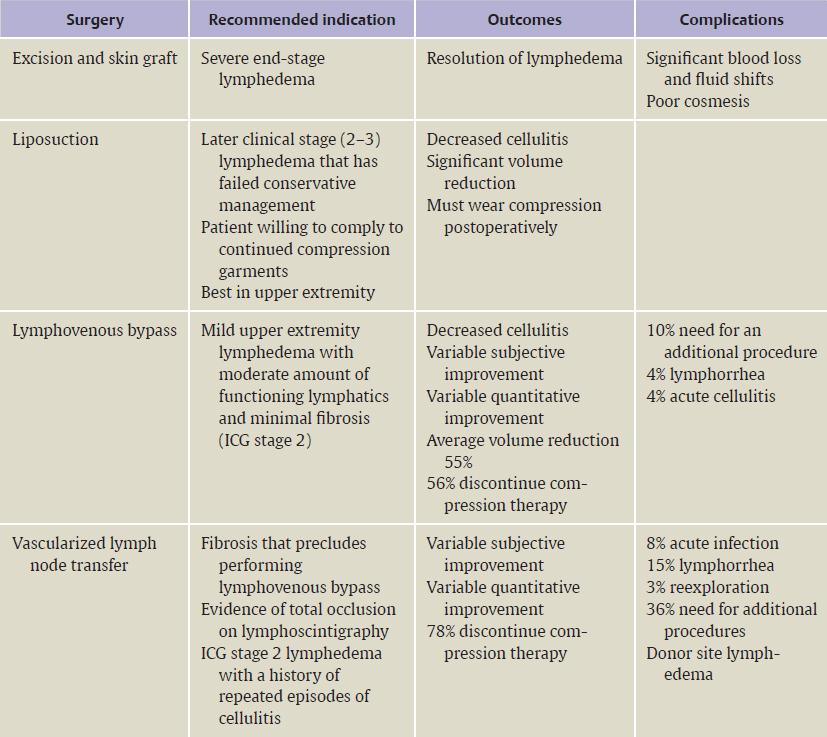
CHAPTER Lymphedema is a chronic and debilitating condition that involves the accumulation of lymphatic fluid, which leads to progressive fibrosis and destruction of lymphatic channels. It can be classified as primary or secondary based on etiology. Primary lymphedema is regarded as genetically determined, with expression occurring at birth (Milroy disease), puberty (lymphedema praecox), or adulthood (lymphedema tarda). In the United States, cancer treatment is recognized as the main cause of secondary lymphedema, whereas worldwide, it is infection with the parasite Wuchereria bancrofti.1 Lymph node dissection for cancer treatment provokes anatomic and physiologic changes. Lymphatic vessels possess vigorous potential for regeneration, and studies have shown that when they are obstructed, a new pathway may be created, which fills the lymphatic gap in the affected region.2 If the new pathway fails to transport enough lymph fluid, lymphedema will slowly manifest. When this occurs, the accumulated high-protein lymph fluid damages the collecting lymphatic vessels that are a mainstay of lymph transport. On electron microscopy, degeneration of the smooth muscle cells in the collecting lymphatic vessels can be seen, and this degeneration appears to progress from proximal to distal after blockage.3 The lymphatic vessels gradually lose their ability to transfer lymph fluid, and this decrease in function corresponds to the stage of lymphedema. Lymphedema may occasionally develop immediately after surgery but most often appears after a latent period. Obesity, postoperative seroma, and radiation represent major risk factors for lymphedema, but the underlying cause of lymphedema is still not fully understood.4 Common symptoms are increased volume and weight of the limb and increased skin tension. The increased volume of the affected upper limb impairs manual dexterity and complicates finding appropriately fitting clothes in addition to affecting patients’ emotional and mental psyche. First-line medical therapies for lymphedema include manual or complete decongestive lymphatic therapy and compression wrapping, usually administered by a certified lymphedema therapist. This mixed therapy includes skin care, compression garments, exercise therapy, and manual lymph drainage. Although conservative therapy is beneficial to some extent in many cases, considerable training is required for patients to master the treatment, and they are expected to continue these rigorous self-care measures for their entire lives.5 When patients fail nonoperative management, surgical treatment can be pursued for the following indications: weight reduction of the affected region, progression prevention, reduction of episodes of infection or inflammation, and improvement of function and cosmesis. Currently, surgical interventions can be broadly categorized into physiological methods and reductive techniques. This chapter focuses on physiologic treatments, including lymph node transfer and lymphatic bypass, which aim to restore lymphatic drainage. Reductive techniques, such as suction-assisted lipectomy (SAL) or direct excision, remove fibrofatty tissue that has been generated as a result of continual lymphatic fluid stasis. With SAL, the patient must always wear a compression garment after the procedure to prevent recurrence of lymphedema. It does not address the cause of lymphedema but treats the sequelae and is often very effective if compression therapy is maintained. In 1912 Charles6 first described the process of direct excision of lymphedematous tissue with subsequent skin grafting for the treatment of scrotal lymphedema. Since then, excisional procedures have been applied to the extremities and have been shown to vastly reduce the size of affected limbs but can have a multitude of complications including destruction of remaining lymphatics, reoccurrence of swelling, aesthetic deformities, and unsightly scars.7 In lymphovenous bypass (LVB), lymphatic fluid is routed back to the venous system to increase the region’s capacity to transport lymph. One of the first attempts at LVB was described by Jacobson8 in the canine model. To treat filariasis, Sedlécek9 used the saphenous vein for end-to-side LVB. However, postoperative concerns over venous hypertension in larger-caliber vessels led to the idea of bypassing venules, which was pioneered by Yamada.10 These procedures can be technically difficult because of the “supermicrosurgery” requirement, which involves anastomoses on vessels smaller than 1 mm. LVB has been shown to be useful primarily in cases of mild lymphedema before the initiation of fibrosis. In 1990 O’Brien et al11 reported long-term follow-up on 90 patients, some of whom received LVB-only and others who had LVB with adjunctive reductive procedures. In the cohort that received LVB-only, subjective improvement occurred in 73% of patients and objective improvement was documented in 42% of patients, with an average 44% reduction in volume. At a 4-year follow-up, 74% of patients were able to discontinue conservative treatment completely, and the incidence of cellulitis decreased by 58%. In a recent report of the senior author’s experience with 100 patients undergoing LVB (89 upper extremity, 11 lower extremity), symptom improvement was reported in 96% of patients, and quantitative improvement was noted by 74%. The overall mean volume differential reduction at 12-month follow-up was 42%. This reduction was significantly larger in patients with earlier stage (stage 1 or 2) versus later stage (stage 3 or 4) lymphedema as determined by indocyanine green (61% versus 17%). LVB alone is best indicated for patients with mild to moderate upper extremity lymphedema who still have some functioning lymphatic vessels and minimal tissue fibrosis. Free tissue transfer of lymph nodes has been the most recent development in the treatment of lymphedema. Autologous or vascularized lymph node transfer (VLNT) was first pioneered successfully in an animal model by Shesol et al in 197912 and used clinically for a patient by Clodius et al in 1982.13 In this procedure, healthy lymph nodes are harvested from one region and transferred to another, either to the original site of injury (i.e., axilla or groin) or within a lymphedematous limb (i.e., dorsum of lower leg). Vascularized lymph node harvest has been described using the groin, thoracic, submental, or supraclavicular nodes. The lymph nodes can be transferred as either a graft or flap. Some have described mincing the donor lymph nodes and transplanting them to the affected region as nonvascularized grafts. This technique, however, has had disappointing results in animal models.14,15 In our practice, we perform vascularized lymph node transfers in which nodes and surrounding fat are kept intact and transplanted with their own venous and arterial supply. This method has been shown to have a greater degree of improvement of lymphedema than nonvascularized grafting. Many theories exist to explain how lymph node transfer augments lymphatic function. One theory postulates that the orthotopically placed lymph nodes act as a sponge or pump to absorb lymphatic fluid and direct lymph into the vascular network.16 Others believe that the transplanted nodes induce lymphangiogenesis that bridges lymphatic pathways.17–19 In general, it has been shown that preserving the vascular supply during transfer results in greater improvement in the degree of lymphedema and in lymphatic function.20 The ideal candidate for lymph node transfer or LVB is one with moderate secondary lymphedema who is compliant with physical therapy and is committed to continued extremity wrapping or wearing a garment postoperatively. During the initial visit, based on a patient’s clinical stage, a recommendation is made to proceed with surgery with the options of LVB, VLNT, or a combination of both. LVB alone is usually reserved for those patients with mild or stage 1 lymphedema. For severe lymphedema, debulking may be necessary (Table 59.1). Preoperatively, our lymphedema therapist evaluates every patient and performs a quantitative volumetric and qualitative analysis of the limb and symptoms. Patients are then also evaluated at 3, 6, and 12 months postoperatively for signs of improvement. We do not typically perform preoperative lymphoscintigraphy studies on patients with secondary lymphedema from a known cause. Patients who present with primary lymphedema always undergo confirmatory lymphoscintigraphy preoperatively to document lymphatic dysfunction. In our experience, patients with primary lymphedema are harder to treat and may not have as favorable outcomes as those patients with secondary lymphedema. This may result from the fact that patients with secondary lymphedema often have scarring and fibrosis that, when released, gives almost immediate relief of symptoms. Also, these patients have a normal lymphatic system but were locally damaged secondarily. Primary lymphedema patients have an abnormal lymphatic system involving lymph nodes or lymphatic vessels or both, and the extent of this abnormality is unclear. High-risk patients are those who are obese with severe stage 4 lymphedema. Overweight patients are encouraged to lose weight so that their preoperative body mass index is at least less than 35. We advocate for patients to be out of bed and ambulating on postoperative day 1, and those patients who are nonambulatory preoperatively are often not considered surgical candidates. Summary Box Recommended Indications, Outcomes, and Complications for Lymphedema Surgery Table 59.1 Surgical recommendations based on clinical stage
59
Microsurgical Treatment of Lymphedema
Reductive Surgery
Physiologic Surgery
Lymphovenous Bypass
Vascularized Lymph Node Transfer
Avoiding Unfavorable Results and Complications in the Treatment of Lymphedema
Preoperative Planning, Counseling, and Patient Selection
Clinical stage | Surgical recommendations |
1 (mild) | LVB alone |
2 (mildmoderate) | VLNT alone or VLNT + LVB |
3 (moderate) | VLNT + LVB |
4 (severe) | Charles procedure Debulking |
Abbreviations: LVB, Lymphovenous bypass; VLNT, vascularized lymph node transfer.
We counsel all patients that surgery does not preclude compliance with limb wrapping and physical therapy, although they may find that lymphatic massage may become more effective postoperatively and their garment size may decrease. If bypasses are performed, all patients leave the operating room with a loose elastic fabric bandage wrapping the operated extremity and are seen immediate postoperatively by a lymphedema therapist for short-stretch bandaging. Short-stretch bandages are worn for 1 month postoperatively and then patients are cleared to wear their own compression garments. Within the first month, LVBs are quite delicate, and the struggle to put on a tight garment may disturb the bypasses; therefore we advocate for atraumatic short-stretch wrapping during this time period. Patients who received only VLNT are cleared to wear their compression garment from postoperative day 1 if it fits appropriately. Patients who undergo LVB or who have extensive scar release may see improvement almost immediately. Patients with lymph node transfer are counseled that it may take up to 1 year to achieve measurable results from surgery.
Intraoperative Considerations
Our preferred lymph node donor site for the surgical treatment of lymphedema is the supraclavicular region.21 The supraclavicular free flap with lymph nodes are harvested with a reliable vascular supply based off of the transverse cervical artery and vein (Fig. 59.1).
For women who are seeking simultaneous breast reconstruction with lymphedema treatment, we harvest lower abdominal flaps (deep inferior epigastric perforator [DIEP] or superficial inferior epigastric artery [SIEA]) with vascularized inguinal lymph nodes, based on the superficial circumflex iliac vessels (Fig. 59.2). For upper extremity lymphedema, the thoracodorsal vessels are usually selected as recipient vessels, thus allowing the positioning of the lymphatic tissue into the axillary space. To treat lower extremity lymphedema, flaps are typically placed in the groin with the femoral vessels or their branches serving as recipient vessels. In certain cases we have transposed flaps to the popliteal or ankle region if lymphedema is limited to these areas.
In the past, a major barrier to LVB was the precise identification of functioning lymphatic vessels in the affected limb. Relying on knowledge of lymphatic anatomy alone is not sufficient for identifying optimal lymphatic vessels for bypass, because these vessels may have been injured during the initial lymph node dissection. Thus we have found that the best way to identify functioning lymphatics is to perform indocyanine green fluorescence lymphography and mapping of vessels just before surgery while in the operating room. Indocyanine green is a water-soluble compound that has been widely used to assess cardiac output, hepatic function, and ophthalmic angiography.22 When it binds to protein in the tissue, it emits energy in the near-infrared region between 750 and 810 nm. After indocyanine green has been absorbed by lymphatic vessels, fluorescence lymphography can detect the near-infrared light emitted by the dye, thereby demonstrating the path of functioning lymphatic vessels. Lymphatic vessels that are damaged do not always uptake indocyanine green, and therefore this method has proven beneficial to identifying functioning lymphatics for bypass targets.
We add 10 mL of sterile water in a 25 mg vial of indocyanine green and then inject intradermally into each finger or toe web of the lymphedematous limb. The extremity is then elevated to enhance uptake of the dye into the lymphatic vessels. Soon after the injections, a Hamamatsu Photodynamic Eye (Hamamatsu Photonics K.K.) is used to visualize fluorescent images of lymphatic vessels, and fluorescent stains are identified proximal to the injection sites. At this point, an objective classification of the severity of the patient’s lymphedema can be made based on indocyanine green lymphangiographic findings22 (Fig. 59.3). As time passes, even after just 10 to 15 minutes, indocyanine green dye refluxes into the superficial and subdermal lymphatics of the limb, creating various patterns of dermal backflow, as described by Yamamoto and Koshima.23 Once this occurs, the linear patterns of functioning lymphatic vessels are often no longer visible, as they are overshadowed by dermal backflow. Therefore we advocate for real-time lymphography at the time of surgery, rather than the day before. With the images as a guide, we use a pen to mark the visible lymphatic pathways and future incision sites for LVBs. Before making each incision, we inject local anesthetic with epinephrine at the incision site to achieve optimal hemostasis. To aid with visually identifying lymphatic vessels during the operation, we use a 30-gauge needle to intradermally inject isosulfan blue dye (Lymphazurin, United States Surgical Corp.) 1 to 2 cm distal to each incision.
Fig. 59.1 The supraclavicular free flap with lymph nodes are harvested with a vascular supply based off the transverse cervical artery and vein.
Fig. 59.2 Lower abdominal flaps (deep inferior epigastric perforator [DIEP] or superficial inferior epigastric artery [SIEA]) with vascularized inguinal lymph nodes based on the superficial circumflex iliac vessels are used for simultaneous breast reconstruction with lymph node transfer.
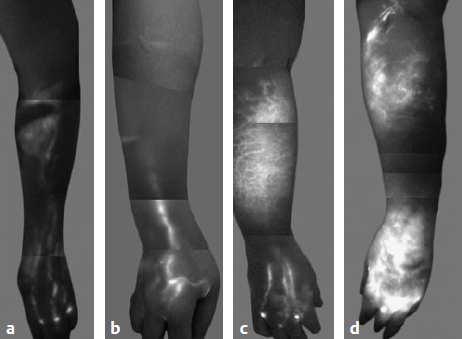
Fig. 59.3 Lymphedema classification based on indocyanine green lymphangiographic findings. (a) Stage 1: many patent lymphatic vessels, with minimal, patchy dermal backflow. (b) Stage 2: moderate number of patent lymphatic vessels, with segmental dermal backflow. (c) Stage 3: few patent lymphatic vessels, with extensive dermal backflow involving the entire arm. (d) Stage 4: no patent lymphatic vessels seen, with severe dermal backflow involving the entire arm and extending to the dorsum of the hand. (Reproduced from Chang DW, Suami H, Skoracki R. A prospective analysis of 100 consecutive lymphovenous bypass cases for treatment of extremity lymphedema. Plast Reconstr Surg 2013;132:1305–1314.)
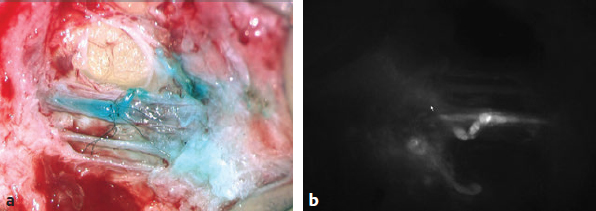
Fig. 59.4 (a) Confirmation of patency of end-to-side lymphatic-to-venous anastomosis with isosulfan blue dye. (b) Confirmation of patency of end-to-side lymphatic-to-venous anastomosis with indocyanine green dye.
The entire operation is performed under a surgical microscope. Using the microscope, 2- to 3-cm incisions are made at the predetermined sites based on lymphography performed earlier. The subdermal region is dissected to identify lymphatic vessels. Lymphatic vessels appear either blue with isosulfan blue dye or clear if no dye was taken up. Once viable lymphatic vessels are identified, they are anastomosed to a similarly sized adjacent recipient venule to create the bypass. End-to-end or end-to-side anastomoses are performed depending on the vessel size match. Some preliminary data suggest end-to-side bypass may result in better outcomes. We use superfine microsurgical instruments for dissection and for creating end-to-end or end-to-side anastomoses with 11–0 or 12–0 nylon sutures with 50-μm needles. Patency of the bypasses is confirmed by observing the indocyanine green (ICGN) pass from the lymphatic vessel through the anastomosis and into the venule under fluorescent views of the microscope (Fig. 59.4).
The use of indocyanine green allows surgeons to avoid unnecessary, “blind” incisions; decreases operative time; and improves outcomes. One of the main challenges of performing LVB in patients before the availability of indocyanine green fluorescence lymphangiography was accurately identifying functioning lymphatic vessels for bypasses. In these patients, we made incisions arbitrarily and conducted random exploration under the microscope to identify the vessels. We conducted an analysis of our own outcomes in 2010 and found that the mean number of bypasses performed in patients who underwent LVB without indocyanine green fluorescence lymphangiography was 3.5; in contrast, the mean number of bypasses performed in patients who underwent LVB with indocyanine green lymphangiography was 5.6.24 From this data we affirmed that indocyanine green fluorescence lymphangiography provides a clear benefit in lymphatic mapping.
Most Common Complications
Although the number of reported cases of secondary lymphedema from harvesting the VLNT is low, it is still the main concern of VLNT. In a study by Viitanen et al,25 a decrease in the lymphatic transport index at the donor leg compared with the contralateral leg after harvesting VLNT from the groin was noted in 6 out of 10 patients. The lymphatic transport index of the donor site limbs ranged from 0. to 12.8, whereas healthy extremities show an index of less than 10. Only two patients had lymphatic transport indices of greater than 10. None of these patients, however, demonstrated any clinical signs of lymphedema such as physical complaints or differences in limb measurement.25 These results were considered unexpected, because it was previously assumed that lymph nodes included in the groin flap were not critical for lower limb lymphatic drainage.
In a retrospective review of 26 patients who underwent VLNT, Vignes et al26 found that 6 patients (4 upper limb, 2 lower limb) had persistent lymphedema in the donor limb at 1 year after surgery. These patients underwent VLNT with donor sites from both the axilla (n = 4) and groin (n = 2). No measurement of clinical symptoms was made, however. Pons et al27 reviewed complications after VLNT at their center, and out of 42 patients, 1 developed swelling in the donor thigh. At 2 years follow-up, the patient showed a 2-cm enlargement in thigh circumference in the donor leg compared with the contralateral one. The percentage increase of the affected thigh was 8.5% (6.2% when considering the entire limb).27
In patients with a history of breast cancer, the most popular VLNT donor site is the inguinal region, because abdominal flaps can be combined with inguinal lymph nodes to simultaneously accomplish delayed autologous breast reconstruction and VLNT. The lymph nodes in the groin have been described as being spread over five regions: central (saphenofemoral junction), superomedial, superolateral, inferomedial, and inferolateral.8 Because nodes that drain the lower limb are located medially and centrally, it is more advisable to harvest the laterally based nodes that drain the suprailiac region.28 The superior row of nodes is supplied by the superficial circumflex iliac artery, whereas the medial column is supplied by branches off the femoral artery. Becker et al17 stressed that the superficial inguinal lymph nodes located along the superficial circumflex iliac vein drain lymph fluid mainly from the abdominal wall and that their procurement did not impair lymph drainage of the lower limb.
To avoid the potential risk of precipitating donor site lymphedema during breast reconstruction, we use reverse mapping to identify the sentinel lymph nodes critical for lower extremity drainage. Preoperatively, a radioactive isotope is injected into the limb, which then travels to the sentinel draining nodes in the groin. An intraoperative gamma probe/Geiger meter counter is used to detect the sentinel inguinal lymph node(s) to avoid removing them with the abdominal flap (Video 59.1). This mapping technique is routinely used by breast surgeons to identify sentinel nodes during axillary node dissections. On the day before or on the morning of surgery, the patient’s donor limbs are injected with Tm99 to facilitate identification of sentinel nodes using gamma probe detection.
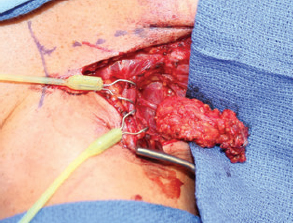
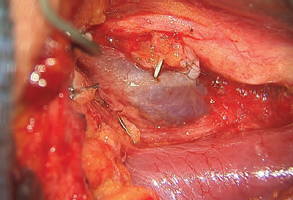
One of the most common problems we have encountered with the supraclavicular lymph node free flap is the development of donor site seroma or lymph leak. To avoid this, a careful surgical dissection with loupe magnification is needed with meticulous clipping of lymphatic vessels. The thoracic duct is often visible within the surgical site and must be preserved. If it is damaged, it needs to be repaired or bypassed to a vein under the microscope (Fig. 59.5). We leave one drain in the donor site, which is pulled when the output is less than 30 mL for 2 consecutive days. If a donor site seroma does develop, we perform serial aspirations in the clinic. Sometimes, in cases of large seromas, the neck incision is opened a few centimeters for drainage followed by a dressing change.
Like any surgical procedure, safe harvesting of this flap requires full knowledge of the anatomy, complete understanding of potential donor site morbidity, and meticulous attention to surgical technique. First, there is a significant anatomic variation of arterial and venous supply in this region, both in terms of the location and the size. The surgeon must be able to adjust the approach accordingly to ensure blood supplies to the lymph nodes are preserved during the dissection. Furthermore, there are many important structures in the region, including the carotid artery, internal jugular vein, phrenic nerve, and thoracic duct (Fig. 59.6). A meticulous surgical approach is required to avoid damaging any key structures and to prevent devastating complications.
The complication of primary concern when harvesting vascularized lymph nodes is causing secondary donor site lymphedema. The supraclavicular lymph nodes mainly drain the breasts, lungs, esophagus, and thyroid. There has been one anecdotal personal communication that upper limb lymphatic drainage to the supraclavicular area has been seen on an injection study. There has been one case report of donor site arm lymphedema after a supraclavicular flap of a same case by two different groups of authors. It is uncertain what type of approach was used in this particular case, but when the photo of the case is carefully examined, the donor site scar appears very different than those we create in terms of the location as well as the extent of the deformity.
The supraclavicular flap is a difficult flap to harvest, because there are many crucial structures in that area and the vascular anatomy can vary. The surgeon must be very familiar with the anatomy in this region. Furthermore, utmost care must be taken with precise surgical technique in harvesting nodes in this area.
Another common complication of surgery is transient numbness at the donor site in the neck or anterior thigh from recipient vessel dissection in the groin. During the dissection, every attempt is made to preserve superficial sensory nerves in the area, although this is not always possible. We have observed that the numbness and neuropathic pain usually resolves within 3 to 6 months after surgery. Patients are managed expectantly, and if pain is bothersome, we have in some instances prescribed gabapentin for symptom relief.
Early in our practice we harvested supraclavicular free flaps with a small skin paddle (Fig. 59.7). However, over time, we found that the perforator to the skin on this flap was not particularly reliable, and we experienced some instances of skin necrosis despite patent anastomoses upon operative exploration. We no longer routinely take a skin paddle with the supraclavicular free flap for lymph node transfer. Instead, flaps are monitored postoperatively with an implantable Doppler placed on the artery. Patients remain in the hospital for 3 days for flap monitoring. If at any point, the Doppler does not function, portending acute anastomotic thrombosis, the patient is taken back to the operating room for flap salvage on an emergent basis. If a patient has severe scarring and requires a large area of skin and subcutaneous tissue to be excised during the recipient site preparation, we use the latissimus dorsi myocutaneous flap with thoracic lymph nodes or lower abdominal flap with groin lymph nodes.