div class=”ChapterContextInformation”>
6. Principles of Urethral Stricture Reconstruction and Its Instrumentation
Keywords
Urethral reconstructionUrethroplastyWound healing6.1 Introduction
Urethral reconstructive surgery is a true hybrid of urologic and plastic surgery. The reconstructive urologist therefore must possess intimate knowledge of the anatomy and function of the genitourinary tract and must be familiar with the principles of tissue transfer, graft take and wound healing. An expert hand is also required, as urethral and transferred tissues are very delicate.
In this chapter, surgical principles of reconstructive urology, as well as the principles of wound healing and tissue transfer will be discussed in detail. An overview of the surgical instruments that should be a part of the reconstructive urologist’s armamentarium will also be performed.
6.2 Surgical Principles in Reconstructive Surgery
As a general surgical rule, valid for not only reconstructive urology but for all surgical specialties, the reconstructive urologist must first do no harm. As previously mentioned, urethral tissues are very delicate and improper handling may lead to further damage and compromised tissue quality. Attention to detail is imperative throughout the entire period of time that the patient is entrusted to your care. This is valid for the entire pre-operative evaluation and the post-operative course, and not only while your patient is in the operating room.
Nomenclature is important when discussing urethral pathology. A urethral stricture refers to urethral luminal narrowing with associated spongiofibrosis, which occurs in the anterior urethra (distal to the external sphincter). Urethral stenosis (or contracture) is an obliterative, fibrotic process that occurs in the posterior urethra or bladder neck, usually secondary to pelvic fracture urethral distraction (PFUD) injuries or surgery such as radical prostatectomy [1].
6.2.1 Pre-operative Evaluation
A proper pre-operative evaluation is key, as good knowledge of the case will help avoid unexpected surprises in the operating room. While taking the patient’s history, knowledge of the mechanism of urethral disease (e.g., straddle injury) may help understand the underlying disease process. Identifying number and type of past treatments will also help plan subsequent procedures, especially if tissue transfer techniques have already been used.
Pre-operative testing, such and endoscopy and imaging, are also essential components of the pre-operative evaluation. Office flexible cystourethroscopy is quick and easy to perform and allows the urologist to determine the severity of the urethral luminal obstruction, as this may also impact treatment decision. Also, cystoscopy may also rule out functional urethral strictures, which means that if the stricture permits the 17-Fr flexible cystoscope to pass, the obstruction caused by the stricture is likely not significant may not have to be treated. Endoscopy also allows the surgeon to rule out a malignant urethral obstruction, and if the lesion looks suspicious, biopsy is recommended before any reconstructive treatments are commenced.
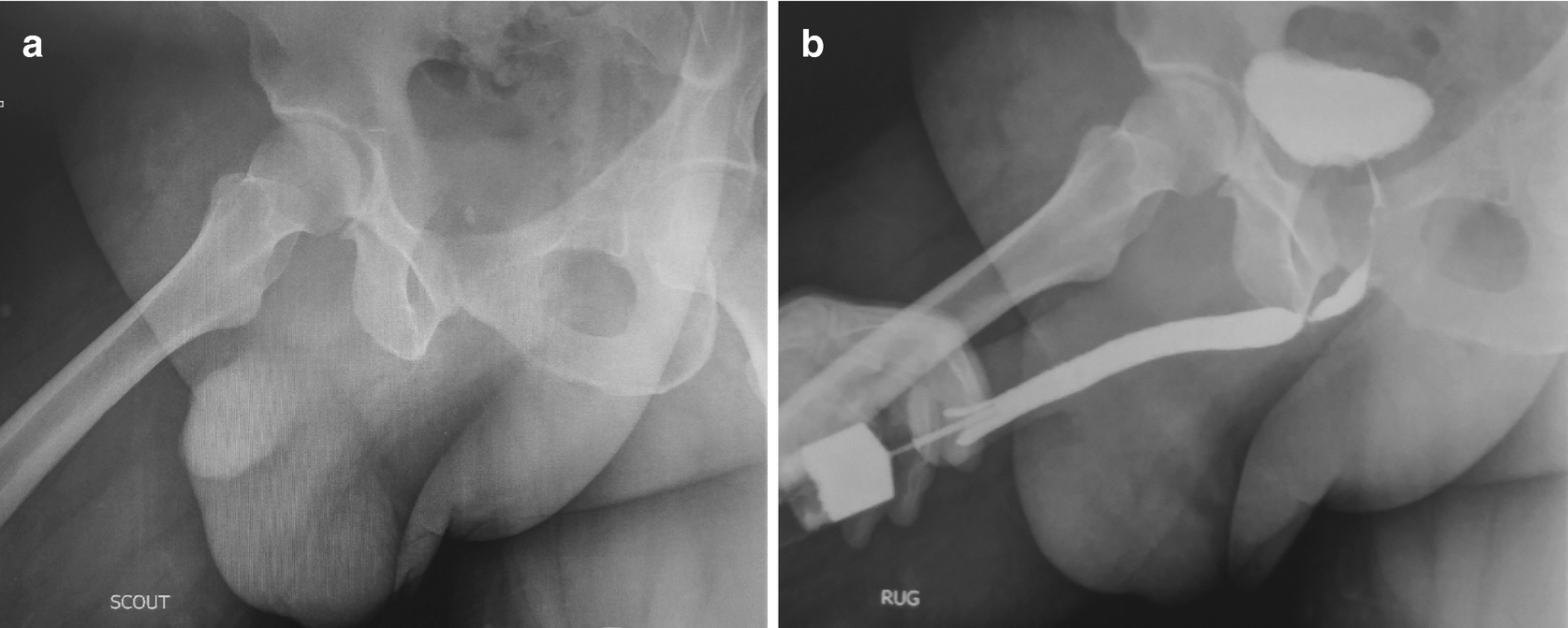
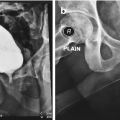
(a) Scout film and (b) retrograde urethrogram showing a bulbar urethral stricture
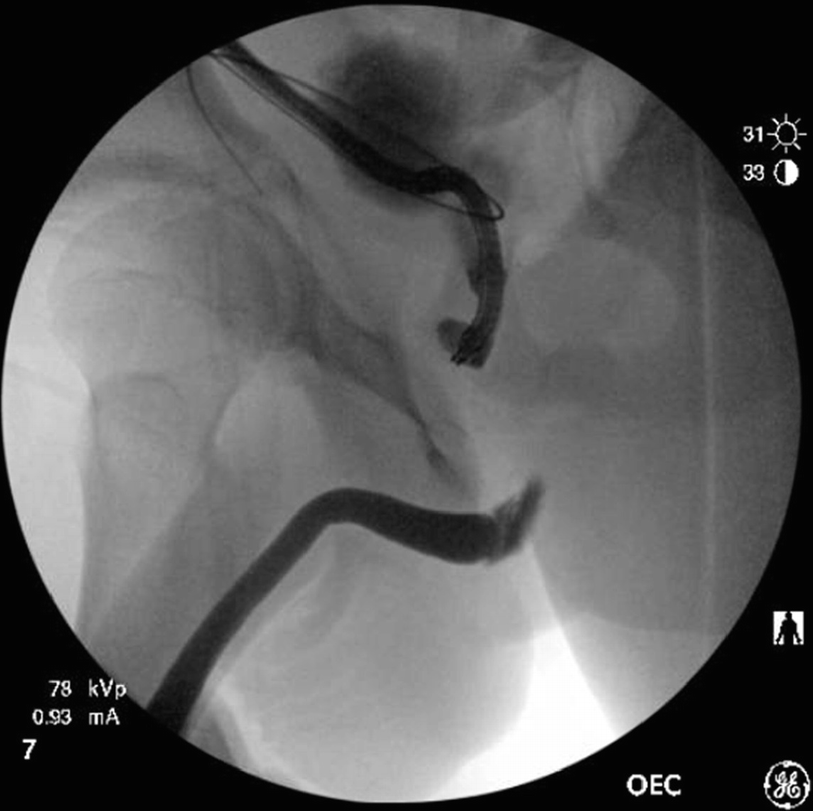
A combined antegrade flexible cystoscopy with a retrograde urethrogram performed under anesthesia reveals the length of the urethral defect in a pelvic fracture urethral distraction (PFUD) injury patient awaiting posterior urethroplasty
All patients must have a pre-operative urine culture performed. If positive, appropriate treatment must be commenced at least 48 h prior to surgery, chosen according to the antibiogram.
6.2.1.1 Urethral Rest
For anterior urethral strictures, the urethra should not be instrumented for 6–12 weeks prior to the urethroplasty. This allows the scar to mature and declare itself appropriately. In cases of very tight strictures causing significant lower urinary tract symptoms (LUTS) or urinary retention, or a severely dilated urethra proximally to the obstruction, placement of a suprapubic tube (SPT) may be indicated. In these cases, we recommend repeat imaging after the period of urethral rest as the surgical plan may differ once the stricture has fully declared itself [3, 4].
For PFUD, a 3-month period of urethral rest is traditionally recommended before proceeding with posterior urethroplasty, although recent data has shown that a 3–6-week resting period has similar outcomes [5]. More importantly, the reconstructive urologist must ensure that the patient’s other injuries have been appropriately addressed, and that the patient is physically able to be placed in the lithotomy position before proceeding to surgery.
6.2.2 Intra-Operative Principles
6.2.2.1 Patient Preparation and Positioning
After proper patient identification and induction of anesthesia, patient preparation for surgery begins. Intravenous antibiotic prophylaxis covering skin flora and urinary pathogens should be given and adjusted according to the pre-operative urine culture and past resistances if indicated. Proper shaving of the perineum, genitalia and the lower abdomen (in case supra-pubic access is required) should be performed using a straight razor, taking care not to injure the skin [6]. Patient is then placed into lithotomy position and the skin is prepped preferably using an alcohol-based solution, as this is more effective than providing in eliminating skin flora [7]. The pre-operative images should be reviewed and available for consultation in the operating theatre at all times.
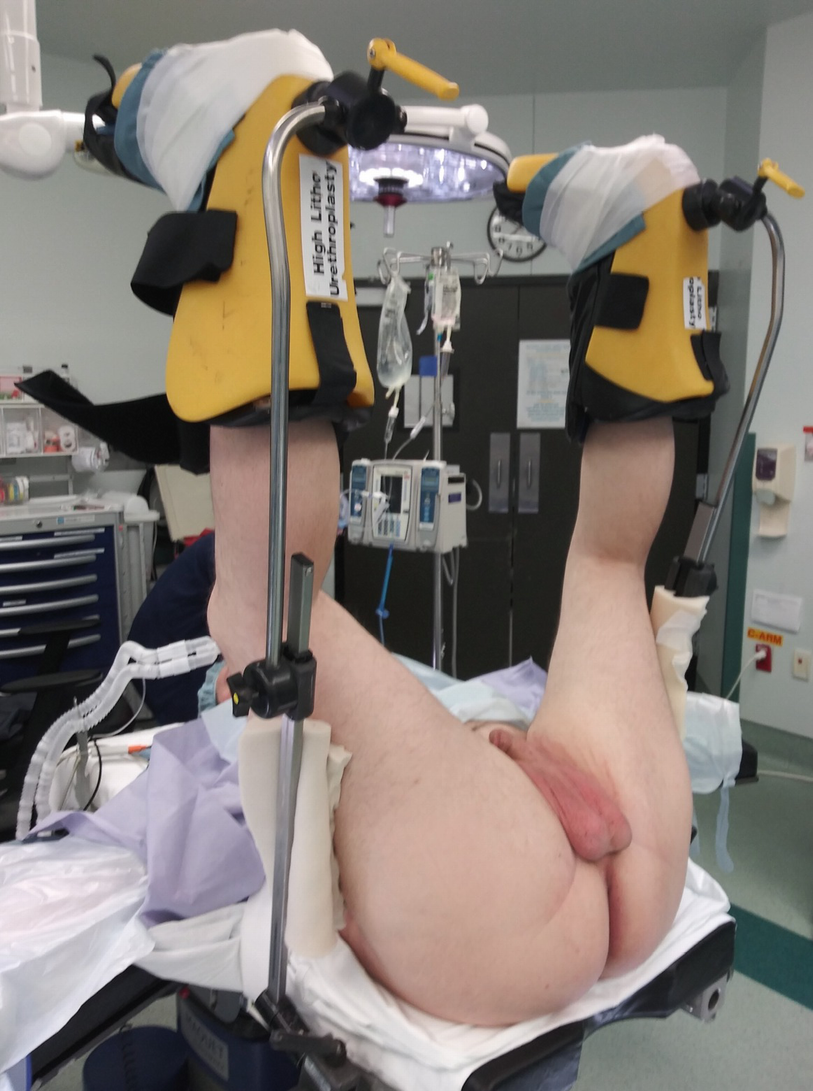
Exaggerated lithotomy position
6.2.2.2 Surgical Equipment
Surgical loupes allow for magnification of the surgical field and proper identification of the very small anatomical structures encountered in reconstructive urology. In the authors’ opinion, 2.5 power glasses are sufficient. A headlight is also a useful tool for proper visualisation.
A detailed review of surgical instruments for urethral reconstruction is presented in the final portion of this chapter.
6.2.2.3 Intraoperative Endoscopy
Endoscopy is a very important tool for the reconstructive urologist. Flexible or pediatric cystoscopy should be liberally used throughout the case. Evaluation of the urethra proximal to the stricture should always be performed to rule out presence of squamous metaplasia – and, if encountered, the urethral spatulation should be extended until healthy urothelium is reached, to prevent stricture recurrence. The bladder should also be fully inspected to rule out malignancy and bladder stones.
Endoscopic placement of a guidewire may be useful in cases with a very tight urethral stricture, to ensure the urethral lumen is opened along the proper plane.
6.2.2.4 Incision
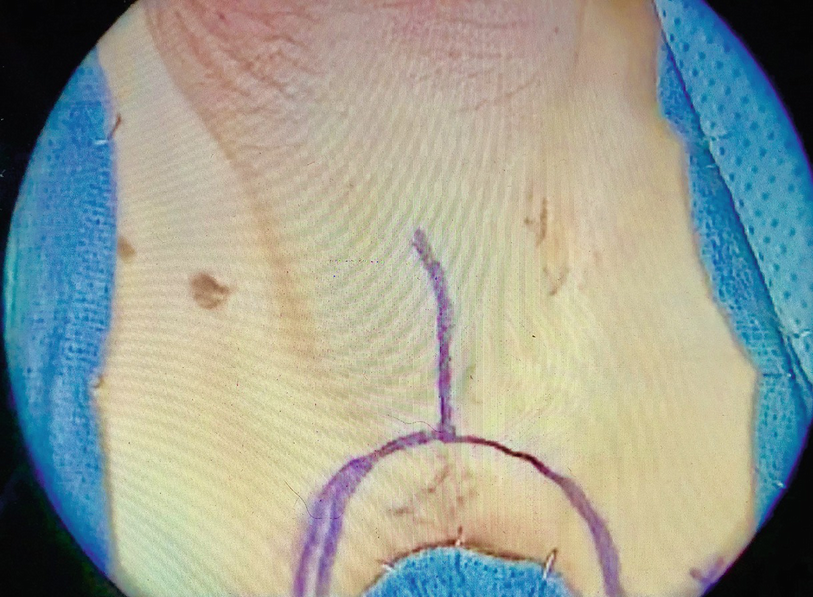
Perineal lambda incision
6.2.2.5 Tissue Handling and Visualisation
As mentioned above, the principle do no harm is primordial when performing reconstructive surgery. One must handle tissues with caution and care, using delicate movements and fine instruments that do not crush or tear the tissues. The tissue planes must be respected, and meticulously dissected layer by layer. Hemostasis must be quick and precise, as a bloody field limits proper visualisation of surgical planes. Bipolar cautery is preferentially used, to limit thermal damage to the adjacent delicate tissues.
Proper visualisation is key. A good retractor is essential in achieving this, as described in the instrumentation segment of this chapter. Surgical loupes, proper lighting and a knowledgeable surgical assistant are all useful adjuncts in achieving good visualisation.
6.2.2.6 Urethral Calibration

Set of bougies-à-boule sized from 8Fr through 30Fr
6.2.2.7 Urethral Anastomosis
The principles of an anastomotic repair are applicable to urethral anastomoses . The reconstructive surgeon must achieve a (1) tension-free, (2) watertight, (3) well-vascularized and (4) stented urethral anastomosis using (5) resorbable sutures.
The first step in achieving a tension-free anastomosis is mobility. Proximally, the entire bulb of the corpus spongiosum is dissected and the central tendon is divided, allowing for mobility of the bulb. Distally, Buck’s fascia is dissected off the corpus spongiosum until the penoscrotal junction. Dissection distal to the penoscrotal junction causes chordee. When performing an excision and primary anastomosis (EPA) urethroplasty, corporal splitting allows the urethra to lie in a straight plane, shortening the gap. Corporal splitting must not be performed if a grafted anastomosis is necessary. For posterior urethroplasty, infrapubectomy and corporal rerouting may also be necessary when large gaps must be bridged.
To achieve a watertight anastomosis , the surgeon must place 8–12 circumferential sutures. The three apical sutures (11, 12 and 1 o’clock) are very important and should be performed at the beginning of the anastomosis. If required, a traction suture at 6 o’clock may be placed before the apical sutures, to aid with visualisation.
The dual antegrade-retrograde blood supply to the corpus spongiosum allows a transecting urethral anastomosis to remain vascularized. However, certain clinical situations – such as anatomic abnormalities like hypospadias, a history of radiation therapy and prior urethral surgery – may warrant a vessel-sparing approach [11]. For patients with PFUD, erectile function must be evaluated pre-operatively. If erectile dysfunction following the injury is present, doppler ultrasound evaluation of the corporal arteries is indicated and if injury is present, a revascularization procedure must be performed before proceeding with the urethroplasty.
A small-caliber urethral stent must always be placed through the urethral anastomosis. This acts not only as a drainage tube, but also as a wick for blood and urethral secretions. A 14Fr silicone catheter is the authors’ stent of choice. If the catheter is placed before completion of the anastomosis, great care must be taken not to suture the stent into place. If a SPT is also in place, it is placed to gravity drainage with a bag, and the urethral catheter is capped.
Finally, as with any suture placed in the urinary tract, the suture of choice must be resorbable to prevent stone formation. The authors’ sutures of choice are either Vicryl® (Ethicon, Somerville, NJ) or PDS® (Ethicon, Somerville, NJ) on an RB-1 taped needle. Suture size varies between 3–0 and 5–0, depending on the clinical situation.
6.2.3 Post-operative Principles
6.2.3.1 Early Post-operative Care
While in the recovery room , patients should be assessed for leg pain. Leg pain out of proportion to the clinical exam is a clinical sign of compartment syndrome, a rare yet severe complication of lithotomy position. This is more common if the surgical time was longer than 4 h.
The majority of urethroplasty patients can be discharged after a short-stay admission (<23 h), and some centers have been practising same-day surgery for young patients with an uncomplicated surgery [12, 13].
Patients are discharged home with a leg bag for their catheter. If a SPT is also in place, it is placed to gravity drainage with the leg bag, and the urethral catheter remains capped. Patients are taught proper catheter care and instructed to keep the perineal incision dry and clean. Patients are also instructed not to place direct pressure on the perineum until they are seen for post-operative follow-up, and rather to sit in a slouched or reclined position. Anticholinergics may be prescribed to prevent bladder spasms, as well as erection suppression to prevent painful erections in young patients. Antibiotic prophylaxis is not indicated in patients with a negative pre-operative urine culture, but a urine culture should be performed at the time of the voiding trial in clinic.
If a buccal graft was harvested, ice packs are very helpful in limiting swelling of the cheek. Patients may increase diet as tolerated, and are instructed to avoid very hot, spicy and salty foods for the first few days or weeks, as they may irritate the harvest site. Viscous lidocaine and Peridex oral rinse may be used as needed.
6.2.3.2 Long-Term post-operative Care
Patients are seen 3 weeks post-operatively in the outpatient clinic for imaging and a voiding trial. In the authors’ practice, the bladder is filled with contrast material via the urinary catheter, then the catheter is removed and a VCUG is performed. Alternately, a peri-catheter RUG may be performed, and the catheter removed if there is absence of extravasation. If a SPT is in place, it is capped and kept in situ for 48 h. If the patient is voiding well, they are instructed on how to remove it at home.
There exists no standardized follow-up protocol after urethral reconstructive surgery. In the authors’ practice, the patient is seen 3 months post-operatively for a uroflow rate and post-void residual (PVR). The patient is then seen at 6 and 18 months for a flexible cystoscopy. At this point, the EPA patient is discharged from our care if he is doing well. In the case of a grafted repair, we continue to see the patient yearly with a flow rate and PVR, and a cystoscopy every 2–3 years, or if symptoms recur.
6.3 Principles of Wound Healing
Knowledge of the principles of wound healing is essential for the reconstructive urologist. Understanding the phases and duration of wound healing allows the urologist to predict timing of recovery after urethral surgery and plan subsequent or sequential procedures.
The three phases of wound healing are described as follows [14]:
6.3.1 Inflammation Phase (0 to 3–5 Days)
The initial inflammatory phase begins immediately after hemostasis. After the clotting process is complete, increased local vascular permeability allows for cellular recruitment of neutrophils and macrophages, which clean the wound via phagocytosis and activate a cytokine response. This phase is clinically evident due to erythema and edema.
Infection, foreign bodies and necrosis impede wound healing by prolonging the inflammatory phase .
6.3.2 Proliferation Phase (3–5 Days to 1 Week)
The second stage of wound healing is characterized by proliferation of basal cells and migration of epithelial cells, causing epithelialization. This new epithelium acts as a protective barrier for the wound but provides little tensile strength. Proliferation of fibroblasts also occurs, initiating collagen matrix formation and myofibroblast production, which allows the wound to begin contracting and closing. Stimulated by collagen production, angiogenesis begins in this phase by forming a small network of fine capillaries.
6.3.3 Remodelling Phase (1 Week to 1 Year)
Important increases in collagen production and tensile strength occur during the final phase of wound healing. Cross-linking of collagen fibers are central to this process. The scar formed achieves approximately 80% of its tensile strength after 6 weeks. The scar continues to mature and gain strength for up to 1 year after the injury, however will never fully recover.
6.4 Principles of Tissue Transfer and Graft Take
The term tissue transfer implies harvesting from a donor site and mobilizing it to a recipient site for the purpose of reconstruction. Knowledge of the anatomy and physiologic properties of both donor and recipient sites is essential, as is the knowledge of the process through which the transferred tissue may survive in its recipient site.
First and foremost, the difference between a graft and a flap must be established. A graft implies tissue that is excised from its donor location and transferred without its own blood supply to a recipient site, where new vascularisation must be formed via a process called take , allowing the graft to survive it its recipient site.
A flap implies that the tissue is excised and transferred with its own blood supply – either preserved or surgically re-established [15].
6.4.1 Grafts for Urethral Reconstruction
6.4.1.1 Phases of Graft Take
As mentioned above, take is the process through which the graft creates a new blood supply within its host bed, allowing it to survive in its new environment. Take requires approximately 5 to 7 days and occurs in three phases , (1) imbibition, (2) inosculation and (3) revascularisation.
Imbibition (0–48 h) is the first phase during which the graft directly absorbs nutrients from its host bed. During the second phase, inosculation (48–96 h), fine capillaries between the graft and the host bed begin to align, initiating the creation of a microcirculation within the graft. With the last phase, revascularisation (5–7 days), the graft is able to survive in its new location due to a completed vascular network.
6.4.1.2 Oral Mucosa Grafts
The oral mucosa harbours multiple favourable characteristics that render it ideal grafting tissue for urethral reconstructive surgery, and it is currently the most common type of tissue used. The buccal, labial and lingual mucosae are all non-hirsute surfaces, with a thick, non-keratinized epithelium, similar to urethral urothelium. Oral mucosa also possesses a richly vascular lamina propria, ideal for graft take. This tissue is also resilient and easily handled and harvested, with minimal donor site morbidity and no cosmetic impact.
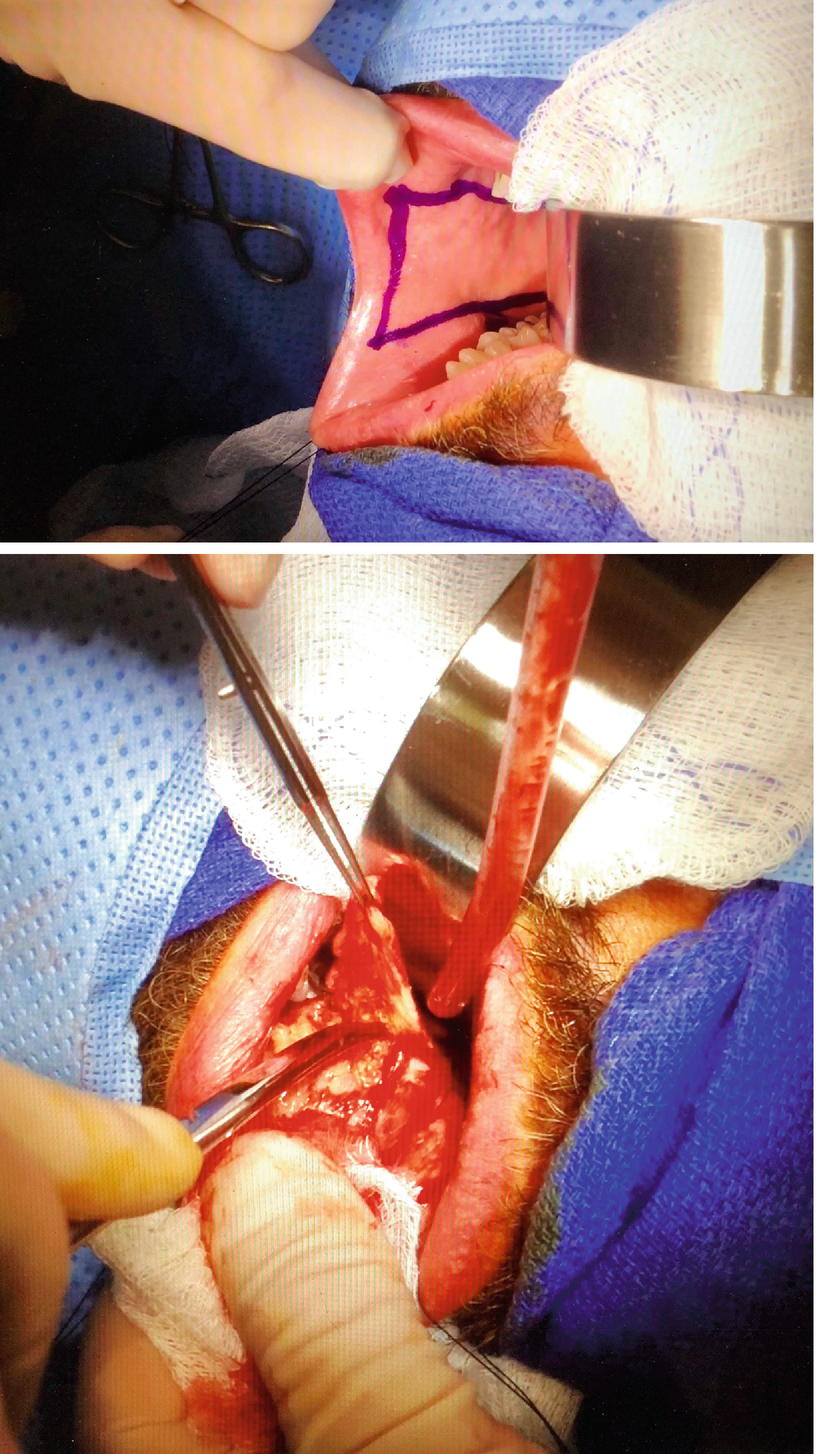
Buccal mucosa graft is the most common type of oral mucosa used and consists of harvesting the mucosa from the inside of the cheek. One can harvest a very large graft from a single cheek, usually up to 5 by 7 cm in size, and mucosa from both cheeks can be harvested if necessary. A few anatomic considerations are important to prevent donor site morbidity; firstly, Stensen’s duct (parotid gland duct) is identifiable as a small yet clearly visible dimple situated adjacent to the second maxillary (superior) molar. Stensen’s duct must be clearly marked out and avoided throughout the harvesting process, as injury to the duct may cause obstructive parotiditis requiring subsequent surgical intervention. Secondly, a margin of a minimum of 5 mm must be kept between the outer limit of the graft and the lip, as straying too close to the lip may cause scarring and poor cosmetic results. Finally, when elevating the graft, the surgeon must take care to stay above the masseter muscle. Injury of the master muscle may cause bleeding and increased post-operative pain [16]. Hydrodissection using 0.5–10% lidocaine with epinephrine aids to stay in the right plane while elevating the graft.
Endonasal intubation is not routinely required to harvest buccal mucosa graft , and it is usually sufficient to tape the endotracheal tube to the contralateral side.
Although small donor sites can be closed with resorbable suture, large donor sites should not be closed due to increased risk of post-operative pain, slower return to normal diet and normal mouth opening, and longer duration of bothersome perioral numbness. Instead, complete hemostasis is achieved, and epinephrine-soaked cottonoids are kept in place until the end of the case [17].

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree