div class=”ChapterContextInformation”>
36. Utility of Hyperbaric Oxygen Therapy in Genitourinary Reconstruction
Keywords
Hyperbaric oxygenHBOTUrethral strictureHypospadiasFistulaRadiation hemorrhagic cystitisFournier’sReconstructionUrologyUrologicHyperbaric oxygen therapyGenitourethral reconstructionTissue hypovascularityRadiation therapyHemorragic cystitis36.1 Introduction
The undersea and hyperbaric medical society approved uses for hyperbaric oxygen therapy |
|---|
Air or gas embolism |
Carbon monoxide poisoning |
Clostridial myositis and Myonecrosis (gas gangrene) |
Crush injury |
Compartment syndrome and other acute traumatic ischemia |
Decompression sickness |
Arterial insufficiencies |
Severe anemia |
Intracranial abscess |
Necrotizing soft tissue infections |
Osteomyelitis (refractory) |
Delayed radiation injury |
Compromised grafts and flaps |
Acute thermal burn injury |
Idiopathic sudden sensorineural loss |
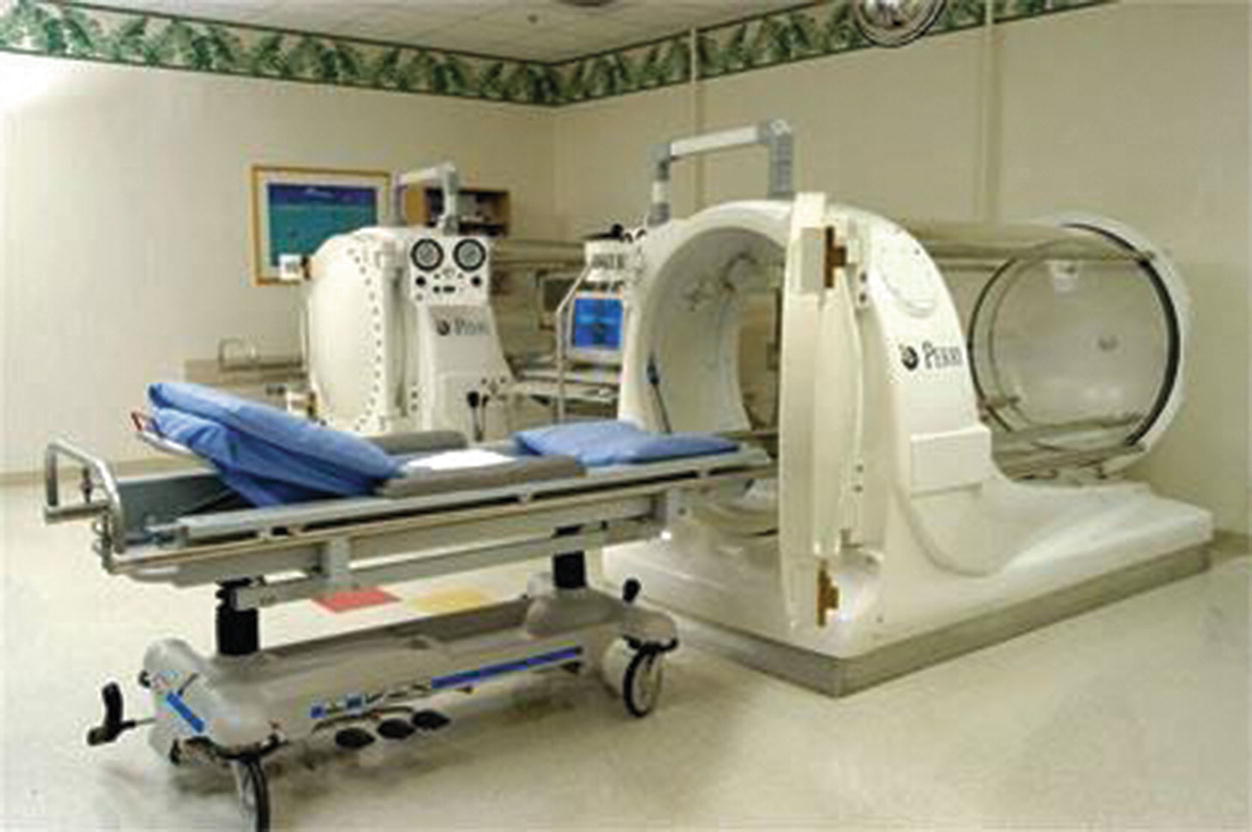
Hyperbaric oxygen chamber. (Photo courtesy of Hyperbaric Medicine & Wound Treatment Center of Utah)
36.2 Use in Surgery
As mentioned, one of the approved indications for HBOT includes compromised grafts and flaps. It’s believed that the enhanced mechanism of angiogenesis described above hastens necessary granulation and thereby improves graft uptake [6]. Since the 1960’s, over 50 animal studies have been performed evaluating the efficacy of HBOT to improve graft and flap healing. These studies have largely shown that HBOT increased capillary network formation when evaluated on microangiography, have earlier graft and flap uptake and improved perfusion [7, 8].
Human studies have largely been limited to case reports and retrospective series but have reported positively on enhanced wound healing afforded by HBOT [5, 7]. Only one randomized control trial has been performed to date and was performed in 1967 on 48 patients undergoing split thickness skin grafts. HBOT was delivered at 2 ATM for 120 min twice daily for 3 days. Patients in the HBOT group had 64% complete graft survival compared to 17% in the control group (p < 0.01). While the results are dramatically in favor of HBOT, the number of sessions are significantly reduced compared to most regimens today in addition to lower average pressure. Furthermore, the 17% graft survival in the control group is remarkably poor and would be considered unacceptable by today’s standards [5].
While the available evidence is practically unanimous in support of HBOT’s availability to improve graft and flap survival, it’s worth noting that its indications are limited to tissues that are threatened, compromised, or otherwise at risk [6]. Therapy should be timely. A retrospective study on free tissue transfer or replantation noted 89% complete graft viability when HBOT was initiated immediately after surgery, 25% when initiated 1–3 days post operatively, and 0% when started more than 3 days post operatively [8]. In concordance with The Undersea and Hyperbaric Medical Society and expert opinion, HBOT for compromised grafts or flaps should be administered at 2.0–2.5 ATM from 90 to 120 min, initially twice per day but can be transitioned to once daily when the flap or graft appears viable and stable. 30–40 sessions are typically required to meet desired outcomes [6].
36.3 Indications in Urology
36.3.1 Hypospadias Repair
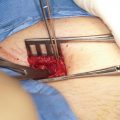
Hypospadias repair is fraught with potential complications including fistula, meatal stenosis, stricture, and dehiscence. Given that in hypospadias repair, adequate vascularity is critical to repair survival and may help reduce the risk of complications, HBOT may have a role in improving outcomes. In a series of 669 pre-pubertal patients undergoing tubularized incised plate (TIP) repair for hypospadias, Bush et al. identified only two independent risk factors for post-operative complications: reoperative TIP (OR 3.07, 95% CI 1.54–6.13) and meatal location (OR 1.79, 95% CI 1.34–2.40) [9]. This has led to some experts proposing HBOT be used in patients undergoing their third or more re-operation [10].
There are a few small series exploring the use of HBOT in hypospadias repair. In a 2014 study out of Turkey, Ateş et al. used 10 sessions of HBOT in 13 patients undergoing proximal and midpenile hypospadias repair with buccal mucosal tube urethroplasty and compared them to 3 patients who did not. In the HBOT group, 5 patients were primary repair and 8 patients were reoperation, with an average graft length of 5.4 cm. In the non-HBOT group, 1 patient was a primary repair and 2 patients were re-operation, with average graft length of 8 cm. The HBOT group experienced 53.8% first stage success and 84.6% second stage success. The three patients in the non-HBOT group had 0% first stage success and 33% second stage success [11]. Though the study is underpowered to make any claims regarding the effectiveness of HBOT in hypospadias repair, it was among the first to demonstrate the potential of its use.
In a 2018 retrospective cohort study , Verhovsky, Bush and Snodgrass et al. compared two groups undergoing two stage hypospadias repairs with either preputial or buccal grafting. Group 1 included 7 patients who underwent HBOT during their repair. These patients had an average of 4 prior operations. Group 2 included 14 patients with an average of 3 prior operations and did not receive any HBOT. Each group was roughly equivalent in severity of hypospadias and prior complications. Group 1 experienced good graft take in 100% (7 out of 7) of patients with two cases neededing reoperation (one for meatal stenosis and another for fistula). In group 2, 14 patients underwent 20 operations graft contractures occurred in 40% (8 out of 20). The 6 patient reoperations were due to 4 cases of fistula, 2 of meatal stenosis and 2 of dehiscence. In all there was a 50% increase in reoperations/complications in group 2 (non-HBOT) [12]. Although, an observational study, this furthers the evidence toward improved outcomes when complimenting repair with HBOT, particularly in patients requiring grafting and reoperation.
While HBOT has shown promise in early studies, its use in hypospadias repair would frequently involve pediatric patients. HBOT in adults is well studied and generally well tolerated; however, its use in the pediatric population can pose challenges. Few centers are equipped to manage pediatric patients, and the patients may not always be able to convey potential complications they’re experiencing. A 54-patient series of patients 3 months to 16 years old found overall HBOT was well tolerated in this age group. Potential complications include oxygen toxicity convulsion (0.3% of treatments), progressive hypoxemia (0.3% of treatments), and brief hypotension (0.6% of treatments) [13]. HBOT appears to be a feasible option in both adults and pediatric patients where available.
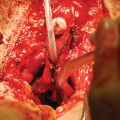
36.3.2 Urethral Stricture
Urethral stricture repair has several known potential complications including re-stricture, post-micturition dribbling, and erectile dysfunction (ED) . Rates for each have been reported to vary from 10–20% depending on stricture location, length, and other risk factors [14, 15]. A 2013 metanalysis investigating de novo rates of erectile dysfunction after anterior urethroplasty found the incidence to be 1–3%, and up to 86% of the time, ED was self-limited and resolved within 6–12 months [15]. Though anterior urethroplasty has a limited durable impact on erectile function, posterior urethroplasty rates carry a 2–5% risk of de novo ED [16].
A 2010 study randomized men to HBOT or nothing following posterior repair. The HBOT group received 14 treatments starting post-operative day 1. International Index of Erectile Function (IIEF) was used to assess erectile function preoperatively and at 3 months. Patients in the HBOT group reported a significantly higher overall IIEF score, as well as scores in the erectile function, overall satisfaction, and intercourse satisfaction. Stricture recurrence rates were not assessed [17]. It’s worth noting that 3 months may be insufficient follow up to suggest HBOT has a role in this setting given historically, ED requires 6–12 months to assess resolution. HBOT has been shown to improve fascial nerve regeneration and it’s reasonable to expect hastened and improved recovery of ED in the setting of stricture repair via enhanced angiogenesis and nerve regeneration [18].
There is paucity of data describing the use of HBOT in treating urethral stricture disease itself or preventing recurrence. A retrospective cohort of 25 patients undergoing HBOT for radiation injuries included 5 patients who were treated for urethral stricture. Four of the 5 patients saw improvement of the stricture with HBOT (1 with “major” response, 3 with “minor” response) while 1 patient experienced stricture progression. The study was not designed to measure stricture specific improvements and no details were provided on how HBOT was used in this setting, and what, if any, other treatments were used [19]. Though the data are limited, extrapolating the known benefits of HBOT toward stricture disease, there is likely a role for HBOT , particularly in threatened tissues.
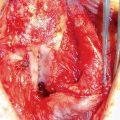
36.3.3 Urologic Fistula
Urologic fistulae are a rare though unfortunate potential side effect of pelvic radiation. Common sites of fistula formation include rectourethral, rectovesical, vesicocutaneous, enterovesical, and fistula from bladder to abscess. Fistula formation is often associated with an inciting event in radiated patients such as rectal biopsy, TURP, and performing clean intermittent catheterization [20]. Management varies on fistula size and location and can involve partial bowel resection, urinary diversion or fecal diversion. There have been few case reports and series evaluating the use of HBOT in treating urologic fistula [21].
Marguet et al. described 4 patients with rectal ulceration at risk for fistula formation. Patients had temporary urinary diversion with urethral or suprapubic catheter and underwent a mean of 50 HBOT sessions. All 4 patients had progression from rectal ulcer to rectourethral fistula after at least 40 HBOT sessions [22]. Another study involved 3 patients with rectourethral fistula following radiation therapy that underwent suprapubic tube placement and HBOT. Two of the 3 patients had eventual resolution of the fistula over 19–21 months [23].
The limited data is mixed regarding the effectiveness of HBOT with temporary diversion for the treatment of rectourethral fistula in patients with history of pelvic radiation. While there may be a role for HBOT in this setting, 19–21 months for complete resolution may be reason for some to favor earlier more definitive, albeit invasive, treatments.
36.3.4 Radiation-induced Hemorrhagic Cystitis
Radiation-induced hemorrhagic cystitis (RHC) occurs with a frequency of 3–6.5% following pelvic radiation and can present from at any time from 6 months to 20 years following radiation therapy [24–26]. RHC can be a tremendous burden to patients requiring multiple hospitalizations for active bleeding, clot urinary retention and transfusions. Some series have reported an associated mortality up to 44% [27]. Therapeutic options are varied but no large prospective study has been performed to date.
A 2018 meta-analysis evaluated HBOT for the treatment of RHC. Sixteen studies with a total of 602 patients who received HBOT for RHC were included. Pooled analysis found 84% (95% CI 76–91%) of patients had at least a partial to complete response to HBOT. Only 14% of patients who responded to HBOT experienced a relapse of symptoms with a median time of relapse of 10 months following HBOT. Predicters of HBOT success included increased number of sessions, a lower (better) Toxicity criteria of the Radiation Oncology Group (RTOG)/European Organization for Research and Treatment of Cancer (EORTC) score, lower RT dose, younger age, and HBOT initiated within 6 months of symptom onset [28].
Though HBOT comes with a significant cost and time commitment, the potential for durable response , particularly when initiated early, makes HBOT an attractive option for RHC.
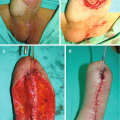
36.3.5 Fournier’s Gangrene
Fournier’s Gangrene is a necrotizing fasciitis of the genitals, perineum or perianal regions. It is an aggressive infection with mortality rates varying from 7–40% across the literature [29]. Infections tend to be polymicrobial and can consist of aerobic and anaerobic organisms [30]. Treatment recommendations are varied but generally consist of early initiation of broad-spectrum antibiotics and surgical debridement of necrotic and devitalized tissue. Following debridement and depending on defect size, there are various options for wound closure including flaps, grafts, and wound VAC [31–33]. HBOT has been utilized by many centers to enhance wound healing and improve infection control [29, 34, 35].
HBOT has been shown to improve efficacy of some antibiotics and can be therapeutic against anaerobic organisms by significantly increasing oxygen delivery to threatened tissues [34]. HBOT encourages aerobic respiration in many pathogens, and increases reactive oxygen species formation, a process which several antibiotics depend on for efficacy [36, 37]. Fluoroquinolones, Penicillins, Aminoglycocides and Carbapenems all have a theoretical potential to be enhanced by HBOT [36–39]. The ability of HBOT to improve infection control in Fournier’s gangrene remains theoretical as no study to date has validated the potentiation of antibiotics by HBOT in this setting.
While there are no prospective randomized trials investigating HBOT use in Fournier’s, retrospective studies do not support its use. Mindrup et al. retrospectively reviewed 42 consecutive patients treated for Fournier’s gangrene at their institution, 26 of the patients were treated with HBOT for a median of 6 treatments (range 2–26) in addition to standard therapy, and the remaining 16 patients underwent standard treatments alone. There were no significant differences in patient characteristics and comorbidities between the two groups, including number of debridements or split-thickness skin graft size. The decision to use HBOT was up to the treating surgeon. The results of the study showed no differences in outcomes between the two groups including graft survival, disease specific mortality, duration of hospital stay or complications. The only significant finding was patients in the HBOT group had a roughly $1000 increase in daily hospital charges. It was also noted that delivering HBOT to patients in the inpatient setting, particularly in the immediate postoperative period can be difficult and dangerous [35].
Given the lack of compelling evidence and significant cost and time burden, HBOT use in Fournier’s remains experimental and is best reserved for refractory cases , or patients at high risk for impaired flap or graft healing.
36.4 Conclusions