Introduction
Local anesthetic agents cause reversible blockade of the neuronal sodium (Na + ) channel, thereby blocking transmission of nerve impulses from nociceptive afferents.
Mechanism of Action of Local Anesthetics
In the un-ionized state, local anesthetics are varyingly lipid-soluble, thereby penetrating the phospholipid cell membrane into the axoplasm. Here, re-ionization of the molecule takes place. It is this re-ionized molecule that then enters and blocks sodium channels, preventing sodium influx into the neuron during depolarization, and thus halting propagation of an action potential.
Local anesthetics have also been shown to exert far-reaching effects on other receptors, independent of their action on sodium channels. These include inhibition of the local tissue inflammation that results in nociceptor sensitization, leading to pain and hyperalgesia. They may also be involved in the priming of descending inhibitory pain pathways, which could show clinical benefit in the reduction of neuropathic pain. Furthermore, local anesthetics have been shown to have antithrombotic, antimicrobial, and neuroprotective actions. ,
Nerve Fiber Characteristics and Differential Blockade
Local anesthetics show a greater affinity for receptors within sodium channels during their activated and inactivated states (i.e., during neuronal stimulation) compared to their resting states. Hence neurons that fire more rapidly are more susceptible to local anesthetic block.
Other nerve fiber characteristics have been shown, in vitro, to govern the sensitivity of nerve fibers to local anesthetics.
- •
Smaller nerve fibers with shorter axons are more easily blocked compared to larger never fibers with longer axons.
- •
Smaller diameter nerve fibers (e.g., 0.4–1.2 μm C-fibers, which transmit pain, temperature, and touch) are blocked at lower concentrations compared to larger diameter nerve fibers.
- •
Myelinated fibers (e.g., A-α fibers, which convey motor and proprioception) are more easily blocked than unmyelinated nerve fibers (e.g., C-fibers), as local anesthetic tends to pool within the myelin sheath.
The aforementioned principles do not, however, consistently correlate with the clinical observations. Nevertheless, when blocking peripheral nerves, there is usually an order in the disappearance of sensing modalities: autonomic, pain, temperature, deep pressure, and finally, motor function.
Physiochemical Properties of Local Anesthetics
Chemistry
Local anesthetics are weak bases. Their basic molecular structure is comprised of a lipophilic aromatic moiety and a hydrophilic amino domain ( Fig. 4.1 ).
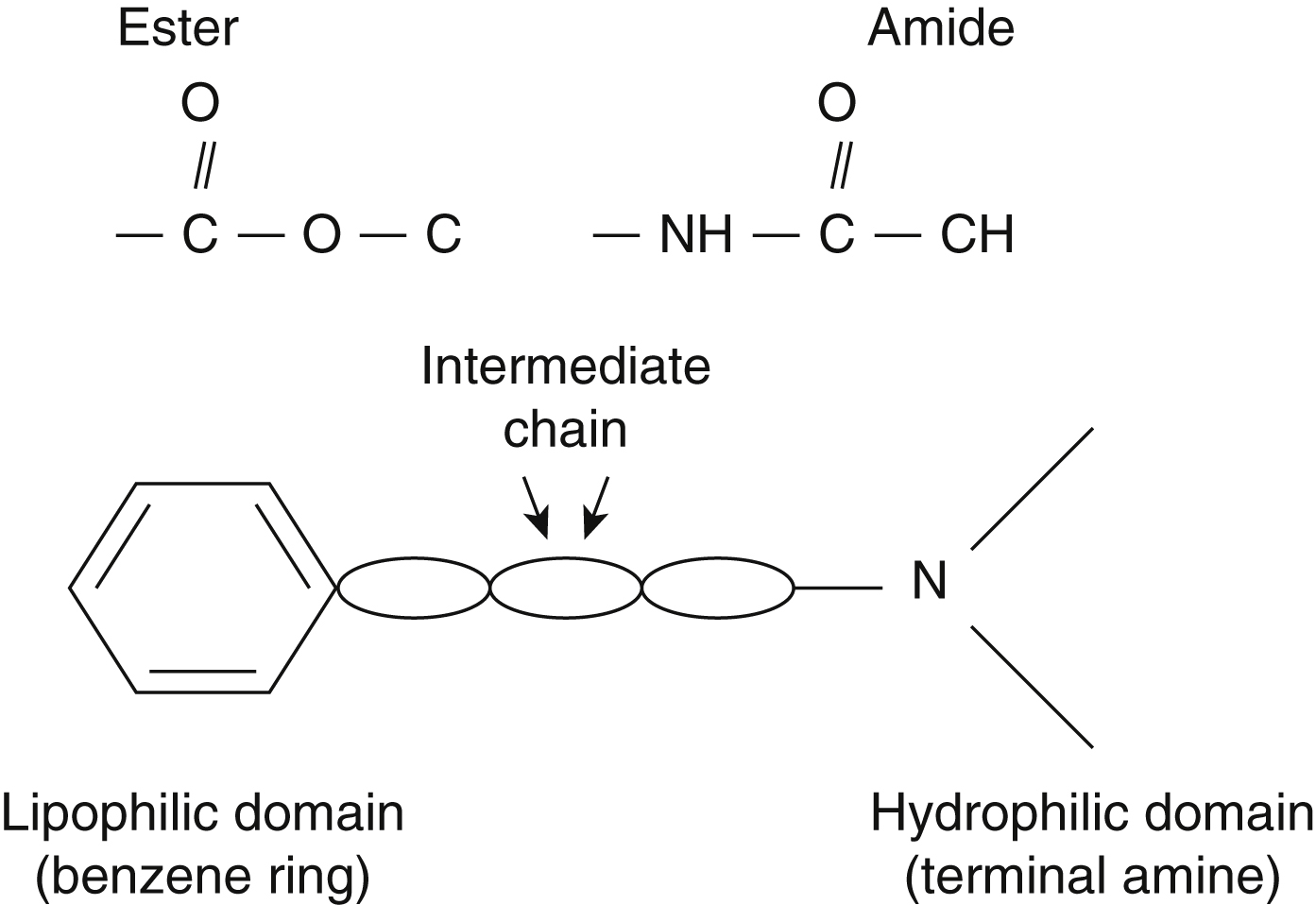
Depending on the chemical linkage between the respective lipophilic and hydrophilic groups, local anesthetics can be classified as amides or esters. This linkage has implications on drug metabolism and allergenicity ( Table 4.1 ).
| Class | Example | Allergenicity a | Pharmacokinetics | Stability |
|---|---|---|---|---|
| Amides | Lidocaine Mepivacaine Bupivacaine Ropivacaine Levobupivacaine | Rare | Extensively protein-bound Hepatic metabolism by amidases; slower than ester metabolism thus potential for accumulation | Long shelf-life and heat-insensitive unless mixed with glucose |
| Esters | Procaine 2-Chloroprocaine Tetracaine (Amethocaine) Benzocaine Cocaine | Rare, but more common with the amides, especially with procaine Cross-reactivity may exist with other esters Implicated metabolite: p- aminobenzoic acid | Minimally protein-bound Hydrolysis by plasma cholinesterase and other esterases | Short shelf-life due to spontaneous hydrolysis |
a Antioxidant additives, or preservatives such as metabisulfite or parabens may be implicated in causing non‐anaphylactic reactions.
Potency
The potency of local anesthetics correlates well with lipid solubility and molecular weight in vitro.
Other factors affecting potency include tissue distribution and direct vasodilator actions of the local anesthetic.
Duration of Action
Local anesthetics are classed as “short-”, “medium-”, or “long-” acting.
The duration of action of local anesthetics positively correlates with the degree of protein binding; the more protein-bound a local anesthetic, the longer the duration of action, with the “unbound” portion of the drug being biologically active. However, local anesthetic molecules remain bound to sodium channels for seconds only. Given such a high dissociation rate, of greater relevance are factors that maintain a high concentration of local anesthetic molecules around the nerve. These factors are:
- •
local anesthetic lipid solubility: there is a direct correlation between lipid solubility and protein binding
- •
tissue vascularity: this depends on regional blood flow and the addition of vasoconstrictors
- •
intrinsic vasodilatory properties of local anesthetics.
Local anesthetics generally cause vasoconstriction at very low concentrations and vasodilatation at higher, clinically useful, concentrations. Considerable variability exists, however. Moreover, ester local anesthetics are more potent vasodilators compared to amides. The exception to this rule is cocaine, which causes vasoconstriction by blocking reuptake of norepinephrine at sympathetic nerve endings. The greater the intrinsic vasodilatory action of a local anesthetic, the more pronounced the effect of the addition of vasoconstrictors.
Onset of Action
The faster the diffusion of local anesthetic across a nerve sheath, the faster the onset of action. The rate of diffusion is determined by:
- •
the site of local anesthetic injection
- •
the concentration of local anesthetic used
- •
the degree of ionization of local anesthetic molecules.
The latter is related to a physiochemical property of a drug known as the pKa. The pKa is the pH at which 50% of the drug is present in an ionized form. For basic drugs, such as local anesthetics, a greater proportion of drug is present in the ionized form at a pH below the pKa; at a pH above the pKa, a greater proportion of the drug will be un-ionized. The reverse is true for weak acids. Therefore, local anesthetics with a higher pKa will have a greater drug fraction present in the ionized state, resulting in a slow onset of action. The most clinically relevant comparison is the faster action-onset of lidocaine (pKa 7.9) compared to bupivacaine (pKa 8.1).
Pharmacokinetics of Local Anesthetics
Absorption
Absorption of local anesthetics primarily depends on:
- •
the site of injection
- •
the addition of vasoconstrictors
- •
the physiochemical properties of the local anesthetic agent itself.
On reaching the systemic circulation, local anesthetics are taken up by distant organs according to vascular density. Highly vascular organs such as the brain, heart, lung, liver, and kidneys, will have greatest exposure to the peak concentration of unmetabolized local anesthetic. Uptake is determined by the tissue-plasma partition coefficient.
Distribution
Local anesthetic molecules can be “free” or bound to plasma proteins.
Alpha-1 acid glycoprotein binds the weakly basic local anesthetic molecules with high affinity. However, albumin represents a greater fraction of plasma proteins, and as such has a high capacity (but comparatively low affinity) for binding to local anesthetic molecules.
Local anesthetic protein binding is pH-dependent and decreases as the pH decreases. This has important clinical relevance. For example, in local anesthetic systemic toxicity (see below), a metabolic acidosis may ensue secondary to tissue hypoxia. As the plasma pH decreases, the proportion of “free” drug increases, potentiating the toxicity. Furthermore, intracellular acidosis increases conversion of the local anesthetic molecule into the cationic state, rendering the molecule unable to cross the cell membrane. This “ion trapping” exacerbates the toxicity further.
Clearance and Metabolism
Esters (except cocaine) undergo rapid hydrolysis by plasma cholinesterases, and other non-specific esterases. Their metabolites are inactive but may be associated with hypersensitivity reactions. Amides are metabolized in the liver by the action of amidases.
Hepatic clearance is dependent on the hepatic blood flow, and the hepatic extraction ratio.
Hepatic extraction ratio depends on the ratio of unbound to protein-bound drug. Drugs that are comparatively less protein-bound (e.g., lidocaine) have a higher hepatic extraction ratio. Clearance is thus only limited by hepatic blood flow. Hence, factors reducing hepatic blood flow may increase toxicity, e.g., heart failure.
Conversely, the clearance of drugs which are more protein-bound (e.g., bupivacaine) is more dependent on free drug concentration, as opposed to hepatic blood flow. Intrinsic liver disease may, in turn, affect free drug concentration, e.g., a reduction in liver enzymes or reduced plasma protein production.
Patients with liver disease and cardiac disease can receive “normal dose” single-shot blocks. However, catheter infusions or repeated blocks should be given at a reduced dose.
Renal failure associated with severe uremia can be associated with reduced clearance and more rapid absorption of local anesthetic, leading to higher peak plasma concentrations.
Some local anesthetics may undergo first pass metabolism by lung extraction due to the lower pH of the lungs compared to the plasma, resulting in an “ion trapping” effect. This effect is lost in patients with right to left cardiac shunts, thus increasing the risk of toxicity.
Local Anesthetics in Clinical Practice
Table 4.2 summarizes the pharmacokinetic and pharmacodynamic properties of the most commonly used local anesthetics.
| Amides | Esters | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Lidocaine | Mepivacaine | Prilocaine | Bupivacaine | Levobupi-vacaine | Ropivacaine | Procaine | Amethocaine | Cocaine | |
| Onset of action | Fast | Fast | Fast | Medium | Medium | Medium | Slow | Slow | Medium |
| Relative potency | 2 | 2 | 2 | 8 | 8 | 8 | 1 | 8 | – |
| Duration of action) | Medium | Medium | Medium | Long | Long | Long | Short | Long | Short |
| Maximum dose (mg/kg) a | 3 | 5 | 6 | 2 | 2.5 | 3 | 7 | 1.0 | 1.5 |
| Maximum dose with epinephrine (mg/kg) | 7 | 7 | 9 | 2.5 | 3 | 4 | 10 | 1.5 | NA |
| Additional notes | Relative lack of vasodilatation Risk of methemoglobinemia | Greatest risk of cardiotoxicity Unpredictable block duration | S -enantiomer of bupivacaine; modestly less cardiotoxic and neurotoxic | Pure S- enantiomer Lower propensity for motor block compared to bupivacaine Marked vasoconstriction at low concentration | Local vasodilatory action Lower propensity for motor block compared to bupivacaine Marked vasoconstriction at low concentration | Moffat’s solution: 2 mL 8% cocaine, 1 mL of 1: 10,000 epinephrine and 2 mL of 8.4% sodium bicarbonate Blocks reuptake of norepinephrine, thus can provoke hypertensive crisis and arrhythmias Potent vasoconstrictor | |||
a Quoted maximum “recommended safe doses” vary. Most available data are extrapolated from animal studies. Other factors, including the site and speed of injection, are of greater relevance (see text).
Concentration of Local Anesthetics
A 1% solution of a drug contains 1000 mg of drug per 100 mL of solution. Thus a 1% solution of lidocaine contains 1000 mg of lidocaine per 100 mL (10 mg/mL). Similarly, a 0.5% solution of bupivacaine contains 500 mg of bupivacaine in 100 mL of solution (5 mg/mL).
Clinically Useful Adjuncts
Vasoconstrictors
Epinephrine is the most commonly used vasopressor. It slows the rate of absorption of local anesthetic, thereby prolonging the duration of action and reducing the risk of toxicity. Furthermore, it can help decrease surgical blood loss, and may identify inadvertent intravascular injection. Absolute contraindications to use include injection close to tissues with inadequate collateral circulation (ankle, wrists, digits, and penis); as well as intravenous regional anesthesia. Relative contraindications to use include the presence or risk of tissue ischemia, cardiac disease, hypertension, and concomitant use of other sympathomimetic drugs (e.g., tricyclic antidepressants).
The maximum safe dose of epinephrine is 4 μg/kg; concentrations in excess of 1:200,000 should not be used ( Table 4.3 ).
| Drug Solution | Drug Concentration (μg/mL) |
|---|---|
| 1 : 10,000 | 100 |
| 1 : 100,000 | 10 |
| 1 : 200,000 | 5 |
| 1 : 400,000 | 2.5 |
Sodium Bicarbonate
Sodium bicarbonate (NaHCO 3 ) raises the pH of the local anesthetic solution, thus increasing the fraction of un-ionized drug able to traverse the neuronal membrane. It may also reduce the pain of injection. Furthermore, the speed of onset of the local anesthetic is theoretically increased. However, the rise in pH seen is only modest, and limited by the solubility of the base form of the drug. This translates to only modest decreases in block onset time.
The recommended dose is 1 mL of 8.4% sodium bicarbonate per 10 mL of local anesthetic.
Topical Local Anesthetics
Topical local anesthetics are available in a variety of formulations and presentations. Their advantages include avoidance of first-pass metabolism, and avoidance of the risks of more invasive local anesthetic techniques.
The “ideal” topical local anesthetic formulation will maximize permeability and absorption via the stratum corneum, while limiting systemic absorption hence toxicity. As a general rule, drug delivery is enhanced by prior skin exfoliation and degreasing, as well as the application of a nonporous dressing (e.g., Tegaderm).
Disadvantages of topical local anesthesia include unpredictable absorption, uneven distribution or spread of the agent during application, and a failure of the occlusive dressings to stick to the skin. These agents should not be applied to broken, irritated, or infected skin.
Topical anesthesia lends itself well to procedures such as skin biopsy and removal, electrocautery, and superficial laser therapy. It also enables painless needle insertion.
Cutaneous Agents
The most commonly used topical agents are summarized in Table 4.4 .