There has been increasing interest in the utilization of autologous platelet-rich plasma (PRP) for the management of various clinical entities. First described for use in tissue repair 1 and hemostasis, 2 PRP has been applied more recently to a multitude of medical and cosmetic conditions, including in orthopedics, 3 dentistry, 4 plastic surgery, 5 and dermatology, 6 as seen in ▶ Table 1.1. Several studies seek to evaluate the effectiveness of PRP for androgenic alopecia, skin rejuvenation, and hair transplantation surgery. However, very few randomized controlled trials exist and as such the literature is lacking qualitatively. 7 That being said, with PRP’s increasing prevalence in aesthetic practices, it is crucial to understand what PRP is and its mechanism of action. The PRP preparation process must be thoroughly understood to effectively distinguish between various available systems ( ▶ Table 1.1). 3, 5, 6, 7, 8, 9, 10, 11
|
|
Abbreviation: PRP, platelet-rich plasma. | |
1.2 Definition of Platelet-Rich Plasma
PRP is an autologous preparation of plasma with high concentrations of platelets derived from whole blood. 12 Normal platelet levels in blood range from 150,000 to 400,000 platelets/μl or 150 to 400 × 109/L. The working definition of PRP today is plasma containing greater than 1,000,000 platelets/μl, based on studies showing bone and soft tissue healing enhancement at this level. 13 Today, most PRP preparations have a concentration that is 4 to 8 times higher than that of peripheral blood, 9 depending on the preparation system.
Studies have shown that the growth factors contained within PRP increase linearly with elevated platelet concentrations. 14 Giusti et al noted the induction of angiogenesis in endothelial cells was optimized with a platelet concentration of 1,500,000 platelets/μl. Additionally, it was also determined that extremely high concentrations of platelets actually decreased angiogenesis. 15 This negative correlation was also seen in studies revealing an inhibitory impact on bone regeneration with extremely high platelet concentrations. 16
Using an autologous preparation of PRP affords multiple advantages. It decreases concern of immunogenic reactions and makes disease transmission unlikely. 17 Thus, the procedure is quite safe, well tolerated, and has minimal side effects.
PRP is differentiated from recombinant growth factors in that they are physiological, derived intrinsically from the humans rather than animal models or cell medium, and thereby contain “pure” growth factors. Additionally, they are delivered via a clot, which is a natural delivery system in humans. Recombinant growth factors are extracted from an external system, usually from other animals, and delivered via synthetic carriers. 13 PRP contains both leukocytes, which are catabolic and proinflammatory, along with platelets and plasma, which produce anabolic functions within the body. It is crucial that these seemingly opposing functions are balanced appropriately, allowing each to accomplish its purpose. Even small changes in the levels of these growth factors could create an imbalance, yielding an increase in inflammation and or pain. 10 It is hypothesized that maintaining an unadulterated balance of anabolic and catabolic functions helps maintain the optimal environment for tissue healing and growth. 9
1.3 Basic Science behind PRP and Proposed Mechanism of Action for PRP
The mechanisms of PRP are not fully understood. However, it is believed that the platelets release signaling proteins, including a multitude of growth factors, chemokines, and cytokines, that result in the promotion of cell proliferation and differentiation. 4, 12, 18, 19 Platelets are known to contain more than 20 growth factors 20 inside their α-granules, which are released upon activation in order to deliver the signaling molecules into surrounding tissue. ▶ Table 1.2 lists important growth factors, such as platelet-derived growth factor (PDGF), transforming growth factor (TGF), epidermal growth factor (EGF), vascular endothelial growth factor (VEGF), insulin-like growth factor (IDGF), and interleukin 1 (IL-1) ( ▶ Table 1.2). 14, 18, 19, 21, 22, 23, 24, 25
Growth factor | Actions |
PDGFαα, PDGF αβ, PDGF ββ |
|
TGF-β1, TGF-β2, TGF-α |
|
VEGF |
|
FGF-2, FGF-9 |
|
EGF |
|
IGF-1 |
|
CTGF |
|
Abbreviations: CTGF, connective tissue growth factor; EGF, epidermal growth factor; FGF, fibroblast growth factor; IGF, Insulin-like growth factor; PDGF, platelet-derived growth factor; PRP, platelet-rich plasma; TGF, transforming growth factor; VEGF, vascular endothelial growth factor. | |
The platelets in PRP secrete most of their growth factors within an hour of activation, 22 with residual factors being released for up to 7 days. 13 The mechanisms by which PRP likely achieves efficacy have been well described in other fields of medicine: orthopedics, surgery, dentistry, and wound healing. PRP increases the release of cytokines, which then bind to the surface of cellular transmembrane receptors, causing intracellular signaling. This effectuates change at the microscopic level including angiogenesis, collagen synthesis, extracellular matrix production, and decreased apoptosis, mimicking the mechanisms needed for dermatologic uses. 3, 26
In dermatology, the proposed mechanism of PRP varies based on its location of use. As noted, growth factors released by platelets in PRP may stimulate different targets based on specific areas treated.
1.3.1 Mechanism of Action for PRP Effects on Hair Growth
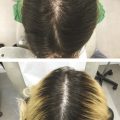
PRP stimulates hair growth via many mechanisms, but one important pathway is anagen-associated angiogenesis. 12, 27 Numerous treatments for hair loss have been aimed at increasing angiogenesis and improving blood flow to the hair follicle unit. 4 Secretion of vascular endothelial growth factor (VEGF) is largely responsible for the anagen-associated angiogenesis and has also been shown to augment growth of dermal structures. 28, 29 Beyond VEGF, the α-granules in PRP release increased levels of PDGF and, platelet-derived endothelial growth factor. These factors are hypothesized to work on the stem cells of hair follicles and stimulate neovascularization. 30 As such, PRP has been used to effectively treat male and female hair loss ( ▶ Fig. 1.1). 19 Improved circulation in the structures immediately surrounding hair follicles indicates a clear mechanism for improved hair growth. 20 Similarly, subcutaneous injections of PRP have improved the survival of skin grafts, likely by a similar mechanism of increasing blood flow. 31

Fig. 1.1 Platelet-rich plasma (PRP) used in female hair loss—before and after. Patient 1 (a) prior to PRP and (b) 4 months post treatment. Patient 2 (c, d) prior to PRP and (e, f) 4 months post treatment.
There are many other mechanisms by which PRP may aid hair growth. Li et al showed increased levels of β-catenin, extracellular signal-related kinase, and Akt signaling, all of which contribute to dermal papilla cell proliferation. 12 Activated PRP increased levels of phosphorylated extracellular signal regulated kinases and phosphorylated Akt, molecules which resulted in human dermal papilla propagation. 12 Interestingly, the effects of PRP were found to be dose dependent in this study, indicating the importance of attaining appropriate levels of PRP to achieve maximum results.
Numerous studies have detailed PRP and its anti-apoptotic effects. 32, 33 This mechanism is based on the ability of PRP to induce activation of Bcl-2 and phosphorylation of Akt, both involved in antiapoptotic regulation. Dermal papilla cells are protected from premature breakdown and remain active, thereby extending the anagen phase of the hair cycle and delaying induction into catagen and telogen phases. 34, 35, 36 Furthermore, Li et al found that PRP treatment almost doubled β-catenin transcriptional activity, which is expressed in the anagen hair follicle. This, along with PRP’s ability to upregulate fibroblast growth factor 7 (FGF-7), also aids in lengthening the anagen or growth phase of the hair cycle, as well. 12, 32 Finally, promoting FGF-7 signaling has also been shown to promote stem cell differentiation into hair follicles. 37
1.4 Mechanism of Action for PRP on Rejuvenation and Repair
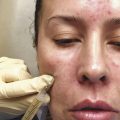
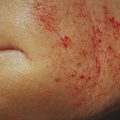
Like hair growth, there are likely several mechanisms by which PRP influences skin rejuvenation. Accumulated fragmented collagen fibrils prevent new collagen growth and lead to extracellular matrix breakdown. 38 Activated PRP increases expression of matrix metallopeptidase (MMP-1 and MMP-3), stimulating extracellular membrane remodeling and removal of damaged collagen fragments, ultimately allowing for improved, more regularly organized collagen synthesis. 39, 40 PRP contains multiple growth factors that stimulate human dermal fibroblasts and boost neocollagenesis. 40 It has also been shown to enhance the secretion of hyaluronic acid. 41 Among its many functions in the dermis, hyaluronic acid avidly binds water, thereby increasing skin volume and hydration. Taken together, these findings suggest that the PRP solution may augment extracellular matrix synthesis and is a possible treatment for skin rejuvenation and acne scaring. 42
PRP has recently been used with resurfacing ablative lasers to treat facial acne scaring. It has also been used for wound care post ablative laser treatments. 23 Ablative carbon dioxide fractional laser resurfacing has been shown to produce similar pathology to that of normal wounds. As such, adding platelets, a key element to normal wound repair, should assist and hasten tissue regeneration after ablative laser therapy. 43 While unproven at this time, it is believed that PRP’s release of α-granules containing large storage pools of growth factors, leads to more efficient and expedited tissue healing. Additionally, PRP was shown to expedite wound healing, reduce erythema, and decrease transepidermal water loss in patients post fractional ablative resurfacing.
1.5 Options for Preparation
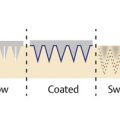
Abstraction of PRP relies on differential centrifugation of whole blood and separation of the desired components based on their specific gravity. PRP preparation can be done either manually or by automated device. Regardless, the basic process begins by drawing peripheral blood from the patient ( ▶ Fig. 1.2a–e). The tubes of blood (often containing an anticoagulant agent) are then spun in a centrifuge, according to proprietary protocols with set speed, spin cycles, and spin times. Rapid spinning layers the different blood cell lines based on mass so that platelets in plasma—hence the name platelet-rich plasma (PRP)—can be extracted from the tubes with variable concentrations of erythrocytes and granulocytes ( ▶ Fig. 1.3). Some protocols add activating substances (see below) prior to use. 44
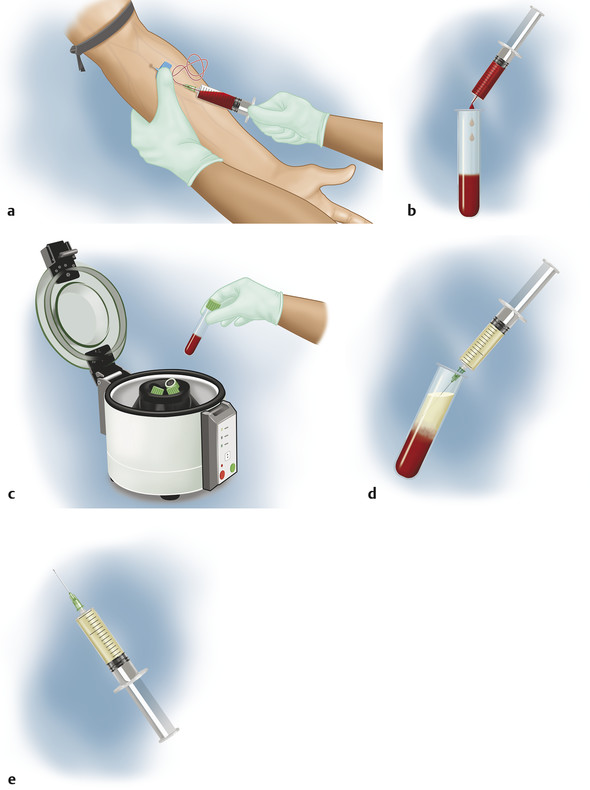
Fig. 1.2 Steps for platelet-rich plasma (PRP) collection in single spin method. (a) Step 1: blood collection—venipuncture from antecubital vein. (b) Step 2: transfer blood into collection tubes for centrifugation. (c) Step 3: place specimens in centrifuge. (d) Step 4: withdrawing PRP layer into syringe post centrifugation. (e) Step 5: syringe with PRP only ready for injection.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree