History of Surgery in Hidradenitis Suppurativa
Hidradenitis suppurativa (HS) was first described by two surgeons in the 1800s, Velpeau and Verneuil. There was much debate surrounding whether HS was primarily a venereal disease, tumor, or abscess. In the 1890s, Barthelemy suggested HS was a folliculitis that led to increased dermatologic interest in HS as a skin disease. For the next 60 years, treatment was largely targeted at medical therapy until the 1950s when Greely proposed the role of excision of chronic disease in the small percentage of cases that evolved beyond the acute setting. The following 60 years focused largely on techniques for excision and reconstruction. In the surgical world, HS was considered primarily as a sweat gland disease that was treated with excision. The most widely popularized operative procedure, the Pollock procedure, focused on primary axillary excision with immediate closure. This chapter will discuss the role of surgery and surgical goals of care in HS, operative techniques for diseased tissue, reconstruction, patient optimization preoperatively, and the role of interdisciplinary care.
Setting Goals for Surgery
Historically, surgery was indicated largely for chronic disease. As our knowledge of HS pathogenesis has evolved, the role for surgical management of HS has also evolved, as has the timing of surgery for acute and chronic lesions. Surgery may be indicated for mild to severe disease and in both the acute and chronic settings, if goals of surgery are established. Surgical management should be viewed as an integral aspect of an HS treatment plan.
Surgery can take on many forms, including incision and drainage, deroofing, and excision with reconstruction when indicated. In the past, surgical management of HS has been met with skepticism stemming from reported high recurrence and postoperative infection rates. However, most of the studies focused on “recurrence” as the primary outcome measure in a disease where surgery should be considered an adjunctive therapy combined with medical therapy rather than a cure. Moving forward, it is important that we define the goal of any surgical intervention in HS. This should be addressed during the initial surgical evaluation. In some patients with focal and limited disease, surgery may provide more definitive therapy for HS lesions. However, in patients with more diffuse disease, surgery may be indicated for symptomatic control of areas that repeatedly flare with the goal of decreasing inflammation and disease burden. In these situations, recurrences would be expected to occur, but hopefully with less frequency, drainage, inflammation, and pain. Surgery in HS has been suggested to work synergistically with medical treatments for HS by decreasing inflammation to allow medications to penetrate more effectively. Surgeons should have preoperative discussions with patients and dermatologists to define the goals of care in each HS patient with the goal of improved outcomes and patient satisfaction.
Operative Techniques
Deroofing
Deroofing is a technique for the management of HS tracts in which the anterior wall of an HS tunnel is removed using a probe to identify the entirety of the tract. This can be used for an abscess, cyst, or tract. Once the probe is inserted, the tunnel is opened, and the anterior wall of the tunnel is removed using scissors, a scalpel, or an electrocautery device. This allows for an open cavity without the risk of the skin edges closing together before the wound has been able to heal by secondary intention. The lining of the tunnel, often the gelatinous material, is removed using a curette and another form of debridement to the level of the epithelial lining of the cavity. The wound is covered with a moist dressing, changed daily, until the wound has completely healed by secondary intention. Techniques can be utilized to fully identify the extent of the tract, including filling the cavity of the tract with methylene blue dye before the tract is open. Once the tract is open and the anterior wall is removed, the tunnel will appear blue and serve as a guide for debridement to ensure the extent of the tunnel has been deroofed and may minimize recurrences from partially unroofed tracts. The benefit of the deroofing technique is that it is a tissue-sparing technique that can be used in the office on small HS lesions or in the operating room on larger or more complex tunnels. Patients do not require inpatient hospitalization, although skilled nursing may be helpful for wound care in more complex cases. Deroofing can be performed in lesions on all anatomic areas of the body.
Most patients heal rapidly with good cosmetic outcomes from deroofing techniques if the tract has been fully identified and deroofed. Deroofed wounds may experience a delay in wound healing if they develop a significant biofilm and bioburden but respond well to mechanical in-office debridement and silver-impregnated dressings. In general, scarring is minimal, and wounds are less noticeable compared to other surgical techniques, such as …. Patients may experience flares within the scar if the extent of the tract has not been fully identified at the time of deroofing or if disease develops adjacent to the deroofed area. There also may be microscopic tunnels forming adjacent to the tunnels that are not identifiable at the time of procedure, which then develop into more prominent and symptomatic disease. These focal areas of recurrence respond well to additional focal deroofing procedures or excision with healing by secondary intention, but for these reasons, it is important that patients undergo concomitant medical therapy to suppress ongoing disease and minimize disease progression. Other complications include bleeding, which responds well to pressure or chemical cauterization. In a study of 88 deroofed lesions, recurrence rates were reported as 17% after a 4.6-month median follow-up. Patient satisfaction was reported as 90%, including patients who experienced a recurrence.
Deroofing has been used to approach more complex wounds in a staged fashion, which would ordinarily require extensive wide excision with reconstruction. Deroofing procedures are performed at approximately 8-week intervals in large tunnels to allow wounds to heal completely prior to additional procedures and are often performed with excisions at the lateral margins of the wound. This approach allows for a series of outpatient procedures, less pain, small open wounds at a given time, and allows patients to return to their daily activities and responsibilities much sooner. Per our experiences, patients who undergo deroofing have less overall cosmetic disfigurement than with wide excision.
Deroofing has been combined with subsequent excision to remove fibrotic tissue that remains once the wound has healed from the initial procedure in patients with moderate to severe hidradenitis. This modified two-staged approach has been postulated to decrease recurrence rates based on the assumption that recurrence originates from epithelialized tracts that may become hidden in the fibrous tissue. It was also hypothesized that recurrence following simple deroofing could arise from baseline progression of disease in adjacent tissue.
Excision
Excision has been considered the gold standard for surgical management of HS. Past recurrence rates, which were considered to be relatively high, were thought to result from incomplete resection of diseased tissue. We now understand that HS is a systemic disease; therefore, excision is not curative. However, excision is an excellent technique to remove isolated areas of active disease. The procedure may be performed under local or tumescent anesthesia in the clinic setting, while larger areas may require sedation or general anesthesia. Active disease should be excised down to the level of subcutaneous tissue to remove all visible tunnels and disease tissue. This can be performed using a variety of devices, including scalpels, CO 2 lasers, or electrocautery devices. There is not enough data to suggest that one tool is superior to the other in terms of excision and recurrence rates, but the use of electrocautery or CO 2 laser may help to facilitate hemostasis during the procedure. It is important to counsel patients that disease recurrence may occur in the postoperative wound adjacent to the scar. This can either be explained by incomplete resection of diseased tissue or the natural progression of HS in surrounding tissue.
Staged Techniques
Patients with diffuse disease may benefit from multiple staged operative procedures. Staged deroofing techniques have been shown to decrease overall inflammation. As each wound heals, there appears to be a decrease in surrounding inflammation, which facilitates control of surrounding HS disease and allows for a targeted, skin-preserving approach requiring less reconstruction. Subsequent deroofings heal with ease, given the decreased inflammation. Patients do not require inpatient hospitalization. Staged excisions can also be utilized and are encouraged in large anatomic areas to allow for manageable wound care, to minimize inpatient hospitalization, and to avoid fluid losses that can occur with loss of skin as a protective barrier. Staged excisions are also useful in perinanal HS to minimize anal retraction and stenosis.
Options for Wound Closure and Reconstruction in Hidradenitis Suppurativa
A variety of different closure techniques have been described in the literature. Aside from primary closure, other common reconstruction techniques include secondary intention healing, vacuum-assisted closure, skin grafts, and various types of flaps. The choice for a specific technique depends on multiple variables such as size and location of the defect, existing perioperative structures within the surgical center (e.g., physiotherapy, wound care), patient compliance, and the surgeon’s preference and training. General guidelines are missing and individual approaches are necessary.
Primary Closure
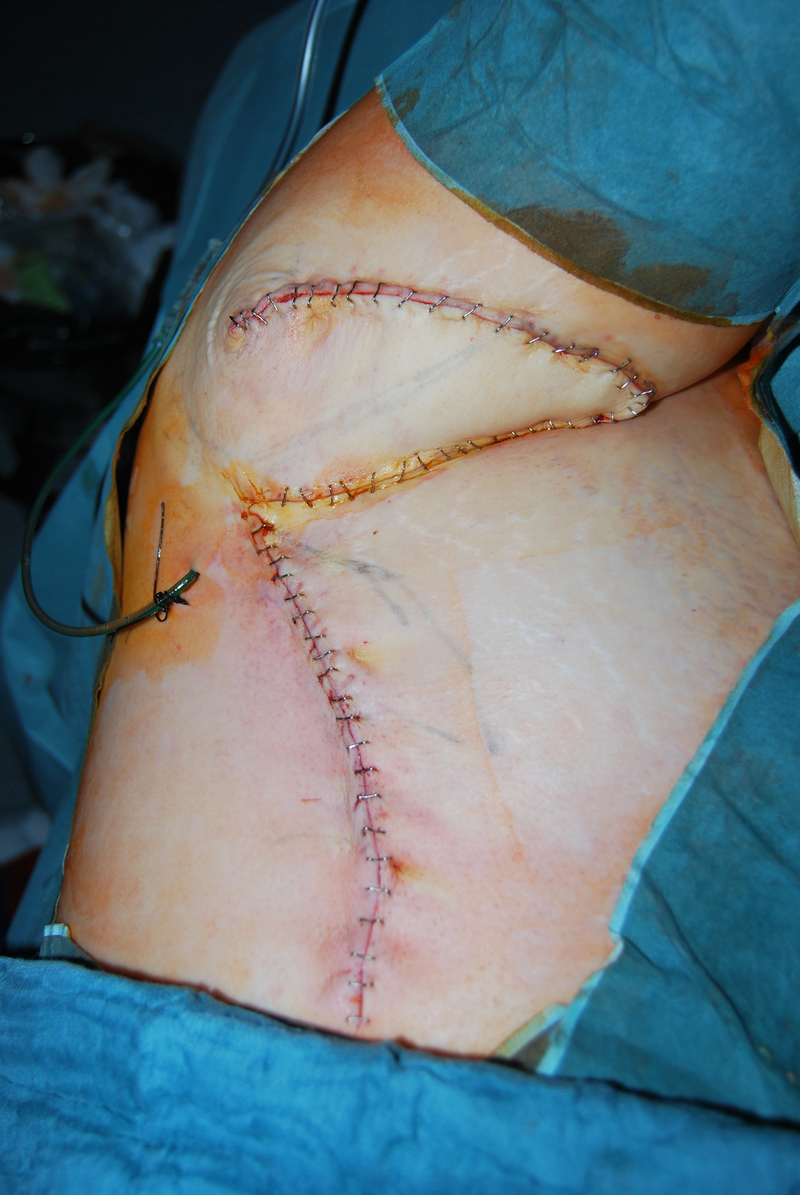
Primary closure can be useful after small excisions, especially in Hurley stage II patients. Some authors have described shorter inpatient stay, lower morbidity, and fewer postoperative complaints about the mentioned technique. The most important limitation for primary closure is the size of the defect ( Fig. 23.1 ). Moreover, immobilization can be necessary, and the risk for wound dehiscence and infection must be discussed. In some situations, partial primary closure with the usage of a drain may be useful to speed up wound healing and reduce the risk for contraction.
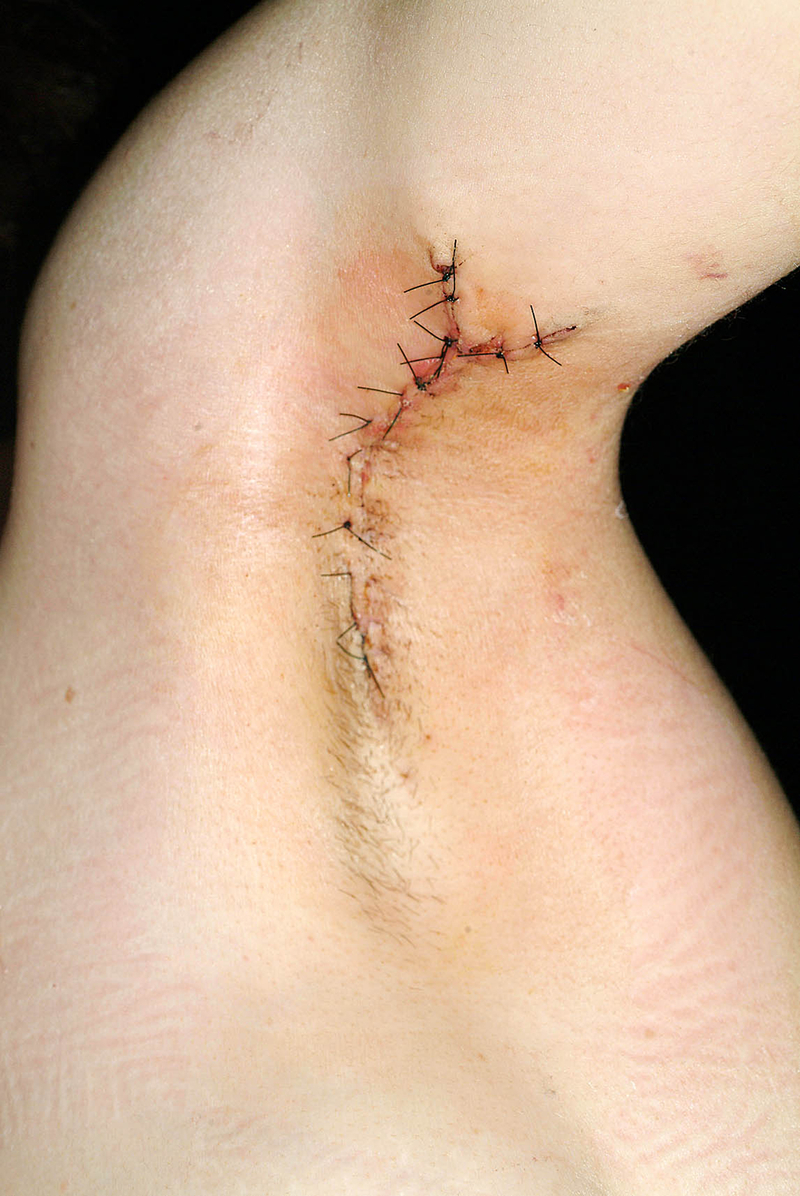
Secondary Intention Healing
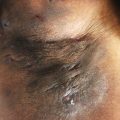
Some surgeons prefer secondary intention healing in the inguinal, anogenital, and the lower abdominal areas ( Fig. 23.2 ). Advantages are immediate mobilization, acceptable scar formation, early reintegration into daily life and occupation, and a low risk for complications and recurrence of HS. These wounds can require complex wound care to be performed by a patient family member or a skilled nurse. Limitations include prolonged wound healing, painful changing of wound dressings, and a risk for contraction, especially in the axillary region. During secondary intention healing, adequate wound care and physiotherapeutic support to prevent prolonged wound healing, contraction, and the risk of restriction of movement are mandatory. Additionally, in the author’s view, the experience-dependent preoperative assessment of risk for contraction is crucial and depends on localization and size of the defect.

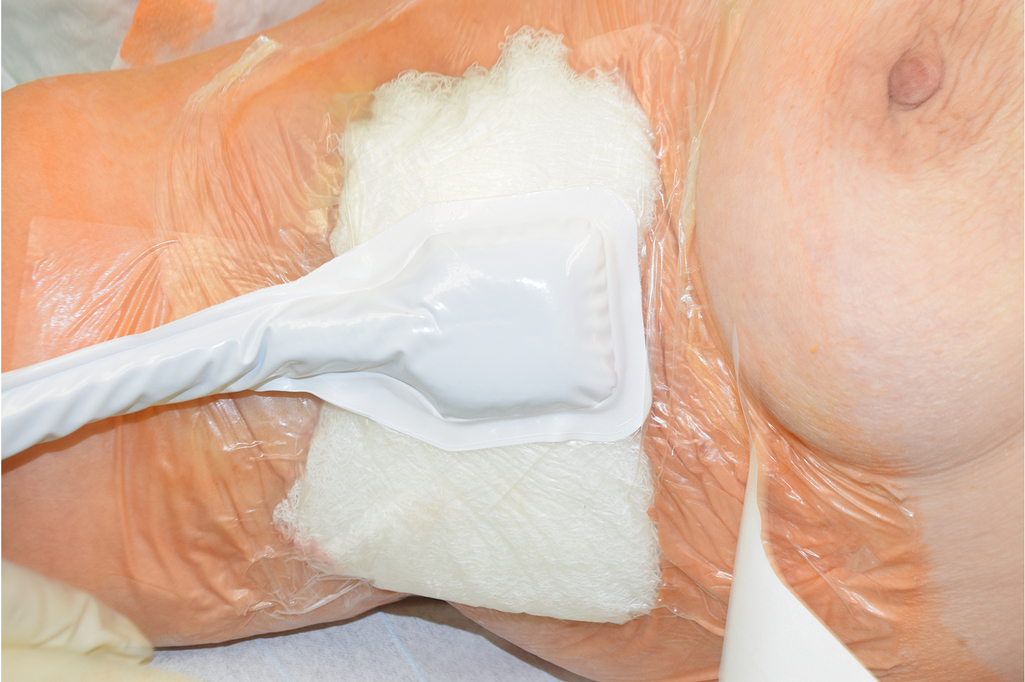
Negative Pressure Wound Therapy
Negative pressure wound therapy (NPWT) has been reported to be associated with faster wound granulation and re-epithelialization, reduction of bacterial load, and increased tissue oxygenation. A small case series illustrated that the combination of NPWT and skin grafting was associated with a higher rate of graft-take and faster wound healing ( Fig. 23.3 ). Additional advantages include added fixation of skin grafts, a drainage of wound secretion, and a reduced number of wound dressings. However, existing data does not allow a final evaluation of NPWT in HS surgery.
Skin Grafting
Most commonly, split-thickness skin grafts (STSGs) are used for skin grafting in HS surgery. STSGs are able to be meshed if larger defects must be covered. Skin grafting can be performed primarily after excision in a one-stage procedure or secondary after wound conditioning ( Fig. 23.4 ). Skin grafting after granulation of the wound to the level of adjacent skin seems to be associated with more favorable aesthetic outcomes (GrDIN ZOTERO_IT). Additionally, the wound contraction leads to smaller graft sizes in the secondary two-stage procedure.
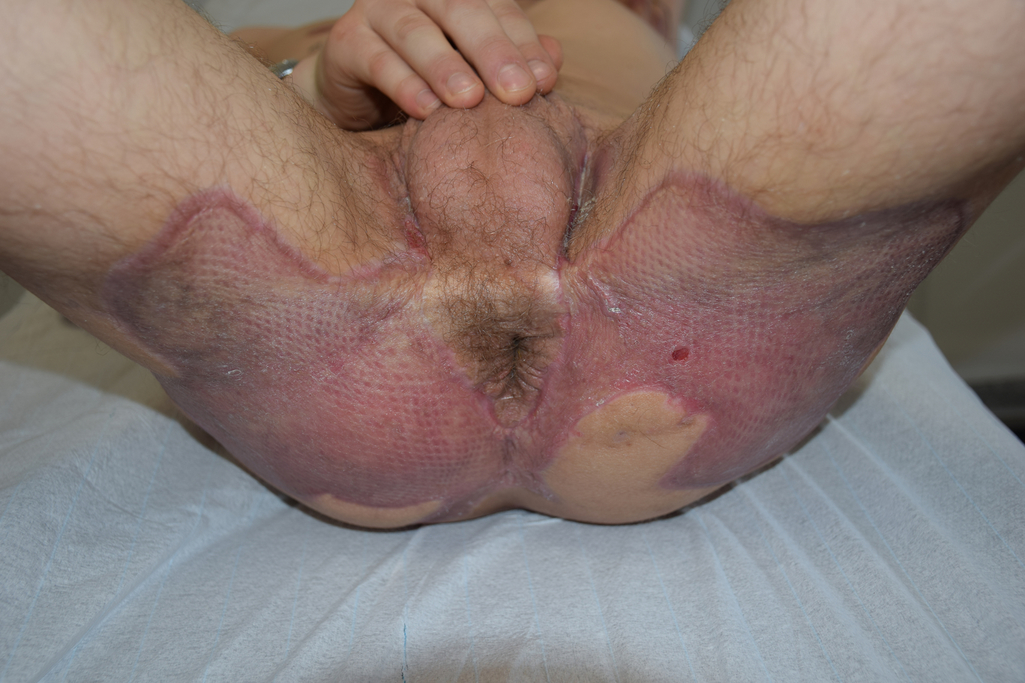
Generally, skin grafts can be applied to defects on all anatomic areas. The most frequently grafted sites are the perianal, gluteal, genital, and inguinal regions. Some authors prefer skin grafting due to low recurrence rates and a favorable cosmetic outcome. a
a References .
Reported limitations include possible risk of contractures and the morbidity of donor sites, especially pain. Successful graft acceptance depends on postoperative wound management with special regard to moisture control and bacterial colonization. After stable graft acceptance, a physiotherapeutic support and scar massage can influence the functional and aesthetic outcome.Based on prior experience, scrotal and penile defects should be covered primarily in a one-stage procedure to prevent deviation. All other locations (e.g., reconstruction of the gluteal area) can be covered after wound conditioning in a second step operation.
A modification of the classic STSG, the “reused skin graft technique,” has been described by Kuo and colleagues. The STSG is taken with an electrical dermatome from the area to be excised; it is then meshed and finally “reused” for coverage of the defect. With this procedure, a classical donor site is avoided. Some reports describe the use of dermal regeneration templates for axillary defects in combination with NPWT and subsequent STSG (ADDIN Ztary: two-stage surgery for hidradenitis suppurativa: staged artificial dermis and skin grafting—PubMed—NCBI,o-stage).
Flaps
A variety of different flaps have been reported for defect reconstruction in HS. In most cases, local flaps are used for coverage of axillary defects, with the intention to reduce the risk of contractions in the shoulder joint—thereby avoiding long-lasting wound healing—or to cover exposed vessels ( Fig. 23.5 ). The advantages of local flaps include shorter healing time, fewer painful dressing changes, shorter inpatient stay and immobilization, reduced risk for contractures, and in some cases, a more favorable aesthetic outcome. Limitations include more difficult application for very large defects, complex surgical techniques, and prolonged surgery times. Janse and colleagues propose the use of flaps for large defects in the axillary and genital region; however, they do not see them as the first option in other locations.