div class=”ChapterContextInformation”>
29. Use of Fasciocutaneous and Myocutaneous Flaps in Complex Urethral Reconstruction
Keywords
Urethral reconstructionUrethral stricturePerineal artery fasciocutaneous flapGracilis flapGluteus maximus flapUrorectal fistulae29.1 Introduction
The great challenge of urethral reconstructive surgery is to create an adequate caliber lumen with durable success in the presence of complex urethral strictures with associated hostile wound environments. Complex urethral pathology has a variety of phenotypes including strictures with or without associated fistulae, extensive periurethral fibrosis, and segmental urethral loss. Furthermore, it may be associated pelvic fractures, perineal trauma, decubiti, radiation damage and re-operative surgery, which may lead to hypovascular and fixed tissues. These account for the most complex and difficult cases confronted by the reconstructive urologist. While these patients represent a minority of cases in a reconstructive urology practice, knowledge and technical proficiency to utilize a variety of fasciocutaneous, myocutaneous and muscular flaps is critical to successful management. The ability to harvest and utilize a variety of flaps requires detailed understanding of the neurovascular and muscular anatomy of the perineum, scrotum, abdominal wall and lower extremity. Often times flaps are used in combination with oral mucosal grafts (OMG) and skin grafts in single staged and planned multi-staged procedures. Furthermore, as with any surgical practice, proper patient selection for these major procedures is critical. It is important to optimize patients prior to extensive reconstruction, including ensuring adequate diabetes control, smoking cessation, and optimizing nutritional status. This chapter will detail the muscular and neurovascular anatomy of reliable fasciocutaneous, myocutaneous and muscular flaps, surgical decision making, surgical techniques, and outcomes related to the use of these flaps in complex urethral reconstruction.
29.2 Flap Characterization
In order to successfully utilize flaps in reconstructive urologic surgery, it is important to understand the basics including the neurovascular anatomic bases of flap constructs and classification of such flaps. The first flaps used in plastic surgery were what would have been considered random skin flaps, relying on a subdermal vascular plexus [1]. Skin flaps were initially characterized by the way in which they were transferred, its shape and whether the flap was local or distant [1]. Rather than relying on a length to width ratio, Milton determined that the viability of a flap was based on the anatomic basis of its vascular supply and this was furthered by Orticochea who described myocutaneous flaps which would allow survival of a larger skin flap relying on vessels perforating the muscle [1, 2]. Ponten then described the inclusion of deep fascia along with a skin flap, which led to longer survival [3].
There are multiple published schemas to classify the various types of skin flaps used in reconstructive surgery. Cormack and Lamberty organized fasciocutaneous flaps into four types based on the origin of circulation to the fascial plexus [1]. Type A is characterized by multiple fascial perforators that are unnamed, akin to a random skin flap. Type B have a large solitary septocutaneous perforator. Type C flaps are composed of multiple segmental septocutaneous branches, which require inclusion of the main source vessel with the flap. Type D is also based on multiple small perforators but is raised as an osteomyofasciocutaneous flap. The Mathes and Nahai Classification is based on the types of deep fascial perforators; Type A is a direct cutaneous flap, whereby the pedicle travels deep to the fascia and then pierces the fascia to supply the skin, Type B is septocutaneous where the pedicle courses within an intermuscular septum, and Type C is a musculocutaneous flap based on a pedicle within the muscle itself [1]. Lastly, Nakajima et al. classified skin flaps into six groups(I-VI) based on perforators of the deep fascia [1]. An angiosome has a specific source artery supplying a three dimensional area of tissue. Perforators were further classified as direct or indirect, depending on if they traveled through a muscle (indirect) to reach the skin or solely perforated fascia only to reach the skin (direct) [1].
Muscle flaps are versatile and may be used in a variety of scenarios in urologic reconstruction. The Mathes and Nahai classifies muscles into Types I-V based on its vascular supply [1, 4]. Type I has one vascular pedicle; examples include gastrocnemius, rectus femoris and tensor fascia lata. Type II include dominant vascular pedicle(s) and small minor pedicles, this includes the gracilis, biceps femoris, and trapezius muscles. Type III is characterized by two dominant pedicles arising from separate regional arteries, examples include serratus anterior, rectus abdominus, gluteus maximus and semimembranosus. Type IV is classified by multiple pedicles entering the muscle in segmental distribution, which includes the sartorius and tibialis anterior muscles. Finally, Type V flaps have one dominant vascular pedicle and secondary segmental vascular pedicles, for example pectoralis major, latissimus dorsi and internal oblique. Furthermore, Taylor et al. classified muscle flaps by their mode of motor innervations, which is salient for dynamic muscle transfers, but is beyond the scope of urologic reconstruction [1]. Additionally, flaps may also be classified as conjoined, which includes multiple flap territories with a common physical junction with retained independent vascular supply or chimeric flaps, which has multiple flap territories with independent vascular supply without physical connection except that they are linked by a common source artery [1]. When designing a flap there are six characteristics that must be considered: circulation , constituents, construction, conformation, contiguity and conditioning [1].
29.3 Fasciocutaneous and Myocutaneous Flap Application
29.3.1 Scrotal and Perineal Flaps
In contemporary practice, scrotal and perineal skin are certainly not the primary choices for reconstruction of complex urethral strictures and fistulae; however, they may be useful if no alternative tissues are available and the patient and surgeon accept the high risk of adverse effects when utilizing these flaps. In modern practice, scrotal skin fasciocutaneous flaps are rarely used; however, it is part of the reconstructive armamentarium and thus included for completeness.
Scrotal skin , while popular decades ago, has fallen out of favor for urethral reconstruction because of its elasticity that results in formation of a urethral diverticulum. Scrotal skin fasciocutaneous flaps may be utilized in a variety of situations, where the use of oral mucosa is not desired or unavailable, the presence of abnormal penile skin or unreliable dartos for vascular support in the case of prior hypospadias repairs, for repair of urethrocutaneous fistulae and in the salvage setting for strictures or fistulae that have been previously repaired.
A hairless scrotal skin flap or epilated scrotal skin (3 months prior) may be utilized in urethral reconstruction and was first described by Gil-Varnet and Blandy et al. [5–9] Scrotal skin is supplied anteriorly by the deep external pudendal artery and posterolateral from perineal and posterior scrotal arteries originating from the internal pudendal artery [5, 6]. The scrotal fasciocutaneous flap includes the skin, dartos, external spermatic fascia, cremasteric fibers and internal spermatic fascia as described by Gil Vernet. Jordan describes using a lateral scrotal skin based flap whereas Gil Vernet has utilized medial scrotal skin island flaps based on biaxial vasculature and the scrotal septum [5, 6]. These scrotal fasciocutaneous flaps may be conformed in a variety of ways; they may be placed as a skin island with de-epithelialization around the island to be used as dorsal, lateral or ventral onlay [5, 6]. Furthermore, the scrotal flap can also be tubularized to replace a diseased portion of the anterior urethra [5]. The width and length of the fasciocutaneous flap and the size of the skin island can be tailored to the length of the urethral stricture or fistula and the width of the remaining urethral plate. At a mean 39.5 months, Gil-Vernet et al., found an 86% success rate overall, and 81% for tubularized urethroplasty and 80% for panurethral strictures [6]. An important technical consideration when using scrotal fasciocutaneous flap is the precise tailoring of the skin flap to fit the urethrostomy defect and keeping the skin and fascia on stretch when performing the anastomosis to prevent diverticulum formation [5].
29.3.2 Perineal Artery Fasciocutaneous Flap (Singapore Flap)
The perineal artery fasciocuteanous flap is another versatile tissue transfer concept, which can be utilized for reconstruction of the aforementioned demanding complex bulbar and posterior urethral strictures and/or fistulae. This vascularized flap not only allows for urethral reconstruction, but also allows for perineal skin coverage and can fill in soft tissue defects. The flap is also valuable for perineal and thigh reconstruction following Fournier’s Gangrene. The flap has a reliable skin island and is easy to harvest with well-defined borders and associated minimal donor site morbidity. History of radiation for prostate cancer and rectal cancer do not prohibit the use of this flap.
The initial motivation behind the creation of the perineal artery fasciocutaneous flap , was the need for a reliable and sensate flap for vaginal reconstruction for patients with vaginal atresia. The flap was first described by Morton et al. in 1986; however, the neurovascular anatomy and characterization of the flap boundaries were further described by Hagerty et al. 1988 who identified the perineal branch of the internal pudendal artery as the basis of this flap construct [10, 11]. In 1989, Wee and Joseph, two surgeons in Singapore, further defined the boundaries and design of the flap and its neurovascular anatomy [12]. Tzarnas et al., in 1994, were the first to describe the use of the perineal artery fasciocutaneous flap for urethral reconstruction, in a transgender woman with a posterior urethral injury [13]. In 1997, Zinman was the first to publish his experience in using the perineal artery fasciocutaneous flap in male urethral reconstruction [14].
The perineal artery fasciocutaneous flap is an axial fasciocutaneous flap, including the underlying fascia as described by Ponten and would be classified as a Cormack Type B with a single septocutaneous perforator [3]. The flap is a construct of skin, subcutaneous tissues and underlying fascia and can be hinged or used as an island flap with predictable measurements [15]. The fascial support preserves the arborizing vascular network originating from septocutaneous, musculocutaneous or direct cutaneous arteries creating a reliable and predictable vascularized flap [15]. The advantage of the fasciocutaneous flap includes less bulk, ease of flap elevation and are thinner and more pliable, making it useful for urethral reconstruction [15].
29.3.3 Flap Design and Elevation Technique for Onlay Urethroplasty
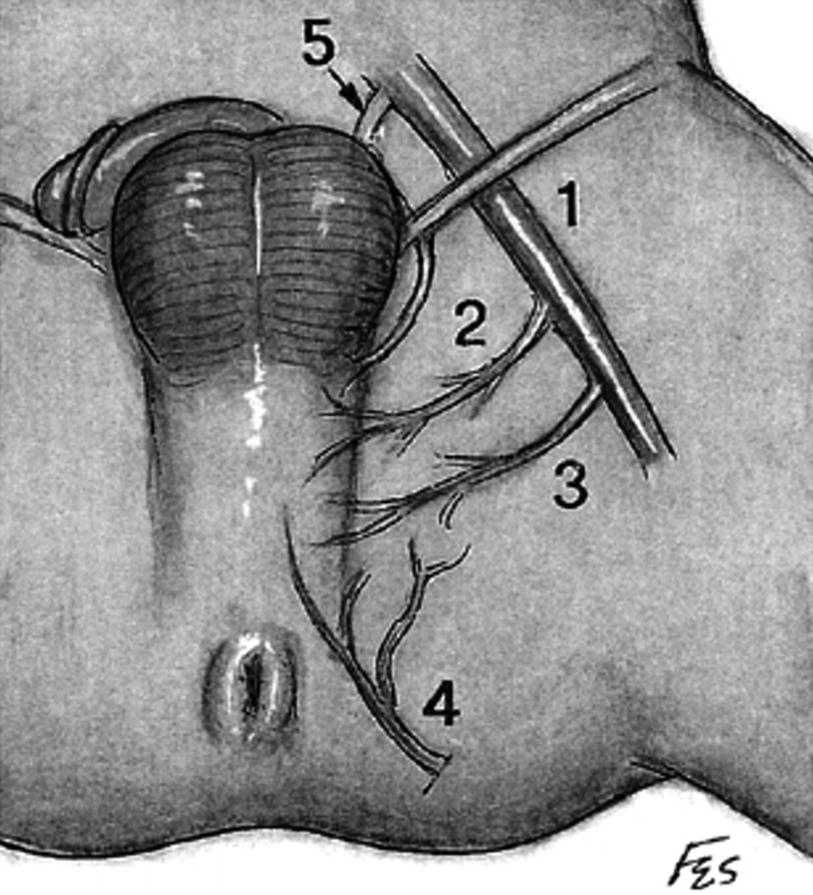
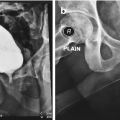
Vascular anatomy supplying perineal artery fasciocutaneous flap. (1) Femoral artery (2) deep external pudendal (3) medial circumflex femoral (4) perineal artery (5) obturator artery (From Zinman [15])
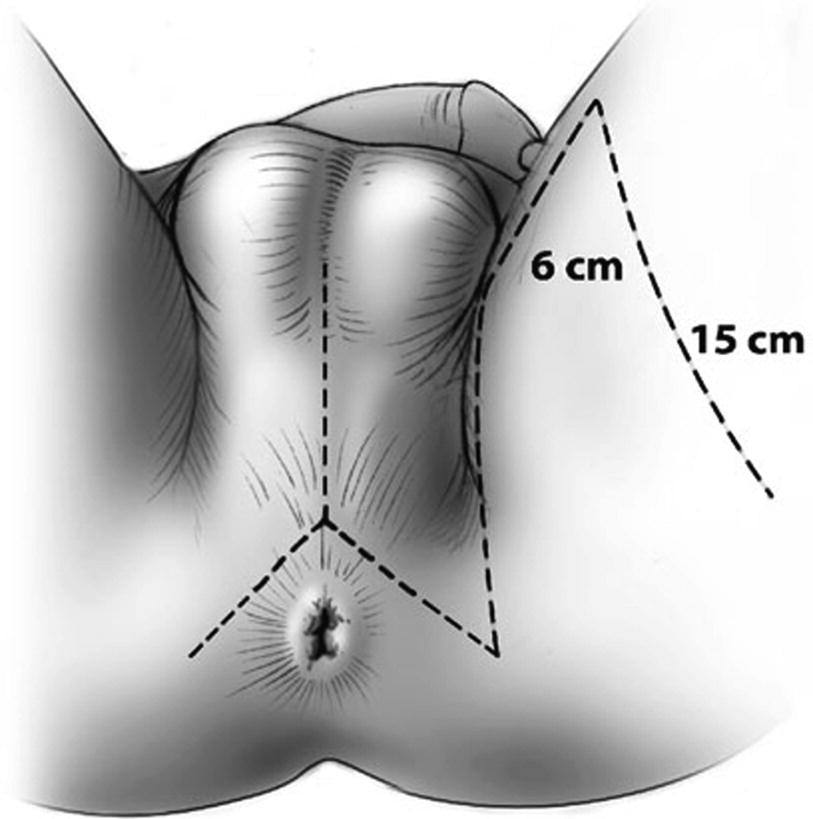
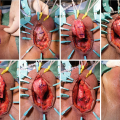
The Singapore flap design and perineal lambda incision (From Zinman LN and Buckley JC [17])
The flap is outlined with the skin on stretch and two parallel incisions are made first to the level of the fascia. The epimysium is raised along with the flap and the fascia is sutured to the dermis to prevent shearing of segmental vessels. The flap is elevated distally, and a distal transverse incision is made and then carried proximally to the base of the flap. Zinman suggests de-epithelializing the very distal 2–3 mm of the flap to confirm a bleeding dermis to ensure adequate vascularization [15, 17]. The bridge of tissue between the flap and the perineal incision is divided to allow rotation of the flap and obviate the need for tunneling the flap.
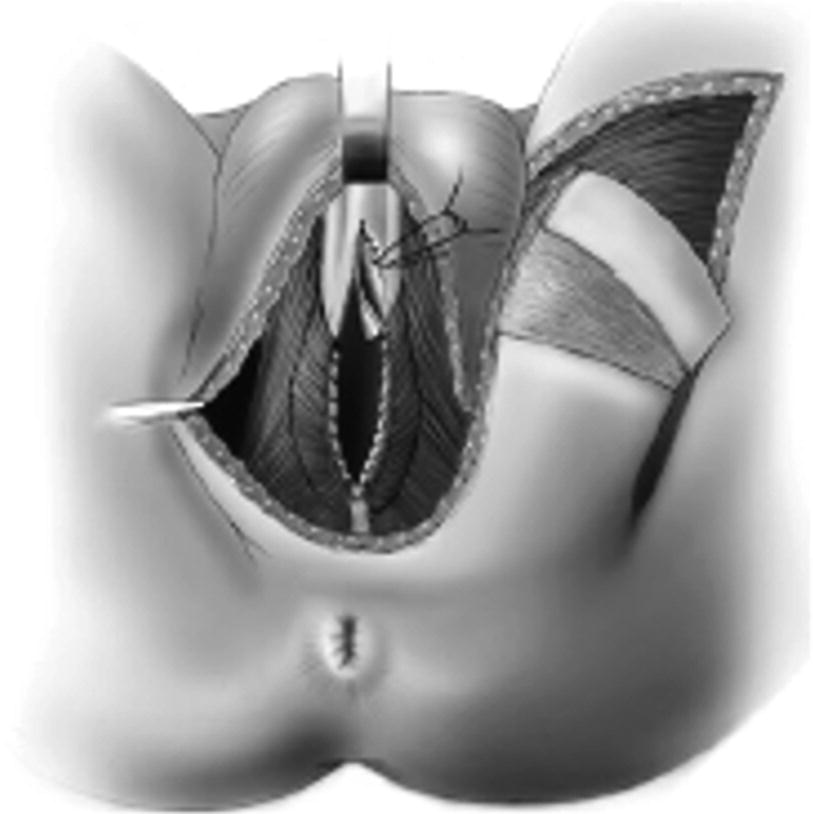
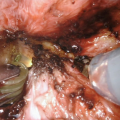
After exposure of the urethra an urethrotomy is shown from the bulbar urethra to the prostatic apex. Here a distally applied BMG is used, although not necessary for strictures 6–8 cm in length, which can be reconstructed with the Singapore flap along. A transverse island flap is outlined distally and a 3 cm wide skin strip is de-epithelialized just proximal to the island leaving a thin layer of dermis intact (From Zinman LN and Buckley JC [17])
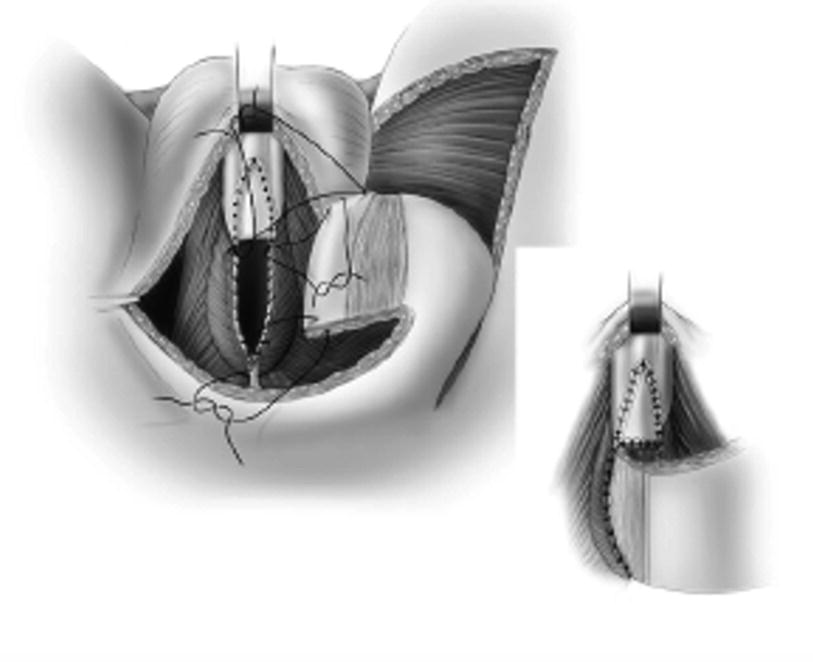
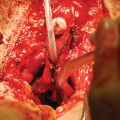
The skin island flap is rotated medially and inferiorly and the skin island is anastomosed to the urethra with running 4-0 absorbable suture. When a longer stricture is encountered and a BMG is used, the flap can be anastomosed to the BMG graft, as shown (From Zinman LN and Buckley JC [17])
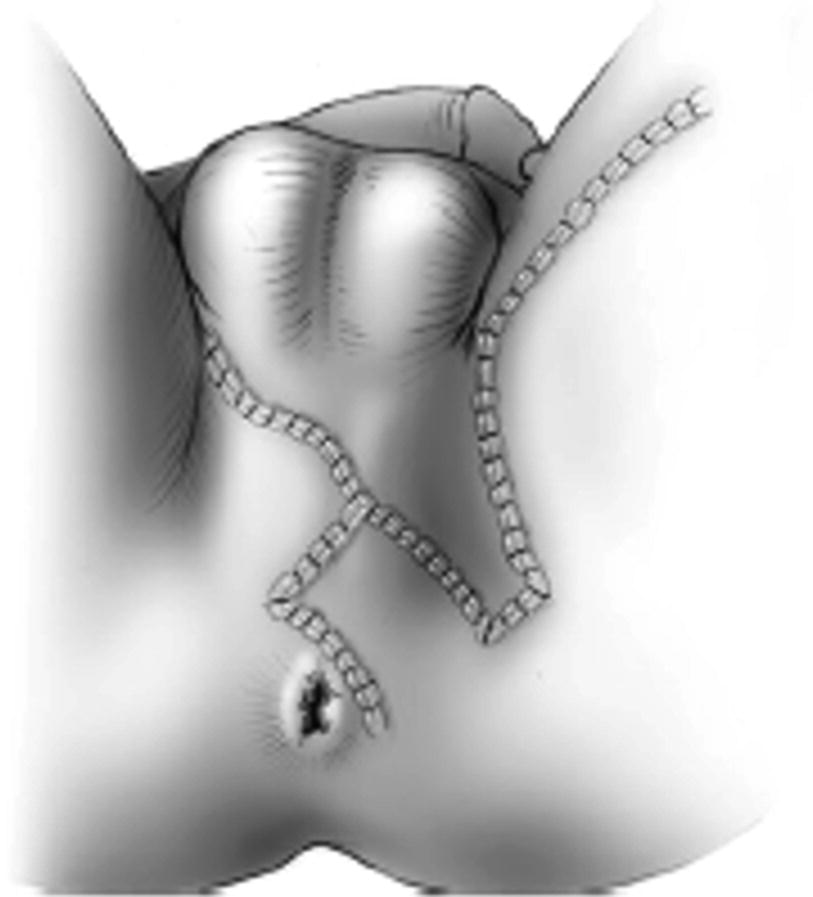
29.4 Alternative Flap Designs for Perineal Wound and Urethral Reconstruction
The Singapore flap is versatile and may be utilized in a number of configurations. The flap can be used for the purpose of covering a perineal wound in the same configuration. Additionally, it may be utilized as a cover during complex urethrocutaneous fistula repair.
Long bulbomembranous urethral defects may occur following pelvic fracture urethral injury (PFUI) that cannot be bridged with progressive maneuvers. Tubularization of the Singapore flap is an option in urethral reconstruction of these long segment defects requiring urethral substitution [14, 15]. This technique may also be useful in cases of bulbar urethral necrosis related to compromised urethral vascularization and prior anastomotic urethroplasty. This flap design may be used as a single stage tubularized flap or multi-stage flap design.
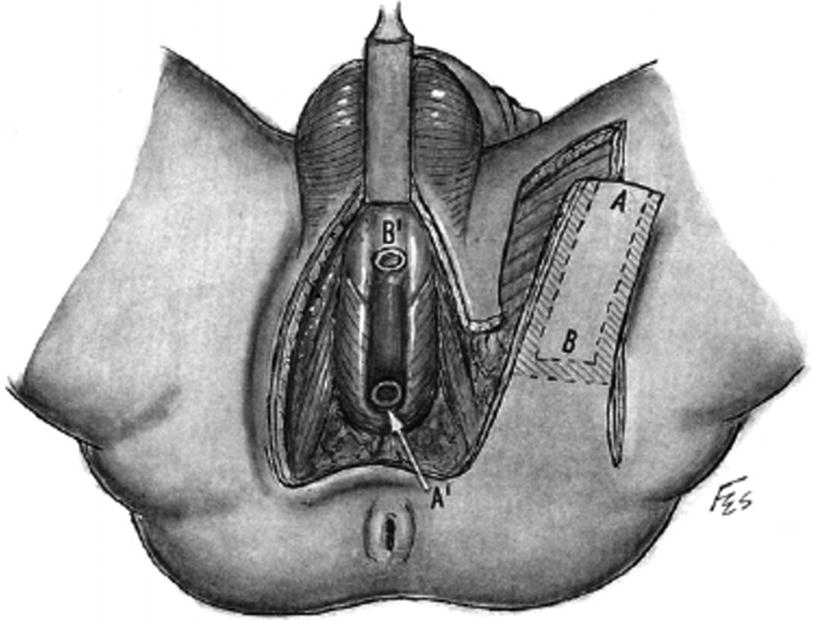
Vertical skin island flap design for tubularized Singapore flap. The distal flap (a) is anastomosed to the proximal urethrotomy and the proximal (b) is anastomosed distally (From Zinman [15])

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree