Dermatitis herpetiformis (DH) is an autoimmune blistering skin disease in which antigen presentation in the gastrointestinal mucosa results in cutaneous IgA deposition and distinct, neutrophil-driven cutaneous lesions. Our findings suggest that the qualitatively different immune response to gluten in the intestinal mucosa of patients with DH results in minimal clinical symptoms, allowing the continued ingestion of gluten and the eventual development of DH. Our model may provide a new way to understand the pathogenesis of other skin diseases associated with gastrointestinal inflammation such as pyoderma gangrenosum or erythema nodosum, or explain association of seronegative inflammatory arthritis with inflammatory bowel disease.
Louis Duhring proposed using the term dermatitis herpetiformis (DH) in 1884 to describe a cutaneous disease that was characterized by violent pruritus. Almost 1 century later, Cormane reported another key feature of this disease: the deposition of immunoglobulins at the dermoepidermal junction. Shortly afterwards, it was described that 85% to 90% of patients with DH had granular and the rest linear, IgA deposits. The third key feature was first described in 1966: these patients had an associated gastrointestinal disease, more specifically, a gluten-sensitive enteropathy (GSE). Not only was it shown that this disease was GSE, but that dietary control was sufficient in abrogating the cutaneous symptoms of DH. The association of GSE with DH was further correlated with the pattern of cutaneous IgA deposits by Lawley and colleagues, who found that patients with clinical DH and an associated GSE had granular IgA deposits only in the skin. This finding defined the significance of DH as a model for an autoimmune disease that linked gastrointestinal mucosal immunity with cutaneous disease. Since that time, much has been done to describe the clinical and immunopathologic features of DH. Although the exact mechanism by which cutaneous lesions appear is still unclear, much has been uncovered in the pathophysiology of the disease.
Clinical presentation
The classic cutaneous manifestations of DH, true to Duhring’s original description, are markedly pruritic, symmetrically distributed papulovesicles that usually affect the extensor surfaces: scalp, especially the posterior hairline, elbows, knees, back, and buttocks. Certainly variations exist, and frankly bullous, pustular, or eczematous lesions can sometimes be found. Patients can have only multiple erosions or small ulcerations, with or without crust, as a result of the severe pruritus. Rarely, atypical morphologies and locations, such as purpuric macules on the palms or tips of fingers, can be found as presenting features, especially in children. The mucous membranes are only rarely, if ever, involved, but have certainly been reported as an initial manifestation of the disease. The patients typically report a tingling or burning sensation 12 to 24 hours before the appearance of clinically evident lesions, which persists until the vesicle is broken and a crust is formed. DH typically presents in the second or third decade of life, with some investigators reporting a mean age of onset in the 40s, but it can certainly appear at any age. The condition is chronic, but waxes and wanes with no clear triggering factors. This disease tends to be chronic, although around 12% of these patients can go into spontaneous remission. Those who were 39 years or older at the age of onset and those who had already had longer duration of disease were more likely to experience remission. It is more prevalent among Anglo-Saxons and Scandinavians, with the estimated frequency between 10 and 39 per 100,000. This frequency seems to be similar among Whites in the United States. It is less common among other ethnic groups, such as African Americans and Asians, presumably because of differences in the frequency of HLA antigens associated with DH. Furthermore, clinical variations may exist among different ethnicities. An increased incidence of fibrillar pattern of IgA deposits in the skin of patients with a clinical presentation consistent with DH has been reported in a Japanese cohort. In addition, these Japanese patients seem to have a decreased frequency of GSE. It is not clear if this situation is because of a true difference in the pathogenesis of the disease, decreased exposure to gluten in the Japanese patients, or a less aggressive diagnostic approach to the potential gut disease. Although DH in the past has not been considered to be a familial disease, recent studies have indicated an indicated prevalence of both DH and isolated GSE in families. The incidence of familial occurrence (ie, a first-degree relative with DH) has been reported at 2.3% to 4.4%. In a series from Finland, as many as 6.1% of patients with DH have first-degree relatives with celiac disease.
Histology and immunopathology
Histology of skin in DH is characterized by a subepidermal blister with a predominantly neutrophilic infiltrate in the dermal papillary tips, although a mixed or even predominantly lymphocytic dermal infiltrate may also be found. Direct immunofluorescence reveals granular deposition of IgA at the dermoepidermal junction in both involved and uninvolved skin as well as the oral mucosa. However, IgA deposition is not evenly distributed in the skin of patients with DH. Deposition is most intense in noninflamed perilesional skin, and is decreased in skin that has never been involved. Erythematous or lesional skin in patients with DH may not show IgA deposition, perhaps as a result of neutrophil destruction of the IgA. Therefore, the ideal site for a skin biopsy in DH is uninvolved, perilesional skin. Patients with isolated GSE without DH do not have cutaneous IgA deposition, showing that this phenomenon is related to DH itself, and not the underlying GSE.
Complement deposition, specifically C3, can be found along with IgA in the skin of patients with DH. There is evidence that IgA activates complement in DH via the alternative pathway.
A fibrillar pattern of IgA deposition in a subset of patients with DH has also been described. Although these patients typically have other clinical features consistent with DH, it has been suggested that these patients may have a higher incidence of atypical features, such as a urticarial or psoriasiform clinical presentation, the absence of GSE, or HLA-B8/DR3/DQ2 haplotype. Some of these patients have been reported to lack circulating antibodies against tissue transglutaminase and endomysium. Whether or not these patients represent a true distinct subset or variant of DH or another disease entity altogether remains to be seen.
Histology and immunopathology
Histology of skin in DH is characterized by a subepidermal blister with a predominantly neutrophilic infiltrate in the dermal papillary tips, although a mixed or even predominantly lymphocytic dermal infiltrate may also be found. Direct immunofluorescence reveals granular deposition of IgA at the dermoepidermal junction in both involved and uninvolved skin as well as the oral mucosa. However, IgA deposition is not evenly distributed in the skin of patients with DH. Deposition is most intense in noninflamed perilesional skin, and is decreased in skin that has never been involved. Erythematous or lesional skin in patients with DH may not show IgA deposition, perhaps as a result of neutrophil destruction of the IgA. Therefore, the ideal site for a skin biopsy in DH is uninvolved, perilesional skin. Patients with isolated GSE without DH do not have cutaneous IgA deposition, showing that this phenomenon is related to DH itself, and not the underlying GSE.
Complement deposition, specifically C3, can be found along with IgA in the skin of patients with DH. There is evidence that IgA activates complement in DH via the alternative pathway.
A fibrillar pattern of IgA deposition in a subset of patients with DH has also been described. Although these patients typically have other clinical features consistent with DH, it has been suggested that these patients may have a higher incidence of atypical features, such as a urticarial or psoriasiform clinical presentation, the absence of GSE, or HLA-B8/DR3/DQ2 haplotype. Some of these patients have been reported to lack circulating antibodies against tissue transglutaminase and endomysium. Whether or not these patients represent a true distinct subset or variant of DH or another disease entity altogether remains to be seen.
Associated conditions
Patients with DH have an associated GSE. In contrast to patients with GSE alone, most patients with DH have little or no gastrointestinal symptoms, in spite of the almost invariable presence of demonstrable pathologic changes in the gastrointestinal tract when there is active cutaneous disease. Even when cutaneous disease is controlled with dapsone or sulfapyridine, the mucosal changes are persistent. Twenty percent to 30% of patients with DH have mild steatorrhea, and even fewer complain of bloating, diarrhea, and malabsorption. The gross and histologic changes of the small bowel in DH are identical, although less severe, to that found in isolated GSE or celiac disease. There is often patchy involvement, with changes confined to the small bowel, typically the jejenum. There is flattening of the intestinal villi, elongation of intestinal crypts, and flattening of the intestinal epithelial cells. A mononuclear infiltrate of plasma cells and lymphocytes is found in the lamina propria and intraepithelially. These changes persist even when cutaneous symptoms are controlled with dapsone and sulfapyridine but normalize with dietary therapy or strict avoidance of gluten.
Aside from GSE, several diseases are associated with DH. It has been reported that up to 41% of patients with DH have hypochlorhydria or achlorhydria, and most of these patients have gastric atrophy. As many as 38% of patients with DH have been found to have thyroid microsomal antibodies, and patients with DH have a higher incidence of thyroid abnormalities, including hypothyroidism, thyroid nodules, and thyroid cancer. Other autoimmune diseases have been reported to be associated with DH as well: systemic lupus erythematosus, dermatomyositis, myasthenia gravis, Sjögren syndrome, and rheumatoid arthritis. IgA nephropathy has also been described in patients with DH, and mesangial changes and IgA deposition can be found even in the absence of overt renal symptoms.
Patients with DH have been reported to have an increased risk of gastric lymphoma 2.3 times higher than the normal population, with the incidence as high as 6.4%. However, others have reported a lower rate. Hervonen and colleagues reported that only 1% of a series of 1104 patients with DH developed gastrointestinal lymphoma 2 to 31 years after the onset of DH and these patients were less likely to have adhered to adhere to a gluten-free diet. More recent reports have found no increased incidence of malignancy or mortality among patients with DH and matched controls.
Immunogenetics
DH has striking HLA associations, specifically with class I antigens HLA-B8 and HLA-A1 and the class II antigens HLA-DR3 and HLA-DQw2. Initial studies showed that 58% of patients with DH had HLA-B8, as opposed to 20% to 30% of normal controls. This same genetic marker was found to be present in 88% of patients with GSE as opposed to 22% of controls. Furthermore, patients with GSE also have an increased frequency of HLA-A1, HLA-DR3 and HLA-DQw2, genetically linking DH and GSE. An even stronger association was found with HLA-DR3 and HLA-DQw2 because 95% and 100%, respectively, of patients with DH with confirmed granular IgA papillary dermal deposition expressed these markers.
Immunopathogenesis
Although it is clear that gastrointestinal inflammation is integral in the pathophysiology of DH, the exact mechanism of antibody production as well as the cascade by which gastrointestinal inflammation translates into cutaneous disease is not known. Our laboratory has been interested in how the inflammatory response to gluten in patients with DH results in an itchy blistering skin disease with rare gastrointestinal symptoms, whereas patients with isolated GSE have no skin disease with significant gastrointestinal symptoms.
Patients with DH can control the appearance of cutaneous lesions by restricting themselves to a gluten-free diet. Moreover, eliminating gluten intake leads to a decrease and eventual clearance of IgA deposits in the skin. Although the presence of IgA in the skin is well documented, we set out to determine if the IgA in the skin could be of gut origin. Evaluation of the IgA subclass present in DH skin revealed that the cutaneous IgA deposits were IgA1 and that joining chain could not be detected. Because IgA1 is the predominant subclass of serum IgA whereas IgA2 is the predominant subclass in mucosal secretions this finding suggested that the IgA was not of mucosal origin. Patients with circulating IgA antibodies against reticulin and endomysium also had these antibodies in their intestinal secretions. These studies showed that the presence of IgA autoantibodies in the serum is concordant with the presence of IgA antibodies in intestinal secretions. Furthermore, whereas normal gut secretions contained more IgA2, gut secretions from patients with DH were predominantly IgA1, showing that the IgA immune response in the gut was predominantly IgA1. Characterization of the serum and gastrointestinal IgA antibodies directed against dietary proteins revealed that both serum and intestinal antibodies had similar isoelectric spectrotypes and IgA subclass composition and that was distinct from the pattern seen with total serum IgA. These studies showed that IgA1 antibodies in the serum can be of gut origin, linking closely the IgA immune response in the gut to the IgA deposits in DH skin. Although attempts to elute IgA from DH skin have not been successful, recent studies by Sardy and colleagues and Donaldson and colleagues have shown that the IgA in DH skin seems to bind to epidermal transglutaminase (eTG) in the dermis.
These studies, together with the clinical studies, have shown that the mucosal immune response to gluten can lead to IgA antibodies of mucosal origin, which can persist in circulation, and that a specific group of these antibodies, IgA antitransglutamase 3 (anti-TG3) (eTG), deposit in the skin. The mechanism of that binding remains unknown. However, these studies do not explain how the IgA deposits may relate to the development of skin lesions, nor do they explain the clinically different presentations of DH and isolated GSE.
eTG or TG3 has been identified as the target autoantigen in DH. It is strongly expressed in the upper epidermis but may also be found in renal basement membrane. Patients with DH have detectable eTG in the papillary dermis, overlapping with the same sites that have IgA deposition. It was not found in sites where IgA was not found. It has been hypothesized that eTG is released from keratinocytes and drops to the basement membrane in response to trauma, and is subsequently bound by circulating IgA. Another hypothesis is that preformed circulating complexes of IgA and eTG deposit in the papillary dermis. Evidence of the presence of these circulating complexes is shown by the precipitation of these complexes in vessel walls of patients with DH. It has also been hypothesized that these dermal deposits are somehow the end product of a reaction against TG3 in kidneys. IgA nephropathy has been associated with DH, and mesangial deposits were detected in as many as 45% of patients with DH without any overt clinical signs of nephropathy. Although IgA deposition in the kidney was not related to epidermal deposits or degree of gut involvement, this was associated with a high frequency of circulating IgA against gliadin and reticulin. In 1 series of patients with DH, Jaskowski and colleagues reported that serum eTG IgA compared with eTG IgA and IgG was more sensitive in detecting GSE; however, sensitivity of IgA anti-eTG was only 71%. Furthermore, dietary intake seems to correlate with eTG IgA; that is, avoidance of gluten resulted in the gradual decrease of antibody levels.
Despite the difference in the clinical presentations of patients with DH and those with isolated GSE, patients with DH and isolated GSE share many common features. In addition to sensitivity to gluten, patients with DH and those with isolated GSE share the same strong HLA association: HLA-A1, B8, DR3, DQ2. Patients with DH and isolated GSE also both have circulating IgA anti-tissue and eTG antibodies and have the same typical histologic features of villous atrophy of the small intestine. In contrast most patients with DH do not complain of the bloating, abdominal cramps, and diarrhea that typically affect patients with isolated GSE. Only around 20% of patients with DH experience steatorrhea, and even fewer (<10%) have bloating, diarrhea, and malabsorption. In addition, patients with isolated GSE do not have cutaneous IgA deposits or skin blisters. These differences in clinical features may be because of a difference in the intestinal cytokine response to dietary gluten. Real-time polymerase chain reaction analysis of small bowel biopsies of patients with DH showed a greater expression of interleukin 4 (IL-4) mRNA and less expression of interferon γ (IFN-γ) compared with patients with isolated GSE. In both DH and GSE, small bowel biopsy often shows a mononuclear infiltrate of plasma cells and lymphocytes if found in the lamina propria and intraepithelially. T-cell lines derived from small bowel mucosa of patients with DH were predominantly CD4+/IL-4+, and less frequently CD4+/IFN-γ+. In contrast, T-cell lines from isolated patients with GSE were predominantly CD8+ with a similar frequency of IL-4+ and IFN-γ+ cells. In vitro culture of these T-cell lines with phorbol myristate acetate and ionomycin revealed that the T-cell lines produced IL-4 whereas isolated GSE T-cell lines produced both IL-4 and IFN-γ. These studies suggest that the increased expression of IL-4 mRNA in the gut of patients with DH when compared with patients with isolated GSE may modulate the inflammatory response and play a role in the lack of symptoms in patients with DH. T-cell receptor V β expression also seems to be more restricted among patients with DH who continue to ingest gluten when compared with patients with isolated GSE on a gluten-containing diet. Therefore, differences in the symptoms exhibited by patients with DH and isolated GSE may be the result of a difference in the local immune response and cytokine production.
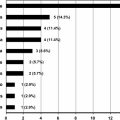
Although these studies showed that patients with DH have an ongoing chronic mucosal immune response that results in mucosal IgA in the serum with deposition of IgA in the skin, the factors that lead to the development of the skin lesions were not clearly understood. Neutrophils play a central role in pathogenesis of the skin lesions seen in patients with DH. Histopathologic evaluation of involved skin shows neutrophil deposits in the papillary dermis. The development of skin lesions is also exquisitely responsive to dapsone, a drug known to inhibit neutrophil function, without changing the small bowel mucosal immune response. Patients with DH have a predominantly asymptomatic, chronic mucosal immune response in the gut, and control of the mucosal immune response through dietary gluten restriction can control the skin disease; it was certainly possible that the ongoing gut small bowel mucosa inflammation could partially prime both the skin and the circulating neutrophil with resultant skin blister formation. Evaluation of serum cytokine levels revealed that patients with DH with inactive skin disease secondary to use of dapsone but on a gluten-containing diet had increased levels of cytokines, including IL-8 and tumor necrosis factor α (TNF-α). We then compared the serum IL-8 and IgA anti-tissue transglutaminase levels in patients with DH before beginning a gluten-free diet and after an average of 24.5 months (range 0.5–40 months) of a gluten-free diet. Patients who were placed on a gluten-free diet had a significant decrease in their serum IL-8 and IgA anti-tissue transglutaminase levels ( Fig. 1 ). In contrast, those who were kept on a gluten-containing diet had persistently increased serum IL-8 and IgA anti-tissue transglutaminase levels even if their cutaneous disease was well controlled on dapsone or sulfapyridine. IL-8 mRNA message in small bowel biopsies of those on a gluten-containing diet was increased, whereas those patients on a gluten-free diet had IL-8 mRNA levels similar to normal controls. Thus, studies showed that the persistent mucosal immune response to gluten in the gut of patients with DH resulted in a chronic increase of proinflammatory cytokines in the circulation.