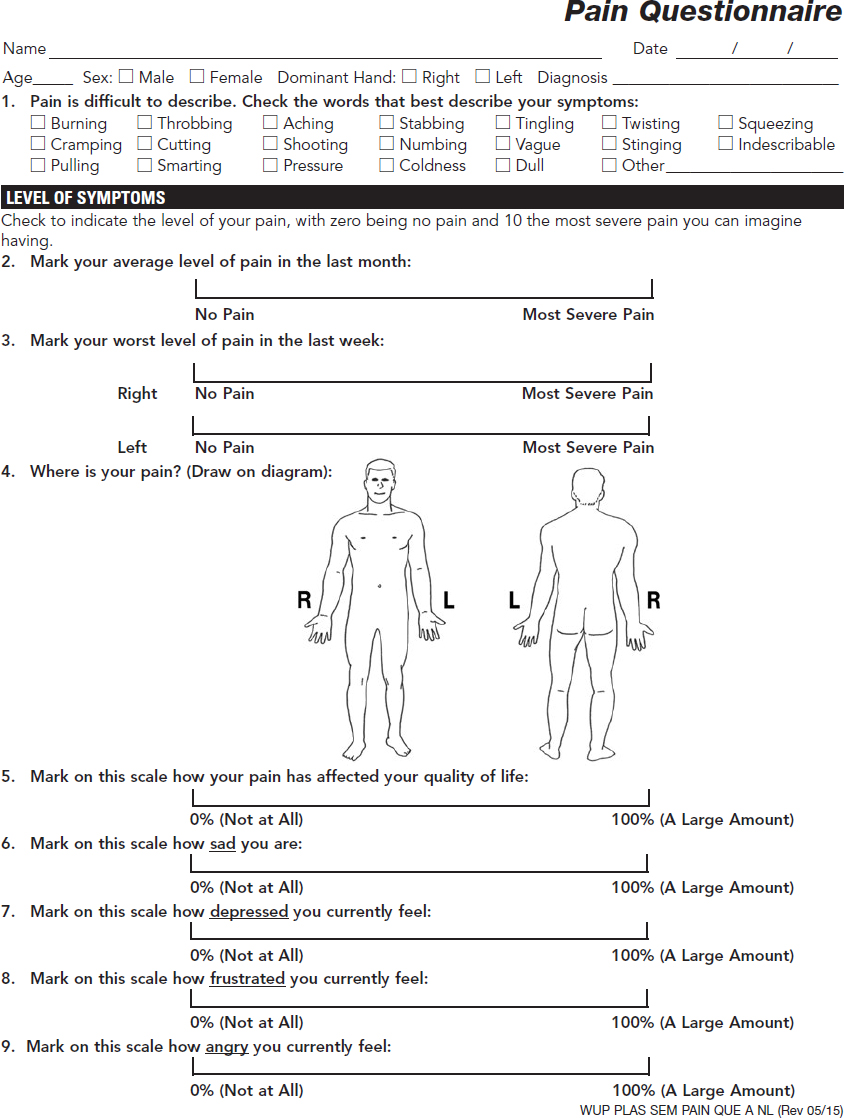
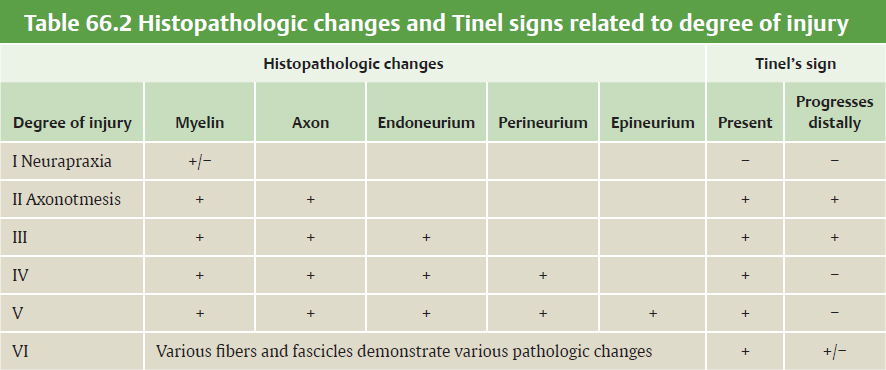
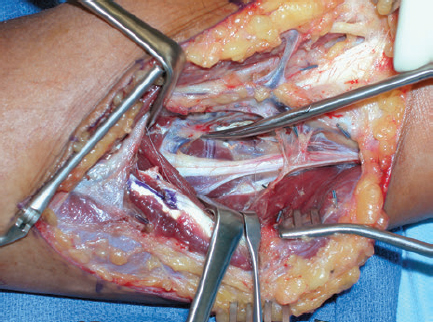
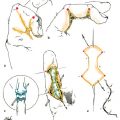
CHAPTER Many factors contribute to the final outcome in nerve surgery. These same considerations apply to reconstructive surgery in general, but the difference in nerve surgery is that success or failure will commonly not be evident for 6 to 12 months or longer because of the slow rate of nerve regeneration and target organ reinnervation and recovery. If restoration of motor function is not successful, there will not be a second chance to reinnervate target muscles because of degeneration of the neuromuscular junction and irreversible architectural changes in muscle structure. Even if there is a significant loss of sensation and function after a nerve injury, it is often the painful sequelae that have the largest impact on patient lifestyle in the long term and are the most difficult to manage. Therefore every effort is made in nerve reconstruction to optimize axonal regeneration and target end-organ reinnervation. Factors that influence functional outcomes after nerve surgery include the following: • Patient selection • Timing and choice of surgical procedure • Technical factors associated with the common techniques of nerve repair and grafting • Rehabilitation In posttraumatic reconstructive surgery, a patient with an anatomically consistent sensory or motor deficit and supporting electrodiagnostic studies is in general a good surgical candidate with realistic expectations. Management of these patients is often straightforward. When treating a patient with chronic pain of potentially neurologic origin from either a previous injury or surgical intervention or a possible compression neuropathy, determining appropriateness for surgical intervention can become more challenging. A patient who presents with symptoms who has a significant psychological barrier or unrealistic expectations is unlikely to have a successful outcome regardless of the surgeon’s skill and the technical success of the procedure. A patient with neurologic symptoms may have secondary gain from their symptoms, may be demonstrating attention-seeking behavior, or may have a psychiatric diagnosis. How the patient responds to pain can have a significant impact on functional recovery and rehabilitation, and therefore a large effect on the final outcome.1 The symptom of pain may have originated from a single incident or traumatic event but may have several sources that are often multiple and coexistent. Therefore it is critical that a patient with an essentially isolated neuroma-related pain be distinguished from a patient with a concomitant pain syndrome. A variety of tools exist to evaluate pain severity, including verbal scales, numeric scales, and the visual analog scale (VAS).2–4 Multidimensional patient-reported questionnaires are commonly used to evaluate pain qualities and severity.5 A pain diagram is helpful to visualize the anatomical location and extent of a patient’s pain, and an appropriate questionnaire will help determine the impact of the pain on a patient’s lifestyle, activities of daily living (ADL), and expectations of how and to what extent possible surgery may improve the symptoms and their lives6 (Fig. 66.1). A pain evaluation questionnaire can also be very helpful in sorting out which patients are most appropriate for surgical intervention. If the patient’s history and examination are not definitive or are unclear and there is a suspicion of possible malingering, numerous examination techniques can be used, including rapid alternating movements, rapid grip exchange, simultaneous tasks, and assessment of intratest variability.7,8 Fig. 66.1 Pain questionnaire that includes a visual analog scale. (Reproduced from Washington University School of Medicine, St. Louis, MO, with permission.) A comprehensive understanding of the patient’s pain, including qualitative nature, location, and severity can help to distinguish its potential sources. Pain from a neuroma is generally focal, brief and intermittent, sharp, and consistent with pressure or contact, whereas nonneuromatous pain is often described as diffuse and inconsistent or of varying quality and duration. The surgeon should proceed cautiously if the patient completes a body pain diagram that does not follow an anatomical distribution or pattern or uses more than four adjectives to describe the pain. In such cases, a referral for a formal and comprehensive psychological evaluation should be considered. The type of adjectives selected to describe pain may also indicate a significant nonneurologic component, and thus any psychological component must also be appropriately managed for a successful outcome. However, in our experience malingerers are exceedingly rare, and we always give all pain patients the benefit of an open, curious mind and a detailed history and physical examination. Electrodiagnostic studies are rarely helpful. A variety of patient factors can also affect the quality of nerve regeneration and outcomes. Age is a significant factor in recovery after nerve injury. Younger patients in general recover faster and more fully even after severely disabling injuries.9 The correlation of age has been demonstrated experimentally in different models of injury and repair. In addition to the shorter distances required for nerve regeneration and target end-organ reinnervation, children also have superior cortical plasticity, which facilitates their rehabilitation and is likely to play a significant role in their functional outcome.10,11 Smoking has also been correlated with delayed functional recovery in experimental models of nerve injury, and medical comorbidities including diabetes, hypothyroidism, and peripheral vascular disease can also have a negative impact12,13 (Video 66.1). Peripheral nerve surgery is rarely an emergent or urgent procedure, even after trauma. The only true emergencies mainly involve acute nerve compression of a posttraumatic or postoperative nature. Examples include acute carpal tunnel syndrome after carpal dislocation or severely displaced distal radial fracture, which may require operative reduction, or after their surgical management secondary to edema and/or hematoma. Otherwise, a sharp nerve transection is certainly time-sensitive and may require early operative intervention within a few days to permit a primary tension-free nerve repair before contraction of the nerve ends and edema precludes their direct coaptation. If there is no tension at the nerve repair site, better functional outcome can generally be expected if a nerve graft can be avoided, but the outcome also depends on the length of a nerve graft, as discussed further in the section on technical factors. If there is an injury to small but critical motor nerves such as branches of the facial nerve, acute or early intervention within 72 hours will permit identification of the distal nerve stumps by electrical stimulation with muscle contraction. Beyond that time, the muscles are no longer responsive and identification of the distal nerve stump may be much more difficult. A nerve that has lost continuity from a crush injury, torsion, avulsion, or a combination of these mechanisms on the other hand will benefit from a delayed approach to operative management. It is important in such cases that the zone of injury is determined, because traumatized and scarred nerve may not regenerate well or may be more likely to result in a painful neuroma. We recommend that operative intervention in such cases be delayed for at least 3 weeks, by which time the zone of injury on the nerve will be more clearly demarcated using a “bread loafing” technique, serial sectioning that can more readily determine the appearance and texture of the normal fascicular pattern of a healthy nerve. An injured, scarred segment of nerve will feel more stiff and harder than nerve outside of the zone of injury, and the appropriate amount of nerve that should be resected and reconstructed is more readily determined. Otherwise residual scar tissue will block axonal regeneration even if nerve continuity is restored, and the functional outcome will be suboptimal. In some cases, operative intervention may be required more acutely because of other associated injuries such as a vascular injury and/or a coverage issue and the surgeon is hesitant to reoperate at a later time because of extensive scarring or prohibitive risk to a vascular repair or reconstruction. In such circumstances, the surgeon will need to make an educated guess as to the zone of injury and should consider a wider nerve resection than might otherwise seem necessary, because the extent of intraneural scarring may not yet be fully evident. This will help to ensure that all injured nerve is resected and nerve graft reconstruction is performed to nerve ends outside of the zone of injury to optimize axonal regeneration. In these instances we assume the proximal and distal extent of injury from the site of transection is similar. We then have the anesthesiologist “lighten” the patient’s anesthetic and use a low setting on the nerve stimulator to stimulate the proximal nerve stump. As we move proximally we assess an increase in the patient’s heart rate as an indicator of healthy nerve and use this as part of the decision-making process to determine how far to resect proximally, then use the measurement distally.16 Surgical intervention that occurs inappropriately early can also be detrimental. An electromyogram (EMG) obtained too early after injury may not be useful, because it takes approximately 6 weeks for electrical evidence of nerve injury to become fully evident. At 12 weeks, if an EMG shows the presence of motor unit potentials, good functional recovery is anticipated even though clinical evidence of muscle recovery may not be evident for months. If reconstruction is performed in such cases, muscle reinnervation and recovery will be substantially delayed and the outcome will be less favorable than expected with spontaneous recovery. However, if the injured nerve has well-established probable distal compression points, surgical decompression should be considered and will facilitate distal regeneration. Examples include median or ulnar nerve injuries in the forearm or higher with release of the carpal tunnel or Guyon canal at the wrist. In many cases, patients are referred too late and nerve reconstruction is no longer helpful. This is more common when the distance required for nerve regeneration to reach the target muscles is long, such as proximal lower and upper extremity injuries, including brachial plexus injuries. Commonly these patients are followed by the referring physician with the expectation that spontaneous recovery will be forthcoming, Typically it takes time for the realization to occur that surgery may be required, and by the time the patient is referred to and seen by the specialist, it may be too late to reinnervate the target muscles. At approximately one year of denervation, there will be deterioration of the neuromuscular junction and motor end plates, and irreversible architectural changes of muscle tissue will begin. If nerve reconstruction including a distal nerve transfer is finally undertaken, it will often still be another 6 months or more before the regenerating axons reach and reinnervate the target muscle. If the referral is made 8 months or longer after the injury, it may already be too late to recover target muscles by surgical reconstruction. Therefore referrals are best made early so surgery can be undertaken as early as appropriate, such as 3 to 4 months after injury for brachial plexus reconstruction. A good understanding of the classification of nerve injury will help to determine the best timing for surgical intervention or whether it is even appropriate. Seddon’s and Sunderland’s classifications14,15 are applied to nerve lesions of all causes; they facilitate care by providing indications for nonoperative management versus surgical intervention and guide expectations for both spontaneous recovery and recovery from reconstruction (Tables 66.1 and 66.2). Seddon’s classification consists of three types of nerve injury: neurapraxia, axonotmesis, and neurotmesis.11 A neurapraxia is the least severe injury and represents a conduction block at a discrete segment of a nerve that may be related to an ischemic insult or demyelination. The nerve remains in continuity, and because there is no axonal injury, there is no distal Wallerian degeneration, so no axonal regeneration is required. On physical examination, Tinel’s sign will not be present and grossly the nerve will look normal in appearance, but the histologic appearance may demonstrate a focal area of demyelination. An axonotmesis is the next Seddon injury level and represents axonal injury in which the epineurium, endoneurium, and perineurium all remain intact. Because there is actual axonal disruption, Wallerian degeneration will occur in the distal nerve segment and axonal sprouting will be seen from the proximal nerve stump. In these favorable axonotmetic injuries, good to excellent recovery occurs because uninjured axons respond to the adjacent denervated neuromuscular end-plates with collateral sprouting. These are detected by motor units at 12 weeks of injury in distal muscles, far too distal to represent injured axonal regeneration from the level of the injury. These early collateral motor units will “babysit” the denervated muscles and provide a supportive environment as the slow regeneration occurs from the injured axons (nascent units). Spontaneous recovery is again expected, because the intact sheaths will guide the regenerating axons to their distal end-organ targets, but will occur slowly at the classic rate of 1 to 1.5 mm/day or approximately 1 inch per month. Because the connective tissue sheaths remain intact, there is minimal to no scar tissue to impede the regenerating axons, whose progression can be followed clinically by an advancing Tinel’s sign. There should be an orderly anatomical progression of functional recovery from the proximal to distal motor and sensory branches. The restoration of neurologic function is expected to be complete because the injury is purely axonal and the Schwann’s cell basal lamina that makes up the endoneurial sheath remains intact. A neurotmesis is the most severe class of injury in Seddon’s classification and involves complete transection of a nerve, including the axons and all of the connective tissue sheaths. Surgical intervention with repair or reconstruction is required for proper nerve regeneration and reinnervation of target end organs. The rate of axonal regeneration will follow the classic rate described previously, but scar tissue at the repair or reconstruction sites will provide variability to the extent of nerve regeneration, reinnervation, and functional recovery. Sunderland’s classification expands the more severe classes of injury in Seddon’s classification.14 His first-degree injury is equivalent to a neurapraxia, a second-degree injury is an axonotmesis, and a fifth-degree injury is the same as a neurotmesis. The third-degree injuries fall under the classification of axonotmesis but with more extensive injury. In a third-degree injury, the Schwann’s cell basal lamina that makes up the endoneurial sheath is violated, causing scarring and fibrosis that impedes axonal regeneration. Consequently, functional recovery is often incomplete because not all of the end organs are fully reinnervated, and the loss of the endoneural guide will also lead to some mismatching of regenerating axons to their proper distal sheath pathways, further downgrading functional restoration. The functional outcome will also depend on the level of the nerve injury. In a proximal nerve, motor and sensory axons are more mixed topographically within a fascicular group and there is more likely to be modality mismatching where motor axons regenerate down sensory pathways or vice versa and reinnervate inappropriate end organs. Further distally there is better fascicular grouping of motor and sensory axons,12 and mismatching is more likely to occur with axons of the same modality. Any deficits in end-organ recovery are therefore more likely to result from incomplete nerve regeneration related to scarring at the lesion and less from modality mismatching as well. In a fourth-degree injury, there is complete disruption of axons and all connective tissue sheaths like a neurotmesis, but the nerve physically remains in continuity by scar tissue. There is no nerve regeneration because it is completely blocked by scar tissue and therefore resection and nerve graft reconstruction are required. A seventh-degree injury has been termed by Mackinnon13 and is a mixed injury pattern representing the existence of more than one or even all of Sunderland’s injury degrees in a neuroma in continuity.16 The variability in the extent of injury exists not only among the different fascicles at a given level but also along the length of the nerve. The technical dilemma lies in appropriately resecting and reconstructing the fascicles with levels of injury that won’t regenerate (fourth degree and fifth degree) while leaving intact those that will (first through third degrees). The identification of the functional and sensory deficits is made clinically preoperatively and coordinated with intraoperative assessment after internal neurolysis and electrical stimulation to identify the functioning components to avoid in the resection and minimize downgrading of any remaining function. Thus the most appropriate timing for surgical management depends entirely on the extent of the nerve injury and the capacity for spontaneous recovery or not. If there is certain or a good possibility of spontaneous recovery (Sunderlund’s first through third degrees), then observation is continued until either the progression ceases or regeneration appears to be held up where there may be a Tinel’s sign at a known area of entrapment, such as the carpal or cubital tunnels, or the proximal fibular head. In such cases, it is then reasonable to proceed with decompression at the entrapment site in an elective manner. If the anatomical progression of recovery stops with persistent functional deficits, then some fourth and fifth degree injury is present and surgical intervention is indicated. With a known sixth-degree level of injury, surgical reconstruction may be planned but will not be scheduled until a period of waiting is recommended to provide time for the less severely injured nerve components to show evidence of recovery. Such evidence may be appreciated by clinical examination with visible or palpable muscle contraction, which may still be very early and subtle, or may not be clinically evident but demonstrated by the presence of motor unit potentials on an EMG study. Examples of such cases include brachial plexus injuries and gunshot wounds in proximity to major nerves. A standard management protocol will include a period of observation of approximately 3 and 4 months, respectively, which may vary somewhat at different institutions, followed by surgery to reconstruct the components that have not shown any evidence of recovery at that point. In these cases, recovery of the components with lesser degrees of injury will still be ongoing, and surgery may even be held off indefinitely as long as the critical surgically reconstructable functions are improving. Some functions such as hand function in brachial plexus injuries are not amenable to reconstructive nerve surgery because their distal level precludes the possibility of muscle reinnervation before the development of permanent structural changes of the motor end-plates and muscle architecture. Reconstruction is still possible with nerve transfers or functional muscle transfers but will be of lower priority than more proximal functions, such as elbow flexion and shoulder abduction, that are recoverable with nerve reconstruction. Electrodiagnostic studies are invaluable to the assessment of the degree of nerve injury and the recovery of muscle function. Proper interpretation requires a basic understanding of the terminology and its clinical correlation. Conduction velocity and distal latency reflects myelination with a demyelinating lesion, such as a neurapraxia causing a decrease in conduction velocity and therefore an increase in distal latency. Waveform amplitude is an indicator of the number of axons functioning, with a decrease representing greater loss of axons. After axonal injury, muscle distal to the level of the lesion may still stimulate for up to 7 to 10 days but thereafter will no longer be responsive. Muscle fibrillations on a needle EMG, however, may not be demonstrated for approximately 3 to 6 weeks after injury and will be present as long as there is some degree of denervation. In a muscle that has had a completely denervating injury and no reinnervation has occurred, fibrillations will generally disappear after approximately 1 year. Once this has occurred, that muscle is no longer recoverable by surgical means or otherwise. In a partially denervated muscle, however, fibrillations may still be seen for years after injury and may indicate the ability of the muscle to respond to further reinnervation. In partial denervation injuries, muscle reinnervation will first begin with collateral axonal sprouting from the intact viable axons to the denervated muscle fibers, causing the existing motor unit potentials to become polyphasic and of longer duration, but the amplitude may not change. These findings may not be evident for up to 12 weeks after injury, and consequently, EMG studies are not very helpful before that time period. Reinnervation from axonal regeneration rather than collateral sprouting of viable axons produces motor unit potentials that contrast those from collateral sprouting. These are known as nascent potentials and have short duration, low voltage, and a more immature form. They may appear up to 2 months before any clinical signs of muscle reinnervation or recovery. As reinnervation progresses to more muscle fibers, the potentials slowly increase in amplitude while the number of polyphasic units shows a corresponding decrease, and fibrillations will also slowly resolve during the process.17 In summary, an EMG obtained before 6 weeks after the injury will be able to distinguish a first-degree injury or neurapraxia from more severe injuries (second through fifth degree) by the absence of fibrillations in muscle distal to the nerve injury and any preexisting nerve injury. However, it will not be able to differentiate lesions for which spontaneous recovery is expected (second and third degree) from those that have no capacity to recover (fourth and fifth degree). A study obtained after 6 weeks from the injury may be able to also show motor unit potentials and nascent units, indicating that at least some recovery is forthcoming. For most closed injuries, including brachial plexus injuries, an observation period of approximately 3 months is advocated (4 months for gunshot wounds), and surgery is recommended if there is no evidence, electrical or otherwise, of reinnervation by that time. Electrodiagnostic studies may also be helpful in determining both whether a patient is an appropriate surgical candidate and, for compression neuropathies, which procedures are most likely to be successful. Although the clinical presentation and examination are the most important in evaluating a patient with a possible nerve entrapment syndrome, the surgeon should proceed cautiously if the electrodiagnostic studies are normal. Neurologic changes and pain in an extremity may also come from a more proximal source, such as spinal root compression associated with degenerative disease or trauma. In such cases, peripheral nerve decompression will not address the primary source of the symptoms and may not be helpful, and it subjects the patient to unnecessary surgery. Nerve compression may also occur at more than one level, such as median nerve compression at both the proximal forearm and the carpal tunnel. Both sites will need to be addressed to provide the patient with an optimal chance of a successful outcome. Wide exposure with good visualization is necessary for successful nerve surgery. For nerve decompression procedures, a small incision with limited exposure may not provide adequate visualization at the most proximal and distal ends of the dissection and may lead to inadequate decompression at either end where a fascial edge or a sharp change in the direction or angle of the nerve course may be present. When a patient is referred to us with persistent symptoms after an initial decompression procedure with a short scar, such as after ulnar nerve transposition, we usually initially suspect ongoing compression at either the proximal or distal end for the reasons previously mentioned. Although safe in experienced hands, we also do not advocate the endoscopic approach to carpal tunnel release. Visualization is certainly more limited, and although the incidence of complications has not been shown to be any higher than the open approach, they do include more severe complications such as complete transection of the median or ulnar nerve.18 With limited exposure and visualization, longitudinal structures such as tendons and nerves may appear similar, but a wider exposure will help to confirm their course and true identity. Technical factors also play a large role in affecting outcome, especially in the management of proximal mixed nerve injuries. The pros and cons of epineurial versus grouped fascicular repairs are generally understood, but the topographical anatomy of larger mixed nerves is still underappreciated. It is important that the two nerve ends are in their most anatomically accurate position and rotation to avoid a mismatch of sensory with motor fascicles and vice versa. Any degree of mismatch will downgrade the anticipated anatomical outcome, but a total mismatch may lead to absolutely no muscle reinnervation, whereas a partial mismatch may lead to abnormal target muscle reinnervation and complicate the rehabilitation. Therefore we feel that an epineurial repair is better than a grouped fascicular repair in most cases, because rarely is the proper topographical alignment of the nerve stumps perfectly clear without a doubt. Only in such circumstances should a grouped fascicular repair be used. Anatomical clues can be used to verify that the two nerve ends are in their most anatomically accurate position before a repair or reconstruction is performed. The position of vasa nervorum running longitudinally can be used to line up the two nerve ends correctly (Fig. 66.2). Also, the configuration of the transected nerve ends may provide clues as to how the ends match up the best. For example, an obliquely oriented nerve laceration will be more accurately lined up than a completely transverse nerve laceration, because the nerve stump configurations will tend to match up better as they are brought together. The fascicular grouping pattern at each of the nerve ends should also be noted and can provide further clues if the patterns can be recognized and matched to each other. In general, all of these techniques should be used together when coapting nerve ends to ensure that regenerating axons reach their appropriate end organs. There should be no tension on the nerve coaptation, and flexed positioning compensation should not be relied on to force a primary repair. For example, the elbow should not be flexed to allow a primary repair of a lacerated ulnar or median nerve. In such cases, the elbow should be extended and nerve grafts used such that the joint can be moved through its full range of motion while the nerve coaptations remain intact and loose. As in nerve repairs, it is critical that motor and sensory fascicles are properly aligned to minimize modality mismatch, which will downgrade the functional outcome. Thus when reconstructing a gap with nerve grafts, it is important that the nerve stumps are anatomically positioned and rotated, and that nerve grafts done in a cabled fashion are matched as closely as possible to the corresponding topographical regions of the proximal and distal nerve stumps. Cabled nerve grafts should therefore also be in linear alignment with each other without any twisting or torsion, which might contribute to fascicular mismatch (Fig. 66.3).
66
Peripheral Nerve Injuries and the Brachial Plexus
Avoiding Unfavorable Results and Complications in Nerve Surgery
Patient Selection


Timing
Classification of Nerve Injury
Seddon’s Classification
Neurapraxia
Axonotmesis
Neurotmesis
Sunderland’s Classification
Electrodiagnostic Studies
Surgical Considerations
Exposure
Nerve Repair
Nerve Grafts
Plastic Surgery Key
Fastest Plastic Surgery & Dermatology Insight Engine