Introduction
“Don’t cut what you don’t see!” This rule mandates large incisions and wide open surgical exposure to safely dissect aponeurotomic fibers and release restrictive scars or fascial structures.
“Borrow from Peter to pay Paul.” This fundamental principle of flap reconstruction in plastic surgery creates defects at the donor site, generates more scars, creates wound healing issues, and often leads to reconstructions that look like patchwork.
We have recently developed the percutaneous aponeurotomy and lipofilling (PALF) procedure as the regenerative alternative to the classic open scar releases and flap transfers ( Figs 7.1–7.3 ). Instead of “borrowing from Peter to pay Paul,” we developed an incisionless way to release contractures and induce Paul to regrow and regenerate the needed tissue himself. PALF is based upon the following fundamental principles and observations:
- 1.
Needle-pricks in the 1–2 mm range leave no scars (this is why intravenous lines usually leave no scars). Therefore, multiple nicks with a 1 mm needle applied in a staggered pattern can go through the skin without leaving a permanent scar.
- 2.
Needles only cut fibers under tension; this selective cutting ability spares nerves and other vital structures that are not tight when the contracture is forcefully extended. This then opens the possibility of safely releasing contractures percutaneously without open exposure.
- 3.
Staggered needle-pricks can generate a pattern of slits that mesh-expand in the direction of the tight traction in a fashion similar to a meshed skin graft expansion.
- 4.
The body can regenerate missing tissue to bridge gaps in the 1 mm range, while gaps in the 2 mm range are good recipients for fat grafts with regenerative abilities.
- 5.
A fine mesh pattern of 1–2 mm gaps is capable of expanding the meshed area by 20–30% (meshing ratio 1 : 1.2). Meshing the tissues also creates a scaffold matrix for fat grafts with regenerative potential.

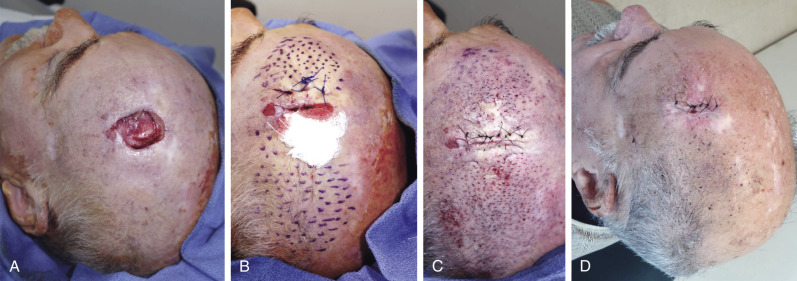
2-D Application of PALF
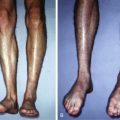
For patients with contractures or localized surface tissue coverage defects, PALF is an alternative to the classic flap. It generates the tissue needed for the reconstruction in situ, without the traditional incision-needing, scar-creating, and complication-prone flaps.
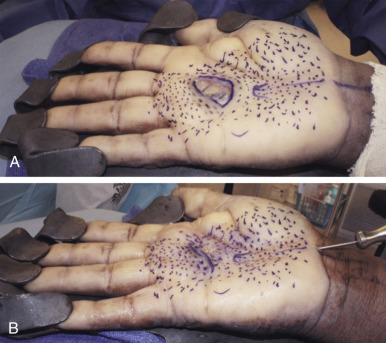
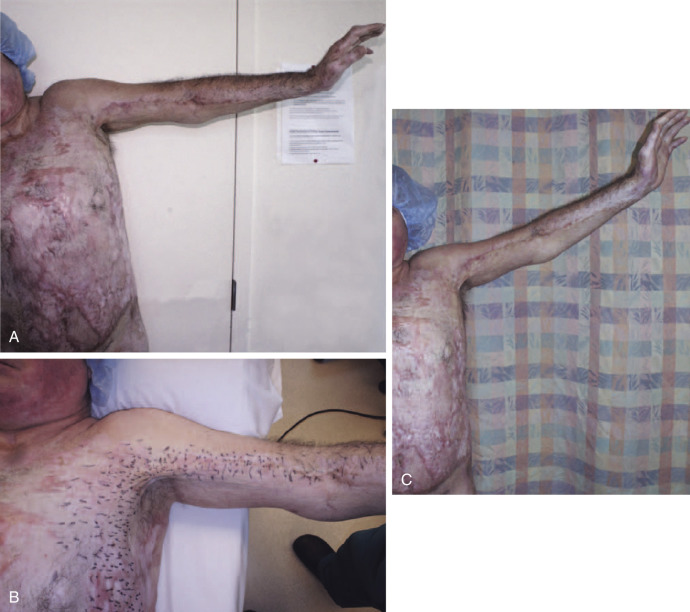
The crucial component of PALF is strong tension maintained in the direction of tissue deficiency throughout the percutaneous release process. For 2-D hand contractures defects ( Fig. 7.4 ), we use a strong lead hand retractor that maintains strong extension force. For other parts of the body ( Fig. 7.5 ), we use hooks or temporary retention sutures to maintain tension. We then adhere to the following rules as we perform the PALF:
- 1.
Since PALF can safely generate only 20–30% tissue gain over the meshed area, we plan the extent of aponeurotomy mesh release based upon the defect size. For example, to gain 4 cm, plan to release 8–10 cm on each side of the defect.
- 2.
In order to maintain the tension throughout the release process, mesh in an orderly fashion from proximal to distal along the tensed areas. And whenever possible, restore tension as it dissipates with the meshing.
- 3.
To preserve perfusion and the underlying vascular network, do not incise and do not undermine.
- 4.
Release tissues evenly along the entire length of the area placed under tension. It is important to avoid relaxing incisions or localized areas of over-release. These lead to cavities where fat grafts will pool and fail to revascularize for lack of intimate graft-to-recipient interface with a stromal/vascular scaffold. Cavity is the enemy of fat grafting and should be avoided at all cost.
- 5.
As it mesh-expands under tension, percutaneous aponeurotomy with the Rigotome® should create a fine honeycomb pattern of tiny, <2 mm cavities. The Rigotome device holds multiple small needles in place, allowing multiple percutaneous nicks to be performed at once.
- 6.
Graft about 0.1 mL of dilute lipoaspirate per 1 cm of reinjection. A 10 cm-long microribbon will contain 1 mL of lipoaspirate. The fat is injected with a blunt cannula in a diffuse, spray-like fashion through 3–4 needle puncture entry sites.
- 7.
Whenever possible, splint the PALF area in extension, or in such a way that maintains open the release without applying pressure on the PALF area. The splint keeps the expanded area in the open position and maintains tension on the expanded area.
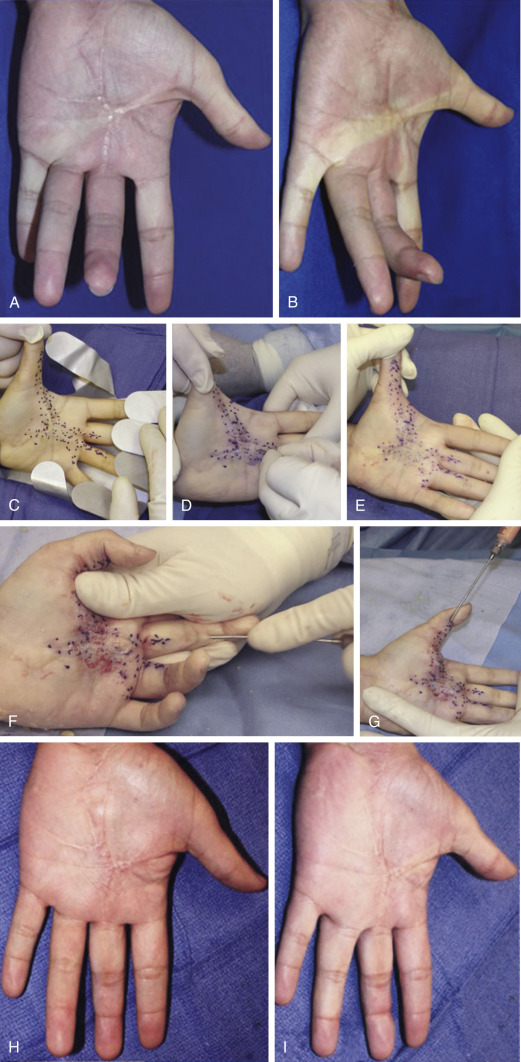
3-D Application of PALF
Cicatrix to Matrix
PALF is also applicable to more complex 3-D contour defects. Whether it is a concave deficiency that needs to be filled or a convex tissue mound that has to be re-created, PALF is an incisionless regenerative alternative to traditional flap transfers. The major application is breast reconstruction, especially the scarred breasts from radiation or from previous failed reconstruction attempts.
Whenever possible, we prefer to stretch-expand in 3-D, the tissue defect prior to the PALF. External expansion of the recipient site creates more space for grafting, creates more area to mesh, and prepares the skin envelope to accept the additional volume without tension. Pre-expansion also increases capillary density to preserve a functional capillary network, despite more extensive mesh release.
Pre-grafting expansion is achieved in the breast with the Brava® external expander device. Brava is a vacuum-based bra that applies isotropic mechanical distraction forces to the breast surface. It was developed to expand breasts preoperatively and induce angiogenesis and adipogenesis. The resultant increased volume and mechanical compliance of the breasts allows the surgeon to spray many more micrografts of fat without coalescence and without significantly increasing interstitial fluid pressure. The added vasculature increases the total blood flow to the tissue and decreases the average distance that oxygen must travel to reach the center of a graft. Brava cycles between 60 mmHg for 2 min and no pressure for 1 min. It is used for 10–12 h/day for 2–3 weeks preoperatively and without interruption for the last 24–48 h, preoperatively. We currently recognize that the minimum therapeutic dose is 200 h of cycling vacuum distraction of the breast over a 15-day period. It is the intensity of use that matters. Accumulating 200 h over a longer time period is much less effective.
After adequate external expansion, we meticulously inject dilute lipoaspirate in a spray-like fashion through multiple puncture wound entry sites. The graft is often injected with the “Jackhammer grafting” technique that helps release contractures. Unlike grafting a healthy recipient site with smooth passes that release the fat while the cannula is retracted, the “Jackhammer” cannula forcefully pokes multiple tunnels into the cicatrix to loosen it, and injects it with dilute lipoaspirate, which induces tumescence and places the restrictive fibrous bands under tension. Remaining fibrous bands are then released with the “Rigottomy” needle release. Similar to the 2-D meshing of a skin graft, we use fine, sharp needles to create many nicks in the contracted tissue to induce a 3-D release that expands the scar to increase the volume.
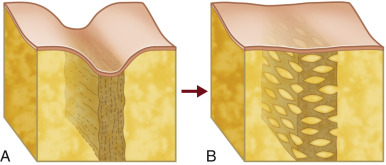
The 3-D Rigottomy of the PALF transforms a restrictive cicatrix into a larger recipient matrix with many microcavities that can support the fat grafts through diffusion until neovascularization occurs. When the microcavities in a brick-like solid scar get filled with micrografts of fat, the scar becomes less brick and more like the fat around it. Repeating the procedure can eliminate the scar and turn it into fat, as the surrounding soft tissue. The process of serial Rigottomy and fat grafting effectively transforms the cicatrix into the recipient matrix scaffold for fat grafts ( Fig. 7.6 ).
Keys to Fat Grafting
Fat grafting is a key component of PALF, but it also has its own benefits for several indications. Patients would much rather undergo minimally invasive procedures with needles, cannulas, and syringes than undergo invasive flap surgeries or be implanted with prosthetics. For decades, patients and surgeons have wanted to transfer excess fat for reconstruction. But until recently, the only reliable method was the invasive, morbidity, and complication-prone flap transfers. As simple as autologous fat transfer sounds, it presents unique challenges, which we have only recently discovered how to overcome. These challenges stem from the fact that fat grafts are avascular and simple bolus injections will form necrotic cysts in most conditions. The four major challenges are listed and discussed below:
- 1.
Surface area-to-volume ratio to ensure vascular inosculation
- 2.
Interstitial fluid pressure build-up that limits recipient capacity
- 3.
Recipient site vascularity
- 4.
Ability to differentiate necrotic cysts from cancer
Surface Area-to-Volume Ratio.
Fat grafts receive nutrition entirely by diffusion for the first three postoperative days, until revascularization occurs. The rate at which oxygen diffuses into the graft is directly proportional to the surface area over which oxygen can diffuse. The rate at which the graft metabolizes oxygen is directly proportional to the volume of the graft. As the radius of a cylindrical fat injection increases, its surface area-to-volume ratio decreases. Therefore, in the central region of a large fat injection, the tissue is metabolizing oxygen more rapidly than it is receiving oxygen by diffusion, leading to necrosis. According to our calculations and our clinical experiences, any injected fat lobule with a radius >1.6 mm is likely to have a region of central necrosis. In a given volume recipient site, there is a limit to how many small grafts can be injected before these small injections begin to coalesce into larger pools.
Therefore, successful large-volume fat grafting to the breast requires craftsmanship and the ability to “spray-deliver a mist” in the 3-D recipient site, as small individual droplets that do not coalesce. Larger blobs delivered as caulk boluses will not survive as well. Mega-volume fat grafts require microfat droplets.
Interstitial Fluid Pressure.
In 3-D grafting, the recipient site enlarges and commensurates with the added graft volume, but there is a limit to how much the recipient can enlarge to accommodate that volume increase. This has to do with the mechanical compliance of the recipient; a factor that is not constant. Healthy, unscarred recipient subcutaneous tissue is most compliant and will expand to accept a 20% volume increase before reaching its compliance limit and becoming very stiff. Beyond that limit, adding even a small drop of fat will increase the interstitial tissue pressure (IFP) to a level that compresses the capillaries, reduces blood perfusion, and the rate of oxygen delivery to the grafts, resulting in more necrosis.
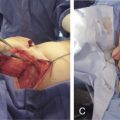
For successful mega-volume fat grafting, we should never graft beyond the physical capacity of the recipient to accept the added amount of tissue. We monitor this by measuring IFP in a fashion similar to compartment pressure measurements ( Fig. 7.7 ).