21 Local flaps in partial breast reconstruction
Synopsis
 Breast conserving therapy (BCT) is applied to most early stage of breast cancers.
Breast conserving therapy (BCT) is applied to most early stage of breast cancers.
 Oncoplastic surgery within multidisciplinary approach is “gold standard” in BCT.
Oncoplastic surgery within multidisciplinary approach is “gold standard” in BCT.
 Using pedicled flaps is indicated in high tumor/breast size ratio.
Using pedicled flaps is indicated in high tumor/breast size ratio.
 Partial breast reconstruction with pedicled flap is alternative to mastectomy in selected patients.
Partial breast reconstruction with pedicled flap is alternative to mastectomy in selected patients.
 Overcorrection is essential when pedicled flap is used in partial breast reconstruction.
Overcorrection is essential when pedicled flap is used in partial breast reconstruction.
 Perforator or muscle sparing latissimus dorsi flaps are the first choice of flap surgical techniques.
Perforator or muscle sparing latissimus dorsi flaps are the first choice of flap surgical techniques.
 Fat grafting and contralateral breast remodeling may be required to achieve breast symmetry at long-term follow-up.
Fat grafting and contralateral breast remodeling may be required to achieve breast symmetry at long-term follow-up.
History
Partial mastectomy includes quadrectomy and lumpectomy. In quadrectomy, a wide excision is performed with an additional 1–2 cm or even more of healthy parenchyma around the tumor, including the overlying skin and underlying muscle fascia. In lumpectomy, the goal is tumor excision with clear surgical margins. The 5-year incidence of breast tumor recurrence was shown to be higher in patients treated by lumpectomy and radiation compared to quadrectomy and radiation patients (8.1% vs 3.1%).1 Excision volumes <60 cm3 have been shown to increase the relative risk of local recurrence in women under 45 years.2
The combination of a quadrectomy or lumpectomy with partial breast reconstruction, so-called “oncoplastic surgery”, is considered a determinant in the advancement of breast cancer surgery today. Oncoplastic surgery was introduced in 1993 and focuses on tumor resection and breast aesthetic restoration through partial breast reconstruction techniques that minimize potential breast deformities.3 The term oncoplastic surgery has been used differently, depending on the specialty in which it is being referred.4 In plastic surgery, it is used in order to define a large partial mastectomy, combined with a volume-replacement technique, with the use of local or distant flaps.
Basic science
Partial mastectomy combined with radiotherapy, often referred to as breast-conserving therapy (BCT) followed by breast irradiation, has replaced modified radical mastectomy as the preferred treatment for early stage invasive breast cancer. The 5-year survival rate of partial mastectomy with radiation is not statistically different when compared with mastectomy alone in patients with stage I or II breast cancer.5,6 Women diagnosed at early stages of invasive breast cancer have equivalent outcomes when they are treated with lumpectomy and radiation therapy or modified radical mastectomy.7
It is interesting to point out that the surgical clearance of pathological margins has the most significant impact upon local recurrence in patients treated with BCT and RT. The presence of DCIS at the surgical margin is associated with the identification of residual DCIS in 40–82% of re-excised specimens and is correlated with margin widths of: 41% at 1 mm, 31% at 1–2 mm, and 0% with 2 mm of clearance.5 A recent meta-analysis concluded that a margin width was significantly superior to lesser margins.8
The incidence of local recurrence depends upon certain factors, such as the tumor margin, nuclear grade-histology, radiation therapy and patient age. Most local recurrences occurred at the site of initial tumor excision (57–88%) or in the same breast quadrant (22–28%). In general, during the first 10 years after lumpectomy with radiation, the recurrence rate is 1.4% per year. The treatment of in-breast tumor recurrence in patients is completion mastectomy.9 Therefore, more radical excision including the tumor with large surrounding tissue (quadrectomy or partial mastectomy) is recommended; however, it may result in an unacceptable aesthetic outcome.
Diagnosis/patient presentation
Breast-conserving approaches may be preferable over a total mastectomy. This is especially true for patients with early stage disease, because the majority of the breast can be preserved and the operation is perceived as less invasive than a mastectomy. Most early-stage cancers (T1 and T2 cancers with or without nodal involvement) are indicated for BCT; however, there are some exceptions. Commonly known reasons for contraindications are: patients with a high probability for recurrence, especially those with multicentric disease; those who are pregnant; have collagen vascular disease; or those who have a history of prior radiation therapy.10 Relative contraindications include: patients with a high probability of subsequent cancers (BRCA mutations) and patients who are likely to have a poor cosmetic result, which includes patients with a high tumor/breast ratio, medially and inferiorly based tumors and tumors that require removal of the nipple-areola complex.
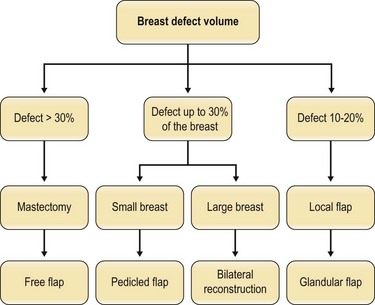
Approximately 10–30% of patients are dissatisfied with the aesthetic result after partial mastectomy with radiation.11 Although the causes may vary, the factors that motivate patients to seek corrective surgery after partial mastectomy are: a volume discrepancy, contour deformity, and nipple malposition.12 The resection of more than 15–20% of the breast parenchyma in a small-volume (A or B cup) and more than 30% in larger breasts will cause volumetric deformities and bilateral asymmetries. In addition, radiation therapy extenuates the image of the breast, initially causing breast edema and skin erythema and eventually causing parenchymal fibrosis, retraction, skin envelope atrophy, hyperpigmentation, hypopigmentation and telangiectasia. The long-term final effects of radiation are difficult to predict but seem to stabilize 1–3 years post-radiation.13,14
Type of reconstruction
1. Volume displacement techniques refer to advancement, rotation or transposition of large local breast flaps into the smaller created defect, redistributing the volume loss. The dissection involves the advancement of a full-thickness segment of breast fibroglandular tissue to fill the dead space. Volume displacement procedures and surgical scars are optimal when combined with mastopexy-reduction techniques. The tumor is excised within the planned markings of the reduction specimen in medium, large, or ptotic breasts, and the remaining parenchyma is sufficient enough to reshape the breast mound.15
2. Volume replacement techniques are technically more difficult and are used in small–moderate sized breasts or when the tumor/breast ratio is large and the remaining breast tissue is insufficient for the rearrangement and the replacement of the defect. Volume replacement with the use of non-breast local or distant flaps provides both tissue for the filling of the glandular defect and the skin deficiency of the reconstructed breast.16
Timing of reconstruction
Reconstruction of partial mastectomies can either include delayed, immediate or immediate-delayed procedures. In delayed reconstruction, at least 6 months to 1 year is allowed to elapse after the last radiation therapy session, in order to evaluate the deformities of the breast and plan the appropriate reconstruction modality. In immediate reconstruction, the goal is to perform co-instantaneously tumor resection in oncologic appropriate margins and partial breast reconstruction. Immediate-delayed partial breast reconstruction is actually a two stage immediate surgical approach with delayed reconstruction (within a few days) until the results of the pathology report are known and the margins of the tumor excision are determined as sufficient or must be re-excised to clear margins before reconstruction. The final aesthetic outcomes are similar to those with immediate reconstruction.17
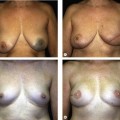
Immediate breast reconstruction offers many advantages over delayed breast reconstruction, such as a better aesthetic outcome due to the preservation of the three-dimensional breast skin envelope. The aim of immediate reconstruction after breast conserving therapy (BCT) is essentially to give better aesthetic results with the same oncological safety. Our target should be the reshaping of the treated breast with the alteration of the opposite breast for better symmetry, as part of the treatment, when indicated. Immediate reconstruction before radiation therapy is also preferred, because the breast can be better manipulated before radiation, with potentially decreased complication rates and improved aesthetic outcomes. In planning the approach on which method should be used to treat the partial mastectomy defect, the primary decision that must be made is whether reparative surgery will be needed after the tumor excision. If oncoplastic surgery is necessary, the next step is to choose the technique. If there is sufficient tissue, reshaping the breast, along with reduction of the opposite breast for symmetry is usually the best option. If there is not enough tissue left for breast reshaping, tissue must be added. If the defect is not too large, local tissue can be used, with simple techniques, such as rotation or transposition fasciocutaneous flaps from the axillary area or superiorly based composite flap of skin, subcutaneous fat and upper pole breast parenchyma. If the defect is too large to be filled by local tissue alone, a loco-regional pedicled or distal pedicled or free flap will be required (Fig. 21.1).
Breast and plastic surgeons must have a thorough understanding of breast anatomy, physiology, and the qualities of an aesthetically pleasing breast shape. Surgeons performing the oncoplastic approach should consider the aesthetic subunits when planning cosmetic quadrectomies, resections, and reconstructions.18 Also, knowledge of the anatomical landmarks, breast proportions, and shape is essential in order to achieve a pleasing outcome.
The base and width of the breast, the width of the NAC, the height of the nipple and the distance from the sternal notch, midline and inframammary crease, must be recorded in detail.18 Any natural breast asymmetry should be pointed out to the patient before surgery. Different body types, skin laxity and fat distribution are important factors in the decision-making process.
Treatment/surgical technique
Large defects in large breasts may be treated by converting a partial mastectomy into a reduction mammaplasty. Planning of skin incisions and parenchymal excisions follow templates using reduction mammaplasty and mastopexy techniques. The pattern can be rotated laterally or medially to fit the location of the defect. The choice of pedicle is related to the tumor location and the blood supply of the breast. It is important that skin incisions be planned, so that if mastectomy is ultimately required for margin control, the incision site can be comfortably included within the mastectomy skin island.6,15
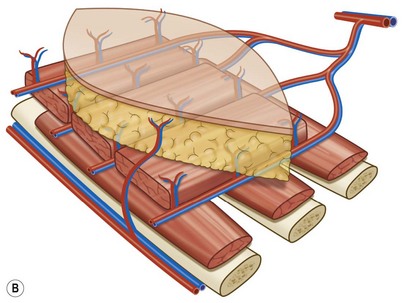
The latissimus dorsi muscle or musculocutaneous flaps have been very popular as a method of choice in partial breast reconstruction.19
Classification and vascular anatomy of flaps
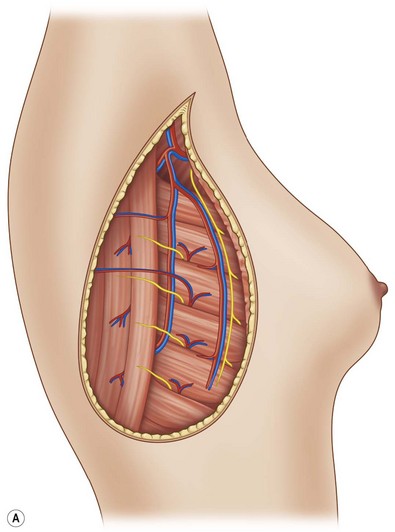
The latissimus dorsi (LD) flap has a constant anatomy.20 The blood supply of the latissimus dorsi comes from a terminal branch of the subscapular artery (Fig. 21.2). The subscapular artery runs about 5 cm before dividing into the scapular circumflex and thoracodorsal arteries. The thoracodorsal artery is about 2–4 mm in diameter and it courses along the posterior portion of the axilla for about 8–14 cm, before it pierces the latissimus dorsi on its costal surface. The thoracodorsal artery gives off one or two branches to the serratus anterior muscle and one branch to the skin. The basic pattern of the thoracodorsal bundle (artery, nerve and 1–2 venae comitantes) branching is bifurcation into a lateral (vertical) and a medial (horizontal) branch. The lateral branch follows a course parallel to the muscle fibers, 1–4 cm medial to the free-lateral border of the muscle and gives off perforating vessels that supply the skin. The smaller medial branch diverges at an angle of 45° and travels medially. A vigorous blood supply to the muscle is also available at its origin. Perforating vessels from the intercostal and lumbar arteries supply the muscle and overlying skin.20,21