Key Terms
Red, white, and blue sign
Chancre
Eschar
Pyoderma
Necrotic and ulcerative disorders encompass a wide variety of disease, ranging from infections to autoimmune to vascular conditions. There are many diseases that can present with ulcers, but this chapter discusses only those conditions that present principally, or exclusively, as necrotic lesions or ulcerations.
Important History Questions
How long has the condition been present?
Infections, whether localized (e.g., chancroid, necrotizing fasciitis) or systemic (e.g., ecthyma gangrenosum), necrotic arachnidism (spider bites) tend to have an abrupt onset, whereas some disorders, such as vascular disorders (e.g., stasis ulcers), tend to have a gradual onset.
Do you have any underlying systemic disorders?
Always investigate any predisposing condition. For example, inflammatory bowel disease, rheumatoid arthritis, and lymphoproliferative conditions may be associated with pyoderma gangrenosum. Diabetes mellitus may predispose someone to necrotizing fasciitis, whereas renal disease is a predisposition for calciphylaxis. The use of chemotherapy may predispose an individual to ecthyma gangrenosum.
Is there any history of a clotting disorder?
Some ulcerative conditions are associated with clotting disorders (e.g., stasis ulcers), whereas others are associated with livedoid vasculopathy.
When was your last sexual contact, and did you use a barrier device?
This question is usually reserved for someone with genital ulcers, but it is important in that setting.
Have you had exposure to animals?
This is important in rare cases of anthrax, in addition to milker’s nodules (cows) and orf (sheep).
Important Physical Findings
What is the distribution of the lesions?
Some ulcerating conditions involve certain areas. For example, sexually transmitted diseases usually affect the genitalia, whereas vascular disorders usually affect the lower extremities. Many infectious causes of ulcers, such as orf or milker’s nodules, involve the hands.
How many lesions are present?
Some ulcers are solitary (e.g., syphilis, necrotizing fasciitis, brown recluse spider bite), whereas other ulcers are multiple (e.g., chancroid, pyoderma gangrenosum, ecthyma gangrenosum).
What is the shape of the ulcer?
Some ulcers are round or oval, with a clean base and sharp circumscription (e.g., syphilis chancre), whereas others often demonstrate ragged edges (e.g., chancroid).
How deep does the ulceration extend?
Some ulcers are superficial (e.g., syphilis, herpes progenitalis), whereas others extend into muscle, tendon, or even bone (e.g., pyoderma gangrenosum, calciphylaxis).
What does the base of the ulcer look like?
The base of some ulcers often resembles granulation tissue (e.g., granuloma inguinale), whereas others are usually quite purulent (e.g., chancroid, pyoderma gangrenosum).
Are the peripheral pulses normal and equal in patients with lower extremity ulcers?
Circulatory abnormalities are important to document when considering arterial insufficiency.
 Necrotizing Fasciitis
Necrotizing Fasciitis
ICD10 code M72.6
BACTERIAL INFECTION
Pathogenesis
Necrotizing fasciitis is a life-threatening infection of the skin that involves the deep subcutis and superficial investing fascia. Fournier gangrene represents necrotizing fasciitis of the genitalia and perineum. The current trend is to refer to these conditions, in general, as necrotizing soft tissue infections (NSTIs). Although laypersons may refer to the condition as “flesh-eating Strep,” and some cases are due to Streptococcal spp. and/or a single organism, most cases are polymicrobial in nature. Cases of necrotizing fasciitis have been attributed to a number of organisms, including Streptococcus pyogenes, Staphylococcus aureus, Bacteroides fragilis, and Clostridium, Haemophilus, Vibrio, and Prevotella spp. Other rare bacteria and even fungi may be involved in some cases of necrotizing fasciitis.
Clinical Features
- •
NSTIs occurs in adults and children, with both genders equally affected.
- •
Patients may be otherwise healthy, but risk factors for NSTIs include diabetes mellitus (≈50% of cases), obesity, alcoholism, intravenous (IV) drug abuse, and recent history of prior surgery.
- •
Patients with NSTIs may report a history of trauma (or surgery) at that site, or they may have another infection that serves as the source of the bacteria (e.g., odontogenic abscess, remote skin abscesses).
- •
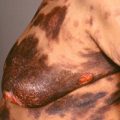
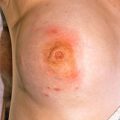
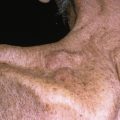
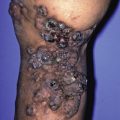
The primary lesion begins as a painful area that looks much like routine cellulitis ( Figs. 14.1 to 14.3 ). The most common sites are the extremities, followed by the trunk and perineum. The pain is often described as being out of proportion to the clinical findings.
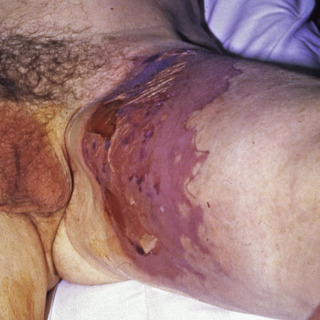
Fig. 14.1
Diabetic patient with Streptococcus -induced necrotizing fasciitis, with epidermal necrosis and hemorrhage.
(From the Fitzsimons Army Medical Center Collection, Aurora, CO.)
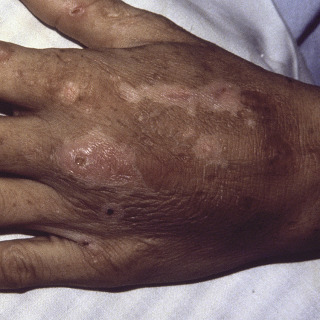
Fig. 14.2
Patient with early necrotizing fasciitis with crepitus due to Bacteroides species.
(From the Fitzsimons Army Medical Center Collection, Aurora, CO.)
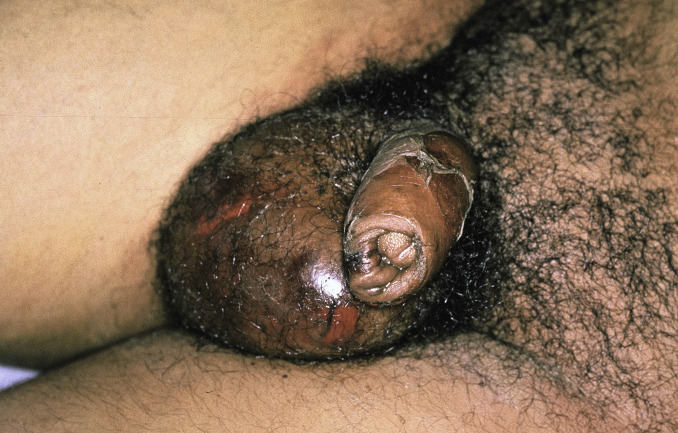
Fig. 14.3
Patient with necrotizing fasciitis of genitalia—Fournier gangrene caused by Escherichia coli .
(From the Fitzsimons Army Medical Center Collection, Aurora, CO.)
- •
More developed lesions may manifest with dusky skin hues, hemorrhagic blisters, hemorrhagic necrosis, and ulceration. Crepitus or focal abscess formation may be present with some species.
Diagnosis
- •
An incision or excision biopsy is preferred that should include a generous amount of subcutaneous fat or soft tissue. A punch biopsy might be useful if done deeply, depending on the clinical circumstances.
- –
Half of the specimen should be sent for bacterial (aerobic and anaerobic) and fungal cultures.
- –
The other half of the specimen can be used for a touch prep with Gram staining if there are facilities and knowledge available for this technique. Typically, there are numerous bacteria; this will provide an immediate indication of the infectious organism(s) likely present.
- –
After doing a touch prep with Gram staining, the rest of the specimen should be submitted for a tradition histopathologic examination and be expedited as much as possible.
- –
- •
Particularly if there is crepitus, consider soft tissue x-rays to see if a gas-forming organism is present (e.g., Clostridium ).
- •
Blood cultures should be performed before initiating antibiotics.
- •
Minimum laboratory studies should include a complete blood count (CBC), chemistry panel—electrolytes, liver function studies, renal function studies—and urinalysis.
Treatment
- •
Start IV antibiotics in the emergency room (or in any other setting in which this is possible). Always admit the patient to the hospital because NSTIs are an emergency (flesh-eating bacteria).
- •
Consult an infectious disease specialist to recommend an empiric antibiotic regimen. If such a consultation is not possible, agents such as ticarcillin-clavulanate, piperacillin-tazobactam, imipenem, or meropenem may be used. IV vancomycin should be added if methicillin-resistant Staphylococcus aureus (MRSA) is a concern.
- •
Immediately call a surgeon because prompt surgical débridement is the treatment of choice. Impress on him or her that this life-threatening situation and time is of the essence.
Clinical Course
In a retrospective study at a major medical center, the mortality rate approached 50% and morbidity was substantial.
Ecthyma Gangrenosum
ICD10 code L08.0
BACTERIAL INFECTION
Pathogenesis
Ecthyma gangrenosum (EG) is an infection consisting of sepsis and localized skin necrosis, with the latter at one or more sites. Classic forms of the disease involve neutropenic patients, with the most frequently implicated bacteria being Pseudomonas aeruginosa, followed by Serratia , Escherichia coli , Klebsiella pneumoniae, and other gram-negative organisms. Some cases of EG are polymicrobial and, in addition to gram-negative bacteria, another gram-positive bacteria may also be involved in the infection.
Pathogenesis
- •
Patients with EG are usually neutropenic, with the total white blood cell count usually less than 250 cells/mcL.
- •
Rarely, patients have a normal white blood cell count, but there may be other contributory diseases (e.g., HIV infection, hypogammaglobinemia, lymphoma).
- •
Signs and symptoms of a systemic infection, including fever, malaise, lassitude, are often present.
- •
EG usually affects the axillary skin, anogenital skin, and skin of the lower extremities.
- •
Single or multiple lesions may be present,
- •
Primary lesions are edematous red macules or patches that rapidly progress to hemorrhagic bullae ( Fig. 14.4 ). Mature lesions demonstrate an eschar ( Fig. 14.5 ) or ulcer ( Fig. 14.6 ), with a rim of erythema.
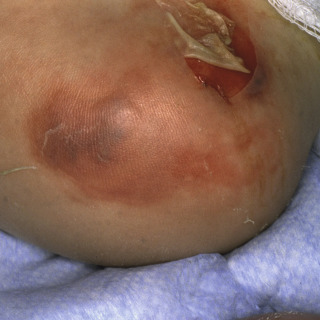
Fig. 14.4
Patient with early ecthyma gangrenosum showing necrosis and hemorrhage.
(From the Fitzsimons Army Medical Center Collection, Aurora, CO.)
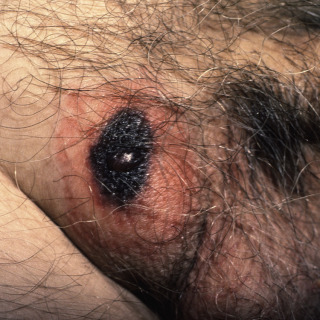
Fig. 14.5
Patient with ecthyma gangrenosum, with black eschar and rim of erythema.
(From the Fitzsimons Army Medical Center Collection, Aurora, CO.)
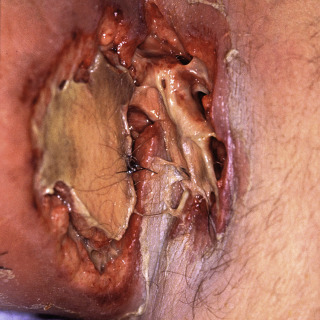
Fig. 14.6
Patient with polymicrobial ulcerated ecthyma gangrenosum.
(From the Fitzsimons Army Medical Center Collection, Aurora, CO.)
Diagnosis
- •
An acute onset of erythematous macules or patches, later with ulceration and hemorrhage, in a patient with neutropenia or another form of immunosuppression, should raise suspicion of EG.
- •
A CBC to determine neutropenia is always indicated.
- •
Gram staining of an aspirate or touch prep will demonstrate numerous bacteria ( Fig. 14.7 ) and provide information as to whether this is a gram-positive, gram-negative, or polymicrobial infection.
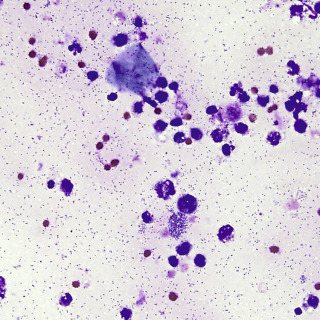
Fig. 14.7
Touch prep from ecthyma gangrenosum showing numerous very short rods of Serratia marcescens (Gram stain: 400×).
- •
A 3- or 4-mm punch biopsy is characteristic, with millions and billions of bacteria typically identified, often with little or no inflammation because the patient is usually neutropenic.
- •
Culture blood, aspirate, and biopsy material.
Treatment
- •
Monotherapy is usually with a antipseudomonal antibiotic, such as ceftazidime, imipenem, or cefepime.
- •
Polytherapy may involve an antipseudomonal aminoglycoside plus an antipseudomonal β-lactamase penicillin (e.g., piperacillin) or ticarcillin-clavulanate potassium.
- •
If an infected catheter is suspected, there are chemotherapy-induced oral ulcers, known gram-positive species are present, or the patient is already on fluoroquinolone, vancomycin is often added.
- •
Correction of the neutropenia or another cause of immunosuppression is useful, if possible.
Clinical Course
EG is a serious infection, with mortality rates in the literature of over 30% and as high as 90% in some series.
Orf
ICD10 code B08.02
VIRAL INFECTION
Pathogenesis
Orf is an uncommon viral infection ( Parapoxvirus, from the family Poxviridae) of the skin that is usually acquired from direct contact with goats or sheep or with fomites in a farm or ranch environment (e.g., fencing, digging in infected soil). Rarely, the infection may also be acquired from deer, musk ox, camel, or pronghorn antelope. A similar but less common viral infection, called milker’s nodule, can be acquired from cattle.
Clinical Features
- •
Orf is acquired through minute breaks in skin and begins 3 to 7 days after inoculation.
- •
Orf usually occurs on acral skin, but any site may be involved.
- •
Orf lesions are usually solitary, but multiple lesions can occur.
- •
Early orf creates round to oval erythematous lesions, with a maculopapular appearance.
- •
Fully developed orf consists of round to oval boggy nodules, with a targetoid appearance ( Figs. 14.8 to 14.10 ). The center is typically eroded and red, with a gray or white middle zone, and with peripheral erythema.
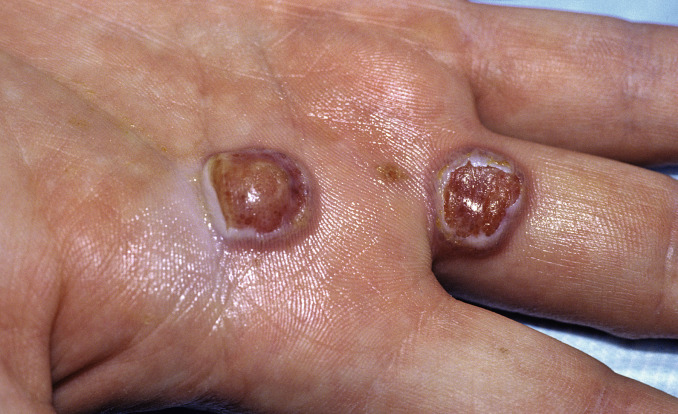
Fig. 14.8
Two superficially ulcerated nodules typical of orf. This was acquired from digging in the backyard of the patient’s home in Colorado that had been built on the site of an old sheep ranch. It is important to realize that the virus can remain dormant in the soil and on fomites for decades.
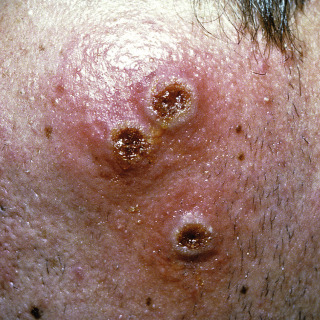
Fig. 14.9
Three ulcerated nodules on the left cheek of a young man from Texas. The source of the infection was never identified.
(From the Fitzsimons Army Medical Center Collection, Aurora, CO.)
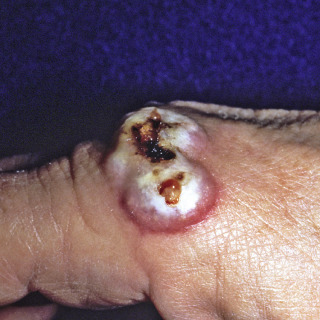
Fig. 14.10
Patient with large linear ulcerated lesion on the dorsum of the hand. The lesion was acquired in Colorado, with no known source.
(From the Fitzsimons Army Medical Center Collection, Aurora, CO.)
- •
Older lesions of orf may demonstrate a verrucous appearance, and resolving lesions may be crusted.
- •
Rare patients may demonstrate lymphangitis or regional lymphadenopathy, or even constitutional symptoms, such as fever, malaise, and chills.
Diagnosis
- •
The presentation of a usually solitary, acral, boggy nodule, perhaps with a targetoid appearance, and in a patient with exposure to farm animals or in an agricultural setting, suggests the diagnosis of orf.
- •
A history of direct contact with sheep, goats, or other more exotic animals or exposure to agricultural settings should be carefully sought because it is thought that the virus may remain dormant for years in soil, wood, or on fomites in an agricultural setting. If there is a history of exposure to cows, then milker’s nodule should be suspected.
- •
A punch or small excision biopsy is usually fairly diagnostic, although it may be difficult to differentiate from a closely related virus (milker’s nodule).
- •
In rare cases, specimens can be submitted for electron microscopy, which is a more specific test. However, this is expensive and rarely done.
- •
Viral culture, complement fixation tests, and immunofluorescent tests are available in research laboratories but are not typically done in routine clinical settings.
Treatment
- •
No treatment is required because this is a self-limited viral infection.
- •
There have been reports of treatment via excision, but this is not recommended.
- •
Topical idoxuridine or topical cidofovir may accelerate healing, although the reports are anecdotal, and controlled randomized trials have not been done.
Clinical Course
Orf is a benign, self-limited disorder that heals without treatment over 4 to 6 weeks. Rare cases result in permanent scarring.
Necrotic Arachnidism
ICD10 code T63.3
ARTHROPOD REACTION
Pathogenesis
The true prevalence of spider bites is unknown, but about 10,000 spider bites are reported to poison control centers in the United States each year. Necrotic arachnidism is the term used for all necrotic spider bites. Loxoscelism is a term reserved for reactions to bites of the brown recluse spider and members of that same genus ( Fig. 14.11 ). Brown recluse spiders are normally found outdoors—under rocks, cliffs, and areas that afford protection—but this spider readily adapts to indoor habitats as well. There are 13 species of Loxosceles , with 5 causing cutaneous loxoscelism ( Loxosceles reclusa , Loxosceles deserta , Loxosceles arizonica , Loxosceles laeta , and Loxosceles rufescens ). The brown recluse is sometimes called the violin spider because of a violin-shaped marking on the dorsum. Brown recluse venom contains at least nine identified protein fractions; the most important to cutaneous reactions is a substance called sphingomyelinase D2.