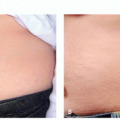
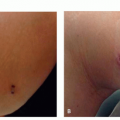
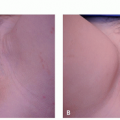
Lentigines
E. Victor Ross
BACKGROUND
The lentigo is one of the most common presenting complaints in a cosmetic dermatology practice, the treatment of red and brown dyschromias accounting for up to 50% of first-time patients (VR, unpublished data). Lentigines occur in over 90% of Caucasians, with genetic predisposition, aging, and chronic sun exposure being the most important contributing factors.1 Solar lentigines should be distinguished from ephelides, lentigo simplex, pigmented actinic keratosis (AK), flat seborrheic keratosis (SK), melanocytic nevus, and malignant melanoma. Some lentigines are actually early SKs, and one can make the case that most lentigines evolve into SKs. The lentigo must be differentiated from other types of epidermal pigmentation with sometimes similar clinical presentations. These can be differentiated based on clinical appearance from other types of so-called static hyperpigmentation. The term “static” hyperpigmentation is used to differentiate the lentigo from inflammatory variants of hyperpigmentation, most notably melasma, a more complicated condition in which histological and physiologic properties are different from the lentigo.
Any cosmetic dermatologist must be able to treat these macular lesions in a predictable fashion. In this chapter, we review the range of both physical and topical therapies.
PRESENTATION
Patients complain of dullness, discolorations, or brown spots to the skin. They may give a history of sudden appearance or worsening of dark spots usually after periods of increased sun exposure. The discolorations involve sun-exposed skin.
DIAGNOSIS
Histopathology
Under the microscope, the lentigo presents as an area of basilar hypermelanosis with mild epidermal acanthosis and variable elongation of the rete ridges, and an increased number of melanocytes.
Subtypes
Solar Lentigines
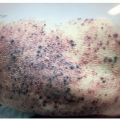
Solar lentigo (eg, actinic lentigo, senile lentigo, sun spot, liver spot) is the most common benign photo-induced lesion. Solar lentigines are photo-distributed on the face, extensor arms, dorsal hands, and V-distribution of the chest. They present <5 mm in diameter, slowly enlarge and darken, and may coalesce to form patches. The lesion surface may be flush, may be atrophic, or may possess overlying crepe-like rhytids. The color may range from yellow-tan, dark brown, and brown-black. On dermoscopy, a faint pigmented network with uniform pigmentation is noted.
On light skin types, solar lentigines may appear in the second or third decade, although most commonly they appear after the age of 30 and increase with each decade.
Lentigo Simplex
Lentigo simplex (eg, simple lentigo, juvenile lentigo) is the most common form of lentigo and not related to sun exposure nor to systemic disease. The lesions are round-to-oval 3- to 15-mm hyperpigmented macules with either irregular or smooth borders. In simple lentigo, pigmentation is evenly distributed, and the color ranges from brown to black. Lesions are few in number and occur anywhere on the skin or mucous membranes.
In contrast to the solar lentigo, lentigo simplex usually appears in early childhood, although it may be present at birth or may occur later.
Familial Lentiginoses
A number of genetic syndromes are associated with the presence of multiple, mucosal or diffuse lentigines; the most prevalent are listed here.
Peutz-Jeghers Syndrome. Lentigines are distributed over the perioral region, oral mucosa, and hands. This disorder is associated with GI polyps and tumors of the GI tract, pancreas, breast, ovary, and uterus. Autosomal dominant.
Carney Syndrome/NAME/LAMB. Carney syndrome, NAME (nevi, atrial myxoma, myxoid neurofibroma, ephelides), and LAMB (lentigines, atrial myxoma, mucocutaneous myxoma, blue nevi) present with lentigines, nevi, and blue nevi and associated with myxomas, myxoid neurofibromas, acromegaly, and breast and testicular tumors. Autosomal dominant.
LEOPARD Syndrome. Lentigines present in early infancy/childhood. Associated with ECG changes, aneurysms, ocular hypertelorism, pulmonary stenosis, abnormal genitalia, short stature, deafness. Autosomal dominant.
Cronkhite-Canada Syndrome. Lentigines of buccal mucosa, face, hands/feet. Typically affects older men and associated with alopecia, nail dystrophy, intestinal polyposis. Sporadic.
Differential Diagnosis
Ephelides (freckles)
Lentigo simplex
Pigmented actinic keratosis
Flat seborrheic keratosis
Nascent seborrheic keratosis
Melanocytic nevus
Lentigo maligna/malignant melanoma
Genetic lentiginosis syndromes (eg, LEOPARD syndrome, Carney complex, Peutz-Jeghers syndrome, Laugier-Hunziker syndrome, xeroderma pigmentosum, and others)
PATHOGENESIS
Solar lentigines appear to be due to chronic UV radiation-induced epidermal hyperplasia, concomitant melanocyte proliferation, and increased transfer of melanosomes to keratinocytes. UV radiation stimulates keratinocyte production of various cytokines including interleukin 1, which has been shown to lead to keratinocyte growth factor production, stimulating pigment production via tyrosinase expression.2 The melanocortin-1-receptor gene (MCR1) variants have
been identified as “the freckle gene,” and variants in MCR1 are implicated in solar lentigo formation.3
been identified as “the freckle gene,” and variants in MCR1 are implicated in solar lentigo formation.3
Mutations in other genes causing inherited lentiginosis syndromes also appear to be involved in the pathogenesis of lentigines (eg, PTPN11, STK1, NF1, PRKAR1A, PTEN, and others), but the mechanisms and pathways are still being delineated.
TREATMENT
Ortonne et al1 reviewed lentigo treatment and assigned an efficacy score based on the literature (Table 1.1.1). Broadly, lentigo treatments may be categorized as (1) physical and (2) topical approaches. Physical therapies may be further subdivided into (a) selective and (b) nonselective interventions. Nonselective therapies (meaning that there is no intrinsic selectivity for the excess pigment) include chemical agents, cryotherapy, electrosurgery, dermabrasion, and laser where water is the chromophore, namely erbium YAG, erbium YSSG, and CO2 lasers. Physical therapies (such as light, laser, cryotherapy, and peels) are frequently used with excellent results, but these interventions must be balanced against associated side effects and recurrence rates.1,4 A treatment algorithm approach to lentigines is presented (Algorithm 1.1.1).
Medical
Medical therapy is the first component of treatment of lentigines and typically includes topical therapy.
Topical Therapies
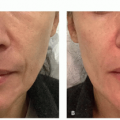
Hydroquinone and Retinoids. This category of treatment refers to multiple applications of an agent to reduce the pigmentation in the lentigo. Hydroquinone (HQ) and retinoic acid (RA) are two of the main ingredients in these products, although over the past 10 years a number of non-HQ products have been introduced as alternatives, much of this driven by papers that suggest a possible mutagenic role for HQ.5 The full spectrum of RA has been applied, from tretinoin, adapalene, and tazarotene. Variants of so-called triple bleaching creams (RA + steroid + HQ) have been applied to lentigines as well.
Although the potential adverse effects of HQ have been noted in the literature, most physicians agree that HQ is a safe and effective compound.6 We particularly find it helpful in the recovery phase after laser treatment of lentigines in some darker skinned patients, where the risk of postinflammatory hyperpigmentation (PIH) is high. Levitt noted the HQ controversy and countered that the typical HQ content from surface applications is less than that from common foods, and that the types of carcinomas found in mice were only after prolonged exposures and higher doses; he also noted that some types of tumors actually decreased in incidence in the animal studies.6
Triple bleaching creams (adding a topical steroid to an RA and HQ preparation) have been used for lentigines, but newer non-HQ-containing creams have recently been applied. In one study, Dreher et al7 used four “actives” in a non-HQ-containing kit. After a washout period of 4 weeks with an SPF 30 sunscreen, Journée Bio-restorative Day Cream (Neocutis, Inc, San Francisco, CA, USA) was applied in the morning. The subjects additionally applied the study cream twice daily containing 5% of the proprietary cream called Melaplex (Neocutis, Inc) to the entire face. This complex contained disodium glycerophosphate, L-leucine, phenylethyl resorcinol, and undecylenoyl phenylalanine, all chemicals of a relative small molecular weight of less than 500 g/mol, which indicates that those chemicals are able to penetrate skin. The sunscreen provided some UVA protection. They found that when using Melaplex (Neocutis), reviewers observed a 32% pigment decrease 12 weeks later.
Fabi-Goldman8 compared two HQ-containing kits for lentigines and general hyperpigmentation. In accordance with a predetermined randomization schedule, subjects were assigned to either the four-product SkinMedica Hyperpigmentation System (SKM) (cleanser, microentrapped HQ 4% with retinol and antioxidants, physical sunscreen SPF 30, and tri-retinol 1.1%) or the seven-product Obagi Nu-Derm System (OMP) (cleanser, toner, “clear”-[HQ 4%], exfoliator, “blender”-[HQ 4%], sunscreen SPF 35, and tretinoin 0.025%). These were applied over 12 weeks. Both “systems” achieved similar improvement in overall pigmentation (from an average of “moderate” to an average of “mild” hyperpigmentation).
In another study,9 an HQ-containing kit was compared with a non-HQ-containing kit in a 12-week evaluation of general hyperpigmentation. In accordance with a predetermined randomization schedule, subjects were
assigned to either the HQ-free four-product kit, which included cleanser, skin brightening complex, sunscreen SPF 30+, and tri-retinol 1.1% products (Facial Cleanser, Lytera Skin Brightening Complex, Daily Physical Defense SPF 30+ Sunscreen, Tri-Retinol Complex ES [SkinMedica]) or the seven-product OMP kit, which included cleanser, toner, HQ 4% (applied twice daily), exfoliant, another HQ 4% (applied once daily), sunscreen SPF 35, and tretinoin 0.025% (Foaming Gel, Toner, Clear Exfoderm, Blender, Healthy Skin Protection SPF 35, Tretinoin Cream [Obagi Medical Products]). The study was conducted over a 12-week period.9 The investigators found equal efficacy among the two commercially available hyperpigmentation kits over the 12-week period.
assigned to either the HQ-free four-product kit, which included cleanser, skin brightening complex, sunscreen SPF 30+, and tri-retinol 1.1% products (Facial Cleanser, Lytera Skin Brightening Complex, Daily Physical Defense SPF 30+ Sunscreen, Tri-Retinol Complex ES [SkinMedica]) or the seven-product OMP kit, which included cleanser, toner, HQ 4% (applied twice daily), exfoliant, another HQ 4% (applied once daily), sunscreen SPF 35, and tretinoin 0.025% (Foaming Gel, Toner, Clear Exfoderm, Blender, Healthy Skin Protection SPF 35, Tretinoin Cream [Obagi Medical Products]). The study was conducted over a 12-week period.9 The investigators found equal efficacy among the two commercially available hyperpigmentation kits over the 12-week period.
TABLE 1.1.1 Level and Quality of Evidence for Solar Lentigines Therapies | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Trenaxemic Acid. An antifibrinolytic drug, tranexamic acid (TXA) has been used for hyperpigmentation topically and systemically. It is a synthetic lysine derivative that blocks lysine binding sites on plasminogen. Although TXA is primarily used for melasma, UV exposure increases plasminogen activator production by
keratinocytes and the blocking of the conversion of plasminogen to plasmin by TXA has shown to be effective in reducing melanogenesis and UV-induced hyperpigmentation. It has been used to prevent recurrence of solar lentigos treated by lasers and in combination cases of melasma and lentigos. Typical protocols involve nightly topical application from 5 to 18 weeks.10
keratinocytes and the blocking of the conversion of plasminogen to plasmin by TXA has shown to be effective in reducing melanogenesis and UV-induced hyperpigmentation. It has been used to prevent recurrence of solar lentigos treated by lasers and in combination cases of melasma and lentigos. Typical protocols involve nightly topical application from 5 to 18 weeks.10
Cosmetic
Chemical Peels
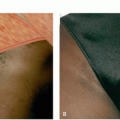
Chemical peels to improve irregular pigmentation have been performed on both facial and nonfacial skin using glycolic acid, TCA (trichloroacetic acid), Jessner solution, salicylic acid ointment, and salicylic acid liquid. Various chemical agents have been used to treat lentigines. A cotton tip applicator is normally used with either 20% to 60% TCA, phenol 88%, or other agents as well. Regardless of the agent, apply until a light uniform frost confined to the lesion is achieved. Solutions of 20% to 40% TCA in combination with 70% glycolic acid have also been used successfully to treat irregular pigmentation of facial and nonfacial skin, including solar lentigines. The principles underlying this treatment are that glycolic acid decreases the incidence of postinflammatory pigment changes, whereas TCA is able to penetrate deeply but uniformly into the skin.1 There have been a couple of comparisons between TCA and LN2 (liquid nitrogen). In one study, 25 patients were treated on the back of their hands with spray LN2 1 to 3 second vs 30% TCA spot application until frost in a split hand study. Both approaches have been shown to work, with more pain with LN2.11
Lasers and Light-Based Technology
The treatment of benign pigmented lesions with light technologies plays to the very strengths of these devices. Selective photothermolysis (SPT) allows for heating of melanosomes and surrounding hyperpigmented areas with relative sparing of the normal skin. Broadly, there are three ways to treat excess epidermal pigment: (1) visible (VIS) light technologies (IPL, KTP, Q-switched lasers, etc) that use the theory of SPT, (2) ablative lasers with confluent spots that remove the lesions from top to bottom using water as a chromophore, and finally (3) fractional lasers, through a series of treatments, can also reduce the amount of pigmentation in the skin
Most providers use VIS light to treat lentigines. The devices rely on color contrast to heat the lesion and spare the background normal skin.
Based on the following formula, the temperature increase (ΔT) of the epidermal lesions is observed, as follows:
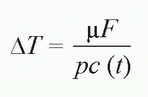
where F is the surface fluence, µ is the absorption coefficient (the relative amount of absorption for melanin for that particular wavelength or wavelength range), pc is a constant, and t represents a reduction in the peak T elevation based on the length of the pulse. It follows that the shorter the laser or IPL pulse, the greater is ΔT. The equation is based on energy conservation and is a simplification of the mathematical foundations of SPT12 whereby spatially selective heating is achieved by:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree