19 Microsurgery Abstract Reconstructive surgery has the ability to offer a reliable and durable solution to patients suffering from conditions, wounds, or defects of increasing complexity. Surgeons can now use microsurgery to transfer tissues of varying compositions to restore anatomy and function with a high rate of success. Moreover, microsurgery allows replantation of amputated segments as well as the successful transplantation of allogeneic tissues. The principles of microsurgery as they relate to tissue handling and suturing have remained relatively unchanged for decades; however, significant improvements have been made in the manufacturing of instruments, sutures, and magnification, which, together with surgical experience and improvements in technique, have contributed to the remarkable rates of success, making microsurgery a safe and highly reproducible technique. The route to becoming a proficient microsurgeon involves commitment, perseverance, and long sessions of laboratory training. After that, maintaining a reasonably steady flow of microsurgical cases during clinical practice is also important to maintain the skills. Despite experience and advances in technique and instruments, failure of a microsurgical anastomosis and subsequent flap loss are still possible; hence close postoperative monitoring is mandatory in order to promptly detect and treat a vascular complication. This chapter describes the fundamentals of microsurgery in relation to training and basic techniques as well as the main aspects of free flap failure. Keywords: flap failure, free tissue transfer, ischemia, microsurgery, thrombosis Microsurgery literally means surgery under the microscope and the term microvascular surgery refers to the suturing of small blood vessels with the aid of magnification. In 1902, Alexis Carrel, a French surgeon, described the technique of triangulation for vascular anastomosis, and 10 years later he was awarded the Nobel Prize in Physiology or Medicine for his outstanding contributions on vascular sutures and the transplantation of organs and blood vessels. In 1921, Nylén was the first to use a monocular operating microscope for eardrum surgery, and soon after Holmgren would use a stereoscopic microscope for otolaryngological procedures. During the early and mid-1960s Jacobson and Katsumura reported the successful anastomosis of vessels of < 1.5 mm in diameter, while Buncke successfully replanted amputated digits and ears in rhesus monkeys and rabbits, respectively. In the clinical setting, in 1963, surgeons in China were able to replant a patient’s hand amputated at the level of the wrist, and 1 year later Malt and McKhann reported two cases of arm replantation. Also in 1964, Nakayama et al used vascularized segments of intestines for esophageal reconstruction, which were attached by anastomosing vessels of 3–4 mm in diameter. Komatsu and Tamai in 1968 performed the first successful digit replantation, and then Cobbett in 1969 reported the first transfer of a hallux to the hand. During the early 1970s the world witnessed the first free flaps in the hands of Antia and Buch, who transferred a superficial epigastric artery skin flap to the face in 1971. McLean and Buncke reconstructed a scalp defect with an omentum free flap in 1972, and Daniel and Taylor, followed closely by O’Brien in 1973, independently reported the use of a free groin flap for lower extremity reconstruction. Microsurgery and free tissue transfer soon became the focus of attraction to numerous surgeons around the world and the technique began to expand rapidly. Although in the beginning microsurgery was performed in a selected number of places and the rates of success hardly reached 80%, today most tertiary units perform free flap surgery on a routine basis, with patency rates 95–98% due to a combination of careful preoperative planning, better instruments and surgical technique, and improved postoperative care. The fact that it is performed routinely and successfully in many institutions worldwide does not imply that microsurgery is a procedure to be taken lightly; rather, it is to be admired as the product of dedicated research, clinical work, and technical advances provided by the pioneering microsurgeons who paved the way to what we know and do today. Learning microsurgery requires both commitment and perseverance. In the authors’ opinion, a 1-week laboratory course with neither cell phone interruptions nor clinical or social obligations is highly recommended to acquire the necessary basic skills. During such time, trainees are usually faced with a number of difficulties that, although irritating and frustrating at first, represent extremely valuable learning opportunities that will someday pay off when similar situations arise during clinical cases. Focusing on the technique and paying careful attention to the details are paramount during the process and should never be shadowed by the feeling of defeat when things go wrong. Much has been said about the best way to prepare for a microsurgical procedure, which requires concentration, fine skills, and the right state of mind. Tobacco may affect concentration and performance for about 30 minutes and is therefore discouraged, whereas sticking to one’s routine dose of coffee is highly recommended—tremor may be exacerbated not only by excess but also by the lack of it. Time spent in the microsurgery laboratory is important and precious, but also limited. To make the most of it, regular breaks of 5–10 minutes every 60–90 minutes must be taken. During these breaks it is important to fully disconnect from the work being done so as to regain focus and concentration. Conversely, working restlessly for several hours will severely influence your judgment and ability to learn as well as significantly reduce your capacity to overcome difficulties in an intelligent and efficient manner. Note Trainees and surgeons wanting to become proficient in microsurgery should do as follows: • Dedicate a great amount of time to lab training. • Learn to deal with frustration and despair. • Not become discouraged by a poor result. • Not struggle under difficulties, but instead learn to identify, accept, and efficiently manage difficult situations. • Learn the anatomy of the working area, including vascular variations. A correct and comfortable posture is an absolute requirement in microsurgery. Good positioning of the feet, body, and hands is important to avoid tremor and remain focused, whereas constant changes and movements usually result in loss of concentration and unnecessary delays, which eventually build up and undermine your surgical skills. A good posture starts with the feet, which should be placed separate at an equal distance from the base of the stool, thus forming a three-point supporting base. Do not cross your legs or put them under the seat. Sit in the middle of the stool; the trunk should align in a way that allows the body to rest in space without effort. The seat must be set at a height that is just right for your eyes to meet the microscope’s eyepieces without the need to flex, extend, or rotate the neck. The ulnar aspect of the elbows, forearms, and hands should rest flat on the table, making sure they are not bearing the weight of a forwardly inclined upper body. This position should allow you to work for a long time under the microscope without fatigue and employing little or no effort in maintaining a good posture. Furthermore, avoiding unnecessary muscle contraction greatly reduces the use of extrinsic hand musculature and helps to reduce tremor. Finally, the instruments should be held between the first three fingers, which will also enhance precision ( Tremor is the rhythmic, purposeless, trembling movement of the hand resulting from the involuntary alternating contraction and relaxation of opposing skeletal muscle groups. There is a myth about surgeons with “steady hands”; everyone has a degree of tremor, although the author (GP-A) agrees that some trainees do show an “unusually stable” hand, which is explained by a higher capacity to control unwanted movement that allows them to perform better. As said earlier, there are a number of measures that help to reduce tremor, including consumption of the usual dose of coffee, a good body posture, proper hand positioning, and correct holding of instruments. After exercise, the resting activity of muscles increases, which is why strong exercises, particularly those involving the upper extremities, are discouraged during the 24 hours prior to a microsurgical procedure. Fig. 19.1 Microinstruments are held using a threedigit grip with the instrument resting between the thumb, index, and middle fingers and the ulnar border of the hand lying flat on the working surface. Nonliving models are an essential part of the microsurgical learning curve because they provide an excellent training opportunity allowing a steady transition on the route from inanimate models to living animals to living patients. Latex gloves, silicone tubes, leaves, petals, artificial vessels, chicken legs, and human vessels obtained from cadaveric dissections or discarded after a surgical operation have all been described and represent valid alternatives. Their use depends mainly on the preference and availability of each laboratory as well as the size of the suture—practicing with 11–0 or 12–0 nylon requires thin, delicate materials due to the small size and fragility of the needles. In our microsurgery laboratory we have designed “Tavi,” a modified stuffed toy with a piece of circular latex glove attached to a plastic ring on its the belly ( The rat is an excellent and relatively inexpensive model to start practicing on live, bleeding vessels. Bearing in mind that the goal is to obtain a patent anastomosis, only a living animal will allow the surgeon to contemplate the smooth and clear pulse of the artery at the end of the procedure. Laboratory rats are easy to obtain, can tolerate prolonged anesthesia times, are clean and pathogen free, and in general very docile animals. Rats weighing around 350 g are ideal because their femoral vessels are approximately 1 mm in diameter (± 0.2 mm), a perfect size to work with. Practicing with the carotid artery and internal jugular vein in the neck is an alternative to the femoral vessels; however, whereas surgical access is not difficult, their proximity to the trachea and heart increases the risk of a breathing problem or major bleeding that may result in the death of the animal. Using the abdominal vessels (aorta and vena cava) requires more advanced microsurgery skills. Despite their larger diameter, these vessels have important branches and are located deep in the retroperitoneal space, which makes their access more challenging, and bleeding is difficult to control. In this sense, it is advisable to progress to these areas once you feel fully comfortable with the femoral vessels. Microsurgery is a technique routinely used in reconstructive surgery and other surgical specialties. Although it has been around for almost 50 years, it is surprising how little the technique itself has evolved. The manufacturing of instruments and sutures has certainly improved over the years, but the surgical principles and actions involved in vessel/nerve suturing have stood the test of time and proven highly efficient and safe, as illustrated by the current > 95% overall success rates. A basic microsurgery set comprises at least six elements: a needleholder, straight adventitia scissors, curved dissecting scissors, jeweller’s forceps, dilator forceps, and microvascular clamps with their accompanying clamp application forceps ( The vessel ends need to be at a distance that allows anastomosis without tension and should rest flat on the surface so that no kinks or twists are seen once suturing is completed. During vessel preparation and throughout the whole procedure it is important to manipulate the vessels gently, always grabbing them by their adventitia and never their full thickness ( Fig. 19.4 The vessels should be grabbed by the adventitia and never full thickness. (Reproduced from Weerda, Reconstructive Facial Plastic Surgery: A Problem Solving Manual, ©2001, Thieme Publishers, New York.) Fig. 19.5 Vessel preparation. (a) Once the clamps have been applied, a straight cut perpendicular to the axis of the vessel is made. (b) The vessels are flushed with heparinized solution or the blood remnants milked out. (c, d) Adventitia is trimmed from the vessel ends by gentle pulling and cutting flush with the vessel edge. (Reproduced from Weerda, Reconstructive Facial Plastic Surgery: A Problem Solving Manual, ©2001, Thieme Publishers, New York.) Several techniques exist for end-to-end vessel anastomosis, including the triangulation technique, one way up technique, continuous technique, and spiral interrupted technique. This chapter describes the triangulation technique; however, it is important to note that irrespective of the technique used, some basic principles must be followed when performing a microvascular anastomosis: • Stitches must be placed at a distance from the edge that is approximately twice the wall’s thickness. • The needle must pass perpendicularly through the whole thickness of the vessel wall, in one pass and avoiding repeated punctures ( • Catching the back wall should be strictly avoided on every stitch and confirmed by direct visualization of the lumen. • Tension of the knot should be sufficient to bring the edges together firmly but without tearing the wall. • The number of stitches should be just enough to obtain a sealed anastomosis without undue trauma from unnecessary stitches. All these measures are directed to minimize injury to the intima, which would otherwise expose the highly thrombogenic subendothelial collagen, likely resulting in thrombus formation at the anastomotic site. Likewise, at the end of the anastomosis the vessels’ end should lie in perfect apposition or be slightly everted, but never inverted, because this exposes the thrombogenic external surface into the lumen. In addition, the lumen should be kept free of blood clots, tissue, and debris at all times throughout the procedure by repeated irrigation with heparinized solution. The triangulation technique starts with two stay sutures, the most important ones placed at 120 degrees from each other on the front wall of the vessel ( Fig. 19.6 Vessel dilation using dilator forceps. (Reproduced from Weerda, Reconstructive Facial Plastic Surgery: A Problem Solving Manual, ©2001, Thieme Publishers, New York.) Fig. 19.7 Sutures are placed perpendicular to the vessel wall and full thickness. (Reproduced from Weerda, Reconstructive Facial Plastic Surgery: A Problem Solving Manual, ©2001, Thieme Publishers, New York.) Caution Remember to always release the vein first followed by the artery. An end-to-side anastomosis is mainly indicated in cases of severe vessel mismatch or when preservation of distal flow is needed (i.e., one vessel leg). Moreover, Godina advocated the use of this type of anastomosis under the observation that, after an arteriotomy, contraction of smooth muscle in the vessel wall tends to open up the space, thus reducing the risk of spasm. However, it is now widely known that the rates of success of an end-to-side anastomosis are no different, and as good as, those of end-to-end ones. The procedure begins by performing an arteriotomy on the recipient vessel that matches the diameter of the donor vessel. Importantly, the adventitia must be completely removed from the site of the arteriotomy before making the cut. First, with the main vessel clamped proximally and distally, a stitch is placed and left long right in the middle of the proposed site. Next, two cuts are made at 45-degree angles in such a way that they meet exactly in the middle, thus creating an oval-shaped opening on the vessel wall ( Fig. 19.8 (a-d) 120-degree suturing technique with back wall hanging down, which helps to prevent its being caught during suturing. (Reproduced with permission from Acland, Sabapathy, Practice Manual for Microvascular Surgery. 3rd edition, ©2008, Indian Society for Surgery of the Hand.) Fig. 19.9 Two or three stitches are placed between the two stay sutures to complete the front wall. (Reproduced from Weerda, Reconstructive Facial Plastic Surgery: A Problem Solving Manual, ©2001, Thieme Publishers, New York.) Fig. 19.10 (a-d) Arteriotomy for end-to-side anastomosis. (Reproduced with permission from Acland, Sabapathy, Practice Manual for Microvascular Surgery. 3rd edition, ©2008, Indian Society for Surgery of the Hand.) Fig. 19.11 End-to-side anastomosis. The procedure begins with one suture on each side, followed by suturing the front wall and finally the back wall. (Reproduced from Weerda, Reconstructive Facial Plastic Surgery: A Problem Solving Manual, ©2001, Thieme Publishers, New York.) Of the different devices developed for microsurgical anastomosis beyond the traditional hand-sewing method, the coupler system has been the most successful. Not surprisingly, this device has been incorporated into the routine practice of most units performing microsurgical reconstructions. The device consists of two rings, each containing six pins, attached to the end of a metallic piece with a turning handle on its back end ( The coupler system works best in pliable vessels with a wall thickness of ≤ 0.5 mm. For this reason, this device has found its main application in venous anastomosis, where, compared to the traditional hand-sewn method, it is four to five times faster, has a shorter learning curve, and yields higher patency rates (e.g., close to 100%). Fig. 19.12 (a, b) During end-to-side anastomosis, stitches are placed radial to the center of the arteriotomy. (Reproduced with permission from Acland, Sabapathy, Practice Manual for Microvascular Surgery. 3rd edition, ©2008, Indian Society for Surgery of the Hand.) A number of techniques have been described to deal with discrepancies in diameter between the donor and recipient vessels. For differences of up to 2:1, careful placement of the sutures farther apart on the larger vessel usually suffices ( The ultimate goal of a microsurgical anastomosis is to obtain a patent unrestricted flow. Different methods to test patency are available, but these should be used judiciously because they imply vessel manipulation and therefore carry a risk of trauma. The least traumatic but least reliable is observation—a patent artery should be seen pulsating distal to the anastomotic site. However, in cases of clotting, a “pulse” may be mistakenly seen due to transmission of the longitudinal wave from the vessel pulsating proximal to the obstructed anastomosis. Besides, this method does not work for veins. In the uplift test, the vessel distal to the anastomosis is gently lifted and as it becomes stretched and occluded, alternating collapse and filling confirm the presence of pulsatile flow. Finally, highly reliable but traumatic is the milking test in which the vessel distal to the anastomosis is gently occluded with forceps and with another forceps blood is “milked” for several millimeters away from the anastomosis. Rapid refilling after releasing the proximal forces confirms patency. Unlike the previous two, this method can be used to assess venous patency ( Fig. 19.14 Size mismatch up to 2:1 can be addressed by careful and proportioned placement of sutures. (Reproduced with permission from Acland, Sabapathy, Practice Manual for Microvascular Surgery. 3rd edition, ©2008, Indian Society for Surgery of the Hand.) Fig. 19.15 Beveling of the small vessel end to overcome size mismatch. (Reproduced with permission from Acland, Sabapathy, Practice Manual for Microvascular Surgery. 3rd edition, ©2008, Indian Society for Surgery of the Hand.) Fig. 19.16 (a-c) The milking test for patency check. (Reproduced from Weerda, Reconstructive Facial Plastic Surgery: A Problem Solving Manual, ©2001, Thieme Publishers, New York.) As Dr. Robert Acland used to say, “the secret of success is preparation.” The procedure begins by making a 3–4 cm incision on the groin area, parallel to the inguinal ligament and reflecting the inguinal fat pad laterally while maintaining its attachment to the epigastric artery ( The primary goal in any microsurgical tissue transfer is vessel permeability and flap survival. Whereas in the early days of microsurgery, patency rates barely reached 80%, today this figure rounds up to 95–98%. Furthermore, the indications for microsurgery have expanded to increasingly complicated cases and scenarios, most of which would have been judiciously denied the possibility of a microsurgical reconstruction in the past. Still, even in the most experienced hands, some flaps do fail. There are a number of biological, physical, and mechanical factors that can affect survival of a microsurgical transfer, which the (micro) surgeon must be aware of in order to anticipate what can go wrong and delineate a salvage plan when needed. Fig. 19.17 Exposure of femoral vessels in the rat. (a) Isolation of the inguinal fat pad leaving it attached to its epigastric pedicle. (b) Vessels are grabbed by their adventitia and not full thickness. (c) The perivascular sheath is incised and dissected with the scissor’s blades parallel to the plane of the vessels. (Reproduced from Weerda, Reconstructive Facial Plastic Surgery: A Problem Solving Manual, ©2001, Thieme Publishers, New York.) The vascular endothelium is a highly dynamic and specialized layer of cells that serve a number of important functions locally and systemically. One of the main tasks of endothelial cells is to maintain blood flow by providing an antithrombotic surface that prevents the spontaneous formation of blood clots. Nevertheless, whenever the endothelial lining is disrupted and the subendothelial elements are exposed to the bloodstream, the coagulation cascade is activated. This chapter provides a summary of the main events and molecules involved in this process.
19.1 Introduction
19.2 Training in Microsurgery
19.2.1 Preconditions in Microsurgery
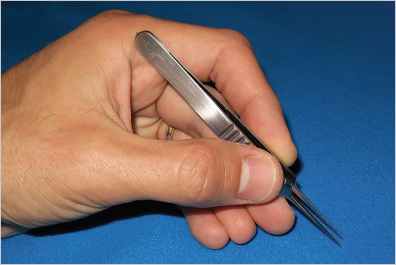
19.2.2 Posture
 Fig. 19.1).
Fig. 19.1).
19.2.3 Tremor
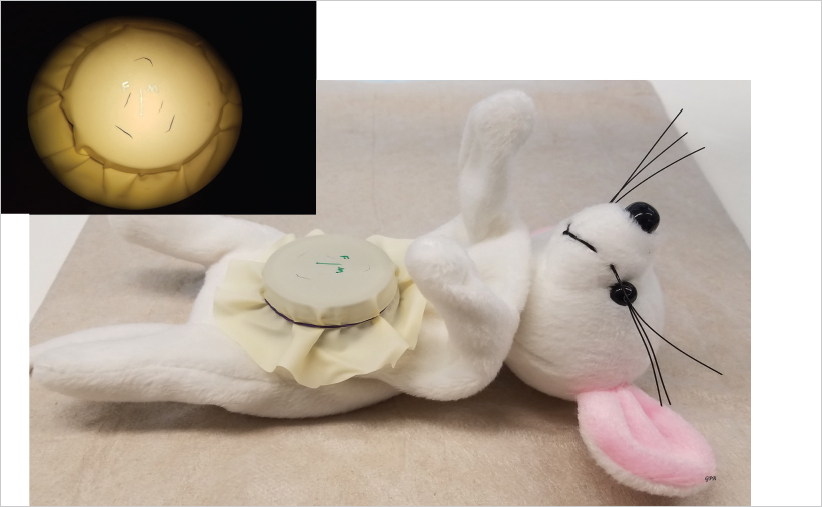
19.2.4 Nonliving Models
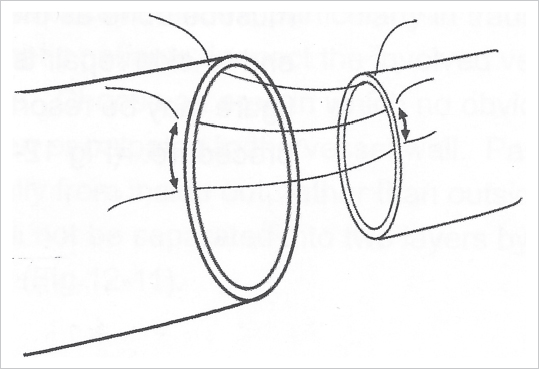
 Fig. 19.2). This model allows suturing in all directions, using both the dominant and the nondominant hand as well as direct visualization of the inner aspect of the suture line by flipping the latex piece. Three to 4 hours of practice on the glove during the first day in a regular 5-day microsurgery course is usually enough to master the use of 9–0, 10–0, and 11–0 nylon suture materials and learn the basics of microsuturing.
Fig. 19.2). This model allows suturing in all directions, using both the dominant and the nondominant hand as well as direct visualization of the inner aspect of the suture line by flipping the latex piece. Three to 4 hours of practice on the glove during the first day in a regular 5-day microsurgery course is usually enough to master the use of 9–0, 10–0, and 11–0 nylon suture materials and learn the basics of microsuturing.
19.2.5 Living Models: The Rat’s Groin
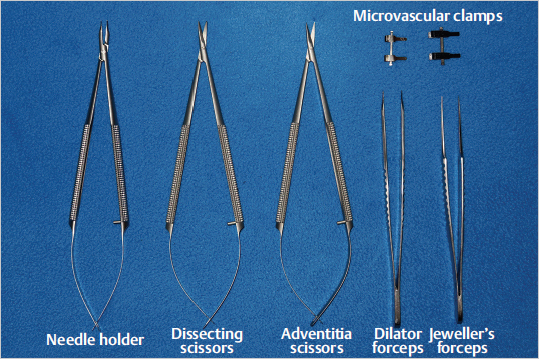
19.2.6 Basic Techniques in Microsurgery
 Fig. 19.3). Today, most units also include in their set of instruments the microvascular coupler system; a device used mainly for vein anastomoses with many advantages and similar patency rates when compared to the traditional hand-sewn anastomosis.
Fig. 19.3). Today, most units also include in their set of instruments the microvascular coupler system; a device used mainly for vein anastomoses with many advantages and similar patency rates when compared to the traditional hand-sewn anastomosis.
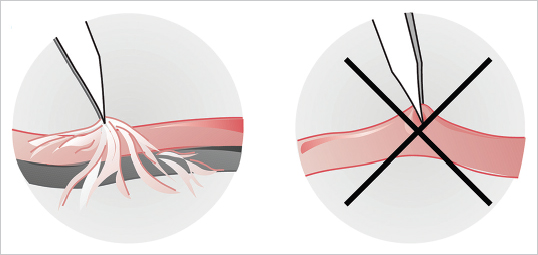
Vessel Preparation
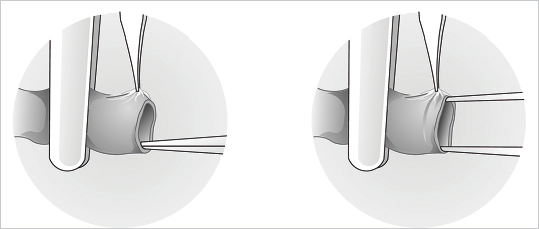
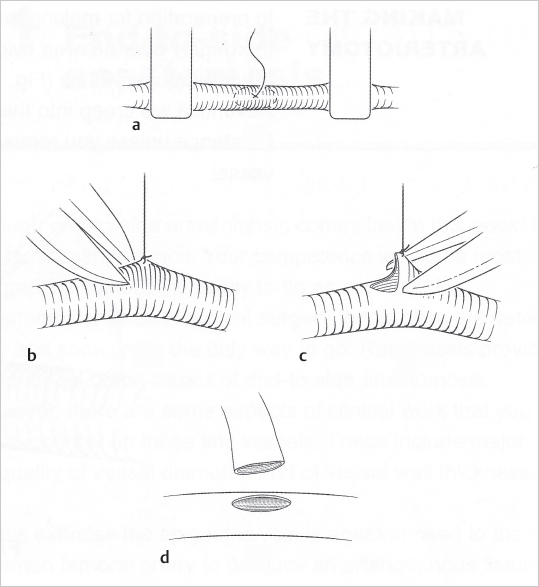
 Fig. 19.4). Clamps (single or double) are applied and the vessels’ ends cut straight and cleanly with straight scissors and irrigated with heparinized lactated Ringer’s solution (10 UI/mL) (
Fig. 19.4). Clamps (single or double) are applied and the vessels’ ends cut straight and cleanly with straight scissors and irrigated with heparinized lactated Ringer’s solution (10 UI/mL) ( Fig. 19.5a, b). The adventitia is then excised just enough to prevent it from getting into the line of anastomosis and is best done by pulling the tissue past the vessel end with forceps and cutting it flush with the vessel edge (
Fig. 19.5a, b). The adventitia is then excised just enough to prevent it from getting into the line of anastomosis and is best done by pulling the tissue past the vessel end with forceps and cutting it flush with the vessel edge ( Fig. 19.5c, d). Next, using dilator forceps, the vessels are carefully dilated. This allows better definition of the lumen and helps in preventing spasm by causing transient paralysis of the smooth muscle of the vessel wall (
Fig. 19.5c, d). Next, using dilator forceps, the vessels are carefully dilated. This allows better definition of the lumen and helps in preventing spasm by causing transient paralysis of the smooth muscle of the vessel wall ( Fig. 19.6). Remnants of blood or tissue inside the lumen are flushed with heparinized solution, and a background contrast material is finally placed under the vessels and clamps. The vessels are now ready for anastomosis.
Fig. 19.6). Remnants of blood or tissue inside the lumen are flushed with heparinized solution, and a background contrast material is finally placed under the vessels and clamps. The vessels are now ready for anastomosis.
End-to-End Anastomosis
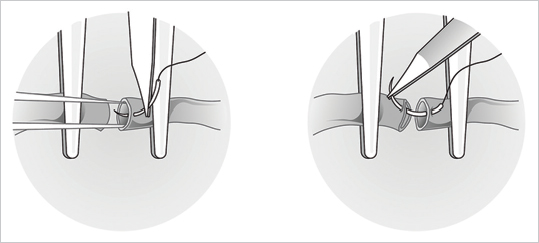
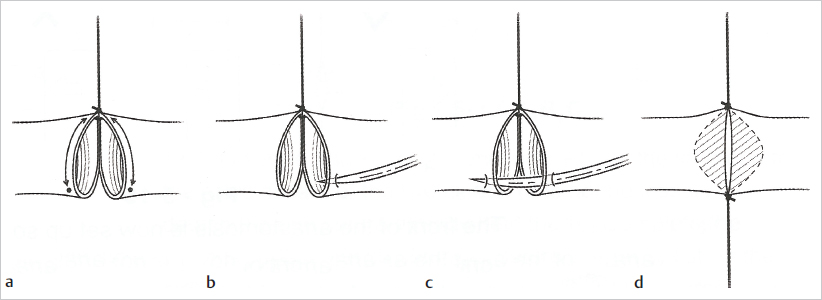
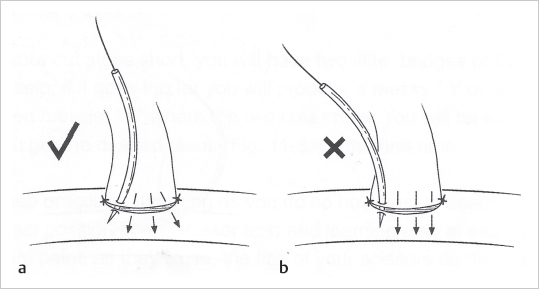
 Fig. 19.7).
Fig. 19.7).
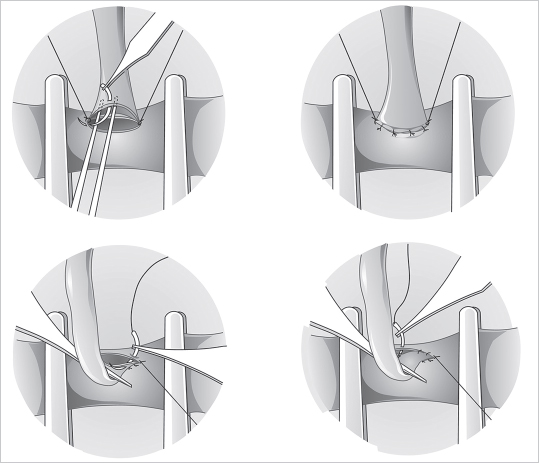
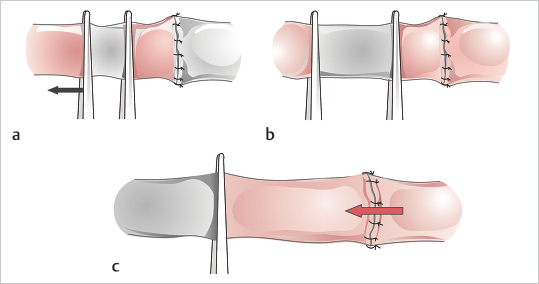
 Fig. 19.8). This allows the back wall to fall away, which minimizes its risk of being caught while one is suturing the front. Next, depending on the diameter of the vessels, two or three stitches are placed between the two stay sutures, and this completes the front wall (
Fig. 19.8). This allows the back wall to fall away, which minimizes its risk of being caught while one is suturing the front. Next, depending on the diameter of the vessels, two or three stitches are placed between the two stay sutures, and this completes the front wall ( Fig. 19.9). The vessel is then turned 180 degrees to address the back wall, which can be sutured in two ways. A stay stitch can be placed in the middle and two or three sutures placed on each side, or, alternatively, the wall can be sutured from one pole to the other.
Fig. 19.9). The vessel is then turned 180 degrees to address the back wall, which can be sutured in two ways. A stay stitch can be placed in the middle and two or three sutures placed on each side, or, alternatively, the wall can be sutured from one pole to the other.

End-to-Side Anastomosis
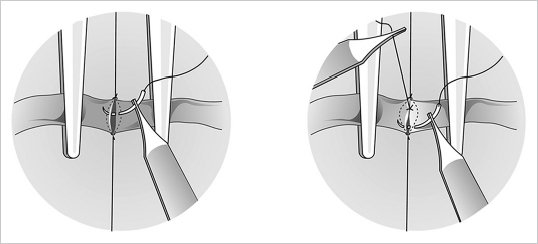
 Fig. 19.10). Alternatively, the arteriotomy can be performed using an arteriotomy set available from various manufacturers (ASSI; S&T), which eases the procedure and increases precision significantly. Next, key sutures are inserted at each extreme of the oval at 180 degrees from each other, followed by suturing of the front and back wall in either an interrupted or a continuous fashion (
Fig. 19.10). Alternatively, the arteriotomy can be performed using an arteriotomy set available from various manufacturers (ASSI; S&T), which eases the procedure and increases precision significantly. Next, key sutures are inserted at each extreme of the oval at 180 degrees from each other, followed by suturing of the front and back wall in either an interrupted or a continuous fashion ( Fig. 19.11). At this stage, it is critical to place the stitches radially in relation to the center of the arteriotomy and not perpendicular to the main vessel (
Fig. 19.11). At this stage, it is critical to place the stitches radially in relation to the center of the arteriotomy and not perpendicular to the main vessel ( Fig. 19.12).
Fig. 19.12).
Microvascular Coupler System
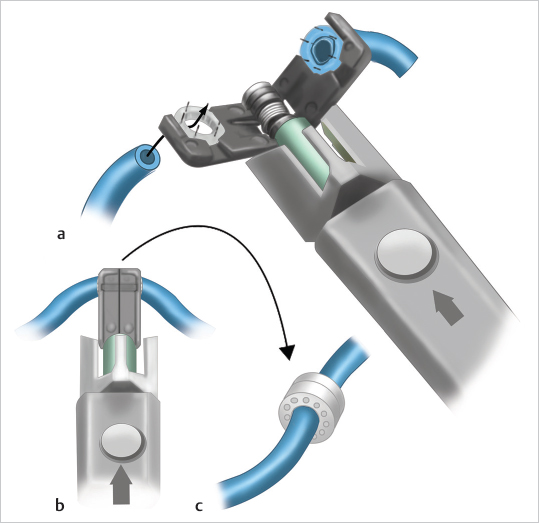
 Fig. 19.13). The rings’ diameter ranges from 1 to 4 mm, including half measures (e.g., 1.5, 2.5, and 3.5 mm), for a total of seven sizes to choose from. Following proper preparation, a “sizer” is placed right next to the vessels in order to select the diameter of the ring. Next each vessel end is passed through the corresponding ring, and the vessel wall is secured to the surrounding pins by gentle eversion and pushing using a specially designed forceps with an opening on its end that allows the passage of the pin. It is advisable to secure the longer or more mobile vessel first (usually the flap’s pedicle) and then the shorter one to allow more freedom of movement when working with the latter. Once both vessels have been “pinned” to their corresponding ring, the handle is turned and the rings are brought together. A hemostat is then used to gently squeeze the rings together, after which the system is released from the anastomotic site by continuous turning of the handle. In cases of size mismatch, the coupler system is able to accommodate a difference of up to 3:1 without the need of any additional maneuvers, always making sure that the selected ring size corresponds to the smaller vessel.
Fig. 19.13). The rings’ diameter ranges from 1 to 4 mm, including half measures (e.g., 1.5, 2.5, and 3.5 mm), for a total of seven sizes to choose from. Following proper preparation, a “sizer” is placed right next to the vessels in order to select the diameter of the ring. Next each vessel end is passed through the corresponding ring, and the vessel wall is secured to the surrounding pins by gentle eversion and pushing using a specially designed forceps with an opening on its end that allows the passage of the pin. It is advisable to secure the longer or more mobile vessel first (usually the flap’s pedicle) and then the shorter one to allow more freedom of movement when working with the latter. Once both vessels have been “pinned” to their corresponding ring, the handle is turned and the rings are brought together. A hemostat is then used to gently squeeze the rings together, after which the system is released from the anastomotic site by continuous turning of the handle. In cases of size mismatch, the coupler system is able to accommodate a difference of up to 3:1 without the need of any additional maneuvers, always making sure that the selected ring size corresponds to the smaller vessel.
Size Mismatch and Patency Tests
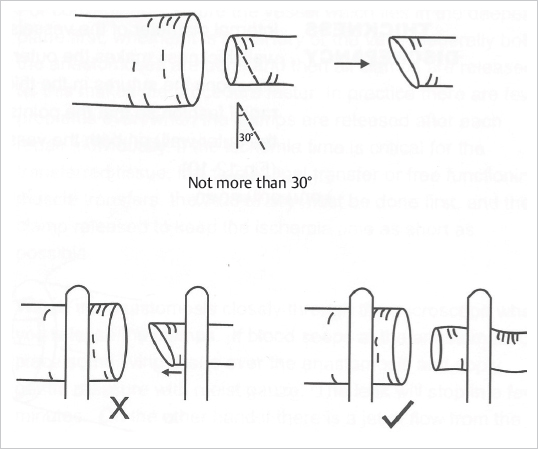
 Fig. 19.14). Higher mismatches require additional maneuvers, such as the fish-mouth technique, beveling or tapering the larger vessel, and end-to-side anastomosis as described earlier. When beveling, an oblique cut of not more than 30 degrees is done at the edge of the smaller vessel, thus increasing the diameter of its opening (
Fig. 19.14). Higher mismatches require additional maneuvers, such as the fish-mouth technique, beveling or tapering the larger vessel, and end-to-side anastomosis as described earlier. When beveling, an oblique cut of not more than 30 degrees is done at the edge of the smaller vessel, thus increasing the diameter of its opening ( Fig. 19.15). Of note, for the anastomosis the beveled end should be placed parallel to the lumen of the other vessel. Tapering implies cutting the excess diameter obliquely and suturing it so that a narrower vessel end is obtained. This technique, however, does inflict more trauma to the vessel by the additional stitches and therefore should be done with utmost care.
Fig. 19.15). Of note, for the anastomosis the beveled end should be placed parallel to the lumen of the other vessel. Tapering implies cutting the excess diameter obliquely and suturing it so that a narrower vessel end is obtained. This technique, however, does inflict more trauma to the vessel by the additional stitches and therefore should be done with utmost care.
 Fig. 19.16).
Fig. 19.16).
Practicing on a Living Model: End-to-End Anastomosis of the Rat’s Femoral Artery
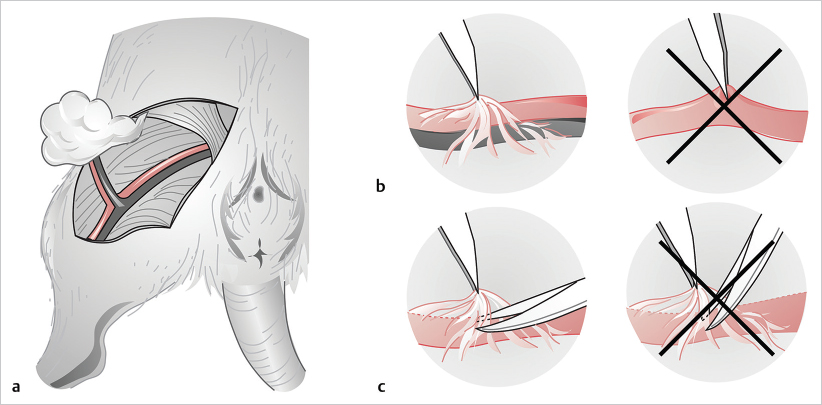
 Fig. 19.17a). The perivascular sheath is then incised with the dissecting (curved) microscissors parallel to the plane of the vessels (
Fig. 19.17a). The perivascular sheath is then incised with the dissecting (curved) microscissors parallel to the plane of the vessels ( Fig. 19.17b, c) and the vessels completely freed by ligation or bipolar coagulation of their side branches. It is important to make sure that enough space is made for the vessel and clamp to turn freely during the procedure. Next, a contrast background material is placed and a double clamp with a frame (Acland’s clamp) brought into the field, and the vessels are prepared as explained earlier, including cutting, flushing, trimming of adventitia, and dilation. The front wall is addressed first, using 10–0 nylon suture, securing the two stay sutures to the suture-holding “cleat” on each side of the clamp, and placing two stitches in between to complete the anterior wall. The clamp is then flipped over to suture the back wall by placing the third stay stitch in the middle and two stitches on each side. Upon completion of the anastomosis, the stay sutures are cut and the clamp turned to its normal position and released, first distally and then proximally. The anastomotic site is covered with gauze, a sponge, or the inguinal fat pad, and gentle pressure is applied for 2 minutes. Finally, the vessel is carefully observed and patency tests performed to confirm permeability.
Fig. 19.17b, c) and the vessels completely freed by ligation or bipolar coagulation of their side branches. It is important to make sure that enough space is made for the vessel and clamp to turn freely during the procedure. Next, a contrast background material is placed and a double clamp with a frame (Acland’s clamp) brought into the field, and the vessels are prepared as explained earlier, including cutting, flushing, trimming of adventitia, and dilation. The front wall is addressed first, using 10–0 nylon suture, securing the two stay sutures to the suture-holding “cleat” on each side of the clamp, and placing two stitches in between to complete the anterior wall. The clamp is then flipped over to suture the back wall by placing the third stay stitch in the middle and two stitches on each side. Upon completion of the anastomosis, the stay sutures are cut and the clamp turned to its normal position and released, first distally and then proximally. The anastomotic site is covered with gauze, a sponge, or the inguinal fat pad, and gentle pressure is applied for 2 minutes. Finally, the vessel is carefully observed and patency tests performed to confirm permeability.
19.3 Basic Science of Free Flap Failure
19.3.1 The Endothelium and the Coagulation Cascade
Plastic Surgery Key
Fastest Plastic Surgery & Dermatology Insight Engine