Melanocytic neoplasms of one type or another are common in the pediatric population presenting as a birthmark or as a later acquired lesion(s). One-third or more of skin biopsies or excisions seen in dermatopathology laboratories from children are melanocytic lesions. Many of these lesions follow a stable course, whereas others are more dynamic in behavior with growth, changes in the clinical appearance, and, in some cases, spontaneous regression, on the basis of a suspected immunologic reaction.
As some measure of the frequency of cutaneous melanocytic lesions in children, we reviewed our experience over a recent 2-year period and found that one-third of all skin biopsies and excisions obtained during the first two decades of life were melanocytic proliferations.
1 Verruca vulgaris and squamous-lined cysts together comprised almost 20% of cases in the same age group and time period so that pediatric and dermatopathologists alike are more than familiar with these lesions in their practice.
Melanocytic nevus is a generic designation for a broad category of lesions that can be characterized clinically and pathologically into a number of subtypes in both children and adults, and several of these are seen predominantly in children and adolescents and are clustered in children 10 years old or less.
It is accepted today, though not consistently in the past, that the melanocyte is one of many derivatives of the neural crest (NC).
2 Progenitor melanocytes are one of the few NC-derived cells that arise from the four major axial designated sites: cranial, vagal, truncal, and lumbosacral; NC cells behave in many respects like stem cells. As NC cells initiate the process of migration, they undergo an epithelial to mesenchymal transition with the development of various differentiated tissue types.
3 Through a study of human embryos, Holbrook and associates determined that proto-melanocytes begin their migration to the epidermis at estimated gestational age of 40 to 50 days.
4 Melanocytes are also present in the mucosal basal layer in sites from the oral cavity, nasal mucosa, and genitourinary tract. It is thought that some melanoblasts fail to arrive at their epithelial destination as a defect in migration to explain some of the uncommon and unusual sites for melanocytic nevi and melanomas.
Given the commonality of the melanocytic nevus, there remain many unanswered questions despite the rather thorough evaluation of these lesions from the molecular genetic perspective.
5 Three competing hypotheses on the histogenesis of melanocytic or nevocellular nevus have been proposed over the last 100 years and still maintain an element of currency in the literature.
6 The oldest of these is the so-called “trickling down” hypothesis of Unna, which postulated that nevi arise from melanocytes in the dermoepidermal junction from a junctional nevus that migrates vertically into the dermis as the intermediate compound nevus and finally as a dermal nevus (DN) with the arrest of junctional proliferation, but Unna thought that the melanocyte arose in the epidermis.
6 It was well into the mid-twentieth century that the origin of the melanocyte remained a puzzle as articulated by Masson
7: “Whatever their origin, schwannian or melanoblasts, all nevus cells share the properties, which are seen only as characteristics of the schwannian syncytium.” It is as though Masson was anticipating the histogenetic relationship of these two cell types as derivatives of the
NC. Later Cramer,
8 acknowledging the NC origin of the melanocyte, also invoked peripheral nerve elements, especially the perineurial cell in an analogous fashion to Masson’s hypothesis, in reference to the histogenesis of the congenital melanocytic nevus (CMN). The plasticity of melanocytes and Schwann cells has been discussed by others.
9 Migration of melanocytes arrested in the dermis explains the family of blue nevi (BN) and dermal melanocytosis (DM). Possibly the pure dermal melanoma is another clinical expression of arrested migration of progenitor preneoplastic cells.
10
ATYPICAL MELANOCYTIC NEVUS
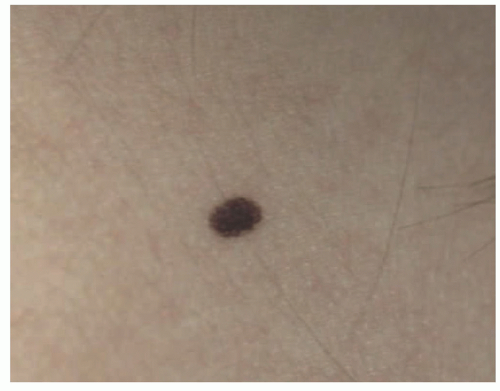
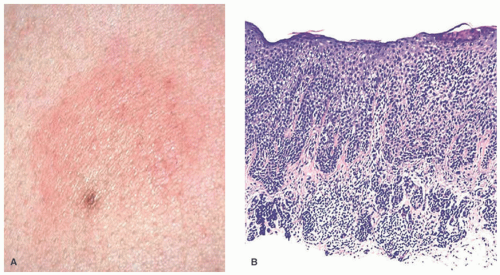
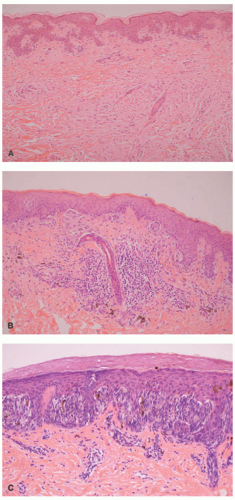
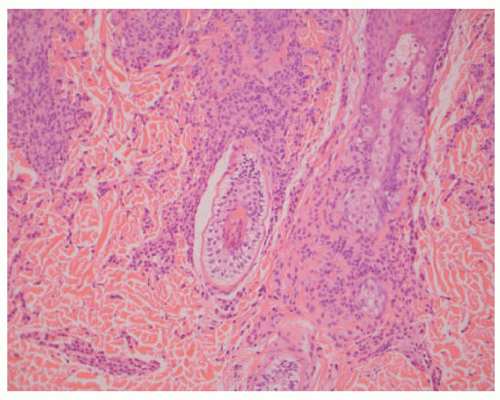
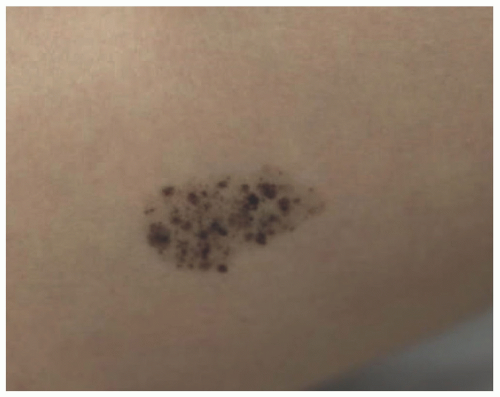
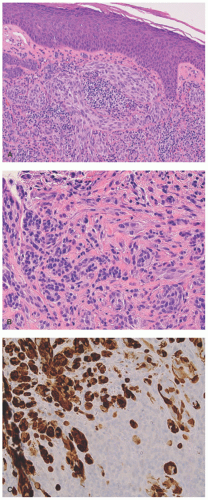
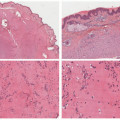
Atypical melanocytic nevus is a generic category based in part upon the clinical appearance and the pathologic features of the nevocellular proliferation. An irregular distribution of junctional nests, lentiginous (single-cell) junctional pattern, and nuclear alterations such as enlargement, density and coarseness of the chromatin, prominence of the nucleoli and mitotic figures, and their location are some of the qualifying histologic features of an “atypical nevus.” In a sense, it does not imply a specific subtype of nevus because these findings occur across the broad spectrum of lesions. Less than 10% of atypical nevi are diagnosed in the first two decades of life in one epidemiologic study.
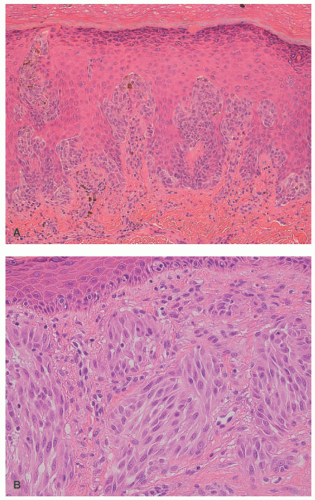
32 The most common context of an atypical melanocytic nevus in our experience is the so-called dysplastic or architecturally disordered nevus whose basic morphologic features include the following: (1) architecture with bridging of rete ridges; (2) nested nevus cells at the tips of the rete and single or lentiginous cells along the rete ridges; (3) continuous lamellar pattern of fibroplasia outlining the superficial papillary dermis; and (4) a shoulder of junctional nevus cells without an underlying
dermal component (
Figure 22-9).
33 The preference of one of us (LPD) is “melanocytic nevus with architectural disorder,” rather than dysplastic or Clark’s nevus. Despite the efforts of several convened consensus conferences, there is still a lack of consensus on many aspects of these lesions including the grading of atypia, its significance, and management.
34,
35,
36,
37,
38 Socalled dysplastic features are not confined to the common AMN, but are seen in other nevocellular lesions such as the SN and combined nevus.
39,
40,
41Another category of AMNs with atypical features includes those from the so-called anatomic special sites such as the scalp, genital area, hand/foot, flexural sites, ear, breast, and lower extremity.
42,
43,
44 There is overlap in the histopathologic features of the special site AMN and the architecturally disordered or dysplastic nevus. AMNs can also be seen in association with pregnancy which particularly have a higher number of mitotic figures and in association with lichen sclerosus.
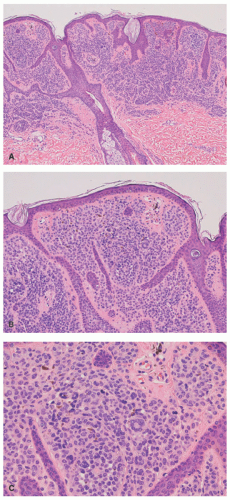
Scalp Nevus
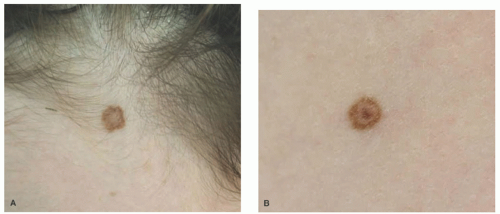
Scalp nevus presenting in children and adolescents may herald the development of additional AMNs on the vertex and parietal scalp and elsewhere.
45,
46,
47 It has been noted that scalp nevi in children are accompanied by an increased number of nevi elsewhere than in children without scalp lesions.
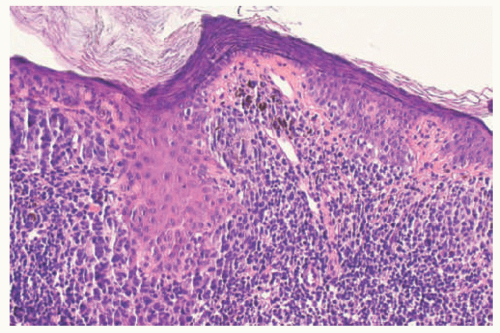
48 The scalp nevus is often elliptical, is variably pigmented, and may have irregular borders. The presence of large, atypical nevus cells, dusty cytoplasmic melanin, and large, irregular nests, and lentiginous proliferation along and between rete ridges are some of the atypical features.
49 Pagetoid spread of nevus cells and dyscohesion of individual nevus cells within the nests are additional features.
50
Genital AMN
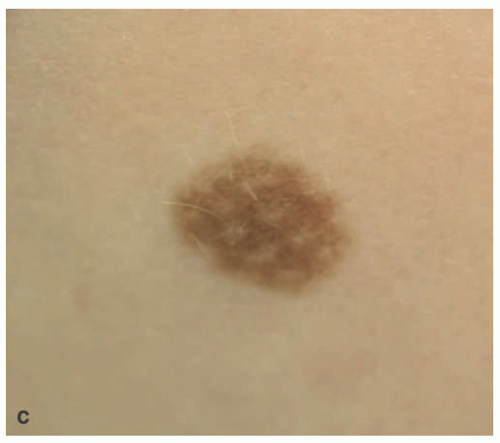
Genital AMNs were detected in 35% of children in the experience of one pediatric dermatology practice.
51 These lesions are generally recognized at or before 5 years of age and even at or shortly after birth. A medium to dark brown symmetrical nevus(s) on the labia majora and minora or penile shaft or scrotum is the clinical appearance. Like the scalp
nevus, the genital nevus has a compound pattern with architecturally disordered or dysplastic features.
52,
53
Acral Nevocellular Lesions
Acral nevocellular lesions include the acral nevus of acquired or congenital types and longitudinal melanonychia. One cohort study of individuals 18 years of age and older noted acral pigmentary lesions in 30% of cases.
54 Skin-of-color individuals are more likely to have acral melanocytic lesions, but when seen in non-Hispanic fair-skinned children, there is also an increased overall count of melanocytic nevi. A study of school-aged children in Colombia revealed 42% had at least one acral melanocytic lesion.
55 Kim et al
56 reported that the most common site of an acral lesion in children was the forefoot of the sole followed by the volar toe, together accounting for almost 85% of cases, whereas approximately 15% of pigmented lesions were found on the volar palm and finger.
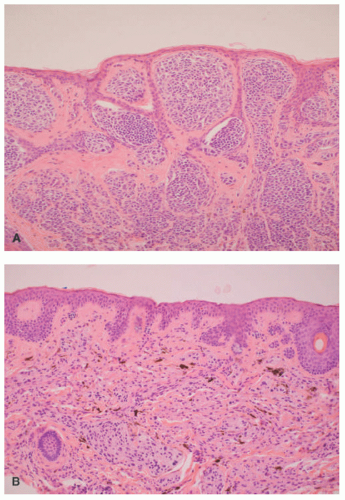
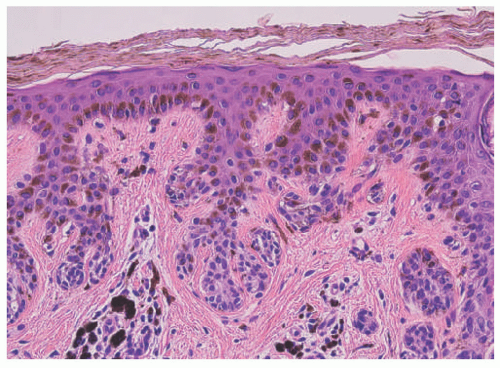
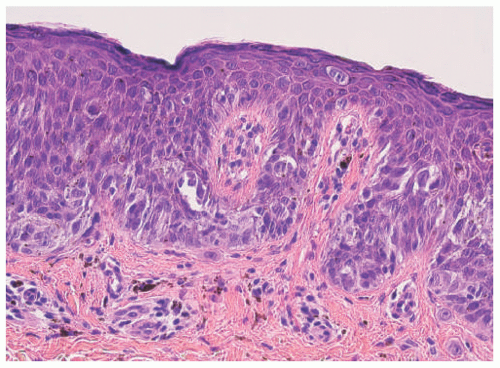
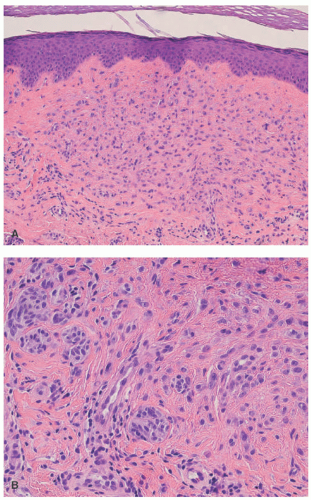
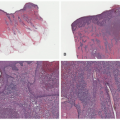
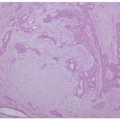
Histopathologically, virtually all cutaneous acral lesions have architecturally disordered features and 60% to 65% are junctional proliferations and 25% to 30% are compound nevi with mixed lentiginous and nested patterns (
Figure 22-10).
57 Cytologic atypia is generally mild in most cases. Pagetoid spread through the epidermis and transepidermal elimination of nevus nests are other findings. SN and BN are also present in acral sites.
58 Eruptive nevi are reported on the sole and/or palm in children after chemotherapy, but their histopathologic features are not unique from other acral nevi in children. In some cases, the pagetoid spreading can be very prominent, particularly in young patients. The term
MANIAC nevus (melanocytic acral nevus with intraepidermal ascent of cells) has been used to described such findings.
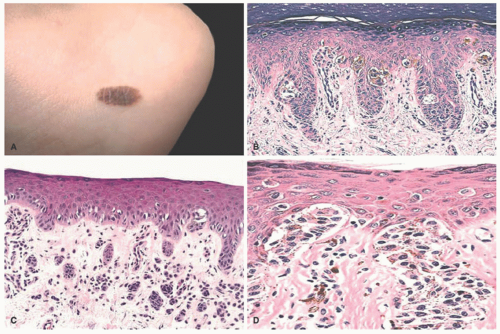
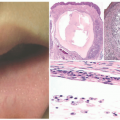
Longitudinal Melanonychia (Melanonychia Striata)
Longitudinal melanonychia, like the cutaneous acral nevus, occurs more commonly in skin-of-color than fair-skinned children. The finger(s), especially the thumb, is the most common site of the longitudinal pigmented band in children and rarely in neonates.
59,
60 Melanocytic activation with increased pigmentation in the basal-parabasal layer without an increased number of melanocytes, melanocytic hyperplasia with an increase in the number of melanocytes, and a disordered junctional melanocytic nevus are the range of histologic findings.
59,
61 Subungual melanoma is rarely reported in children, but apparent progression of longitudinal melanonychia discovered in childhood to a melanoma in adults has been reported.
62,
63