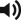Introduction
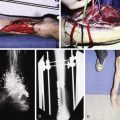
Flap prefabrication and prelamination are distinct techniques designed to address complex reconstructive needs. While these techniques are generally not primary reconstructive options, their usage has increased in response to demands for more sophisticated reconstructive efforts. In reconstructive surgery, a delicate balance exists between the availability of matching donor tissue and the complexity of the recipient defect. Prefabrication and prelamination techniques have been applied to areas where a special surface, contour, or structure is desired and reconstructive goals cannot be met by conventional means. Areas of the body where prefabrication ( Fig. 5.1 ) and prelamination have played a role in reconstruction include facial subunits, facial cartilage, facial skeleton, oropharynx/esophagus, and the penis ( Tables 5.1–5.5 ). This chapter focuses on problems of the head and neck region, where aesthetic, structural, and functional needs are complex and demanding, with scarce reconstructive options.

| Defect | Technique | Flap location | Technique description | References |
|---|---|---|---|---|
| Lips | ||||
| Direct axial flap Prefabrication Prelamination Prelamination | Scalp/submental scalp Submental forearm | Staged transfer of hair-bearing flap from scalp/submental Vascular pedicle implant + secondary transfer Tissue expander + skin graft on underside of submental platysma flap Subfascial skin grafts in radial forearm flap | Hyakusoku et al. Pribaz and Guo Pribaz and Fine Costa et al. Baudet |
| Cheek | ||||
| Direct axial flap Prefabrication Prelamination | Submental neck/upper chest or distant forearm | Submental island flap to cheek Implant vascular pedicle beneath skin and over a tissue expander with secondary transfer Subfascial skin graft over a tissue expander in radial forearm territory, or subfascial mucosal graft and silicone sheeting in radial forearm territory (± nerve) | Martin et al. Faltaous et al. Kim Khouri et al. Pribaz et al. Pribaz et al. Rath et al. Rath et al. |
| Neck | ||||
| Burn contracture | Prefabrication | Thigh forearm upper chest | Implantation of pedicle subcutaneously placed over a tissue expander and subsequently transferred to neck | Khouri et al. Pribaz et al. |
| Defect | Technique | Flap location | Technique description | References |
|---|---|---|---|---|
| Nose | ||||
| Existing laminated flap Prelamination Prelamination | Ear Forehead Forearm | Ascending helical free flap based on superficial temporal artery Skin graft for lining and cartilage for support in paramedian forehead flap Skin graft for lining and cartilage for support in radial forearm flap | Pribaz and Falco Gilles Pribaz et al. Costa et al. Baudet |
| Ear | ||||
| Absent ear | Prelamination | Forearm | Carved costal cartilage graft or silicone framework covered with radial forearm fascia and skin graft with secondary transfer to ear | Costa et al. Baudet Hirase et al. |
| Trachea/Larynx | ||||
| Prelamination Prefabrication | Radial forearm Radial forearm fascia | Mucosa or ear cartilage prelaminated onto antebrachial fascia for tracheal defects Free radial forearm fascial flap wrapped around upper trachea, which is subsequently moved for hemilarynx reconstruction | Vranckx et al. Delaere et al. |
| Defect | Technique | Flap location | Technique description | References |
|---|---|---|---|---|
| Maxilla | ||||
| Existing laminated flap Prelamination | Second toe proximal phalanx Scapular or fibula | Osteointegrated implants placed into proximal phalanx with composite flap based on dorsalis pedis artery and transferred to premaxilla Prelaminate scapular bone or fibula dermal graft + wrap with silicone sheeting ± osteointegrated implants | Pribaz and Guo Holle et al. Rohner et al. |
| Mandible | ||||
| Segmental or total loss | Prelamination | Scapular | Cancellous bone in carrier tray placed in scapular flap territory + secondary transfer to mandible | Orringer et al. |
| Defect | Technique | Flap location | Technique description | References |
|---|---|---|---|---|
| Intraoral | ||||
| Mucosa-lined soft tissue loss | Prelamination | Forearm | Mucosal grafts placed over radial forearm and later transferred for intraoral reconstruction | Rath et al. Carls et al. Chiarini et al. |
| Esophagus | ||||
| Prelamination Prelamination | Radial forearm Tensor fascia lata (TFL) | Skin surface of the radial forearm flap is rolled into a lumen during first stage to allow healing of suture line; during second stage 2 weeks later, microvascular transfer is completed Longer defect requires TFL flap with skin lumen prelaminated the same way | Chen et al. |
| Defect | Technique | Flap location | Technique description | References |
|---|---|---|---|---|
| Penis | ||||
| Absence secondary to tumor or trauma | Prelamination | Lateral arm radial forearm fibula | Prelamination with skin tube for neo-urethra reconstruction with subsequent transfer of osteocutaneous flap when using fibula | Young et al. Capelouto et al. |
They terms flap “prefabrication” and “prelamination” are two distinctive entities in reconstructive surgery. Flap prefabrication , first introduced by Shen in 1982, describes a two-stage process: the introduction of a vascular pedicle into a body of tissue that bears desired characteristics of the area to be reconstructed, followed by a transfer of this neovascularized tissue into the defect based on its implanted vascular pedicle. Flap prelamination , a term coined by Pribaz and Fine in 1994, also refers to a two-stage process, whereby one or more tissues are engrafted into a reliable vascular bed to create a composite flap. This flap is subsequently transferred on its original vascular supply, en bloc , for reconstruction. A clear understanding of these two techniques is helpful in choosing the appropriate method to deal with a specific clinical problem and also for scientific communication.
Flap Prefabrication
Concept
The first step in planning a reconstruction is to delineate the specific needs. It is desirable to use flaps that provide a good color match and restore surface and contour. Regarding head and neck reconstruction, in particular, the recipient site may require further specialized flaps, such as hair-bearing or mucus-producing flaps, for optimal reconstruction. Although there may be local flap options with the desired characteristics, these may not have a reliable axial blood supply on which they can be transferred. The technique of flap prefabrication provides this by implanting an axial blood supply into the donor tissue, rendering that tissue transferable once neovascularization has occurred.
Technique
A vascular pedicle includes at least an artery and its venae comitantes, surrounded by an adventitial cuff. It may be available locally or, if not, imported as a small free flap and implanted beneath the intended donor tissue. The distal end of the pedicle is ligated. To prevent scarring around the base of the pedicle and to facilitate secondary harvest of the prefabricated flap, a short segment of Gore-Tex (polytetrafluoroethylene) tubing or thin silicone sheeting can be placed around the pedicle up to the undersurface of the tissue that the new pedicle is expected to support ( Fig. 5.1 ). A nonadhesive sheeting may be placed under the implanted pedicle, away from the proposed flap, not only to facilitate secondary elevation of the flap but also to shunt the direction of neovascularization from the pedicle toward the flap to be prefabricated. A tissue expander is frequently used in this capacity. In experimental animal models, a neural island flap can also be prefabricated. In this model, a peripheral nerve can be placed under subcutaneous tissue and the intrinsic vasculature of the nerve can supply the flap following a delay period.
Flap Maturation
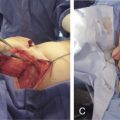
Neovascularization between the implanted pedicle and the donor tissue matures by 8 weeks in humans. However, experimentally, maturation may possibly be hastened. Angiogenic factors, such as basic fibroblast growth factor (bFGF) and vascular endothelial growth factor (VEGF), have been shown in animal models not to improve flap survival by increasing its vascularity. Flap delay has also been successfully employed, experimentally, to accelerate neovascularization. By virtue of the need to prefabricate the donor tissue (which lacks a good axial blood supply), the delay is accomplished by progressively raising the flap tissue off its non-axial blood supply, thus rendering it dependent on the implanted pedicle. Although the contact area of the vascular pedicle did not seem to be significant in our initial experiments, others have found that there is a proportional relationship of pedicle size and the rate of neovascularization.
Another helpful adjunct is the use of tissue expansion . Tissue expansion and flap prefabrication both are two-stage procedures and thus complement each other nicely. The implanted pedicle is placed directly underneath the donor tissue and above the expander. Expansion can start as early as 1 week and may be monitored by Doppler ultrasonography. Expansion can be as aggressive, as allowed by the continued presence of the Doppler signal of a patent pedicle and clinical observation of flap color. Tissue expansion accomplishes four things: (1) it provides an abundance of tissue that facilitates donor site closure following final transfer; (2) it thins out the donor tissue flap, which is helpful in head and neck reconstruction; (3) it provides mechanical stretch that stimulates endothelial cells to proliferate, thereby enhancing the rate of neovascularization; and (4) it focuses the direction of the neovascularization from the implanted pedicle toward just the side of the prefabricated flap. Assessment of flap vascularity and viability by modalities designed to evaluate perfusion may be useful in preoperative planning and optimizing flap harvest.
Flap Transfer
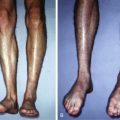
During the second stage of flap prefabrication, the prefabricated flap is transferred to its final location, based on its newly acquired axial blood supply. This can be done locally as a pedicled flap if close to the defect or via microvascular anastomosis if prefabrication is at a remote site ( Fig. 5.2 ). A commonly observed problem seen after flap transfer is transient venous congestion. This may be caused by unequal neovascularization of the lower-pressured venous system compared with the higher-pressured arterial system of the same pedicle. This problem can be ameliorated in several ways. All maneuvers that enhance neovascularization, including flap delay, lengthening maturation time, or increasing the contact area between the pedicle (usually in the form of a fascial flap) and the donor tissue, would help. Also, other strategies such as delayed insetting, temporary leeching (chemical or medicinal), avoiding flap folding or, if possible, performing an additional venous anastomosis using a native subcutaneous vein in the prefabricated flap can all help to minimize flap venous congestion. Finally, there was a recent report of a further surgical delay performed at 6–7 weeks after pedicle implantation and 2 weeks before flap transfer that was shown to minimize venous congestion in that series.