Key points
- •
Poly -l -lactic acid works by a mechanism that it is completely different from other products for soft tissue augmentation; it produces effects that are long-lasting, but not permanent.
- •
Poly -l -lactic acid has been shown safe and effective for the treatment of facial lipoatrophy through many studies in immunocompromised and immunocompetent patients.
- •
Many patients with human immunodeficiency virus experience facial lipoatrophy, which is a debilitating condition that appears to be brought on by components of highly active anti-retroviral therapy.
- •
Poly -l -lactic acid is used more commonly for off-label cosmetic purposes in the United States; this off-label use is legal when the off-label use is consistent with the common practices of other dermatologists and plastic surgeons.
- •
Poly -l -lactic acid has been shown to be effective for treating localized lipoatrophy in medical conditions.
- •
The formation of granulomas and nodules can be avoided by using correct technique that includes complete reconstitution with adequate dilution of poly -l -lactic acid; proper injection technique with a strategy of treating facial units and not wrinkles; a wait-and-see approach with adequate time of 3–4 weeks between sessions; and appropriate training.
Introduction
Poly -l -lactic acid (PLLA; Sculptra®; Dermik, Bridgewater, NJ, USA) is categorized as an injectable medical device with approval by the US Food and Drug Administration (FDA) for the treatment of facial lipoatrophy in patients with human immunodeficiency virus (HIV). Facial lipoatrophy can develop due to (1) the natural course of aging in healthy adults; (2) surgical procedures to cause extensive weight loss; and (3) specific disease states, either inherited or acquired, in which lipodystrophy syndrome causes metabolic disturbances that lead to abnormalities in adipose tissue ( ). Although the etiology is unclear, studies suggest a strong link between lipodystrophy syndrome and treatment with protease inhibitors and nucleoside reverse transcriptase inhibitors, an effect that may result from individual or synergistic drug activity ( , , , ). Lipodystrophy syndrome has proven difficult to define, leading to widely varying estimates of prevalence among patients with HIV that range from 15% to over 80% ( ). The highest estimate reported that lipodystrophy syndrome developed in 83% of patients within 8–10 months of initiating protease inhibitor therapy ( ). Several scales for grading the severity of lipoatrophy have been proposed. However, the Facial Lipoatrophy Grading Scale ( Table 5.1 ) that was introduced in 2006 by the Facial Lipoatrophy Panel is the current standard used to grade the severity of lipoatrophy in all patients. Validation of an improved lipoatrophy scale has just been completed by the Facial Lipoatrophy Panel and the new scale should be available for use in 2009.
| Grade 0 | No facial lipoatrophy |
| Grade 1 | Mild flattening or shadowing of one or more facial regionsNo prominent bony landmarksNo visibility of underlying musculature |
| Grade 2 | Intermediate point between Grade 1 and Grade 3 |
| Grade 3 | Moderate concavity of one or more facial regionsProminence of bony landmarksMay have visibility of underlying musculature |
| Grade 4 | Intermediate point between Grade 3 and Grade 5 |
| Grade 5 | Severe indentation of one or more facial regionsSevere prominence of bony landmarksClear visibility of underlying musculature |
Benefits
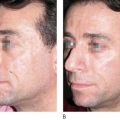
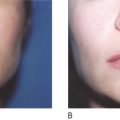
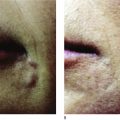
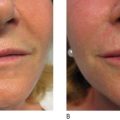
Poly -l -lactic acid is a biodegradable bioabsorbable synthetic polymer used for several decades in resorbable medical devices, such as sutures, surgical sealant meshes, screws, plates, and membranes for guided tissue regeneration ( , ). In soft tissue augmentation, PLLA is used exclusively to produce volume in areas of ‘sinking and sagging’ skin. It is not intended for the focused treatment of specific wrinkles. Rather, PLLA is intended for the treatment of areas requiring contouring or sculpting. Poly -l -lactic acid can be used with other soft tissue fillers for a variety of applications in soft tissue augmentation. It can be used to build up the maxillary fat pads, but should not be injected into the lips ( ). The effects of volume restoration can persist for as long as 18–24 months, but are not permanent ( , ). After this period, only one booster treatment is needed to maintain the cosmetic effect ( ).
Mechanism of action
Poly -l -lactic acid differs in mechanism from other fillers because in point of fact, it is not a dermal filler. To the contrary, PLLA is a biocompatible, biodegradable synthetic polymer that serves as a catalyst that stimulates the process of neocollagenesis ( , , , , ). Poly -l -lactic acid causes an immediate effect by physically occupying space. However, this initial response is temporary, lasting only 1 week or less ( ). Once the carrier solution is absorbed, a delayed but progressive volumizing effect begins. Poly -l -lactic acid causes a foreign body giant cell reaction that leads to the gradual production of collagen. This occurs through a process of hydration, loss of cohesion and molecular weight, and solubilization and phagocytosis of PLLA by the host’s macrophages. Poly -l -lactic acid is slowly degraded into lactic acid microspheres and CO 2 through non-enzymatic hydrolysis. Respiratory excretion eliminates CO 2 , leaving behind crystals, which stimulate collagen and a granulomatous reaction. This inflammatory reaction elicits (1) resorption; (2) formation of fibrous connective tissue about the foreign body; and (3) dermal fibroplasia that leads to the desired cosmetic effect ( ). Over the course of several months, the process of dermal thickening is gradually achieved ( , , , , ).
Safety history
Since 1999, over 150 000 people in more than 30 countries have been treated with PLLA for a variety of facial volume and contour deficiencies ( , ). In our own clinic, The Center for Dermatology and Dermatologic Surgery, Washington DC, USA, PLLA has been successfully used to treat lipoatrophy of the hands, feet, face, and buttocks in both immunocompetent and immunocompromised patients. In fact, we conducted a study of 61 HIV-infected patients treated with PLLA and found excellent responses to treatment in 100% of patients at 6 months follow-up. After treatment was complete, the patients were followed every 6 months from 2001 to 2005, with no reports of allergic reactions, infections, or adverse events ( ). Several other studies have shown safety and efficacy in both immunocompetent and HIV-infected patients ( , , ). For example, a study in 102 HIV-positive patients receiving highly active antiretroviral therapy (HAART) and 620 cosmetic (presumed immunocompetent) patients, treatment with PLLA resulted in optimal volume enhancement in all the patients, even at two years post-final injection. In addition, adverse events were minimal, and included five cases of palpable, non-visible papules, and one possible foreign body reaction found 1-year post-injection, resulting in a mild visible nodule. No allergic reactions were observed ( ). Clinicians have begun to identify other conditions for the use of PLLA. For example, a recent publication has reported that the use of PLLA to treat acne and varicella scars yielded excellent safety and efficacy results. Also in development, is the novel use of PLLA as an injectable craniofacial implant in a supraperiosteal location to address both soft tissue volume loss and loss of craniofacial support ( ).
In the United States, the safety record of PLLA has continued to be excellent; only a small number of adverse events have been reported in connection with PLLA treatment. In Europe, the earlier experience with PLLA was marked by a much greater propensity for adverse events. When adverse events have occurred with the use of PLLA, insufficient reconstitution and incorrect injection technique have typically been implicated. Physicians must remember that PLLA is not a filler, and requires a different approach to treatment and injecting. Thorough knowledge and training is absolutely required. Additionally, because of the gradual mechanism of dermal thickening, physicians must employ patience, as well as an artistic approach to restoring volume.
Soft tissue augmentation with PLLA is not permanent. Since the effect of augmentation is limited to approximately 24–30 months, there is less concern for errors in technique to become permanent. There is also less concern for problems that result as facial contours continue to change with age. For example, permanent fillers that initially provide satisfactory results may become more visible with age or create an unnatural appearance as patients grow older (Saylan 2005).
Indication: treatment of lipatrophy in patients with HIV
New advances in antiretroviral therapy have enabled patients with human immunodeficiency virus (HIV) and acquired immune deficiency syndrome (AIDS) to live longer, healthier lives ( ). This therapy, however, is not without adverse effects. Patients receiving HAART often develop lipodystrophy syndrome, a condition characterized by insulin resistance, hyperlipidemia, and fat loss/redistribution. Lipodystrophy-associated fat redistribution consists of fat loss from the limbs, buttocks, and face; fat accumulation in the abdomen and the dorso-cervical fat pad (the so-called ‘buffalo hump’), and sometimes breast hypertrophy ( ). The most visible component of fat redistribution, the component that causes the patient the most distress, is facial lipoatrophy ( ). Facial lipoatrophy begins in the nasolabial region, progresses outward to the temples, and finally affects the eye sockets. As fat loss progresses, the facial skin comes to rest directly on the underlying muscles, resulting in a gaunt, anorexic appearance ( ). For the patient, the psychosocial effects of facial lipoatrophy are significant. Collins surveyed HIV-infected patients regarding their feelings about their body fat changes ( ). The study found that patients often felt their doctors expressed much less concern about the effects of lipoatrophy than the amount of concern felt by the patient ( , ). Poor body image, low self-esteem, and depression were frequently described. Patients reported being socially withdrawn and that sexual relationships were adversely affected. Some patients were forced by their appearance into disclosing their HIV status. Perhaps most significant, some patients stopped taking their antiretroviral regimen in order to avoid the adverse psychosocial affects of fat wasting ( ).
Patient selection: HIV patients
PLLA can be used to treat HIV patients with any level of severity ( Table 5.1 ). Prior to beginning treatment, the physician should carefully discuss details of (1) patient satisfaction and expectations; (2) potential discomfort during or after injection; (3) risk for adverse events; (4) the results that should be expected immediately after the initial treatment and throughout subsequent sessions; and (5) typical durability expected after all sessions have been completed. The patient’s medical history should be reviewed, with attention to the use of prescription and nonprescription medications, allergies, autoimmune disorders, history of cold sores, and prior facial operations or soft tissue augmentation. Patients should be counseled to avoid medications, herbs, or supplements that might increase bleeding. Informed consent should be obtained.
PLLA-injection preparation: HIV patients
Unlike many other products for soft tissue augmentation, PLLA does not come ready to use. It must be reconstituted prior to use. Experts recommend reconstituting in a minimum of 5 ml of sterile water to allow for light-to-moderate plunger pressure, and to help distribute the product evenly ( ). In highly mobile areas, such as around the eye and around the lip, it is recommended to reconstitute PLLA in 6–8 ml. However, it is not recommended to use large dilutions in large areas or in non-mobile areas ( ).
Treatment plan: HIV patients
Physicians must fully comprehend the differences in technique between this product and dermal fillers before attempting to use PLLA. Therefore, physicians must adjust their approach from treating a ‘specific line’ to a strategy of ‘returning volume to a facial area’ ( ). Poly -l -lactic acid is not injected in the typical place where fillers such as collagen or hyaluronic acid are injected. Poly -l -lactic acid should be placed in the deep dermis or subcutaneously for creating supportive structure and foundation. Correct placement in the deep dermal and/or deep dermal subcutaneous plane is crucial; too shallow and visible nodules and/or blanching of the skin occurs ( ). Poly -l -lactic acid, due to the thickness of the hydrogel or insufficient reconstitution and preparation, tends to clump in the syringe and is somewhat difficult to inject. In addition, the gradual mechanism of dermal filling means that overcorrection is not required and must be avoided ( ). Several treatments, administered three or more weeks apart, are required to achieve maximum benefit; the exact number depends on the severity of the lipoatrophy. Mild cases may require three treatment sessions; moderate cases, approximately four treatment sessions; and severe lipoatrophy could require five to seven treatment sessions ( ). In our treatment of HIV patients with lipoatrophy, we found that the required amount of PLLA varied from 1–4 vials, depending on the degree of lipoatrophy severity and areas of involvement. The number of treatment sessions also varied. Mild cases required one to two treatment sessions; moderate cases, approximately four treatment sessions; and severe lipoatrophy required four or more treatment sessions. The amount of PLLA was gradually decreased over subsequent sessions ( ).
Injection technique: steps
Several treatments, administered four or more weeks apart, are required to achieve maximum benefit; the exact number depends on the severity of the lipoatrophy. Mild cases may require three treatment sessions; moderate cases, approximately four treatment sessions; and severe lipoatrophy could require five to seven treatment sessions ( ).
Injection steps
- •
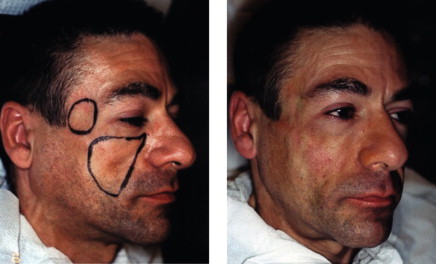
Obtain pre-operative photographs.
- •
Prior to injection, the areas of correction are prepped with isopropyl alcohol.
- •
The treatment areas are anesthetized with topical lidocaine, nerve blocks, or 1% lidocaine with or without 1 : 100,000 epinephrine infused into the reconstituted vial of PLLA.
- •
Areas of concavity are outlined with a wax marking pencil or eyeliner pencil ( Figure 5.1 ).