light B light (NB-UVB), and intermittent courses of topical steroids under plastic occlusions are therapeutic alternatives.
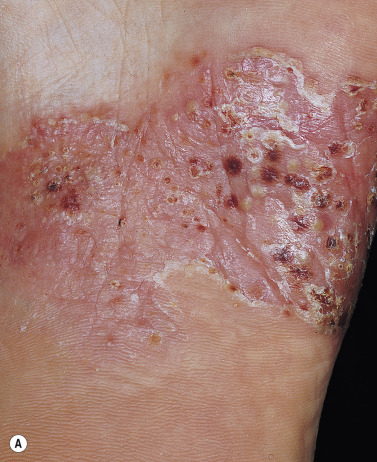
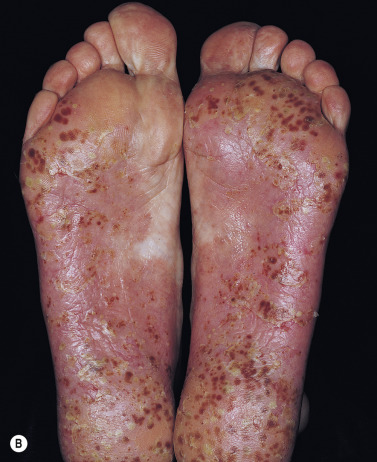
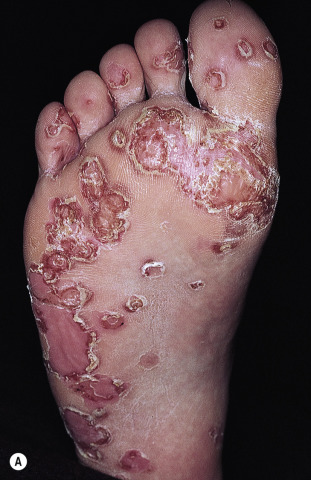
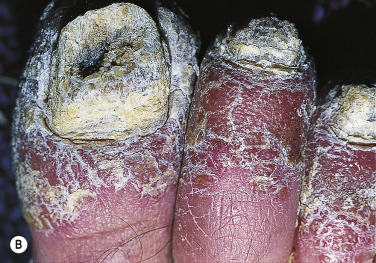
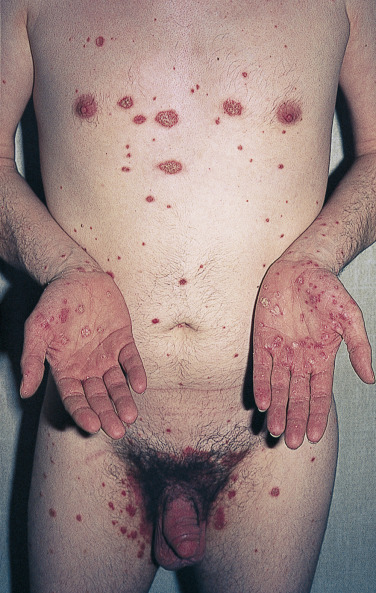
Keratoderma Blennorrhagicum (Reiter Syndrome)
Reiter syndrome appears to be a reactive immune response that is usually triggered in a genetically susceptible individual (60% to 90% of patients are HLA-B27 positive) by any of several different infections, especially those that cause dysentery or urethritis, such as Yersinia enterocolitica and Yersinia pseudotuberculosis . Psoriasiform skin lesions develop in patients with Reiter syndrome (urethritis and/or cervicitis, peripheral arthritis of more than 1 month’s duration) usually 1 to 2 months after the onset of arthritis; conjunctivitis develops in 25% of patients. The distinctive lesions, keratoderma blennorrhagica, typically appear on the soles ( Fig. 8.18A ) and extend onto the toes ( Fig. 8.18B ) but also occur on the legs, scalp, and hands. Nail dystrophy, thickening, and destruction occur. The plaques are psoriasiform with a distinctive circular, scaly border ( Fig. 8.19 ). The scaly, scalloped-edged plaques develop from coalescence of expanding papulovesicular plaques with thickened yellow, heaped-up scale. Similar lesions occur on the penis. Skin and joint symptoms have responded to MTX, acitretin, and ketoconazole.
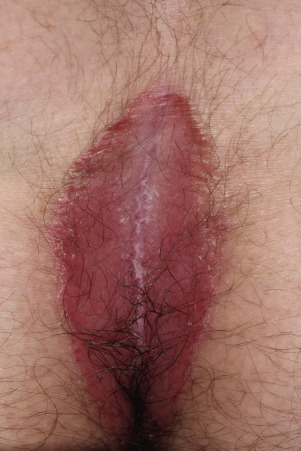
Psoriasis of the Penis and Reiter Syndrome
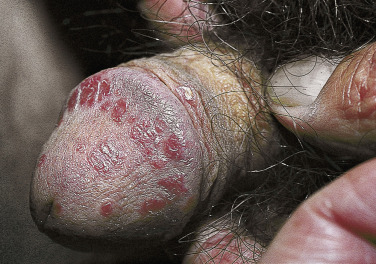
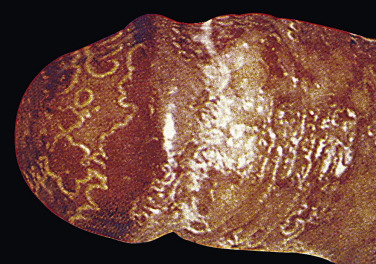
Typical psoriatic scaling plaques with white scale can appear on the body and circumcised penis ( Fig. 8.20 ). Scale does not form when the penis is covered by a foreskin. A highly characteristic psoriasiform lesion, balanitis circinata, occurs in Reiter syndrome when erosions covered by scale and crust on the corona and glans coalesce to form a distinctive winding pattern ( Fig. 8.21 ). A biopsy helps confirm the diagnosis. A potassium hydroxide examination excludes Candida infection.
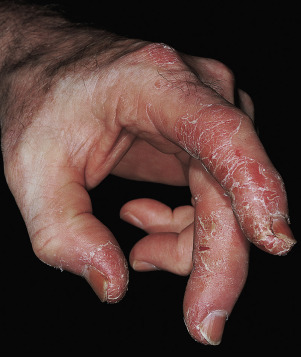
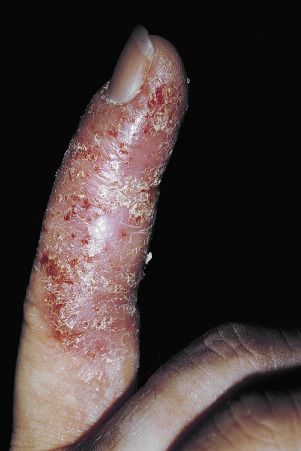
Pustular Psoriasis of the Digits
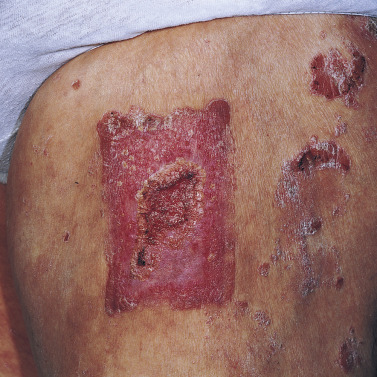
This severe localized variant of psoriasis, also known as acrodermatitis continua of Hallopeau (see Generalized Pustular Psoriasis ), may remain localized to one finger for years. Vesicles rupture, resulting in a tender, diffusely eroded, and fissured surface that continually exudes serum. The loosely adherent, moist crust is easily shed, but recurs ( Fig. 8.22 ). Localized pustular psoriasis is very resistant to therapy.
Human Immunodeficiency Virus (HIV)–Induced Psoriasis
Psoriasis may be the first or one of the first signs of human immunodeficiency virus (HIV) infection. Psoriasis in the setting of HIV may be mild, moderate, or severe. It can be atypical and unusually severe with involvement of the groin, axilla, scalp, palms, and soles. An explosive onset with erythroderma or pustular lesions that rapidly become confluent should lead one to suspect HIV. Treatment of psoriasis in the setting of HIV is challenging because the depleted T cells in HIV may be worsened by systemic psoriasis therapy. Topical steroids and vitamin D analogues (see Formulary) may be utilized for mild limited plaque psoriasis. Phototherapy, such as NB-UVB and antiretroviral therapy are the first-line therapy for moderate to severe psoriasis. Acitretin may be utilized next and third-line therapies for recalcitrant psoriasis include CS, MTX, and tumor necrosis factor (TNF)-α inhibitors. Apremilast has been utilized as well for severely affected patients. The efficacy and safety of biologic agents in the treatment of HIV-infected individual is unknown, since HIV-infected individuals are not included in clinical trials.
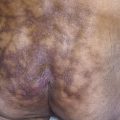
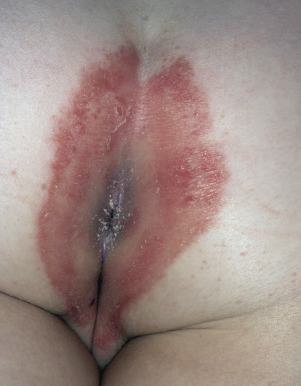
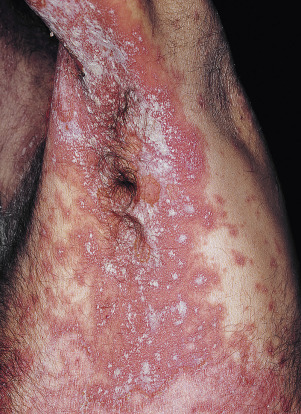
Psoriasis Inversus (Psoriasis of the Flexural or Intertriginous Areas)
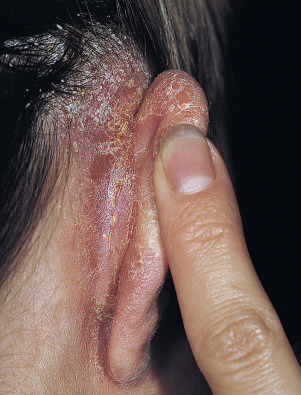
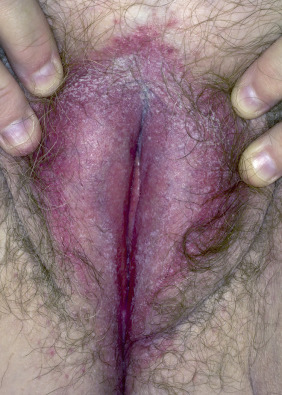
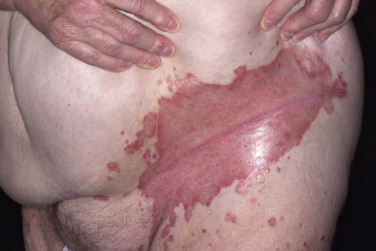
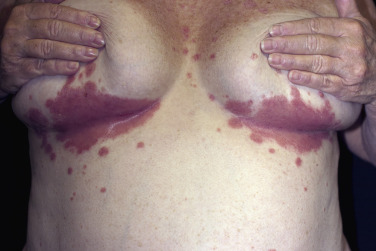
The gluteal fold, axillae, groin, submammary folds, retroauricular fold, and the glans of the uncircumcised penis may be affected. The deep red, smooth, glistening plaques may extend to and stop at the junction of the skin folds, as with intertrigo or Candida infections. The surface is moist and contains macerated white debris. Infection, friction, and heat may induce flexural psoriasis, a Koebner phenomenon. Cracking and fissures are common at the base of the crease, particularly in the groin, gluteal cleft, and superior and posterior auricular folds ( Figs. 8.23 to 8.26 ). As with typical psoriatic plaques, the margin is distinct. Pustules beyond the plaque border suggest secondary yeast infection. Infants and young children may develop flexural psoriasis of the groin that extends onto the diaper area.
Psoriasis of the Nails
Nail changes are characteristic of psoriasis and the nails of patients should be examined (see Chapter 25 ). These changes offer supporting evidence for the diagnosis of psoriasis when skin changes are equivocal or absent.
Onycholysis.
Psoriasis of the nail bed causes separation of the nail from the nail bed. Unlike the uniform separation caused by pressure on the tips of long nails, the nail detaches in an irregular manner. The nail plate turns yellow, simulating a fungal infection ( Fig. 8.27 ).
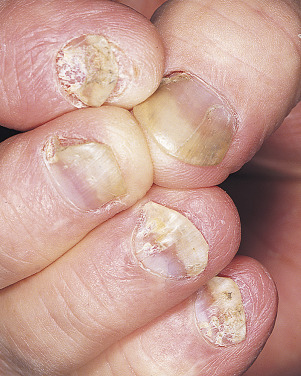
Subungual Debris.
This is analogous to fungal infection; the nail bed scale is retained, forcing the distal nail to separate from the nail bed (see Fig. 8.27 ).
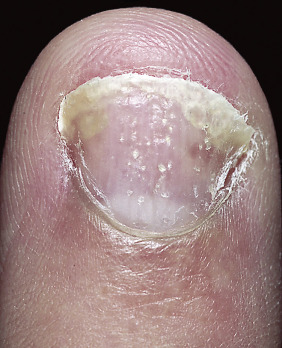
Pitting.
Nail pitting is the best known and possibly the most frequent psoriatic nail abnormality ( Figs. 8.28 and 8.29 ). Nail plate cells are shed in much the same way as psoriatic scale is shed, leaving a variable number of tiny, punched-out depressions on the nail plate surface. They emerge from under the cuticle and grow out with the nail. Many other cutaneous diseases may cause pitting (e.g., eczema, fungal infections, and alopecia areata), or it may occur as an isolated finding as a normal variation.
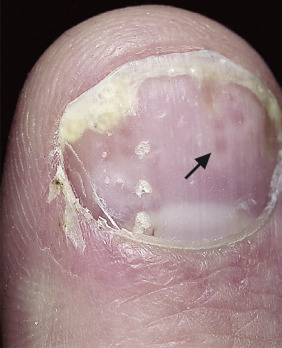
Oil Spot Lesion.
Psoriasis of the nail bed may cause localized separation of the nail plate. Cellular debris and serum accumulate in this space. The brown-yellow color observed through the nail plate looks like a spot of oil (see Fig. 8.29 ).
Nail Deformity.
Extensive involvement of the nail matrix results in a nail losing its structural integrity, resulting in fragmentation and crumbling.
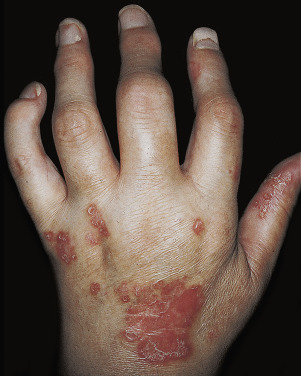
Psoriatic Arthritis
Psoriatic arthritis (PsA) is a chronic inflammatory arthropathy of the peripheral joints, spine, and entheses; it is associated with psoriasis in which rheumatoid factor (RF) and anti–cyclic citrullinated peptide (anti-CCP) measurements are usually negative . It may precede, accompany, or, more often, follow the skin manifestations. Onset may occur at any age, but peak occurrence is between ages 20 and 40; women and men are equally affected. Symmetric polyarthritis with joint pain and joint swelling often indicates erosive progressive disease. Unlike in rheumatoid arthritis (RA), the distal interphalangeal (DIP) joints are regularly involved. The presence of inflammatory arthritis in patients with psoriasis varies between 5% and 42%. Approximately 15% of patients with PsA have an onset of arthritis before the onset of psoriasis. The prevalence of PsA is higher among patients with more severe cutaneous disease. Nail involvement occurs in more than 80% of patients with PsA, compared with 30% of patients with uncomplicated psoriasis ( Fig. 8.30 ). The prevalence of nail psoriasis is highest among patients with PsA who have arthritic involvement of their fingers, but the presence of nail disease does not have predictive value in determining if a patient is at risk for PsA. Cases of arthritis have been reported to develop following trauma. Patients with PsA who become pregnant improve or even show diminishment of symptoms in 80% of cases. Despite active treatment and a reduction in joint inflammation and the rate of damage, PsA may be a progressively deforming arthritis. Fig. 8.31 highlights the key cell types and inflammatory pathways involved with PsA.
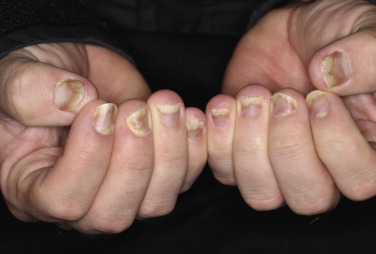
Defining Psoriatic Arthritis (PsA)
The spondyloarthritis (SpA) family of diseases includes PsA, RA, ankylosing spondylitis (AS), reactive arthritis, and enteropathic arthritis. PsA is characterized by infrequent seropositivity for RF and anti-CCP, and an association with HLA-B27 alleles, particularly in those patients with axial involvement.
Other features can include enthesitis, dactylitis, iritis, peripheral arthritis (oligoarticular asymmetric and polyarticular symmetric), spondylitis, and a variable clinical course. The heterogeneity in clinical presentation and course makes PsA difficult to classify and differentiate from other forms of SpA and inflammatory arthropathies.
Dactylitis presents as the “sausage digit” – diffuse swelling of the entire digit. Enthesitis – inflammation at the site of ligamentous and tendinous insertion – is characteristic of all the HLA-B27-associated spondyloarthropathies.
Clinical Features.
There are five recognized presentations of PsA ( Fig. 8.32 ).
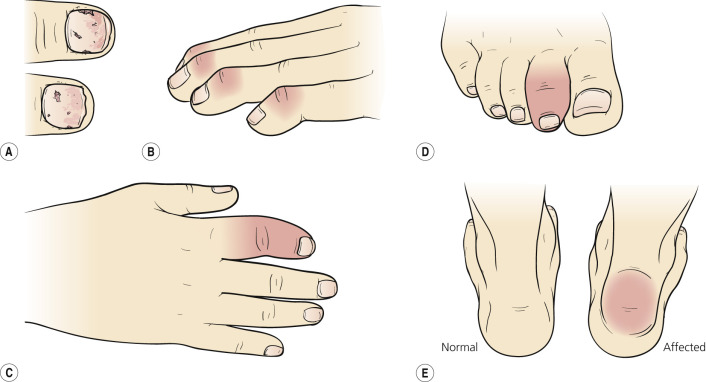
The most common pattern is an asymmetric arthritis involving one or more joints of the fingers and toes ( Fig. 8.33 ). Usually one or more proximal interphalangeal (PIP), DIP, metatarsophalangeal, or metacarpophalangeal joints are involved. During the acute phase, the joint is red, warm, and painful. Continued inflammation promotes soft tissue swelling on either side of the joint (“sausage finger”) and restricts mobility. HLA-DR7 level is significantly increased in this group with peripheral arthritis.
Moll and Wright Classification.
The Moll and Wright classification was developed more than 30 years ago and is still used today to divide PsA into five clinical subtypes ( Table 8.6 ). These patterns are often mixed and not well defined.
| Type | Percentage of All Psoriatic Arthritis Patients | Features |
|---|---|---|
| Oligoarticular, asymmetric arthritis (one or more joints) | 30–50 | Joints of fingers and toes (“sausage finger”) are involved. |
| Polyarticular, symmetric arthritis (RA-like) | 30–50 | Clinically resembles rheumatoid arthritis, rheumatoid factor negative. The small joints of the hands and feet, wrists, ankles, knees, and elbows may be involved. |
| Distal interphalangeal joint predominant | 25 | Mild, chronic, nondebilitating, and associated with nail disease. Involves hands and feet. This is the most characteristic presentation of arthritis with psoriasis. |
| Destructive polyarthritis (arthritis mutilans) | 5 | The most severe form of psoriatic arthritis involves osteolysis of any of the small bones of the hands and feet. Gross deformity and subluxation are attributed to this condition. Severe osteolysis leads to digital telescoping, producing the “opera glass” deformity. This deformity may be seen in rheumatoid arthritis. |
| Ankylosing spondylitis and sacroiliitis | 30–35 | Occurs as an isolated phenomenon or in association with peripheral joint disease. Association of HLA-B27 and spondylitis. The strongest association is in males with sacroiliitis. Asymptomatic sacroiliitis occurs in as many as one third of cases of psoriasis. It is usually asymmetric and may be associated with spondylitis. |
* To meet the Moll and Wright 1973 classification criteria for psoriatic arthritis, a patient with psoriasis and inflammatory arthritis who is seronegative for rheumatoid arthritis (RA) must present with one of the above five clinical subtypes. Criteria specificity is 98% and sensitivity is 91%.
Classification Criteria for Psoriatic Arthritis (CASPAR).
A new Classification Criteria for Psoriatic Arthritis (CASPAR) was published by the Classification Criteria for Psoriatic Arthritis Study Group ( Table 8.7 ). These criteria were established for patients with long-standing disease duration (mean, 12.5 years). Classification criteria are not yet validated for early disease.
| Inflammatory articular disease (joint, spine, or entheseal) plus the following: | |
| Clinical Finding | Score |
| Psoriasis | Current (2), history of (1), family history of (1); patient-reported history in a first- or second-degree relative |
| Nail dystrophy | (1) Including onycholysis, pitting, and hyperkeratosis |
| Negative rheumatoid factor | (1) |
| Dactylitis | Current (1), history of (1); defined as swelling of an entire digit, or a history of dactylitis recorded by a rheumatologist |
| Radiographic evidence | Juxtaarticular new bone formation, appearing as ill-defined ossification near joint margins (but excluding osteophyte formation) on plain radiographs of the hand (1) |
* To meet the Classification Criteria for Psoriatic Arthritis (CASPAR, 2006), a patient must have inflammatory articular disease and 3 points from the remaining categories; the assigned scores are in parentheses. Criteria specificity is 98.7% and sensitivity is 91.4%.
Differentiating PsA and Rheumatoid Arthritis (RA)
PsA affects men and women equally; RA affects women more commonly ( Table 8.8 ![]() ). RA affects metacarpophalangeal and PIP joints; PsA affects the DIP joints in at least 50% of patients. RA tends to have a symmetric distribution, PsA an asymmetric distribution. PsA often affects all joints of the same digit, leading to a “ray” distribution. The affected joints of patients with PsA are less tender than in patients with RA. The spine is not affected in RA; 50% of PsA patients have spinal involvement manifested as sacroiliitis and/or syndesmophytes. Enthesitis and dactylitis are features of PsA. Skin psoriasis is a defining feature of PsA. Psoriatic arthritis occurs in 2% to 3% of patients with RA, the same frequency as in the general population. Nail lesions are more common in patients with PsA than in patients with just cutaneous psoriasis.
). RA affects metacarpophalangeal and PIP joints; PsA affects the DIP joints in at least 50% of patients. RA tends to have a symmetric distribution, PsA an asymmetric distribution. PsA often affects all joints of the same digit, leading to a “ray” distribution. The affected joints of patients with PsA are less tender than in patients with RA. The spine is not affected in RA; 50% of PsA patients have spinal involvement manifested as sacroiliitis and/or syndesmophytes. Enthesitis and dactylitis are features of PsA. Skin psoriasis is a defining feature of PsA. Psoriatic arthritis occurs in 2% to 3% of patients with RA, the same frequency as in the general population. Nail lesions are more common in patients with PsA than in patients with just cutaneous psoriasis.
| Psoriatic Arthritis | Rheumatoid Arthritis | Inflammatory Osteoarthritis | Gout | |
|---|---|---|---|---|
| Distal interphalangeal joint involvement | Common | Uncommon | Common | Uncommon |
| Symmetry | Less common | Common | Uncommon | Uncommon |
| Erythema of joint | Common | Uncommon | Uncommon | Common |
| Stiffness | In morning and/or with immobility | In morning and/or with immobility | With activity | Uncommon |
| Tenderness | Mild | Severe | Mild | Severe |
| Back involvement | Common | Uncommon | Uncommon | Uncommon |
| Skin lesions | Always | Uncommon | Uncommon | Uncommon |
| Nail lesions | Common | Uncommon | Uncommon | Uncommon |
| Dactylitis | Common | Uncommon | Uncommon | Uncommon |
| Enthesitis | Common | Uncommon | Uncommon | Uncommon |
| Rheumatoid nodules | Never | Common | Uncommon | Uncommon |
| Rheumatoid factor | Uncommon | Common | Uncommon | Uncommon |
| RADIOLOGIC CHANGES | ||||
| Osteopenia | Less common | Common | Uncommon | Uncommon |
| Periostitis | Common | Rare | Uncommon | Uncommon |
| Pencil-in-cup change | Common | Rare | Uncommon | Uncommon |
| Ankylosis | Common | Rare | Uncommon | Uncommon |
| Sacroiliitis | 50%, asymmetric | Rare | Uncommon | Uncommon |
| Quality of life | Reduced | Reduced | Reduced | ? |
| Function | Reduced | Reduced | Reduced | ? |
| HLA association | CW6, B27 | DR4 | No | B14 |
| Female to male ratio | 1 : 1 | 3 : 1 | Hand/foot more common in female patients | 1 : 3.6 |
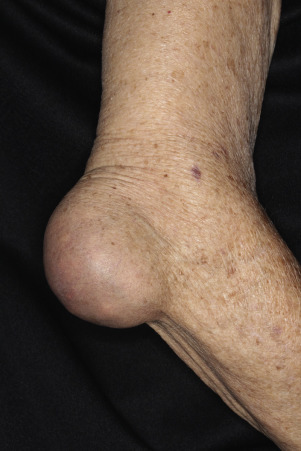
Differentiating PsA From Osteoarthritis (OA) and Gout
The presence of DIP involvement requires differentiation between PsA and OA (see Table 8.8 ![]() ). PsA is inflammatory; OA is usually not. Gout is often associated with periarticular inflammation, which is not seen in PsA. Sometimes large nodules (tophi) filled with uric acid crystals are seen in gout ( Fig. 8.34 ). Tophi may look like rheumatoid nodules or lipomas.
). PsA is inflammatory; OA is usually not. Gout is often associated with periarticular inflammation, which is not seen in PsA. Sometimes large nodules (tophi) filled with uric acid crystals are seen in gout ( Fig. 8.34 ). Tophi may look like rheumatoid nodules or lipomas.
Differentiating Psoriatic Arthritis From Other Spondyloarthropathies.
The spondylitis seen in PsA is not as severe as with AS. Patients with PsA have better mobility and less grade 4 sacroiliitis than patients with AS ( Table 8.9 ). Peripheral arthritis is more common among patients with PsA. Only 2% of patients with PsA have isolated spinal involvement, whereas 10% of patients diagnosed with AS have psoriasis.
| Feature | Psoriatic Arthritis | Ankylosing Spondylitis | Reactive Arthritis | Inflammatory Bowel Disease |
|---|---|---|---|---|
| Male to female ratio | 1 : 1 | 3 : 1 | 8 : 1 | 1 : 1 |
| Age at onset (years) | 35–45 | 20 | 20 | Any |
| Peripheral distribution | 96% Any | 25% Axial Lower limbs | 90% Lower limbs | Common Lower limbs |
| Dactylitis | 35% | Uncommon | Common | Uncommon |
| Enthesitis | Common | Common | Common | Uncommon |
| Sacroiliitis | 50% | 100% | 80% | 20% |
| Syndesmophytes | Classic and paramarginal | Classic | Classic and paramarginal | Classic |
| HLA-B | 50% | >90% | 80% | 40% |
| Psoriasis | Always | Uncommon | Uncommon | Uncommon |
| Other skin lesions | Nail changes | Uncommon | Keratoderma blennorrhagica | Erythema nodosum, pyoderma gangrenosum |
| Quality of life | Reduced | Reduced | Likely reduced | Reduced |
| Function | Reduced | Reduced | Reduced | Reduced |
Laboratory Tests.
There are no laboratory tests that specify a diagnosis of PsA. PsA patients have normal acute phase reactants, 4.6% are RF positive, and 7.6% are anti-CCP antibody positive. Erythrocyte sedimentation rate (ESR) is the best laboratory guide to disease activity ( Table 8.10 ![]() ).
).
| Laboratory Test | PsA | OA | Gout | RA | AS | ReA |
|---|---|---|---|---|---|---|
| ESR | ± | Normal | Normal (elevated during attack) | Elevated | ± | ± |
| CRP | ± | Normal | Normal | Elevated | ± | ± |
| RF | Usually absent | Usually absent | Absent | Usually present | Usually absent | Usually absent |
| CCP Ab | Usually absent | Absent | Absent | Present | Usually absent | Usually absent |
| HLA-B27 | 15%–70% | 4%–8% | 4%–8% | 4%–8% | 90% | 50%–85% |
| Synovial aspirate | Inflammatory | Noninflammatory | Inflammatory | Inflammatory | Inflammatory | Inflammatory |
Imaging Studies.
The CASPAR study identified juxtaarticular bone formation as the only radiologic differentiation between RA and PsA. PsA patients have less severe radiologic changes compared with patients with AS. PsA shows narrowing of joint space, joint erosions, and bony proliferation. Magnetic resonance imaging and ultrasonography detect enthesitis (inflammation at the point of insertion of skeletal muscle to bone).
Treatment of Psoriatic Arthritis.
Evidence-based treatment algorithms have been published ( Table 8.11 ). A step up approach is recommended. For peripheral arthritis, conventional disease modifying antirheumatic drugs are considered early in the treatment of PsA. These drugs include MTX, leflunomide, and sulfasalazine. If conventional disease-modifying antirheumatic drugs (DMARDs) fail, TNF, IL-12/23, IL-17, and PDE4 inhibitors are prescribed. Nonsteroidal antiinflammatory agents are the mainstay of therapy and usually provide adequate control, but they do not induce remissions. They are first-line agents for mild PsA. Intraarticular injections with corticosteroids may be effective. Neither treatment is capable of inhibiting the development of structural joint damage ( Fig. 8.35 ).
| Clinical Involvement | GRAPPA | EULAR |
|---|---|---|
| PREDOMINANT PERIPHERAL ARTHRITIS | ||
| DMARDs naive | csDMARDs (MTX, LFN, SSZ) Poor prognostic factors: TNFi | CsDMARDs (MTX: preferred) |
| DMARDs failure | TNFi; IL-12–23i; IL-17i; PDE4i | Adverse prognostic factors: TNFi preferred; IL-12–23i; IL17i (bDMARDs): if TNFi contraindicated PDE4i if bDMARDs contraindicated |
| bDMARDs failure | Switch bDMARDs | Switch bDMARDs or PDE4i |
| PREDOMINANT AXIAL | ||
| NSAIDs naive | NSAIDs | NSAIDs |
| NSAIDs failure | TNFi; IL-12–23i; IL-17i | TNFi preferred; IL-12–23i; IL17i: if TNFi contraindicated |
| bDMARDSs failure | Switch bDMARDs | Switch bDMARDs |
| PREDOMINANT ENTHESEAL | ||
| NSAIDs naive | NSAIDs | NSAIDs |
| NSAIDs failure | TNFi; IL-12–23i; IL-17i; PD4i | TNFi preferred; IL-12–23i; IL-17i: if TNFi contraindicated PDE4i if bDMARDs contraindicated |
| bDMARDs failure | Switch bDMARDs or PDE4i | Switch bDMARDs or PDE4i |
Methotrexate.
Methotrexate is an effective second-line agent for PsA. It is frequently used as the primary DMARD in PsA, because of its efficacy in treating both skin and joint involvement in patients with psoriatic disease. Pain and function improve dramatically 2 to 6 weeks after starting MTX therapy with 5 mg every 12 hours in three consecutive doses once a week. Lower dosages may not be effective. Methotrexate may also be given as a single dose or divided into two doses taken 12 hours apart. The amount is increased to 25 to 30 mg/week, until control is obtained, and then tapered to a maintenance dose of around 5 to 15 mg/week.
The risk of liver toxicity in patients undergoing long-term, low-dose MTX therapy for PsA is substantial, and that risk increases with the total cumulative dose and with heavy consumption of alcohol.
Etanercept (Enbrel), Adalimumab (Humira), Infliximab (Remicade), Golimumab (Simponi).
These anti–tumor necrosis factor (anti-TNF) agents bind and inhibit the activity of TNF. They are highly effective for PsA. The cost is very high. Guidelines for use are discussed under Treatment of Psoriasis. These agents are often combined with conventional DMARDs, particularly MTX.
Cyclosporine.
Cyclosporine at daily doses usually ranging from 1.5 to 5.0 mg/kg provides impressive relief from arthralgias and improvement of joint function (see Fig. 8.35 ).
Apremilast.
Apremilast, an oral inhibitor of phosphodiesterase, is effective for moderate to severe plaque psoriasis and PsA.
Treatment of Psoriasis
Many topical and systemic agents are available. None of the topical medications are predictably effective. Treatment dissatisfaction and nonadherence are high. All require lengthy treatment to give relief that is often temporary. Compliance is a problem. Patients become discouraged with moderately effective expensive topical treatment that lasts weeks or months. Limited disease can be managed with topical therapy ( Table 8.12 ). One intralesional steroid injection (5–10 mg/mL triamcinolone acetonide) can heal a small plaque and keep it in remission for months. This is an ideal treatment for patients with a few small plaques. Topical steroid creams and ointments, calcitriol, calcipotriene, tazarotene, and tar are the mainstays of topical treatment. These agents are used with or without ultraviolet light exposure. Effective programs can be designed for patients who do not have access to a therapeutic light source and for patients who have limited disease. Without light, tar is moderately effective, but persistent use of calcipotriene or tazarotene can clear the disease and offers the patient substantial remission periods. Topical steroids work quickly, but total eradication of the plaques is difficult to accomplish; remission times are short, and the creams become less effective with continued use.
| Treatment | Advantages | Disadvantages | Comments |
|---|---|---|---|
| Topical steroids | Rapid response, controls inflammation and itching, best for intertriginous areas and face, convenient, not messy | Temporary relief (tolerance occurs), less effective with continued use, atrophy and telangiectasia occur with continued use, brief remissions, very expensive | Best results occur with pulse dosing (e.g., 2 weeks of medication and 1 week of lubrication only); plastic occlusion is very effective but is not used in intertriginous areas |
| Calcipotriene (Dovonex) Calcitriol (Vectical) | Well tolerated, long remissions possible | Burning, skin irritation, expensive | Best for moderate plaque psoriasis; newer combination product is more effective (calcipotriene hydrate and betamethasone dipropionate) |
| Tacrolimus and pimecrolimus | Facial and intertriginous psoriasis, does not cause atrophy | Most common side effect is burning and itching | Only effective for plaque psoriasis if occluded |
| Anthralin (Dritho Cream) | May induce remissions | Irritating, avoid face and intertriginous areas. Stains hair and clothes | More effective when applied with phototherapy. Apply and leave on for 30 minutes and then wash off |
| Tazarotene (Tazorac) | Effective, long remissions possible | Irritating, expensive; pregnancy category X; excreted in mammalian milk | Topical steroids can control irritation and enhance effectiveness |
| Tar | New preparations are pleasant | Only moderately effective in a few patients | Most effective when combined with UVB |
| UVB and lubricating agents or tar | Insurance may cover part or all of treatment, effective for 70% of patients, no need for topical steroids | Expensive, office-based therapy | Used only on plaque and guttate psoriasis, travel and time required |
| Tape or occlusive dressing | Convenient, no mess | Expensive, only for limited disease | May be used to occlude topical steroids |
| Intralesional steroids | Convenient, rapidly effective, long remissions | Only for limited areas, atrophy and telangiectasia occur at injection site | Ideal for chronic scalp and body plaques when small and few in number |
| Excimer laser | Useful for palmoplantar, scalp and nail psoriais | Only for limited areas, office-based treatment | Best utilized for isolated plaques and as a adjunct treatment for topical and systemic agents |
Patients with psoriasis covering more than 5% of the body need special treatment programs (see Table 8.1 ).
Determining Degree of Inflammation.
The most common form of psoriasis is the localized chronic plaque disease involving the skin and scalp. It must be determined whether the plaque is inflamed before instituting therapy ( Fig. 8.36 ). Red, sore plaques can be irritated by tar, calcipotriene, and anthralin. Irritation can induce further activity. Inflammation should be suppressed with topical steroids and/or antibiotics before initiation of other treatments.
Determining the End of Treatment.
The plaque is effectively treated when induration has disappeared. Residual erythema, hypopigmentation, or brown hyperpigmentation is common when the plaque clears; patients frequently mistake the residual color for disease and continue treatment. If the plaque cannot be felt by drawing the finger over the skin surface, treatment may be stopped. Patients should apply an emollient to the previously treated areas to keep the skin healthy and prevent recurrence.
Duration of Remission.
Among topical monotherapy, anthralin and tazarotene induce longer remissions than calcipotriene and corticosteroids; among systemic agents, longer remissions occur with acitretin than CS or MTX, but compared with the remission rate of phototherapeutic modalities, the remission rates are much less ( Fig. 8.37 ). Traditional Goeckerman therapy, conducted in a day treatment setting, is more likely to induce prolonged remissions than simple UVB phototherapy, “home Goeckerman therapy” using liquor carbonis detergens (LCD), or heliotherapy. PUVA phototherapy also induces prolonged remissions.
Some treatments are better suited for rapid clearing; others are better suited to be maintenance treatment. The optimum management involves the sequential use of therapeutic agents involving three steps, namely: the clearing phase, the transitional phase, and the maintenance phase.
Topical Therapy
Topical Steroids
Topical corticosteroids are first-line drugs for limited disease ( Box 8.1 ). There are many strengths and formulations. Corticosteroids are antiinflammatory, antiproliferative, immunosuppressive, and vasoconstrictive. Lower potency corticosteroids are used for limited periods of time on the face, intertriginous areas, areas with thin skin, and in infants. In other areas and in adults, mid- or high-potency agents are used. Patients with thick, chronic plaques require treatment with the highest potency corticosteroids, such as clobetasol. For class I corticosteroids, the available data allow for 2 to 4 weeks of use with increased risk of both cutaneous side effects and systemic absorption if used continuously for longer periods of time. A gradual reduction in the frequency of usage following clinical response is accepted practice.
- •
Indication: All forms of psoriasis
- •
Dosing
- •
Can be used as monotherapy 1 to 2 times daily.
- •
Can be combined with other topical agents, UV light, and systemic agents.
- •
- •
Potency of topical steroids
Stoughton–Cornell classification system divides steroids into seven classes (see back cover of book).
- •
Duration of dosing
- •
Class I steroids (e.g., clobetasol): available data for 2–4 weeks of treatment
- •
Less potent agents: optimal endpoint unknown
- •
Gradual reduction in usage recommended following clinical response; although optimal endpoint is unknown, unsupervised continuous use is not recommended
- •
For clobetasol and halobetasol, maximal weekly use should be 50 g or less
- •
- •
Short-term results
- •
Highly potent agents have greater efficacy than less potent agents
- •
Vehicle, usage area, patient preference, patient age, and cost after efficacy
- •
- •
Long-term results
- •
True efficacy and risks associated with long-term use are unknown since most clinical trials are of short duration.
- •
Tachyphylaxis, although not demonstrated in clinical trials, may affect the long-term results achieved in a given patient.
- •
Combination with other topicals and variations in dosing schedules may lessen risk of long-term side effects.
- •
- •
Toxicities
- •
Local – skin atrophy, telangiectasia, striae, purpura, contact dermatitis, rosacea
- •
Systemic – hypothalamic–pituitary–adrenal axis suppression may occur with use of medium- and high-potency topical steroids. This will be lessened by intermittent or localized use. Unilateral or bilateral avascular necrosis of the femoral head rarely occurs. Increased intraocular pressure, glaucoma, and cataracts have been reported with use around the eye.
- •
Risks increase when used with excessive frequency or duration.
- •
It is unknown if there is an increased risk of infection with long-term use.
- •
- •
Ongoing monitoring
- •
Assessment of growth in children using long-term topical corticosteroids
- •
Regular skin checks for all patients receiving long-term therapy to assess for atrophy
- •
- •
Pregnancy: Category C
- •
Nursing: Unknown safety
- •
Pediatric use: Because of the increased skin surface/body mass ratio, the risks to infants and children may be higher for systemic effects secondary to enhanced absorption. Growth retardation is also a potential concern. Fluticasone is commonly used in pediatric patients.
Topical steroids (see Chapter 2 ) give fast but temporary relief. They are most useful for reducing inflammation and controlling itching. Initially, when the patient is introduced to topical steroids, the results are most gratifying. However, tachyphylaxis, or tolerance, occurs, and the medication becomes less effective with continued use. Patients remember the initial response and continue topical steroids in anticipation of continued effectiveness. Long-term use of topical steroids results in atrophy and telangiectasia.
A group I through V steroid applied one to four times a day in a cream or ointment base is required for best results. Plastic occlusion of topical steroids is much more effective than simple application. Augmented betamethasone dipropionate and clobetasol are extremely potent, and occlusion is not used with these drugs. Group V topical steroids applied once or twice a day should be used in the intertriginous areas and on the face. Some plaques resolve completely, but most remain only partially reduced with continued application. Continual application for more than 3 weeks should be discouraged. Remissions are usually brief and the plaques may return shortly after treatment is terminated. Topical steroid creams applied under an occlusive plastic dressing promote more rapid clearing, but remissions are not extended.
The rapid appearance of atrophy and telangiectasia occurs when the group I topical steroids are occluded. Topical steroid solutions and sprays are useful for scalp psoriasis. Multiple small intralesional injections of plaques with triamcinolone acetonide 5 to 10 mg/mL almost invariably clears the lesion and accords long-term remission. Atrophy can occur at higher concentrations.
Betamethasone valerate foam and clobetasol propionate foam are available in 50-g and 100-g containers. The foam becomes a liquid upon contact with the skin. Clobetasol is available as a spray. These formulations are very effective and preferred by many patients to creams, ointments, and solutions for treating scalp lesions and plaque psoriasis on the trunk and extremities. Transient stinging occurs in some patients. The foam emollient formulations are very well tolerated. Moisturizers can be applied soon after application of the foam.
Intralesional Steroids
Patients with a few, small, chronic psoriatic plaques of the scalp or body can be effectively treated with a single intralesional injection of triamcinolone acetonide 5 to 10 mg/mL. The 10 mg/mL solution may be diluted with saline or 1% lidocaine with epinephrine. A few drops should be injected superficially about 1 cm apart. Most injected plaques clear completely and remain in remission for months. Atrophy and telangiectasias may appear at the injection site. The face and intertriginous areas are avoided.
Topical Calcineurin Inhibitors
The topical calcineurin inhibitors tacrolimus and pimecrolimus block the synthesis of inflammatory cytokines. They are effective for plaque psoriasis when used under occlusion. They are useful for thinner skin areas such as facial and intertriginous psoriasis. They do not cause atrophy or hypopigmentation as do topical corticosteroids in these regions. The most common side effect is burning and itching that reduces with ongoing usage and can be reduced by not applying immediately after bathing. This side effect is worse with tacrolimus. Both are pregnancy category C, are found in human milk, and are not recommended for nursing mothers.
Vitamin D Analogues (Calcipotriene, Calcitriol)
Vitamin D 3 analogues inhibit epidermal cell proliferation and enhance cell differentiation. They are effective and safe and well tolerated for the short- and long-term treatment of psoriasis. Up to 100 g/week can be used. Calcipotriene is more effective than the group II corticosteroid ointment fluocinonide 0.05%. Tachyphylaxis (tolerance) does not occur with calcipotriene. Calcipotriene solution is used for scalp psoriasis. It is not as effective as betamethasone valerate, but it does not have the corticosteroid side effect of atrophy. It is valuable for long-term scalp treatment programs.
Calcipotriene is not as effective as group I corticosteroids, but regimens using calcipotriene and group I corticosteroids are superior over either agent alone. Most patients now use the following regimen: calcipotriene is applied in the morning and a group I corticosteroid is applied in the evening for 2 weeks. Then a maintenance regimen is begun using group I corticosteroids twice daily on weekends and calcipotriene twice daily on weekdays. Application for 6 to 8 weeks gives a 60% to 70% improvement in plaque-type psoriasis. Remission is maintained with long-term use of this program. Application of calcipotriene twice a day is much more effective than once-a-day application. Calcipotriene treatment can produce a mild irritant contact dermatitis at the site of application. The face and intertriginous areas are prone to this side effect.
Calcipotriene is not very effective at improving the response to UVB or NB-UVB. UVB does not inactivate calcipotriene. Hypercalcemia is reported with excessive quantities of calcipotriene applied over large surface areas. Parameters of calcium metabolism do not change when less than 100 g/week is used.
Calcitriol has similar properties to calcipotriene used in the same manner. It is limited to 200 g/week and is a pregnancy category C drug. Use with caution in patients receiving medications known to increase serum calcium levels, such as thiazide diuretics. Caution should also be exercised in patients receiving calcium supplements or high doses of vitamin D.
Calcipotriene Hydrate and Betamethasone Dipropionate.
The two-compound solution containing calcipotriene plus betamethasone dipropionate combines a vitamin D analogue and a corticosteroid. It is applied once each day and is indicated for the topical treatment of psoriasis in adults 18 years of age and older. The solution can be used acutely for 4 to 8 weeks to bring psoriasis under control. After this, control of the disease can be maintained with once-daily application as needed.
The maximum weekly dose should not exceed 100 g. Treatment of more than 30% body surface area is not recommended. It should not be applied to the face, axillae, or groin. Some patients fail to respond to the treatment. It is well tolerated in repetitive use as required for up to 52 weeks. It is very expensive.
Tazarotene
Tazarotene (0.05%, 0.1%) is available as a gel and a cream. Irritation develops in most patients. The stronger formulation is more effective but more irritating. Irritation may be reduced by use of the cream formulation, use of the lower concentration product, combination use with moisturizers, application on alternate days, short-contact (30 to 60 minutes) treatment, and application in combination with topical corticosteroids.
Topical steroids can control irritation and enhance effectiveness. Group I, II, and IV topical steroids are all effective when used in combination with tazarotene. The retinoid may prevent corticosteroid atrophy. Treatment consists of application of tazarotene once a day and a topical steroid approximately 12 hours later.
Remission of psoriasis may be maintained for at least 5 months with a regimen of tazarotene gel 0.1% applied Mondays, Wednesdays, and Fridays and clobetasol ointment applied Tuesdays and Thursdays.
Some clinicians believe that a short-contact regimen is effective. Tazarotene is applied for 5 minutes and then washed off. This regimen minimizes irritation but maintains efficacy.
Patients treated with UVB and tazarotene responded more favorably than patients treated with UVB alone. Tazarotene causes thinning of the stratum corneum of the epidermis, allowing patients to burn more easily. UV doses are reduced by at least one third if tazarotene is added to a course of phototherapy. Tazarotene remained chemically stable when used in conjunction with UVB or UVA phototherapy. Tazarotene is a teratogenic retinoid and is pregnancy category X; therefore use in pregnancy is avoided.
Halobetasol and tazarotene (Duobril lotion).
Duobril lotion (halobetosol 0.01% and tazarotene 0.045%) is indicated for daily topical application in adults. Thirty-six to 45% of patients experience clear to almost clear skin after 8 weeks of application. Contact dermatitis occurs in 7% of patients.
Coal Tar
Coal tar suppresses DNA synthesis. The Goeckerman regimen consists of the combination of crude coal tar along with ultraviolet light. It is very effective treatment for patients with severe psoriasis. Many formulations of coal tar are available over the counter.
Coal tar formulations are often poorly tolerated because of staining of clothes and tar odor. Newer preparations are more cosmetically acceptable. They may be applied daily for extended periods of time. The response is unpredictable but many patients are gratified with this safe, inexpensive treatment.
Anthralin
Antralin cream (Anthralin 1% cream) is applied to the skin and scalp daily for 30 minutes and then washed off. The mechanism of action is not known. Antralin is more effective when used along with phototherapy and or a topical steroid. Skin irritation and staining of the clothes and hair are side effects. Avoid the face and intertriginous areas.
Nonmedicated Topical Moisturizers
Nonmedicated topical moisturizers may be effective treatment. They are applied one to three times daily. Patients who are dissatisfied with irritating, expensive prescription topical medications often turn to use of just topical moisturizers with a gratifying effect. They are more effective at maintaining skin health and thereby decrease skin inflammation.
Ultraviolet Light Therapy
Ultraviolet light therapy has been used for decades to successfully treat psoriasis. It is cost-effective and safe when used appropriately. Many dermatologists continue to offer this treatment option. The treatment is inconvenient in that it requires many visits to the physician’s office. Light therapy is therefore used less often in this era of biologic therapy. There is a significant positive correlation between patients’ responses to sunbathing and their responses to short-wave ultraviolet light (UVB) phototherapy. Sunlight nonresponders have a 70% chance of failure with UVB phototherapy; sunlight responders have an 80% chance that clearance treatment will succeed. UVB is delivered in broadband (UVB) and narrowband forms (NB-UVB). It can be combined with MTX or acitretin for enhanced effectiveness. Guidelines of care for the treatment of psoriasis with phototherapy and photochemotherapy have been published.
Photochemotherapy
The treatment known as PUVA is so designated because of the use of a class of drugs called psoralens (P), along with exposure to long-wave ultraviolet light (UVA). Patients ingest a prescribed dose of methoxypsoralen approximately 2 hours before being exposed to a carefully measured amount of UVA in a uniquely designed enclosure. A major advantage of PUVA is that it controls severe psoriasis with relatively few maintenance treatments, and it can be done on an outpatient basis. Substantial exposure to PUVA increases the risk of nonmelanoma skin cancer and melanoma. PUVA is used less frequency in this age of biologic treatments.
Tape or Occlusive Dressings
One study showed that adhesive occlusive dressings applied and changed every week were therapeutically superior to a group V topical steroid and comparable to UVB therapy. Complete clearing occurred in 47% of the cases in an average of 5 weeks; another 41% of those tested showed improvement. Waterproof tape with low-moisture vapor transmission applied continually for 1 week gave similar results. Two or more applications were required. This treatment may be appropriate for treating localized chronic plaques. An adhesive hydrocolloid dressing (Duoderm) may be applied alone or over topical steroids and changed every 1 to 7 days.
Treating the Scalp
The scalp is difficult to treat because hair interferes with the application of medicine and shields the skin from ultraviolet light. Symptoms of tenderness and itching vary considerably. The goal is to provide symptomatic and cosmetic relief. It is unnecessary and impractical to attempt to keep the scalp constantly clear.
Removing Scale.
Scale must be removed first to facilitate penetration of medicine. Superficial scale can be removed with shampoos that contain tar and salicylic acid (e.g., T/Gel). Thicker scale is removed by applying Baker’s P&S or 10% LCD in Nivea oil to the scalp and washing 6 to 8 hours later with shampoo or Dawn dishwashing liquid. Combing during the shampoo helps dislodge scale.
Baker’s P&S liquid (phenol, sodium chloride, and liquid paraffin) applied to the scalp at bedtime and washed out in the morning is moderately effective in reducing scale. Baker’s liquid is pleasant and well tolerated for extended periods.
LCD (10%), a tar extract of crude oil tar, is mixed with Nivea oil by the pharmacist. The unpleasant mixture is liberally massaged into the scalp at bedtime. Warming the mixture before application enhances scale penetration. A shower cap protects pillows and also encourages scale penetration. An impressive amount of scale is removed in the first few days. Nightly applications are continued until the scalp is acceptably clear.
Mild to Moderate Scalp Involvement.
When used at least every other day, tar shampoos (see the Formulary) may be effective in controlling moderate scaling. Corticosteroid solutions are expensive, but a few drops can cover a wide area. Steroid gels (e.g., fluocinonide gel, clobetasol gel, desoximetasone gel), which have a keratolytic base and penetrate hair, are effective for localized plaques. Fluocinolone acetonide 0.01% oil (peanut oil) is an effective topical steroid that can be applied to the entire scalp and occluded with a shower cap. The scalp is dampened before application. The oil base penetrates and loosens scale. Treatment is repeated each night for 1 to 3 weeks until itching and erythema are controlled.
Betamethasone valerate foam and clobetasol foam become a liquid upon contact with the skin. Foam formulations are effective and pleasant, and easily penetrate through hair. Temporary stinging may occur during application.
Small plaques are effectively treated with intralesional steroid injections of triamcinolone acetonide 5 to 10 mg/mL. Remissions following use of intralesional steroids are much longer than those following topical steroids.
Ketoconazole cream is sometimes useful. Oral ketoconazole (400 mg daily) may be effective in some cases. The possibility of drug toxicity limits its usefulness.
Treatment of Diffuse and Thick Scalp Psoriasis.
Calcipotriene 0.005% and betamethasone dipropionate 0.064% is a topical suspension for the treatment of moderate to severe psoriasis of the scalp in adults. Apply the suspension to affected areas once daily for 2 weeks or until cleared. Treatment may be continued for up to 8 weeks. The maximum weekly dose should not exceed 100 g. Patients should shake the bottle before using the product.
The following medications can be used, all of which use oil or ointment-based preparations for scale penetration. They are applied at bedtime and washed out each morning with strong detergents such as Dawn dishwashing liquid. Topical steroid solutions can be applied during the day.
Tar and Oil.
When 10% LCD in Nivea oil is applied to the scalp, covered with a shower cap, and washed out each morning, scales are removed and inflammation is suppressed. This preparation must be prepared by a compounding pharmacist.
For patients who fail topical measures, systemic immunosuppressive agents (e.g., methotrexate (MTX), CS) or biologic agents may be administered.
Treatment of Inverse/Intertriginous Psoriasis and Genital Psoriasis
Inverse psoriasis can involve the axillae; inframammary areas; abdominal, inguinal, and gluteal folds; groin; genitalia; perineum; and perirectal area. It is erythematous, less indurated, and well demarcated with minimal scale. Genital psoriasis can be distressing. Penetration of medications is facilitated with moist opposing skin surfaces. The risk of atrophy by potent topical corticosteroids is increased. These areas respond to weaker topical steroids and calcineurin inhibitors (topical tacrolimus and topical pimecrolimus). Inflammation in these areas is often misinterpreted as a yeast infection.
Systemic Therapy
Topical treatment has its limits. Many patients do not respond to the most vigorous topical programs, or the disease may be so extensive that topical treatment is not practical.
Moderate to severe psoriasis, variably defined as patients with 5% or more involvement of body surface area or patients unresponsive to topical therapy, can be treated with several modalities including phototherapy, retinoids, MTX, or biologic agents (see Table 8.1 ).
A number of systemic drugs are available, some of which have potentially serious side effects. Methotrexate is highly effective, relatively safe, and well tolerated, but the need for periodic liver biopsies discourages some patients and physicians. Acitretin is used as a monotherapy for plaque, pustular, and erythrodermic forms of psoriasis. Acitretin may cause dry skin, alterations in triglycerides, and muscle soreness. Cyclosporine is rapidly effective, but long-term use may be associated with loss of kidney function. Biologic drugs are safe and effective for moderate to severe psoriasis.
Rotational Therapy
Rotational therapy of systemic agents and phototherapy can also be used to minimize duration of treatment with individual drugs and reduce cumulative toxicities. Rotational therapy is useful for CS and MTX, where adverse effects are related to duration of treatment with interruption of treatment allowing for recovery from reversible side effects. The use of rotational therapy has diminished since the advent of the biologic agents.
Combination Therapy of Systemic and Biologic Agents
Combination therapy improves efficacy while decreasing toxicity. The combination of MTX and CS is effective for recalcitrant psoriasis, resulting in a lower cumulative dose of each agent. MTX can also be combined with phototherapy or with low-dose acitretin. MTX can be combined with all the biologic therapies. Mycophenolate mofetil has been used in combination with CS, allowing for dose reduction of CS.
Treatment of Pregnant or Lactating Women.
Emollients and low- to moderate-potency topical steroids are first-line therapy. Second-line treatment is NB-UVB phototherapy or broadband ultraviolet B, if NB-UVB is not available. Tumor necrosis factor-α inhibitors including adalimumab, etanercept, and infliximab may be used, as may CS and systemic steroids (in the second and third trimesters).
Methotrexate
Indications and Use.
Methotrexate has been used for more than 40 years to treat severe psoriasis and is particularly effective in controlling erythrodermic and generalized pustular psoriasis ( Boxes 8.2 to 8.7 ![]() , Tables 8.13 and 8.14
, Tables 8.13 and 8.14 ![]() ). It induces remissions in the majority of treated patients and maintains remissions for long periods with continued therapy. MTX is also effective for PsA. It is relatively safe and well tolerated. Methotrexate is used alone or in combination with biologics. Safe use of MTX requires appropriate monitoring. Awareness of the risk factors for hematologic toxicity, primarily decreased renal function, will significantly reduce this side effect. Awareness of the risks for hepatic toxicity is crucial. Patients without hepatic risk factors may not require routine liver biopsies. Folic acid supplementation is recommended to increase the safety and decrease the potential side effects.
). It induces remissions in the majority of treated patients and maintains remissions for long periods with continued therapy. MTX is also effective for PsA. It is relatively safe and well tolerated. Methotrexate is used alone or in combination with biologics. Safe use of MTX requires appropriate monitoring. Awareness of the risk factors for hematologic toxicity, primarily decreased renal function, will significantly reduce this side effect. Awareness of the risks for hepatic toxicity is crucial. Patients without hepatic risk factors may not require routine liver biopsies. Folic acid supplementation is recommended to increase the safety and decrease the potential side effects.