div class=”ChapterContextInformation”>
15. Role of Transection and Anastomosis in Bulbar Urethral Reconstruction
Keywords
Urethral strictureBulbar urethroplastyEPAAnastomotic urethroplasty15.1 Etiology of Bulbar Urethral Strictures
The incidence of male urethral stricture disease is greatest in the bulbar urethra, comprising at least 47% of all strictures treated [1, 2]. It is therefore important for the reconstructive urologist to become familiar with effective and durable treatment solutions for bulbar urethral stricture disease. We favor the technique of excision and primary end-to-end anastomosis (EPA) whenever possible.
Contemporary series suggest the four most common stricture etiologies include idiopathic (37%), iatrogenic (28%), post-traumatic (15%), and inflammatory (11%), the latter including lichen sclerosis [1, 3–6]. While idiopathic urethral strictures remain poorly understood, they tend to be found in the bulbar urethra (91%), relatively short (60% ≤ 2 cm), and associated with a remote history of repetitive trauma [3]. These findings suggest that the most common etiology of bulbar urethral stricture disease, idiopathic strictures, may be the eventual consequence of unrecognized urethral injury and activities associated with repetitive subacute perineal trauma such as cycling, motocross, and horseback riding [3, 7]. Fortunately, idiopathic urethral strictures tend to be associated with excellent reconstructive success (>95% five year success) after urethroplasty [4].
Iatrogenic anterior urethral strictures are most commonly associated with previous endoscopic instrumentation, particularly transurethral resection (TUR), and prolonged urethral catheterization [6, 8]. Additional iatrogenic risk factors include prior pelvic radiotherapy and hypospadias surgery. Iatrogenic strictures more frequently occur in the penile urethra, though lesions in the bulbar urethra – specifically the bulbopendulous and bulbomembranous junction – may be amenable to anastomotic urethroplasty [6].
Acute traumatic strictures of the bulbar urethra most often occur following a straddle injury, during which blunt perineal trauma causes compression of the urethra against the pubic symphysis. This leads to some combination of loss of urethral continuity, urine extravasation, and hemorrhage, thereby precipitating inflammation and spongiofibrosis [9]. Patients may present with history of acute perineal trauma, inability to void, and blood at the urethral meatus. Resultant bulbar urethral strictures are commonly focal and obliterative in nature, often with complete transmural spongiofibrosis . These patients have excellent results with delayed primary anastomotic urethroplasty following a period of urethral rest.
15.2 Patient Selection
To optimize success and minimize risk of morbidity, appropriate patient selection is paramount. The technical objectives of anastomotic urethroplasty include complete excision of the diseased segment with formation of a wide spongiosal sparing anastomosis. A 1 cm spatulation of both proximal and distal segments can help facilitate formation of a wide (>26 Fr), oblique anastomosis, thereby reducing the risk of annular retraction and stricture recurrence. When considering the length of the excised segment, as well as the degree of proximal and distal mobilization needed to facilitate a spatulated, tension-free anastomosis, stricture length, location, and degree of spongiofibrosis are the most important considerations.
The penile urethra lacks elasticity, has a paucity of ventral spongiosum, and obtains substantial collateralizing blood supply from perforating vessels along the corpora cavernosa. These characteristics in combination with the often lengthy and multi-focal nature of penile urethral stricture disease limit the use of anastomotic urethroplasty techniques. In highly select men with focal (<1 cm) obliterative strictures of the proximal penile urethra, primary excision, distal bulbar urethra mobilization, and reanastomosis may provide a durable reconstructive solution without a detrimental impact to sexual function [10]. Nonetheless, despite a possible evolving role for EPA in highly selected men with focal penile urethral strictures, this is uncommon in practice, and therefore will not be discussed here in detail.
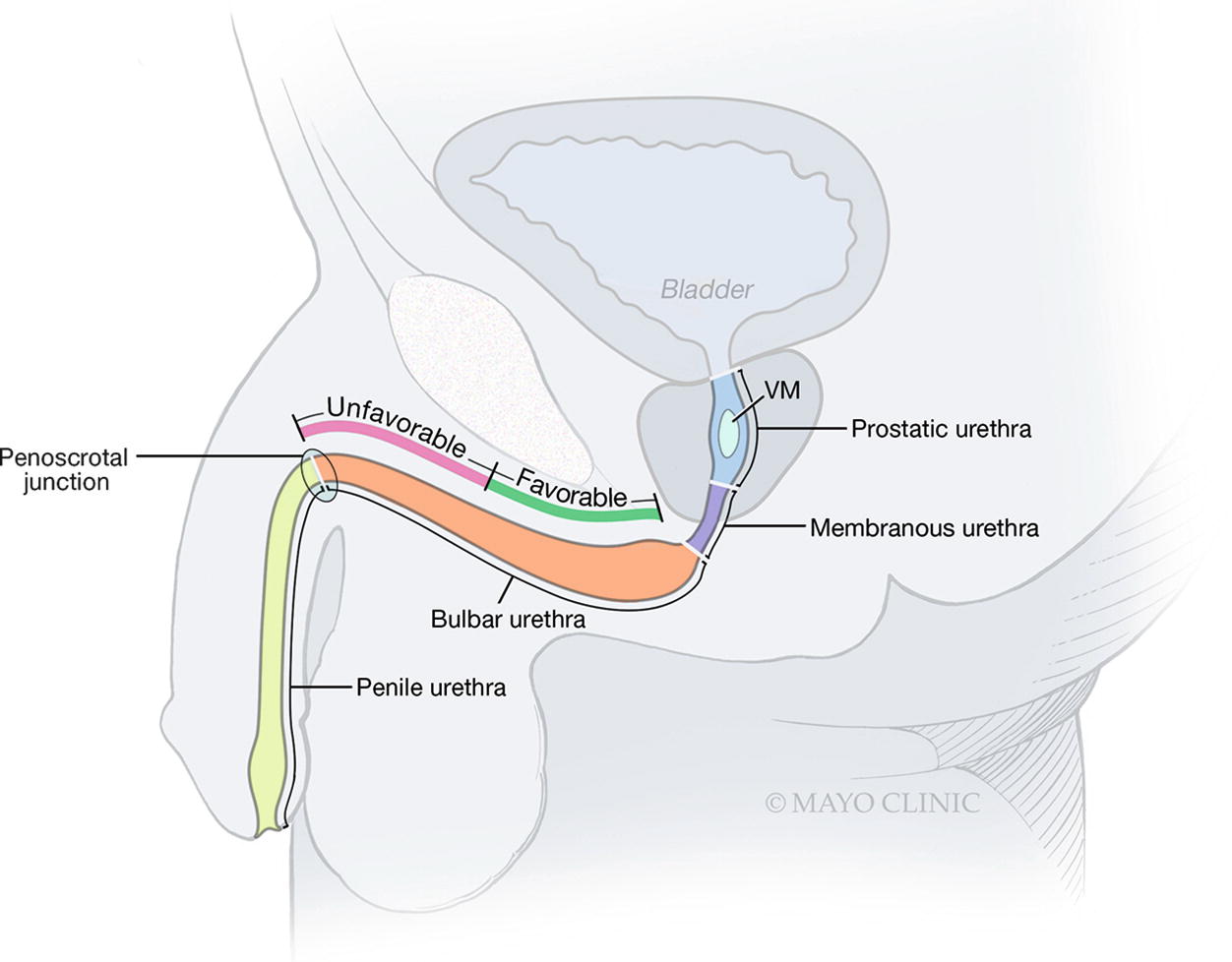
The proximal bulbar urethra is favorable for extended anastomotic urethroplasty for strictures up to 5 cm; the distal bulbar urethra is unfavorable for extended anastomotic urethroplasty and anastomotic urethroplasty in this location should be limited to strictures <2 cm; VM verumontanum
Despite a well-executed preoperative staging evaluation, the decision to proceed with EPA is finally determined with intraoperative assessment. Patients should be counseled that inability to effectively excise all scar and achieve a tension-free anastomosis may necessitate an augmented reconstruction with substitution techniques to optimize outcomes.
15.3 Preoperative Evaluation
Meticulous preoperative evaluation is necessary to facilitate surgical approach and optimize outcomes after anastomotic urethroplasty. A thorough preoperative evaluation consists of several clinical and radiographic considerations as described below.
15.3.1 History
History should include an assessment of urinary symptoms and sexual function, creating a documented baseline for postoperative comparison and to guide counseling on postoperative expectations. Standardized questionnaires (American Urological Association Symptom Index, International Index of Erectile Function, etc.) are useful to objectively quantify pre and postoperative urinary and sexual function.
While the majority bulbar urethral strictures are idiopathic, it is essential that the surgeon attempts to determine stricture etiology as this will influence candidacy for anastomotic urethroplasty and expected outcomes. Historical elements of significance include any prior perineal or pelvic trauma and relative timing, urinary tract instrumentation including urethral catheterization, and review of prior urologic and non-urologic surgeries, eliciting detail regarding any history of perioperative bladder catheterization or traumatic catheterization. Men with iatrogenic strictures abutting the membranous urethra following BPH surgery must be counseled that any EPA technique may predispose to de novo stress incontinence postoperatively. Additionally, men with inflammatory and radiation induced urethral stricture disease should be counselled on a greater incidence of stricture recurrence relative to those with idiopathic, iatrogenic, or traumatic stricture etiologies [3].
Urologic instrumentation history should be specifically evaluated, with special attention to history (including frequency and timing) of indwelling or intermittent catheterization, urethral dilation, internal urethrotomy, and prior stricture surgeries. Corresponding operative reports should be reviewed if available, as these procedures may have implications on anticipated anatomic relationships, vascular supply, and stricture complexity [13]. It is crucial to determine the timing of most recent urethral instrumentation. We suggest 6–8 weeks of “urethral rest” following any urethral intervention prior to preoperative staging and consideration of urethroplasty [14] in order to accurately characterize the urethral stricture.
Additional risk factors for impaired urethral perfusion should also be elicited, including history of diabetes mellitus, peripheral vascular disease, tobacco use, vasculogenic erectile dysfunction, or prior pelvic radiation. In patients with history of pelvic trauma, details regarding any prior pelvic angioembolization; compromise to even a unilateral internal pudendal artery, which gives rise to the ipsilateral dorsal, cavernous, and bulbar arteries, will have implications on anastomotic perfusion and erectile function postoperatively. Any neurologic disease or prior surgery (spine surgery, abdominoperineal resection, low anterior resection, etc.) placing the patient at risk for voiding and erectile dysfunction, should be noted.
15.3.2 Physical Examination
A focused genitourinary exam is crucial. Examination of the urethral meatus, fossa navicularis, ventral penile shaft, and perineum, both visually and by palpation, should enable documentation of phimosis, lichen sclerosis, prior scars, fistulae, erythema, or palpable induration, the latter frequently correlating with underlying spongiofibrosis. Testicular atrophy and general health of the surrounding genital tissue are a good indication of occult hypogonadism, a process that has been associated with urethral atrophy and decreased vascularity of the male urethra [15, 16]. Evidence and degree of stress urinary incontinence is useful to guide patient expectations postoperatively, direct operative approach (transecting vs non-transecting), and the need for future interventions which may require perineal dissection for male stress incontinence surgery. Chronic urine soilage of the perineum and intertriginous folds can be associated with fungal and/or bacterial infection, which may complicate incisional healing postoperatively. Stretched penile length (SPL) is useful to establish a baseline prior to reconstruction and guide patient selection for extended bulbar anastomotic urethroplasty, which is facilitated by increasing penile length [12].
BMI should be considered, as obesity can pose challenges in achieving adequate surgical exposure, increases the risk of positioning-related neuromuscular complications, and may be associated with increased likelihood of stricture recurrence [17]. Given the high lithotomy position which is required for bulbar urethral exposure, mobility of the bilateral lower extremities, particularly at the hips and knees, and neuromuscular function should be evaluated.
15.3.3 Radiographic and Endoscopic Evaluation
Radiographic and endoscopic evaluation provides the surgeon with an understanding of patient-specific urethral anatomy; this is crucial to appropriately select candidates for anastomotic bulbar urethroplasty. It is necessary to stop all urethral instrumentation for a period of time before surgical reconstruction. In men presenting with indwelling catheters, chronic intermittent self-catheterization, or recent endoscopic instrumentation (dilation, DVIU, etc.), we recommend suprapubic cystostomy or a trial of spontaneous voiding for at least 6–8 weeks to allow for scar maturation and stabilization, before stricture staging and definitive reconstruction [14].
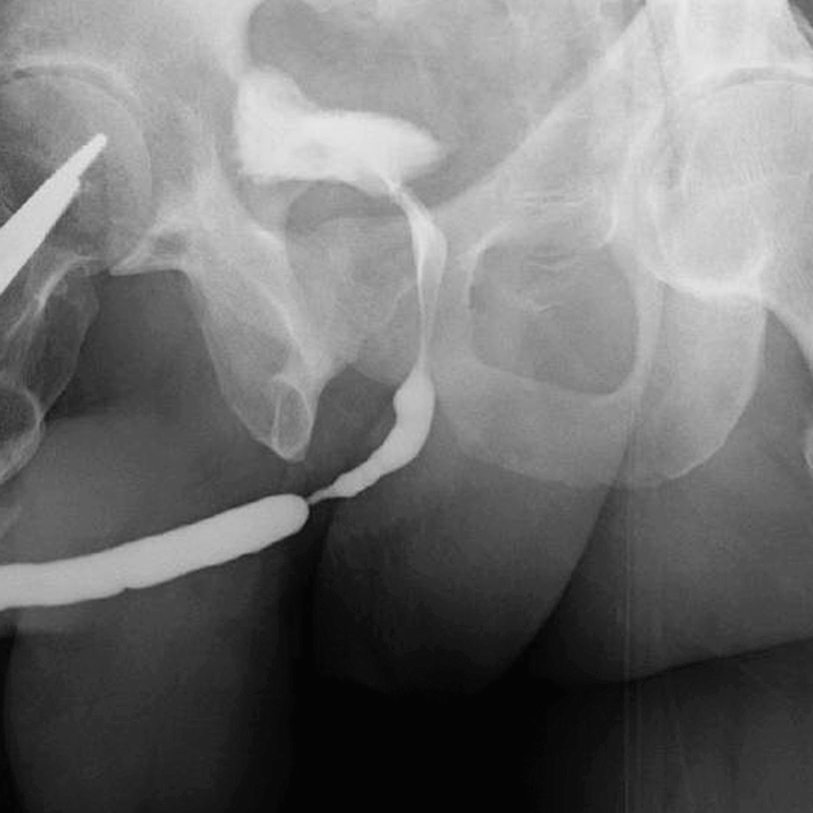
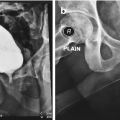
Retrograde urethrogram demonstrating a focally high-grade 2 cm mid bulbar urethral stricture
Preoperative flexible urethroscopy is a useful tool to assess urethral anatomy , characterize stricture density, and identify any suspicious lesions that warrant biopsy. We reserve preoperative urethroscopy for those men with stricture recurrence following prior urethroplasty and those with rapid stricture recurrence following endoscopic management to rule out malignant transformation. Urethroscopy is useful to characterize the relationship between the stricture and the membranous urethral sphincter in those with prior bladder outlet surgery, as well as those men with prior radiation to assess adjacent urethral integrity and vascularity. In men with indwelling suprapubic catheters, antegrade cystourethroscopy can also be performed. While not routinely used in our practice, ultrasonography and MRI provide accurate stricture staging evaluation and may be useful adjuncts in the identification of associated pathology such as fistulae, diverticulae, abscesses, and false passages [19].
15.4 Presurgical Preparation
Prophylactic guideline specific parenteral antibiotics are administered prior to incision, and in men with positive preoperative urine cultures, targeted antibiotic therapy should be implemented. Men with incomplete bladder emptying, recent instrumentation, or indwelling suprapubic catheters, often have urinary bacterial colonization. In these patients, we prefer to initiate culture directed prophylaxis starting three days before surgery to sterilize the urine. For those men without such risk factors, we do not initiate preoperative antibiotic therapy, as prophylaxis administered the day of surgery is typically sufficient to prevent postoperative infection. Venous thromboembolism prophylaxis includes lower extremity sequential compression devices intraoperatively and early postoperative ambulation.
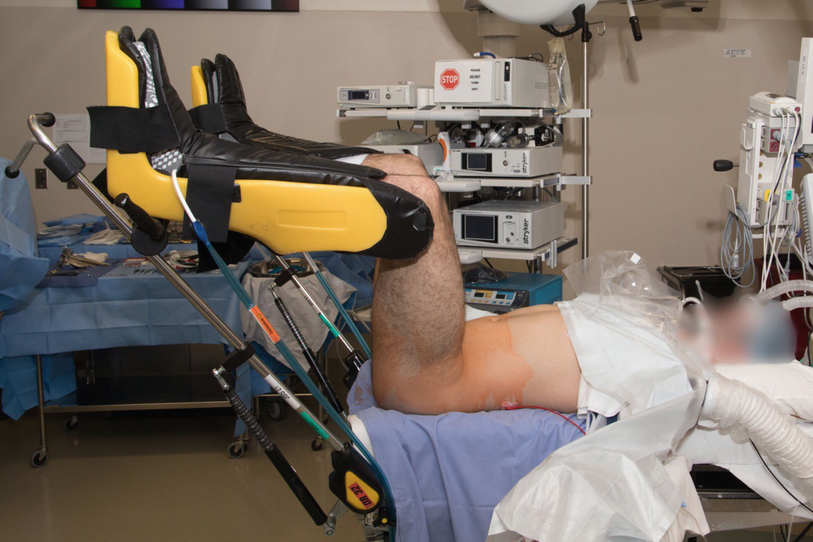
Positioning for transperineal anastomotic bulbar urethroplasty
The risk of positioning-related complications , such as rhabdomyolysis, compartment syndrome, and peroneal neuropraxia, can be minimized by limiting the duration of exaggerated lithotomy positioning; these complications are uncommon when operative duration does not exceed 5 hours [20]. Relative to alternative reconstructive techniques, anastomotic urethroplasty of the bulbar urethra is a time efficient operation with operative times that rarely exceed 4 hours. Nevertheless, we recommend limiting the duration of high and exaggerated lithotomy positioning whenever possible. A low or standard lithotomy position may provide sufficient exposure for more distal bulbar lesions. In our practice during portions of the operation such as wound closure, where high lithotomy is not necessary, we routinely lower the legs into standard or low lithotomy position .
Using electric clippers, all hair is carefully removed from the perineal and genital region. Epidermal abrasions should be avoided as these may be a potential entry point for bacteria, thereby increasing the risk of surgical site infection. Chlorhexidine skin preparation is performed from the umbilicus to the mid-thigh, including the genitalia and perineum. The sterile field includes the suprapubic region to facilitate antegrade access if a suprapubic tube is in place, or in the event that one is needed.
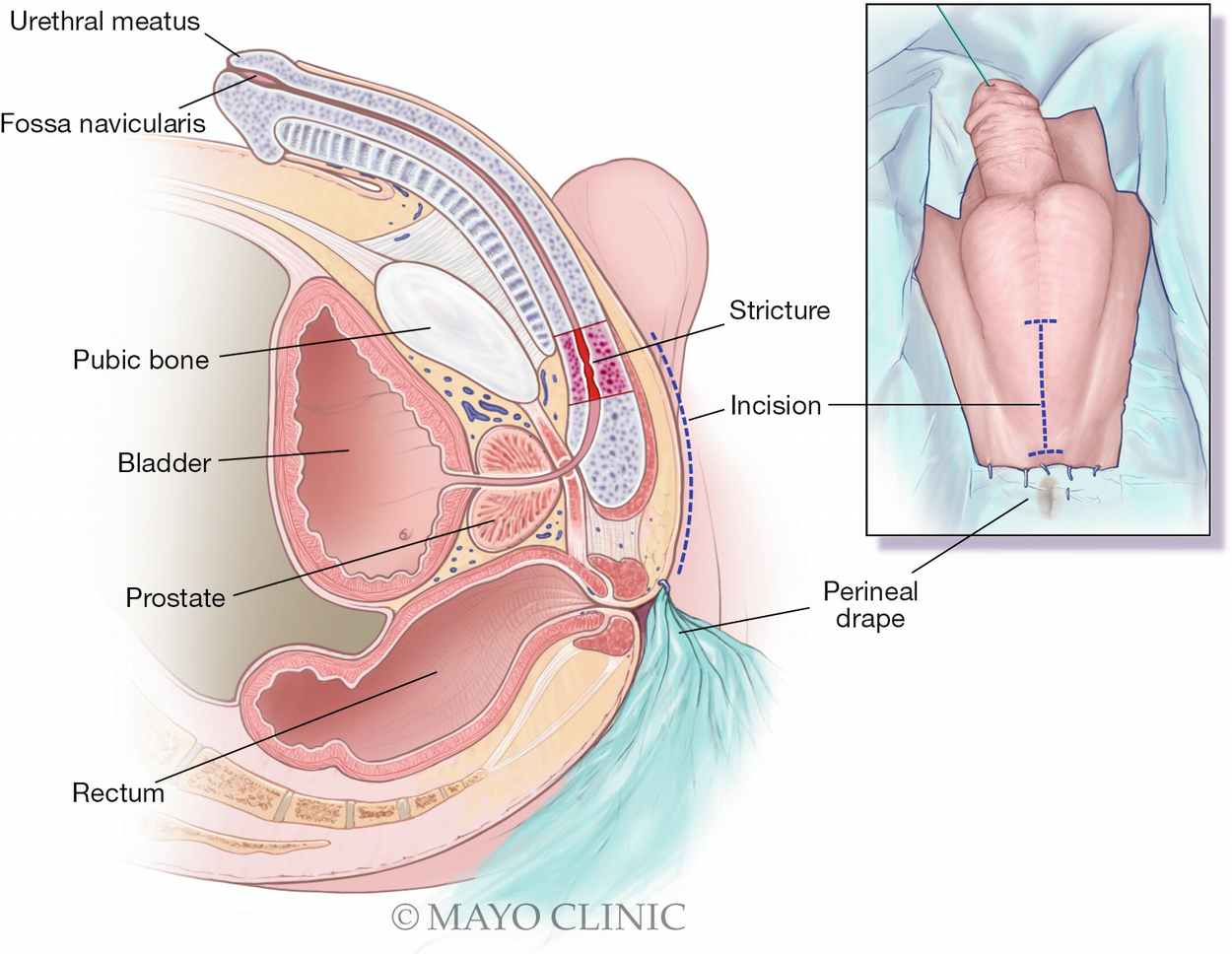
The perineal drape is secured just anterior to the anal verge as shown, maintaining surgeon orientation during initial transperineal dissection in a plane parallel to the rectum
15.5 Operative Technique
Retrograde endoscopic evaluation with a flexible cystoscope allows for a final evaluation of the anterior urethra, and precise confirmation and localization of the distal aspect of the stricture. Aggressive attempts to negotiate the scope proximal to the stricture are not made, but if the surgeon is able to smoothly negotiate a 17 Fr cystoscope through the stricture without resistance, the procedure should be terminated. For non-obliterative strictures , proximal access is secured by passing a flexible guidewire (0.035 in. diameter) into the bladder. Palpating the wire can expedite initial perineal dissection, particularly in obese patients and reoperative fields. If the stricture is obliterative, intraoperative antegrade urethroscopy can facilitate localization of the proximal non-diseased urethra. We caution against the practice of only securing proximal access after transection for proximal bulbar lesions; in a bloody operative field this can be highly challenging, risking loss of tissue planes and potential adverse outcomes.
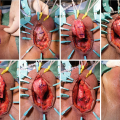
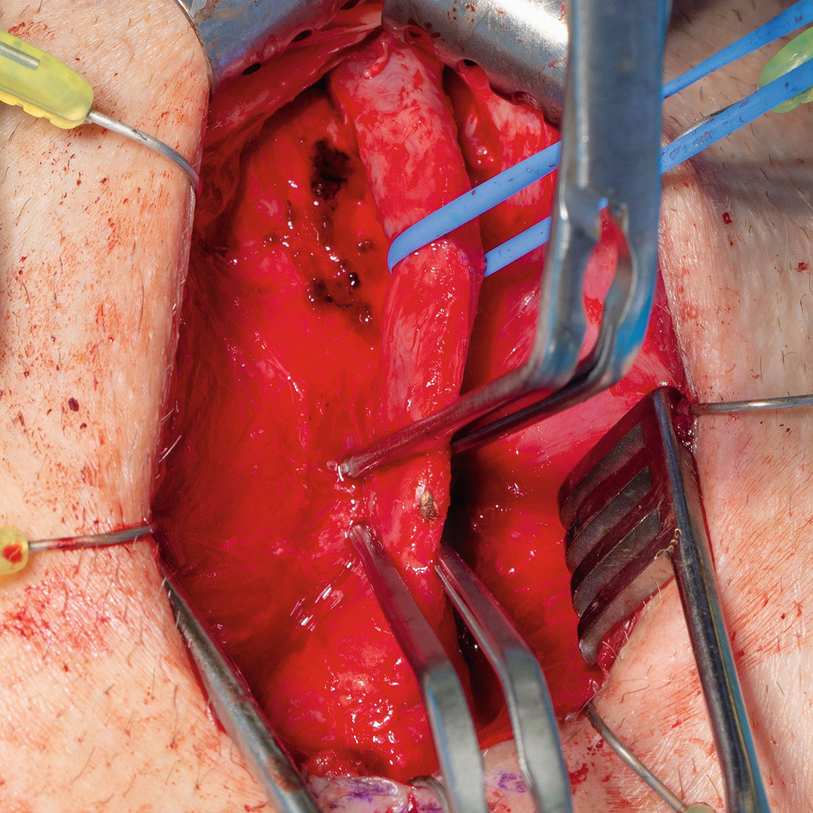
The cystoscope is removed and the safety wire is secured out of the field. A vertical midline incision is made sharply along the perineal raphe from the perineoscrotal junction anteriorly, and carried down to approximately 1 cm above the anal verge (Fig. 15.4). For mid to distal strictures where additional distal urethral mobilization may be needed, this incision can be extended anteriorly onto the base of the scrotum. We have abandoned use of the lamba incision for transperineal urethroplasty due to increased risk of postoperative wound complications [21]. The incision is deepened though the subcutaneous tissue with electrocautery, following which a self-retaining retractor is placed. We prefer use of the Lone-Star self-retaining retractor system (Cooper Surgical, Trumbull, CT, USA), and tend to place this promptly. Eight blunt hooks (sharp hooks for reoperative cases) are placed circumferentially, with laterally placed hooks applying excellent lateral traction thereby facilitating efficient electrocautery dissection through Colles fascia and the perineal fat down to the bulbospongiosus muscle. In men with an abundance of perineal or scrotal fat, it can be helpful to dynamically reposition the hooks and utilize small hand-held retractors as the dissection is deepened, maximizing visualization until there is satisfactory exposure. Two four-finger rake retractors are secured to the retractor at the 10 and 2 o’clock positions to elevate the scrotal contents out of the field.
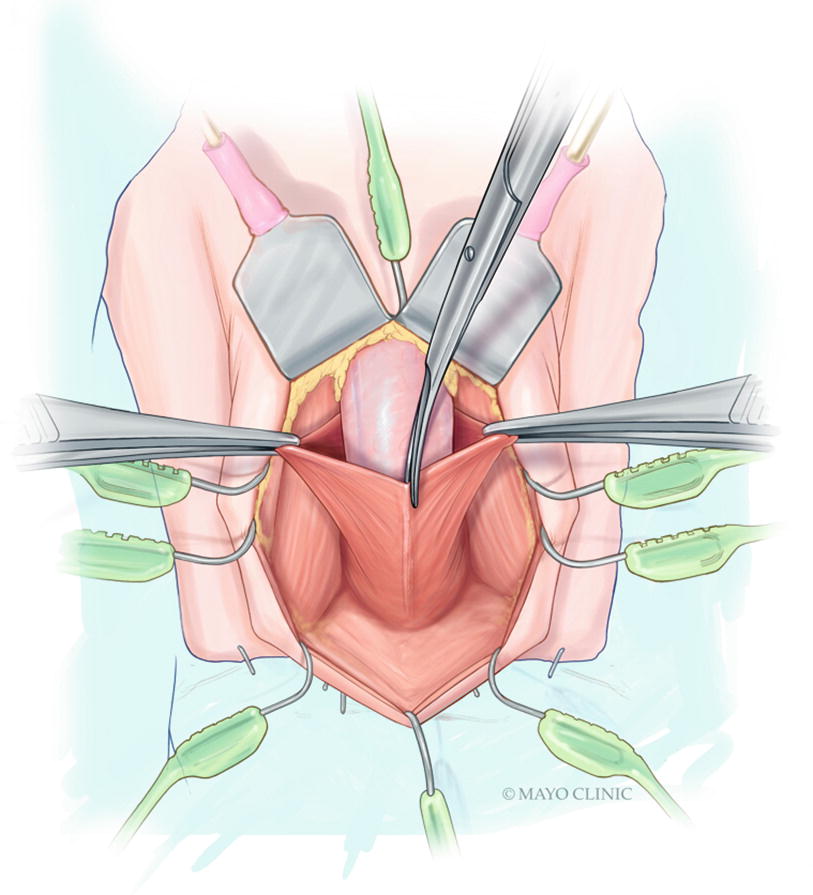
Sharp midline division of the bulbospongiosus muscle
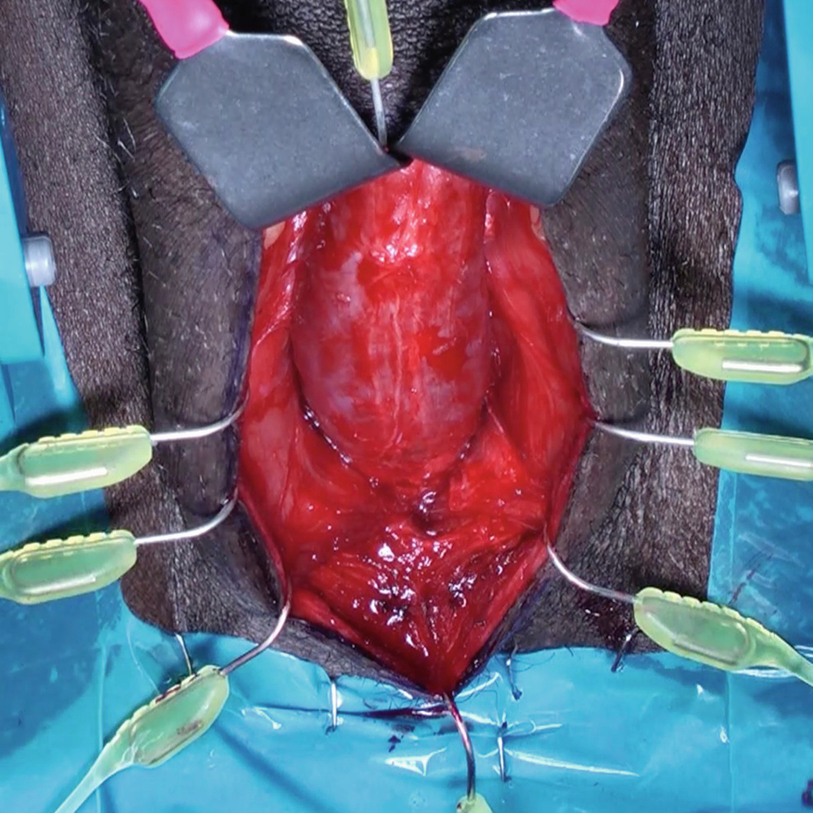
Blunt hooks for the self-retaining retractor are repositioned, incorporating the divided bulbospongiosus muscle, facilitating exposure of the underlying corpus spongiosum
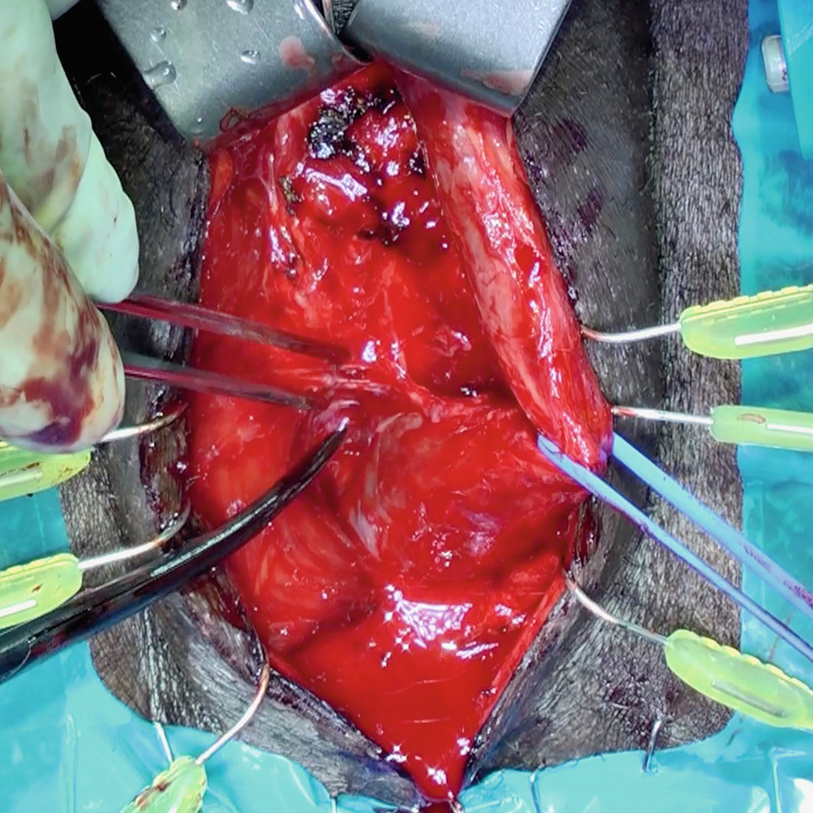
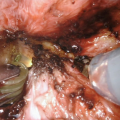
Following circumferential exposure distally, a vessel loop facilitates atraumatic retraction during additional proximal bulbar mobilization
A Beckman-Weitlaner self-retaining retractor can be utilized to further aid with proximal bulbar exposure
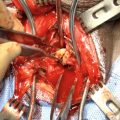
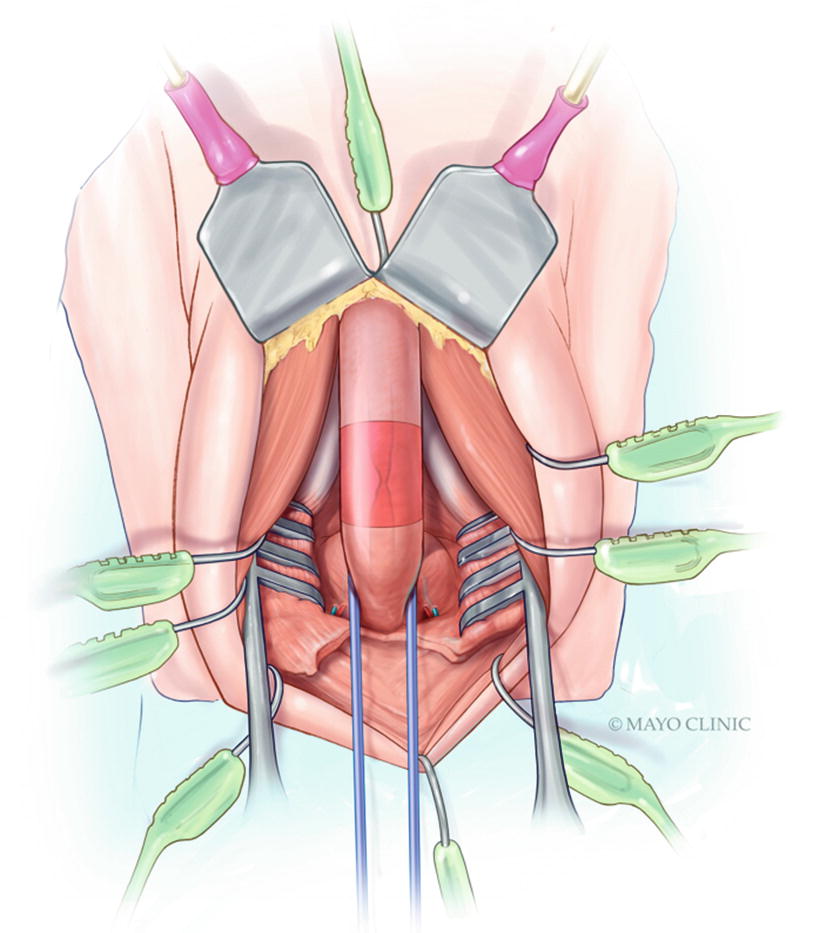
The paired bulbourethral arteries , branches of the paired internal pudendal arteries, perforate the anterior pelvic floor triangle and enter the base of the bulbar urethra at the 4 and 8 o’clock position. These vital bulbar arteries should be prospectively identified and avoided when possible; this will maximize antegrade anastomotic perfusion, theoretically reducing the risk of ischemic recurrence, particularly in men with impaired urethral vascularity [22]. In men with concurrent stress urinary incontinence requiring staged artificial urinary sphincter, maximizing bulbar urethral perfusion is important to reduce the risk of urethral cuff erosion [23]. Though rarely necessary in our practice, the natural curve of the bulbar urethra, largely generated by the proximal fusion of the corporal bodies superiorly with the bulbar urethra curving in concave fashion below and coursing cephalad towards the bulbomembranous junction, can be straightened by incising and splitting the intercrural septum, affording additional mobilization [24].
After sufficient mobilization of the bulbar urethra is complete, attention is turned to localization of the stricture. Multiple techniques exist for precise identification of the distal aspect of the stricture, including passing a sound via the meatus (soft urethral catheter, or Bougie-à-Boule), as well as flexible urethroscopy. We prefer the latter, and once the distal aspect of the stricture is identified endoscopically, the corresponding location on the ventral bulbar spongiosal tunica is identified and marked with electrocautery. Regardless of the technique chosen, it is important to avoid erring distally, which would otherwise sacrifice healthy urethral length. Likewise, it is crucial to avoid dilating the stricture, which can obscure estimation of the true margins of the diseased segment resulting in incomplete excision, a common cause of stricture recurrence.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree